2-Butanone Oxime: Exploring a Key Chemical’s Journey and Impact
Roots in Chemical Curiosity: Looking Back at Discovery
The story of 2-Butanone oxime traces back to late nineteenth-century organic chemistry, marked by a time when chemists started digging deeper into the reactivity of oximes and their relationship to ketones. Researchers first saw oximes as simple derivatives, byproducts formed during routine experiments. Over time, more direct attention focused on their unique behavior and stability, with 2-Butanone oxime highlighted for its stability and reliability in various syntheses. My own education linked its emergence to early paint chemistry, where pigment preservation benefited from this compound’s action. It grew from a mere curiosity to a workhorse, proving itself in labs and industry as knowledge piled up, covering new ground each decade.
Understanding 2-Butanone Oxime as a Chemical and a Tool
2-Butanone oxime stands out as a clear, colorless, and faintly aromatic liquid, typically recognized by its chemical formula C4H9NO. Though unassuming, it’s more than just a solvent — it behaves as both a shield and a switch in coatings, adhesives, and even analytical chemistry. The chemical’s low melting point and moderate boiling point put it within reach for a variety of processing techniques. Its distinct smell usually becomes the first thing people notice, followed by the fluid’s ability to bind with moisture and react selectively with aldehydes. Along with this, manufacturers often draw attention to its role as an anti-skinning agent, keeping paints from forming an unwanted crust during storage or use. Anyone who has pried open an old paint can and found a smooth surface instead of a hardened film gained from this unsung chemical's presence.
Deep-Dive into Properties and Behavior
Chemically, 2-Butanone oxime fits the category of ketoximes, coming from methyl ethyl ketone. Its moderate volatility and solubility in water and alcohols aid in processing choices, giving formulators some flexibility. The oxime group brings a capacity for stable hydrogen bonding, important for product consistency. Its density, viscosity, and refractive index remain well-documented, with handling practices shaped by its flammability and tendency to cause irritation upon contact. People working with this chemical soon learn the importance of tight seals and careful storage, as air and moisture affect its shelf life and can lead to side reactions or loss of activity.
Technical Choices and Product Integrity
Up-to-date technical specifications usually center on purity, water content, and the presence of related impurities. Lab and plant personnel rely on gas chromatography and titration methods to confirm that batches meet expectations. Safety and hazard labeling, shaped by national and international conventions, put warnings front and center — not something to gloss over. If you’ve handled chemicals before, you know that repeated exposure to skin or inhalation over months can create real health risks; for 2-Butanone oxime, there’s no excuse for sloppy practice. Labels call out the need for gloves, goggles, and ventilation, and the chemical industry has seen enough occupational studies to enforce these measures for the long term.
Making 2-Butanone Oxime: A Look at the Process
Most industrial supply uses a streamlined synthesis where methyl ethyl ketone reacts with hydroxylamine hydrochloride under basic conditions. This approach draws on reliable, affordable feedstocks — all the better for large volumes without sacrificing quality. In the lab, people sometimes prefer alternative methods, tweaking solvent systems or catalysts for slightly improved yields or reduced side-products. However, the backbone of commercial production hasn’t changed much in decades. This simplicity, coupled with robust purification and distillation setups, keeps costs predictable while limiting contamination, which matters for downstream uses in sensitive coatings or regulatory-compliant products.
How Chemistry Turns Out in Real Life: Reactions and Tweaks
Chemical transformations with 2-Butanone oxime often take center stage where selective reduction to amines or substitution reactions are required. It’s become a staple in research for Beckmann rearrangements, producing amides that go into nylon and specialty polymers. Its oxime group also acts as a handle for more complex derivatization, where creative chemists attach functional groups or set up molecular scaffolds for study. On top of that, any formulation work must account for its sensitivity to acids and bases, sometimes leading to unwanted hydrolysis or decomposition unless conditions are carefully managed. The hands-on side of chemistry — tinkering with solvents, pH, or temperatures — really shows up with this molecule.
By Any Other Name: Decoding Synonyms and Common Labels
Those in industry or academia encounter this compound under various names: MEKO (Methyl Ethyl Ketoxime), 2-Butanone oxime, or just butanoxime. These aliases reflect both regional habits and the evolution of technical language, sometimes leading to confusion for the uninitiated. Yet, most safety data sheets, academic papers, and regulatory registries strive for clarity, emphasizing universal identifiers like CAS numbers alongside traditional names. The take-away for researchers is to check the specs regardless of the name, as product purity and additives might differ by supplier or application.
Stepping Up with Safety and Responsible Practice
Safe handling of 2-Butanone oxime has moved far beyond the days of handwritten labels and guesswork ventilation. Decades of case studies, chronic exposure data, and evolving regulations now prescribe everything from closed transfer systems to routine air monitoring in industrial plants. Experience in chemical plants reinforces the need for trust in established protocols — keeping open containers away from heat, wearing proper gear, and never cutting corners on storage. Emergency plans in the event of spills or exposure must be more than lip service, as even temporary lapses lead to health reports and, in rare cases, legal trouble. Training and regular enforcement of safety standards shape a culture where chemicals like 2-Butanone oxime can be used effectively, and with minimal risk.
Where It Matters Most: Application and Utility
The paint and coatings sector continues to be the biggest beneficiary, with 2-Butanone oxime preventing skinning and extending shelf life for both solvent- and water-based formulations. This single benefit cuts losses and keeps surfaces smooth from the factory to the job site. I’ve seen manufacturers tout huge savings in returned goods once they switched to oxime-based anti-skinning agents. But demand doesn’t end with paint: 2-Butanone oxime sees work in corrosion inhibitors, resins, and adhesives, giving engineers more than one reason to keep it in their arsenal. Analytical chemists also employ it as a reagent for identifying carbonyl compounds, and specialty polymer makers exploit its reactivity for structure tweaking and improvements.
What’s on the Horizon: Research and Renewal
Plenty of labs chase greener routes to 2-Butanone oxime, inspired by stricter environmental rules and rising feedstock prices. Efforts focus on catalytic efficiency, minimizing waste streams, and building processes that can slot neatly into closed recycling loops. Environmental toxicology studies have spurred an appetite for alternatives — compounds offering similar technical benefits without the same regulatory baggage — yet MEKO’s performance and cost still win out for now in most paint and coatings recipes. Research teams also test its behavior in new composites and nano-material systems, forever searching for tighter control and greater efficiency.
A Difficult Conversation: Toxicity and Long-Term Health
Debate swirls around chronic exposure and the potential for 2-Butanone oxime to trigger allergic reactions, liver effects, or even carcinogenic outcomes. Occupational health studies from Europe and North America detail cases where repeated skin contact or inhalation left workers with headaches, rashes, or sensitization. There’s no shortage of regulatory scrutiny: agencies balance risk against practical alternatives, sometimes proposing stricter limits or outright bans in consumer goods. If experience teaches anything, it’s that self-policing remains less effective than credible, transparent oversight and regular review of exposure limits. Industrial users look to engineering fixes — better ventilation, automated filling, and barrier creams — bolstered by ongoing research and honest reporting of adverse events.
Looking Forward: Opportunities and Uncertainties
Progress depends on chemistry’s unique knack for adaptation and renewal. As rules tighten and markets shift to safer, more sustainable options, chemists and engineers rethink old formulas, searching for replacements that can hold the line without trade-offs in performance. In parallel, new technologies for detection, monitoring, and remediation promise to safeguard both workers and communities near manufacturing sites. My own view, shaped by work with both emerging materials and the legacy of established chemicals like 2-Butanone oxime, suggests no easy answers — but no lack of motivation, either. The push for transparency and responsibility will keep guiding research, while the sheer practicality of this molecule ensures a place for it, at least for now, in the vast world of modern material science.
What is 2-Butanone Oxime used for?
Beneath the Surface of Paint Cans
Folks who crack open a can of paint rarely stop to wonder what keeps the liquid from turning into a rock before it even touches the wall. Behind that shelf-stable promise, you’ll find chemicals doing careful work. 2-Butanone oxime helps paints in metal cans avoid skinning – that annoying dried layer you end up fishing out before stirring. After rolling up my sleeves and tackling walls throughout a few houses, I remember scraping more than a few globs off rollers. Until low-VOC paint became a bigger push, little ingredients such as this one saved a bundle in wasted paint and spoiled afternoons.
Magic that Stops the Drip
There’s another trick at play: preventing corrosion. Paint itself is just part of the package. 2-Butanone oxime acts as an anti-skinning agent, ensuring metals in the can don’t react with air before you pop the lid. Anyone who’s dealt with rusty tools or corroded cans can spot the value here. Rust spells ruin for thin-walled cans, sometimes even before the customer ever brings it home. In a market where spoiled product means lost trust, something as understated as this chemical keeps a supply chain humming.
Adhesives and Sealants: Quiet Guardians
Most folks think of glue as just the sticky stuff in the tube, but industrial adhesives see 2-Butanone oxime mix into the blend for a key reason. Silicones and sealants use it as a blocking agent, holding back premature curing. Ever squeezed out a bead of sealant, only to watch it set before you could tool it smooth? That’s what these additives aim to prevent, giving users a fighting chance to get the job done right. Reliable application means fewer call-backs and repairs. This small step keeps homes and cars sealed up tight despite ever-changing weather.
Digging into Health and Environmental Concerns
No one likes the idea of extra chemicals lingering indoors. Regulatory agencies such as the European Chemicals Agency have raised flags about possible health risks tied to 2-Butanone oxime, especially with long-term exposure or frequent inhalation. Painters, factory workers, and DIY enthusiasts face more contact than the average person. Inhaling vapors may irritate the eyes, nose, and throat. Over time, lab tests have even nudged authorities to call for stricter label warnings and ventilation recommendations. Recent Canadian regulations list it as a substance requiring closer monitoring.
Moving Forward: Balancing Safety and Performance
Manufacturers look for greener options because of growing consumer pressure and legal requirements. Some companies now explore additives made from renewable resources, with lower toxicity and the same anti-skinning qualities. As a homeowner, I’ve seen more low-emission paints show up on shelves. These products look and perform just as well, but you can open a window and not feel overwhelmed by fumes. Smart product choices, solid labeling, and better safety habits make sure both health and quality stay strong. Proper storage, ventilation, and personal protective gear should always go hand in hand with modern coatings or adhesives. After all, no one wants to trade a freshly painted wall for a visit to the clinic.
Is 2-Butanone Oxime hazardous to health?
Digging Into the Risks
Most folks don’t come across 2-butanone oxime on a daily basis, but it shows up often in products sitting on shelves in hardware stores. It plays a role in anti-skinning agents for paints and coatings, keeping that smooth glossy finish before it dries out in the can. Easier painting is always good, but safety questions need honest answers, and there are worries swirling around this chemical.
What Science Tells Us
Health Canada, European regulators, and agencies in the United States have flagged 2-butanone oxime as a probable human carcinogen. Tests on animals exposed to high doses show increased cancer risks. In real world terms, people using oil-based paints or working with products that contain this chemical take in low amounts, mostly through breathing fumes or from skin. Most serious studies center on people in manufacturing jobs with much more exposure than the public. Still, headaches, dizziness, and respiratory irritation can all pop up with even moderate contact.
The Occupational Angle
As a writer who’s spent afternoons scraping painted porch railings, the irritation around strong paint smells feels personal. The nose tingles, eyes water, and sometimes breathing feels rough after an hour in a badly ventilated spot. Direct exposure increases many symptoms—dryness in the throat, sneezing fits, and sometimes even a slight burning sensation. Ventilation lowers those risks fast, but not every DIYer pops open every window.
Institutional exposure runs deeper. Plant workers dealing with big batches of paint or adhesives get more concentrated forms. The National Institute for Occupational Safety and Health (NIOSH) sets strict exposure limits, and personal protective equipment—respirators, gloves—comes as standard on those jobs, for good reason. Chronic exposure in such settings links to possible organ damage, especially liver or kidney, as animal tests make clear. Absorbing this chemical over months or years sets up much bigger health trouble than the occasional home repair project.
Reading Labels, Making Choices
The biggest challenge for most people comes with paint projects or renovations. Many don’t inspect labels or understand technical jargon. If a can says ‘contains 2-butanone oxime,’ it’s smart to treat it with more caution than water or soap. Good air flow, gloves, and careful cleanup help a lot, but the best option is swapping for water-based paints when possible. The industry has pushed for lower-emission coatings, partially because of the troubling data around solvents like this.
Big box stores in North America now stack more “low-VOC” or “zero-VOC” paint, which cuts down dramatically on chemical off-gassing. They’re not absolutely risk-free but less harsh by any measure. Using safer alternatives lines up with what doctors and scientists recommend, especially in homes where kids, seniors, or anyone with lung conditions live.
Solutions for a Safer Space
Regulators recommend limits, but change always moves slow. For now, personal accountability steps in. Choose ventilated spaces, wear gloves, and never ignore odd symptoms during or after a paint job. A quick look at a label and a willingness to swap out old habits helps everyone breathe easier. Paints and coatings improve each decade, and safer options stand waiting on shelves. Simple choices lead to safer homes and healthier hobbyists, all without sacrificing those fresh coats of color.
What are the safety precautions when handling 2-Butanone Oxime?
The Real Risks Behind 2-Butanone Oxime
Handling chemicals at work always calls for extra care, but some products demand even more respect. 2-Butanone oxime, often found in paint and coatings, carries a sharper edge. Folks who deal with it can run into eye and skin trouble. Breathing its vapors sometimes leaves you dizzy or wheezy, and there’s research warning about links to cancer with long-term high exposure. In industrial settings, you can’t count on luck. Safety becomes personal when a single whiff or spill can mess up your day — or your health down the line.
Glove Up and Cover Up
Touching 2-Butanone oxime without protection doesn’t end well. Latex won’t cut it; chemicals slip through. I always reach for nitrile or neoprene gloves because they last longer and hold up better. Long sleeves, pants, and tight cuffs give splashes nowhere to go. After years in manufacturing, I double check seams and make sure nothing’s torn before starting. Face shields or goggles must fit tight—regular glasses don’t block sneaky vapors or spray. I’ve watched coworkers take shortcuts, only to rush to the sink minutes later. No spill is too small to treat like a big deal.
Keep Your Air Clean
Vapors from 2-Butanone oxime gather fast, especially indoors or in tight corners. I’ve seen workshops turn hazy after a few minutes without fans running. Good ventilation is non-negotiable. At my last job, we cracked windows and used exhaust fans near every workstation. If the smell lingers or people feel headaches coming on, it’s time to stop and clear the air. In cramped spaces or near big batches, air-purifying respirators with organic vapor cartridges outdo paper masks every time. You want a filter built for the chemical, not a random dust mask from the hardware store.
No Food, No Drinks, No Excuses
I remember one warehouse where a guy ate his sandwich near the mixing table, saying he hadn’t gotten sick yet. That promise doesn’t last. Oils and alcohols from 2-Butanone oxime find their way onto bread, hands, and water bottles, turning every snack into a hazard. Eating and drinking stays out of any work zone with this stuff around. Once work wraps up, that’s the time for handwashing—soap and water, with more than a quick rinse. Contamination can ride home on clothes and skin even if nothing looks dirty.
Storage and Emergency Smarts
Nothing saves the day like being prepared. Chemical storage often gets ignored, but one tipped-over jug can force a whole floor to evacuate. I never store 2-Butanone oxime near acids, oxidizers, or heat sources, since mixing those ups the danger. Metal containers corrode and leak over time, so the label always guides container choice. Signage matters for everyone’s sake: the more obvious the warnings, the better chance people notice and steer clear if something spills.
Accidental splashes or inhalation sometimes happen, even when everyone’s careful. Eyewash stations and emergency showers belong within reach—those few seconds between splash and rinse make an enormous difference. I’ve helped coworkers flush eyes and skin right away, and it always lessens the aftermath.
Ongoing Care and Training
Training brings all these steps together. Everyone working with 2-Butanone oxime should know what to do when things go off-script. Safety data sheets need to stay updated and easy to find. Emergency contact numbers belong posted near every station. I’ve seen that when teams run through drills and ask questions, people respond faster and cut down on mistakes.
With the right habits and tools, people can handle tough chemicals and head home at the end of the day in one piece. It’s that simple and that important.
How should 2-Butanone Oxime be stored?
Understanding What’s in the Drum
2-Butanone oxime pops up in paints, primers, and chemical labs—not because it’s exotic, but because it prevents paint from skinning over. Those who handle it learn quickly that despite its practical use, this stuff needs respect. Experience in chemical storage tells me: treat every container as if a mistake could happen any time, because it does. Waiting for trouble won’t solve anything, but a solid plan will.
Keep the Heat Away
Temperature jumps mess with 2-butanone oxime more than many realize. Plenty of people stash chemicals in whatever distant corner of a shed or storage room happens to be free. Moisture, humidity, and direct sunlight all speed up spoilage and raise the chance of a hazardous reaction. I once watched a temperature spike in a poorly ventilated storeroom cause lids to warp. Fumes were everywhere, and the clean-up took hours. That could have been much worse if the material caught fire—2-butanone oxime burns quickly, with toxic smoke.
A well-ventilated, cool, shaded spot avoids danger. Anyone who’s worked in a paint shop knows that a small exhaust fan and a reliable thermometer make a difference. Fire inspectors and the Global Harmonized System agree: keep these chemicals below 30°C, don’t let them freeze, and never let containers heat up above room temperature.
No Room for Rust or Spills
It’s tempting to use any available drum for short-term storage. Steel rusts, though, and corrosion finds holes in the best plans. Seals and gaskets wear out. Plastic jugs sometimes crack. I’ve met folks who lost a week’s profits because a slow leak went unnoticed. Choosing containers rated for organic compounds and double-checking those lids are tight keeps accidents from becoming emergencies. Secondary containment—pans or trays to catch leaks or spills—means if something does go wrong, everything doesn’t run into the drains or onto someone’s shoes.
Keep It Apart from Food and Acids
2-Butanone oxime shouldn’t hang out with acids, oxidizers, or whatever’s in the lunchroom fridge. One jobsite suffered a whole shift with burning eyes because someone stored the chemical behind the break-room table as a shortcut. Cross-contamination ruins food, poisons water, and invites regulatory trouble. Segregating chemicals isn’t just a legal check-box: one misstep ruins both supplies and trust.
Label Everything—Nobody Remembers It All
Handwritten masking tape labels invite mistakes. I’ve run into five-gallon pails marked “solvent” that carried wildly different contents. Clear, chemical-resistant labels, up-to-date safety data sheets, and color-coded storage go further than rules written in binders. A quick glance should answer: What is this? How old is it? Is it ready to throw away or safe to move?
Waste and Emergency Planning
Getting rid of unused 2-butanone oxime can’t be an afterthought. Local hazardous waste pickup is better than dumping, and cleanup kits—a spill mat, nitrile gloves, goggles—cost less than even a minor emergency visit. Staff education beats panic. Teams who don’t practice their response wind up fumbling or calling 911 once things go sideways. Preparedness secures health and business alike.
Staying Diligent Keeps Everyone Safe
Real-world safety around 2-butanone oxime rests on daily habits. No shortcut pays off. Staging each step—containing, separating, labeling, planning for mistakes—builds a workplace where nobody gets sick, and nothing explodes. Every drum matters because one bad decision multiplies risk for everybody in reach.
Is 2-Butanone Oxime regulated or restricted in any countries?
What 2-Butanone Oxime Means for Consumers and Workers
2-Butanone oxime isn't a common name for most folks, but a lot of people use products with it every day. You find this substance helping to keep paints in liquid form. That smooth coat you see on your neighbor’s freshly-painted fence or your living room wall—there’s a pretty good chance 2-butanone oxime plays a part. This convenience for homeowners and painters comes with a catch: studies link this chemical to possible health problems. Extended exposure can irritate eyes and skin, and some research flags it as a potential carcinogen. That’s not something you want floating around when there are safer options out there.
Global Approaches to Regulation
Strict rules about 2-butanone oxime aren’t universal, but certain countries treat it with a lot more caution than others. Canada has taken a lead role. Seeing health concerns, its government shaped a law called the Volatile Organic Compound Concentration Limits for Certain Products Regulations. Here, 2-butanone oxime falls under a group of chemicals flagged as toxic. Products such as paints and coatings must meet lower threshold limits. Companies making or selling paint in Canada need to either reformulate or provide safety labels with serious clarity. Retailers and paint brands have put notice on cans, warning users in plain language.
The European Union added 2-butanone oxime to its Candidate List of substances of very high concern under REACH (Registration, Evaluation, Authorisation and Restriction of Chemicals) back in 2018. This step doesn’t flat-out ban the chemical across Europe, but it lets everyone—from manufacturers to importers—know that close monitoring is underway. Its presence triggers strict reporting rules, and proposals to restrict or phase out the chemical are on the table. The EU flagged its potential for cancer and negative impacts on wildlife.
When looking at the United States, things get complicated. The Environmental Protection Agency (EPA) tracks 2-butanone oxime through programs like the Toxic Substances Control Act. So far, it isn’t banned or tightly limited nationwide, but certain uses fall under worker safety controls. The Occupational Safety and Health Administration (OSHA) urges employers to give crews decent ventilation and respirators if they’re working in places with heavy fumes.
Why Rules Lag Behind Science
Delays in setting tough standards for 2-butanone oxime often come down to economics. The paint industry is big, and changing chemical recipes costs money and effort. Every time new testing or reformulation enters the scene, companies debate the balance between public health and the bottom line. Officials want strong science to back up laws. This pulls out the time it takes for new rules to hit the books, despite growing research about risks. The result? Big differences in how local or national laws treat the same paint product from one place to another.
Better Choices Moving Forward
Most folks don’t want to wade through pages of chemical labels or research papers. They want paints that don’t make workers sick or pollute the air at home. Change can start with clearer warnings and labels. Leading paint brands have already found newer additives that cut out the need for 2-butanone oxime without hurting performance. Simple steps, like offering more low-emission paints or promoting safer substitutes, could pull the industry in a healthier direction. Governments have a duty to monitor science and respond when evidence mounts, not just when it gets loud in the media. Regular reviews of chemical safety standards will keep things moving closer to true public protection.
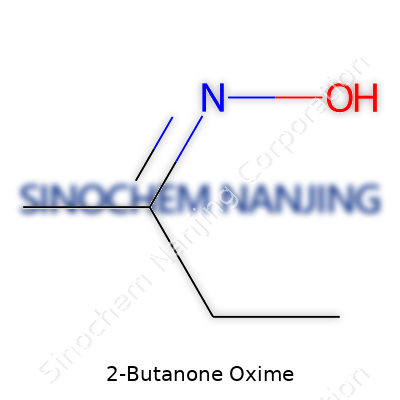
| Names | |
| Preferred IUPAC name | (NZ)-butan-2-one oxime |
| Pronunciation | /ˈbjuː.təˌnoʊn ˈɒk.saɪm/ |
| Identifiers | |
| CAS Number | 96-29-7 |
| Beilstein Reference | Beilstein Reference: 0633176 |
| ChEBI | CHEBI:39248 |
| ChEMBL | CHEMBL33038 |
| ChemSpider | 8666 |
| DrugBank | DB14019 |
| ECHA InfoCard | 31ee5b6c-a79f-4acd-8e18-c2e7b8b22cdf |
| EC Number | 202-496-6 |
| Gmelin Reference | 82880 |
| KEGG | C01915 |
| MeSH | D015217 |
| PubChem CID | 13547 |
| RTECS number | EL1550000 |
| UNII | E73049JGL5 |
| UN number | UN3334 |
| CompTox Dashboard (EPA) | DTXSID4020716 |
| Properties | |
| Chemical formula | C4H9NO |
| Molar mass | 87.12 g/mol |
| Appearance | Colorless transparent liquid |
| Odor | Characteristically sweet |
| Density | 0.962 g/mL at 25 °C |
| Solubility in water | Soluble |
| log P | 0.28 |
| Vapor pressure | 5.3 mmHg (25 °C) |
| Acidity (pKa) | 11.1 |
| Basicity (pKb) | pKb = 10.71 |
| Magnetic susceptibility (χ) | -6.38×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.462 |
| Viscosity | 0.88 mPa·s (20 °C) |
| Dipole moment | 2.90 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 203.3 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -51.5 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2144 kJ/mol |
| Pharmacology | |
| ATC code | V03AB38 |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS06, GHS08 |
| Pictograms | GHS07,GHS08 |
| Signal word | Warning |
| Hazard statements | H351, H317, H319, H412 |
| Precautionary statements | P201, P202, P261, P264, P270, P271, P272, P280, P302+P352, P304+P340, P305+P351+P338, P308+P313, P312, P314, P321, P332+P313, P337+P313, P362+P364, P405, P501 |
| NFPA 704 (fire diamond) | 2-3-1-" |
| Flash point | 59 °C |
| Autoignition temperature | 315°C |
| Explosive limits | Explosive limits: 2.3–12% |
| Lethal dose or concentration | LD50 Oral Rat: 930 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat 930 mg/kg |
| NIOSH | K125 |
| REL (Recommended) | 1 ppm |
| IDLH (Immediate danger) | IDLH: 300 ppm |

