2-Butanethiol: A Sharp Note in Chemistry and Industry
Historical Development
Few chemicals strike a memory like 2-Butanethiol. People working in labs or industries dealing with organic synthesis often remember their first encounter, mostly because of its powerful, skunky odor. Looking into the pages of chemical history, 2-Butanethiol turned up in the late 1800s during early work on thiols and their odd relationship with sulfur and carbon. Back then, researchers worked with little protection, driven by raw curiosity and a sense of duty to unravel organic chemistry’s smelly secrets. Over decades, interest in thiols took hold, from basic research in how sulfur participates in biochemistry to industrial uses in rubber and oil refining. Each stride built on the gritty work of early scientists who braved the stink for knowledge.
Product Overview
For folks outside chemistry, 2-Butanethiol might sound alien. For those in the game, it’s a familiar compound, notable for its presence in fuels, flavorings, and industrial chemistry labs. Its chemical formula, C4H10S, delivers a simple structure: a four-carbon backbone lined with a single sulfur atom. In the world of organosulfur compounds, this simplicity gives rise to tasks from acting as a flavoring, contributing to skunky beer, and warning of gas leaks. From every angle, 2-Butanethiol shows up not as a mystery but as an everyday tool with very real applications.
Physical & Chemical Properties
This compound emerges as a colorless to pale yellow liquid, holding a density lighter than water and a boiling point within the lower mid-range for organic liquids. With a melting point well below freezing, it pours even in a chilly lab. The odor deserves repeating: A powerful, aggressive scent that sticks in the air and on skin, unmistakable and nearly impossible to mask. Solubility leans to organic solvents — alcohol, ether, and benzene absorb it easily, while it hardly mixes with water. The reactivity isn’t just about the smell. Its sulfur-hydrogen bond likes to break in the presence of strong oxidizers, and the molecule attaches itself to metals, which underpins its use in chemical synthesis and analytical science.
Technical Specifications & Labeling
Labeling for 2-Butanethiol focuses on safety, given its hazardous volatility and low odor threshold. In regulated environments, bottles bear hazard pictograms flagging its flammability and toxicity. Chemists respect strict storage: cool, dry places, away from acids and oxidizers. Labels in the lab spell out clear instructions on handling, with suppliers sticking to purity levels most suitable for research or industrial requirements. Regulatory standards, like GHS, tie labeling to clear, immediate hazards, helping everyone in the lab spot risk at a glance.
Preparation Method
Old chemistry routes for making 2-Butanethiol trace back to alkyl halide reactions with sodium hydrosulfide, a tried-and-true method for synthesizing thiols in bulk. In a modern industrial context, the process stays simple but robust, demanding care in excluding oxygen and water to keep product purity high. I’ve seen variants that start with butylene and hydrogen sulfide over an acid catalyst, a method pulled from sulfur chemistry playbooks used in fuel and rubber industries. Each method needs precision — unchecked variations can pump out unwanted byproducts, cranking up the foul smell and cutting into usable yield.
Chemical Reactions & Modifications
In organic synthesis, 2-Butanethiol acts as both a building block and a modifying agent. Reactions with oxidizers transform it into sulfoxides or sulfones, both valuable in specialty chemical work. Many lab technicians, myself included, have used it to create thioethers by alkylation, lending new groups to molecules for custom properties. 2-Butanethiol’s sulfur plays a role in analytical chemistry too, complexing with metals for detection or purification methods. Its reactive group enables all sorts of swaps — good for creating tailored molecules, bad when you spill it and get stuck with the lingering stench for days.
Synonyms & Product Names
2-Butanethiol goes by other names, often listed as sec-butyl mercaptan in older literature. You’ll also spot it as sec-butylthiol or secondary butyl mercaptan, especially in databases and chemical catalogs. Each alias keys into the same pungent, four-carbon thiol recognized by chemists and safety officers worldwide. Chemical abstracts and regulatory listings stick to CAS numbers for clarity, but the varied synonyms show up often in both industry and academia.
Safety & Operational Standards
Safety drives everything with this compound. In all my experience, handling 2-Butanethiol without gloves or decent ventilation is a no-go. Exposure at low concentrations leads to headaches, nausea, and respiratory irritation. For storage, steel or glass containers with secure seals stop the odor from escaping and avoid unwanted reactions. Standard operating protocols call for fume hoods, eye protection, and double-checks with spill kits nearby. Regular training and drills are essential, with safety data sheets posted prominently. Fire risks are also real given the volatility; avoiding static discharge and open flames is not negotiable. Because the smell travels far, even minor spills prompt evacuation until full cleanup is verified by trained personnel.
Application Area
2-Butanethiol works across fields, from the chemical industry to environmental science. Oil refineries rely on it to add warning scents to otherwise odorless gases. That same nose-wrinkling quality gets harnessed in natural gas distribution, giving a clear marker for leaks. Food scientists use it sparingly to mimic flavors in processed foods, aiming for authenticity when creating savory or sulfurous notes. In analytical labs, chemists use it for chemical tagging and in calibration standards, trusting its strong signal for accurate detection. From automotive antifreeze to pest deterrents, applications keep popping up wherever sulfur chemistry makes a difference.
Research & Development
Development keeps pushing 2-Butanethiol beyond familiar territory. In academic settings, new applications appear as analytical standards or bizarre flavor mimics in model systems, helping clarify how sulfur affects taste or volatile emissions. Researchers focus on reducing odor during application, seeking treatments or derivatives that deliver the same technical function with less smell. Some industrial chemists investigate routes for greener production or improved selectivity during synthesis, responding to environmental and regulatory pressures. Others explore ways to recycle or detoxify thiol waste streams after use, a response to stricter pollution laws and community standards.
Toxicity Research
Scientists have documented both short-term and chronic health effects of 2-Butanethiol. Inhalation studies on animals reveal central nervous system depression and liver effects at sustained exposures. Lower doses cause eye and airway irritation — a fact many old-school lab workers learned firsthand. Regulatory agencies cite occupational exposure limits based on these findings. In environmental studies, researchers found the compound breaks down slowly, raising concerns for runoff near chemical facilities. Toxicologists keep watch on metabolites, probing whether long-term, low-level exposure links to cancer or reproductive problems. Real-world data stays thin for now, but caution dominates any risk assessment. In my time working with toxicologists, concerns focus less on theoretical danger and more on real incidents: workplace spills, improper storage, or accidental ingestion.
Future Prospects
The future for 2-Butanethiol splits on two tracks. One path sees regulation nudging the industry to minimize odor releases, cut waste, and develop less hazardous alternatives. Breakthroughs may come from greener production routes or modified derivatives carrying the functional benefits without as much risk. The other path sees new uses popping up, driven by advances in chemical sensing, flavor chemistry, and materials science. Researchers target better containment systems, smarter delivery technologies, or detection methods that spot leaks at parts-per-trillion levels. As society leans harder on safety while keeping industrial output strong, 2-Butanethiol will keep its place as a chemical everyone wants contained but nobody wants to do without.
What is 2-Butanethiol used for?
More Than Just a Foul Smell
2-Butanethiol rarely makes the headlines, yet people cross paths with its influence more often than they think. The pungent, nauseating aroma clings to it like a warning sign. Even a tiny whiff lingers and dominates the senses. In my time working near industrial labs, the sharp stink drifting from a misplaced sample once cleared out a whole corridor—nobody forgets that experience fast. This little molecule, though, serves real purpose beyond its shocking scent.
No Substitute in Odorants and Leak Detection
2-Butanethiol shines in one field above all: gas warning systems. Utility providers mix it into odorless natural gas supplies. That rotten, sulfurous edge helps people spot gas leaks before any real danger, since natural gas on its own carries no warning signal. Incidents in homes and businesses drop sharply with this approach. Across North America, regulations require strong-smelling warning agents for good reason, and 2-butanethiol answers the call.
Role in Industry and Chemical Manufacturing
Chemists value 2-butanethiol for its role as a starting material. It reacts easily with various chemicals, giving it a place in the creation of pesticides and specialty solvents. Oil refineries depend on it for sulfur-related processes and to study chemical breakdowns. In analytical labs, its presence helps calibrate instruments designed to track sulfur compounds, which often relate to pollution concerns or product purity. When I worked with research teams monitoring water sources, sulfur signals from similar thiols often helped locate points of industrial discharge early on.
Applications in Scent and Taste Training
2-Butanethiol pops up in places you might not expect, such as wine and beer quality control. Some training kits for professional tasters include samples infused with this molecule. Trainees learn to identify the off-note of 2-butanethiol among dozens of other aromas, helping spot problems in batches before they reach customers. Winemakers and brewers keep an eye on these “fault compounds” using chemical traces and human noses alike. I joined a few tastings as an observer; it always amazed me how quickly pros zeroed in on this molecule, a skill built with repeated, guided exposure.
Hazards and Safe Handling
Working with 2-butanethiol takes real attention to safety. The smell sticks to skin, clothing, and labware, and the chemical can irritate eyes and airways. Accidental releases never go unnoticed, but cleanup means more than just airing out the room. Proper ventilation, gloves, and storage away from oxidizers keep mishaps rare. Regulatory bodies lay out strict rules for transporting and storing this chemical. I’ve seen what happens when those steps get skipped—nobody enjoys spending hours neutralizing lingering odors in shared workspaces.
Looking to the Future
With so many uses based on its strong smell, finding alternatives isn’t simple. Synthetic chemicals must match the safety margin and reliability that 2-butanethiol delivers. Efforts to design warning signals without the downsides continue. Safer storage containers, improved leak sensors, and community education all help manage the risks. Public awareness about why natural gas smells so bad comes down to this little-known but life-saving chemical.
Sources and Trust in Scientific Discussion
Organizations like the Occupational Safety and Health Administration outline handling standards, and research published in analytical chemistry journals backs up these claims. As scientific understanding grows, so does our capacity to use and manage this molecule wisely. Keeping up with the latest guidance ensures that both workers and communities stay protected despite the challenges 2-butanethiol presents.
What are the safety precautions when handling 2-Butanethiol?
Why 2-Butanethiol Demands Attention
Anyone who’s worked in a chemistry lab remembers “skunk compounds.” 2-Butanethiol lands right on that list. It smells rough—a single spill in a room or on fabric sticks around for days. This chemical is more than a stinky inconvenience, though. It can irritate the skin, eyes, and lungs in a hurry. The vapors travel fast and don’t hide. I learned early on that even a cracked container sends people running for fresh air.
Personal Protection Isn’t Optional
A lot of folks think splashing on a tiny amount isn’t a big deal. This stuff gets under gloves, creeps through thin lab coats, and the fumes bother your nose and eyes in minutes. Tight-fitting goggles, thick nitrile gloves, and proper lab coats keep people safer. A double layer of gloves helps if you’re measuring or transferring the chemical for an extended period. You’ll regret skipping shoe covers if a few drops hit the floor, too.
Ventilation Shapes the Experience
I’ve seen people turn on a regular room fan, thinking that’s enough. It really isn’t. 2-Butanethiol’s vapors are heavy—they spread across a countertop and hang low. Fume hoods do the heavy lifting here. If you use this compound often, an efficient exhaust system pays off. The air smells better, and your coworkers keep their sense of humor.
Storage Speaks for Itself
Plastic caps and glass bottles age fast. 2-Butanethiol can seep around a worn cap and even react with common metals, so dedicated containers matter. Always keep the chemical below eye level on a labeled shelf, somewhere cool and dry. Squeeze bottles make accidents more likely—always use pipettes or syringes inside the hood. Anything that reduces splashing makes cleanup easier if something does go wrong.
Spill Response: Fast and Decisive
The first time I had a bottle tip over, I learned that paper towels just spread the stink. Spill kits with activated charcoal and sand lock up the mess better than anything else. Once you cap the spill, wheel the waste out to the designated area right away. Every minute counts if a strong smell starts filling the room. Always open up windows or use extra fans for a few hours afterward.
Fire Safety Takes Priority
This chemical doesn’t just irritate eyes—it’s flammable. One spark from static or a nearby Bunsen burner can start trouble. Have a fire extinguisher ready and clear flammable items before a single drop hits a beaker.
Training and Culture Help
No online quiz replaces hands-on safety drills. In my own work, quick walk-throughs before lab sessions made a huge difference. Everyone should ask questions and walk through an emergency plan. It’s easy to forget the simple steps—closing bottles, checking for leaks, making labels clear. Repetition helps those practices stick.
Community Knowledge Matters
Chemists, students, and technicians share tips and tricks online and in break rooms. I found out the hard way that lemon juice on gloves helps mask a lingering odor. Sharing what works—and learning from near misses—keeps everyone sharper for next time.
What is the odor of 2-Butanethiol like?
Getting to Know the Stench
Step into a room where someone has just opened a bottle of 2-butanethiol and your sense of smell might beg for relief. I still remember the first encounter in a college chemistry class—students joked about rotten eggs, burnt rubber, and even skunks crammed into a single room. There wasn’t a hint of exaggeration. 2-Butanethiol has a reputation in both industry and pop culture as one of the strongest, most unpleasant odors our noses can pick up.
What Does It Really Smell Like?
Calling it “bad” doesn’t cover the full story. Take the rotten eggs you might recall from a sulfur-rich hot spring, toss in a whiff of garlic that's been left too long on the kitchen counter, and top it with the sulfurous funk from a skunk’s defensive spray. That comes close to the stench this compound throws at you. Scientific literature supports these claims: research on olfactory responses notes that thiols like 2-butanethiol trigger strong, often reflexive disgust due to their resemblance to natural warning odors in decaying food or dangerous animal defense systems.
Why Should You Care?
A strong, foul odor like this isn’t just an inconvenience. The body responds for good reason. Smelly sulfur chemicals can signal spoiled substances or dangerous environments. 2-Butanethiol might not make headlines like some pollutants, but its presence impacts labs, factories, and even homes where gas leaks get artificially “odorized” for safety. Natural gas can’t be detected on its own, so industry experts add compounds like these to let people know when danger lurks.
Researchers don’t take the effects lightly. Studies suggest even tiny concentrations of 2-butanethiol—a few parts per billion—can offend the nose. The Occupational Safety and Health Administration (OSHA) and other health organizations highlight its psychological and physiological effects. Exposure can cause headaches, nausea, and in rare cases, can irritate the eyes and respiratory tract due to its potency.
Factoring in the Science
Scientists classify 2-butanethiol as a low molecular weight thiol. The molecule’s structure helps it stick to olfactory receptors like glue. This trait explains the persistence: airing out a room only goes so far once it’s cut loose. Compared to other bad-smelling chemicals—think hydrogen sulfide or ethyl mercaptan—2-butanethiol ranks among the top offenders for both threshold (how little you need to notice it) and staying power.
Solutions Make a Difference
Chemistry labs and industrial sites never drop their guard with 2-butanethiol around. Proper ventilation stands as the first line of defense, since opening a window or running a fume hood can dilute the stench quickly. Lab coats and gloves get more than symbolic use. They protect bodies and clothing from lingering odor. For the rare spill, activated charcoal, bleach, or hydrogen peroxide break down the culprit, restoring air quality faster.
Communities rely on education and preparedness when these materials come into play. Knowing what you’re smelling, why it matters, and how to react can help avoid panic and keep everyone safer. Smell recognition isn’t just a chemistry anecdote—it serves as an early-warning system that deserves respect and attention.
Tough Lesson from a Chemical Oddity
Anyone who’s worked with 2-butanethiol won’t soon forget it. The chemical doesn’t only test the nose—it reminds us of the power and importance of our senses, and of the safety protocols built around them. The strong, almost unbearable stench means every whiff carries a story about vigilance, human health, and the ways we train ourselves to interact safely with the materials that science and industry bring into our lives.
Is 2-Butanethiol toxic or hazardous?
The Smell That Follows You
You never forget your first encounter with 2-butanethiol. It's not famous because of complex chemistry or a long-winded industrial history; it's famous because the stench sticks around, burrowing into your clothes and memory. Most people recognize 2-butanethiol from a whiff of skunky beer or a regrettable encounter with a lab bottle. The compound doesn’t just knock you back with its odor—it actually carries real risks worth talking about.
Risks Lurk Beneath the Odor
Getting a noseful is more than a nasty surprise. According to the National Institute for Occupational Safety and Health, exposure to 2-butanethiol can irritate the eyes, throat, and lungs. One time, a tiny splash landed on the sleeve of a lab coat, speaking from experience, and the cough that followed wasn’t just from disgust. Those are acute effects; headaches and dizziness can sneak up later, especially in stuffy, unventilated rooms. There's something unsettling about a substance the body instantly rejects with tears and gagging, and it’s usually a good sign to take extra care.
Beyond the Immediate Reaction
2-Butanethiol is flammable, adding a whole layer of risk whenever it sits out in the open. Knock over a bottle in a crowded storeroom, and the vapors don’t just fill the air—they can ignite. For labs and plants that use it, fire risk needs a real plan, not some afterthought. MSDS sheets don't mince words: keep 2-butanethiol away from sparks.
Only a fraction of people exposed develop chemical burns, but that's little comfort when it happens. It's corrosive to skin and mucous membranes; gloves and goggles become tools for survival, not just basic compliance. Respiratory protection isn't a nicety, either. Those who skimp on gear invite more than watery eyes—they risk lung damage that doesn't fade after a cold shower.
Hidden Hazards Over Time
No long parade of headlines draws attention to long-term 2-butanethiol exposure, but animal studies suggest it brings more than short-term misery. Chronic inhalation leaves rats with damaged kidneys and livers, and regulatory agencies haven’t set any “safe” daily dose for people. Industry workers—janitors, chemists, custodians—deal with these risks regularly, which makes good training and engineering controls crucial.
Smarter Handling for a Safer Environment
Toxicity in a classroom or a worksite turns into a people problem, not just a paperwork issue. Fume hoods, gloves, ventilation—these measures make a world of difference. One day in a well-run lab, everything clicked: the right hood shut out the worst fumes, nobody skipped handwashing, and no headaches followed. That’s no coincidence, just the result of real-world protocols working as designed.
Education and regulation build a firewall between accidents and safe practice. Agencies set limits, but success hinges on daily culture: staff asking questions, reporting spills, and checking storage. Clean-up kits (with neutralizers) stop panic in its tracks, and regular drills remind everyone that safety is more than a box-ticking exercise. Digital inventory management helps too, so bottles don’t age and leak, forgotten on a back shelf.
Take Safety Personally
Walking past a spill kit in a corridor may seem ordinary, but it’s a sign of a space that takes chemical hazards seriously. 2-Butanethiol doesn’t care about ignorance; it only rewards caution and respect. Experience teaches that one careless moment can haunt for days—a lesson worth learning before the next whiff hits.
How should 2-Butanethiol be stored?
Understanding the Beast: What Makes 2-Butanethiol Tick
Few chemicals can clear a room quite like 2-Butanethiol. If you’ve come across it in the lab, you know why folks respect the stuff. That rotten cabbage stench? It sticks, drills into your memory—and your nose—long after you think it’s gone. This brings up bigger issues than a bad smell. 2-Butanethiol also happens to catch fire easily. Sparks, static, even a hot surface turn a messy day into an emergency. Anyone who works with it quickly learns that all storage rules exist for a reason.
Why Proper Storage Matters More Than You Think
One day in a shared university lab, I watched a visiting chemist carelessly return a half-full bottle of 2-Butanethiol to a desktop cart. A week later, an entire bench reeked of rotten eggs, and the blame landed fast—improper sealing, wrong location, and a bottle that shouldn’t have been left out in the first place. Freshmen and researchers alike lost work hours that week trying to air out the place. The lesson: not even a seasoned chemist is immune to lazy habits.
This isn't just about comfort. According to the National Institute for Occupational Safety and Health (NIOSH), 2-Butanethiol can irritate the eyes, skin, and lungs, and inhaling too much leads to headaches, dizziness, or worse. The Environmental Protection Agency (EPA) puts it high on the list of substances that demand respect from anyone handling it. Fires and toxic spills are realities, not abstract risks.
Locking Down Safe Storage
After years of wrangling nasty-smelling chemicals, I’ve come to rely on three key guidelines. First, glass bottles with ground-glass stoppers or Teflon-lined lids keep that stench locked down tighter than most plastics ever will. Plastic containers often let tiny amounts seep through, and 2-Butanethiol doesn’t need much to stink up a closet.
Stashing it in a tightly closed, clearly labeled bottle sits right next to placing it in a cool, dry, well-ventilated spot, far away from sunlight and ignition sources. Never warehouse this chemical near acids or oxidizers. Not only does that set the stage for spills or chemical reactions, but fire regulations are clear—mixing incompatible materials leads to trouble you don’t need.
Metal cabinets designed for flammable liquids come in handy here. Using flame-arresting storage, grounding every metal container, and keeping spill kits close by shows respect for this compound. Some facilities even pair storage with explosion-proof refrigerators, guarded entry, and fume extractor fans.
Why Staff Training Beats Any Lock or Label
Regular chemical safety briefings pay off. It’s easy to imagine everyone knows the drill, but fatigue or turnover chips away at safe routines. New staff stumble onto shortcuts, or old staff forget with time. Learning about hazards helps keep a healthy level of caution alive. The moment a bottle gets sticky or a label starts to fade, workers see the risk, not just another day at the lab.
Emergency procedures also need regular refreshers. Nobody wants to thumb through a binder mid-crisis. Fire extinguishers for chemical fires, eye wash stations, and quick escape routes should feel familiar. Building muscle memory saves time and, sometimes, lives.
Solutions Worth Keeping
A few fixes make life easier. Always store 2-Butanethiol in small amounts rather than bulky containers to minimize risk. Keep inventories updated and run scheduled checks—the chemical equivalent of an oil change, but more important. Strong ventilation systems earn their keep, especially with notorious chemicals like this.
For organizations stretched thin, teaming up with outside safety consultants for annual audits pulls blind spots into focus. Nothing like a fresh set of eyes to spot an overlooked hazard.
Bottom line: Respect for 2-Butanethiol starts with storage, but good habits, solid containers, and regular training keep both noses and workplaces safe.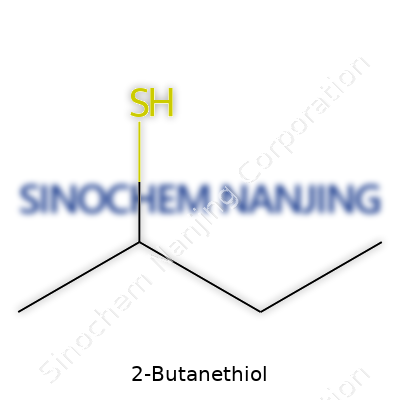
| Names | |
| Preferred IUPAC name | butane-2-thiol |
| Other names |
Ethyl methyl sulfide sec-Butyl mercaptan 2-Butylmercaptan 2-Butylthiol sec-Butanethiol |
| Pronunciation | /tuː bjuːˈteɪnˌθaɪɒl/ |
| Identifiers | |
| CAS Number | 107-03-9 |
| 3D model (JSmol) | ``` /* 2-Butanethiol JSmol model */ C[C@@H](CC)S ``` |
| Beilstein Reference | 1718734 |
| ChEBI | CHEBI:44101 |
| ChEMBL | CHEMBL15825 |
| ChemSpider | 7923 |
| DrugBank | DB14096 |
| ECHA InfoCard | 100.004.755 |
| EC Number | 211-198-2 |
| Gmelin Reference | 84790 |
| KEGG | C02724 |
| MeSH | D001685 |
| PubChem CID | 6364 |
| RTECS number | EH1575000 |
| UNII | TE6P08N98X |
| UN number | UN2347 |
| Properties | |
| Chemical formula | C4H10S |
| Molar mass | 90.19 g/mol |
| Appearance | Colorless liquid with a strong, unpleasant odor |
| Odor | Unpleasant, skunk-like |
| Density | 0.813 g/mL at 25 °C (lit.) |
| Solubility in water | insoluble |
| log P | 1.98 |
| Vapor pressure | 3.9 kPa (at 20 °C) |
| Acidity (pKa) | 10.7 |
| Basicity (pKb) | pKb = 3.77 |
| Magnetic susceptibility (χ) | -54.5·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.440 |
| Viscosity | 0.836 cP (20°C) |
| Dipole moment | 1.66 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 198.3 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -161.1 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2857.7 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS06, GHS07, GHS08 |
| Pictograms | GHS02,GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H225, H302, H315, H319, H332, H335, H400 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P264, P271, P273, P280, P303+P361+P353, P304+P340, P305+P351+P338, P312, P321, P331, P370+P378, P403+P235, P405, P501 |
| NFPA 704 (fire diamond) | 2-2-4-W |
| Flash point | -18 °C (closed cup) |
| Autoignition temperature | 285 °C |
| Explosive limits | 2.1–12.5% |
| Lethal dose or concentration | LD50 oral rat 160 mg/kg |
| LD50 (median dose) | LD50 (median dose): 340 mg/kg (oral, rat) |
| NIOSH | TIQ3500000 |
| PEL (Permissible) | PEL = 0.5 ppm (1.5 mg/m3) |
| REL (Recommended) | 0.5 ppm |
| IDLH (Immediate danger) | 100 ppm |

