2-Bromopropionic Acid: A Look at Its Path and Impact
Tracing Its Roots
Chemistry never moves in a straight line. 2-Bromopropionic acid came to the scene in the early 1900s as researchers dug into the reactivity of halogenated carboxylic acids. The bigger story has always been about finding ways to manipulate small molecules, giving chemists tools to chase down new reactions. Labs working out of Europe drove early publications, but within a few decades, its presence spread to manufacturing settings in North America and Asia, each finding unique value in bromo-substituted building blocks. Some of the earliest notable work involved using it to alter peptides and in agriculture as a precursor. Few compounds of this class have stayed as relevant both in specialty synthesis and ongoing research.
What Sets 2-Bromopropionic Acid Apart
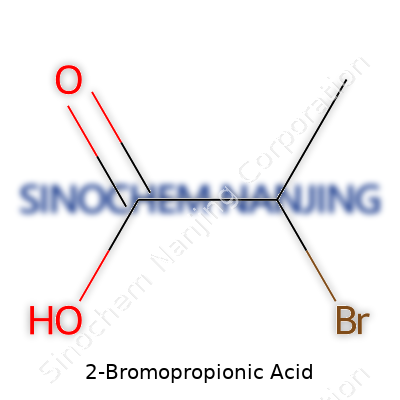
This molecule shows up as a colorless to pale-yellow liquid or crystalline solid with a sharp, penetrating scent. Chemists know it by the formula C3H5BrO2, and folks who’ve worked with it in a lab remember handling a dense, corrosive substance. The molecule features a bromine atom tethered to the second carbon of propionic acid, giving it properties that matter in practical synthesis — both the ability to act as an electrophile and as a source of the propionic acid segment. The branched nature means it often behaves differently from simpler bromoacetic acid.
Physical and Chemical Identity
2-Bromopropionic acid boils around 196 to 198°C, with a melting point near 46°C for the pure form. Its density hovers around 1.6 g/cm³, making for easy phase separation in mixed organic reactions. It mixes moderately with water and most polar organics but separates easily in nonpolar solvents. Beyond the basic numbers, the acid group proves reactive with bases, while the bromo substituent opens doors for nucleophilic substitution or metalation. Stability changes if exposed to high heat or strong bases, sometimes producing toxic fumes. On the shelf, containers need airtight seals, kept away from light and moisture.
Technical Standards and Clear Labeling
In the industry, every batch comes with tight spectroscopic checks, often with NMR confirming the placement of bromine. Key standards include purity above 98%, minimal moisture, and no detectable signs of decomposition by IR or GC-MS. Labels highlight the need for gloves, goggles, and ample ventilation; spill instructions usually emphasize neutralizers and containment, given its corrosive and environmental risks. Regulations in Europe and the United States encourage barcode tracking and documentation from source to shipment, which helps build trust in quality and safety.
How Chemists Make It
Labs often make 2-bromopropionic acid from lactic acid or alanine via hydrobromic acid bromination. Lactic acid reacts with phosphorus tribromide or HBr under controlled reflux, swapping a hydroxyl for a bromine atom with reasonable yields. Alternatives involve starting with propionic acid and using radical bromination, but this usually leads to undesired mixtures, so selectivity suffers. Purifying the product usually takes a simple distillation followed by crystallization from chilled solvents. Commercial producers operate at larger scales, managing temperature closely to avoid runaway reactions and byproducts.
Chemical Behavior and Ways to Tweak It
Reacting 2-bromopropionic acid with amines or alkoxides yields useful amides and esters — a favorite trick in drug development. Metal-catalyzed coupling, such as Suzuki or Negishi reactions, opens up carbon–carbon bond formations beyond the basics. In some studies, the bromo group gets swapped for azides or thiols, prepping the acid for joining peptides or specialty polymers. Its best-known move, nucleophilic substitution, gets harnessed for synthesizing a range of alpha-substituted acids, delivering both research and industrial value.
Names That Follow It Everywhere
The chemical goes by several aliases in research and commerce: alpha-bromopropionic acid, 2-Bromopropanoic acid, 2-propionic acid bromide. Chemists sometimes refer to it by its registry number, but most vendors keep things clear by pairing the name with molecular structure diagrams. In pharma, sometimes short-hand labels show up — “BPAcid” or “alpha-BPA” — although clarity is critical when buying for regulated labs.
Getting Safety Right
Direct contact risks both skin and eye burns, and the vapor stings if inhaled. In my own grad school days, we used this acid in a small-fume hood, with nitrile gloves and a dedicated chemical-resistant apron. Good air flow matters, because accidental spills evaporate quickly, making exposure more likely. Spilled liquid foams slightly with bases, so simple neutralization works. Fire training isn’t just a checkbox: 2-bromopropionic acid can break down into dangerous gases if heated with strong oxidizers. Regulations demand restricted access for storage, annual reviews of SOPs, and emergency decontamination showers in easy reach.
Industries and Real-World Use
Pharmaceutical companies rely on this acid as a stepping stone to synthesize active small molecules, especially those demanding precision at the alpha-carbon. Agrochemical producers work with it in herbicide development, building blocks, and even in flavor-modifying agents. Polymer chemists sometimes use it to introduce functionality to backbone chains, giving certain plastics and resins special properties. While its use in direct consumer products stays low for safety reasons, its footprints are found in the intermediates inside everything from medicines to adhesives.
Pushing the Boundaries: Research and Development
In the past decade, new work centers on stereoselective synthesis, allowing chemists to build one mirror-image version of the acid over the other. Catalysis with custom ligands or enzymes lets researchers hit high yield and purity, avoiding waste. In drug development, the acid acts as a key intermediate for chirally pure molecules — a big deal in medications where left- and right-handed versions behave differently. Environmental chemists track breakdown pathways, aiming for better waste treatment. The acid also serves in hydrogels for medical diagnostics, enhancing the function of bioactive surfaces.
Toxicity: Risks at the Forefront
Studies on rats and rabbits show the acid can cause burns on contact, and if swallowed, disrupts the gastrointestinal tract. Experimental inhalation sometimes leads to respiratory distress. Chronic exposure remains rare given its use in controlled settings, but animal studies guide safety policies for labs and plants. Ecotoxicity findings prompt companies to handle waste with formal treatment, keeping brominated organics out of streams. Good personal records show accidents drop in sites with safety culture and real training, not just rulebooks.
Peering Into What’s Next
As more industries dial in on green chemistry, 2-bromopropionic acid could see its production routes shift toward less hazardous reagents, cutting waste and dip into feedstock recycling. It could end up at the center of new biotech applications for building enantioselective drugs or smart-materials precursors. Researchers eye automation and continuous-flow synthesis, both for safer operation and as a way to drive down energy and solvent requirements. Upcoming years may bring tighter regulations, more open data sharing on toxicity, and smarter disposal solutions. Yet, the molecule’s versatility probably means advancements will keep it relevant across many technical fields.
What is 2-Bromopropionic Acid used for?
2-Bromopropionic Acid in Labs and Industry
2-Bromopropionic acid may not get much attention outside of chemistry circles, but this compound plays a big part in how research moves forward and how products take shape. With its simple structure—essentially a two-carbon chain with a bromine atom—this acid lands on many lab benches. Many organic chemists rely on it to start building more complex molecules. Anyone who has worked in a synthetic chemistry lab can tell you how a basic chemical like this opens up routes for a variety of syntheses.
I remember those endless days in the undergraduate lab, turning to substances like 2-bromopropionic acid to prepare chiral building blocks. This acid often acts like a seed in a larger reaction, helping form useful ingredients in pharmaceuticals and agrochemicals. People in drug development focus on such building blocks because chiral molecules can make or break a drug’s safety and effectiveness. Once, a research group I knew used this compound to produce beta-amino acids, which then became precursors to larger biologically active molecules. That sort of work sets new drugs in motion.
How 2-Bromopropionic Acid Makes Synthesis Work
The real draw comes from its reactivity. Chemists working to introduce a chiral center—basically, a part of a molecule that provides handedness, or asymmetry—often grab for 2-bromopropionic acid since its structure makes it useful for this purpose. Pharmaceutical manufacturers use it to create enantiomers, which are mirror-image molecules. In simple terms, imagine left and right hands: you want one for treating disease, but the other might be inactive or even harmful. Making the correct form with 2-bromopropionic acid's help is a major focus in safe drug creation.
People also use this acid outside the medical field. It shows up when companies make specialty chemicals. Take agricultural chemicals, for instance—some herbicides need chiral intermediates, and this compound is written directly into those reaction recipes. Polymer researchers sometimes use it to introduce specific functional groups into polymer chains, often improving properties like strength or flexibility. If you work in a material science lab, chances are you’ve run into this compound, maybe without giving it much thought. These interactions, small as they seem, ripple out to all sorts of consumer goods.
Risks, Safety, and Responsible Science
Some folks forget to think about downside. 2-Bromopropionic acid is hazardous, like many fine chemicals. Direct exposure can cause skin and eye irritation. It demands respect in the lab—gloves, goggles, and good ventilation. Chemical manufacturers and academic labs run risk assessments and train new researchers carefully. Regulatory bodies such as OSHA in the United States list it with guidance on handling and disposal. Any lab pro worth their salt remembers the acrid smell and knows to store it away from incompatible substances, especially bases or oxidizers.
Ethical responsibility extends past safety. Proper waste treatment means 2-bromopropionic acid doesn’t wind up contaminating water systems. Universities and large companies use advanced disposal processes and regularly audit their chemical management to keep both workers and the environment protected. More people are asking questions about sustainability, so research teams want to cut down on hazardous reagents. There’s always talk about using greener chemistry approaches, but so far, the unique properties of this acid keep it in the lineup for specific niches.
Alternatives and the Road Ahead
Some research teams keep their eyes peeled for safer or more environmentally sound substitutes, but the unique features of 2-bromopropionic acid—its ability to create chiral centers efficiently—keep it in play. That said, new synthetic techniques may one day shift demand. The steady progress toward greener manufacturing—using renewable feedstocks or safer solvents—gives hope for the future. For now, this basic compound stays locked into many industrial and laboratory processes, energizing the search for better drugs, materials, and technologies.
What is the chemical formula of 2-Bromopropionic Acid?
Chemistry Beyond Textbooks
2-Bromopropionic acid might sound like a mouthful, but its structure is simple if you’ve spent any time with basic organic chemistry. This compound carries the formula C3H5BrO2. Three carbons, five hydrogens, one bromine, and two oxygens shape its backbone. In the lab, it’s as common as a pipette for researchers working with pharmaceuticals or organic synthesis. That’s not just trivia—a lot depends on getting these formulas right, and plenty goes wrong when mistakes slip through.
Diving Into the Structure
Picture the parent molecule, propionic acid—C3H6O2—a three-carbon chain with a carboxylic acid group. Put a bromine atom in place of one hydrogen on the second carbon and you have 2-bromopropionic acid. This swap changes much more than you might think. The molecule doesn’t just look different; its reactivity shifts dramatically. Scientists call this sort of chemical fine-tuning “functionalization.” With just one switch, chemists open up new doors for creating materials or tailoring how a drug molecule behaves in the body.
Real-World Impact
That formula—C3H5BrO2—matters outside textbooks. It gets used in the synthesis of drugs and advanced materials. Anyone who’s worked in a chemistry lab knows the challenge of finding just the right building blocks for a reaction. 2-Bromopropionic acid is one such block. I remember a grad student who swapped out acetic acid for 2-bromopropionic acid and ended up with a totally different reaction outcome. One small change in the starting formula led to weeks of new experiments and eventually a more efficient synthesis. That moment reinforced how formulas aren’t empty numbers. They carry weight in dollars and time, not just molecular mass.
Safety and Responsibility in Handling Chemicals
It’s easy to gloss over the hazards involved with halogenated acids. 2-Bromopropionic acid brings a bromine atom along, and that brings risks. Brominated compounds can irritate skin, eyes, and lungs. Early in my own career, I saw someone get careless—just a splash, a split-second lapse, and they were off to the emergency room. No reaction is worth a health setback, no matter how trivial the volume or concentration looks. Understanding what’s inside a bottle means understanding its risks, not just reading the label or trusting your memory.
Quality Control and Environmental Care
Each chemical formula connects to environmental safety as much as to research accuracy. 2-Bromopropionic acid is no exception. If this compound escapes into wastewater streams, bromine atoms don’t just disappear. Waste treatment plants can face headaches because these molecules persist and sometimes bioaccumulate. Responsible disposal isn’t a box you check—it means learning how a substance like C3H5BrO2 can break down, and finding partners who take stewardship seriously. Labs that cut corners with their waste hurt everyone, from the next person at the bench to communities downstream.
Better Communication in Chemistry
Knowing a chemical’s formula helps ground our work in reality. There’s no shortcut through careful recordkeeping. Sharing results, updating protocols, and keeping a reliable inventory all depend on getting the basics right. From my experience, the difference between a good day and a bad day in the lab often comes down to those basics. That single, specific formula—C3H5BrO2—is more than numbers. It serves as the starting line for discovery and the foundation for responsible lab work.
What safety precautions should be taken when handling 2-Bromopropionic Acid?
Understanding the Risks
2-Bromopropionic acid sits among the more reactive and irritating chemicals you’re likely to see in a lab or an industrial setting. I still remember the time a colleague underestimated a chemical’s volatility; it taught our whole team to respect reactive compounds. Cutting corners never paid off. Inhaling fumes, getting it on your hands, or worse, on your face can lead to chemical burns, respiratory distress, and lasting skin problems. The acid stings sharply, and its vapors creep up fast if ventilation is poor.
Gear Up: Personal Protective Equipment
Working with this kind of corrosive requires proper gear every time. Nitrile gloves beat latex when handling 2-bromopropionic acid. Latex gives up too easily with halogenated compounds. Splash-resistant goggles block out vapors and liquid contact. In my own experience, basic lab glasses just don’t seal enough, and one poorly-timed splash could risk your eyesight.
Add a chemical-resistant lab coat or apron and closed shoes—no open toes allowed, no matter how quick you plan to be. The acid travels fast on surfaces and reacts with careless fingertips even faster. Handling in a lab means using a fume hood. I’ve seen people try to bypass this, but the first cough or eye-watering moment usually changes their tune.
Workspaces Kept Clean
Keeping your workspace uncluttered gives you more ways out if you spill. I make a habit of storing unrelated containers and personal items far from my station. Paper towels, glassware, and equipment should only be on the bench if needed, with everything else tucked safely away. Any accidental spill deserves immediate attention, and absorbent pads rated for acids manage these small disasters. A chemical-resistant tray beneath your reaction gives extra insurance.
Emergency Tools Ready to Go
Emergency showers and eye wash stations aren’t just for show. I check their location at the start of every day. You never want to scramble while acid burns your skin. Neutralizing solutions, such as sodium bicarbonate, come in handy, though water is your first response for most exposures. Phone numbers for emergency response and poison control need to stay visible. I keep mine taped up near the fume hood—time wasted finding help leads to permanent harm.
Safe Storage and Waste Disposal
Storage for 2-bromopropionic acid means a tightly sealed, chemically compatible bottle, usually glass with a securely fitting cap. The storage cabinet should be ventilated, acid-rated, and far from anything that reacts to strong acid, like bases or organic compounds. Accidental mixing with incompatible chemicals can cause fires or toxic releases.
Waste must follow hazardous chemical disposal rules. I log every container, label it, and never pour it down the drain. Improper disposal could land you in serious legal trouble—something I’ve seen ruin careers. Trained specialists pick up the waste for incineration or treatment.
Continuous Learning and Training
Working safely means staying current on procedures. I join refresher courses and review SDS details often; the chemistry world keeps evolving, and regulations change. Discussing near-misses with coworkers builds a culture where everyone watches out for hazards. Changing habits after a close call once saved me from repeating an ugly mistake.
No matter how routine the task, handling 2-bromopropionic acid with focus and care makes all the difference. Precautions build confidence, not just for yourself, but for the entire lab community counting on you to get it right.
How should 2-Bromopropionic Acid be stored?
Understanding the Risks: Handling 2-Bromopropionic Acid
2-Bromopropionic acid, a halogenated organic acid, appears as a crystalline solid or sometimes a pungent liquid. Its applications in synthesis are well-documented, yet with usefulness comes risk. Exposure can cause respiratory, eye, and skin irritation, even at low concentrations. Improper storage brings environmental hazards and safety threats at work. At the bench, I once saw a poorly sealed bottle corrode a metal shelf. That smell lingered for days—a sharp reminder that chemical stewardship is more than a box-ticking exercise.
Smart Choices: Storage Practices That Matter
Shelving a bottle just anywhere could result in disaster. I learned early on to keep 2-bromopropionic acid in a dedicated corrosives cabinet. No shortcuts—an acid-corrosive proof liner, preferably polyethylene, stops surprise leaks from ruining infrastructure. Acids like this break down thin plastics and metal, so putting the container in a secondary tray catches spills before they spread. Once, a cracked cap let the liquid drip out overnight; only because of the tray did we avoid an expensive clean-up.
Glass, HDPE, or PTFE containers outlast most plastics and show less tendency to degrade. The original manufacturer’s bottle usually wins for reliability. After seeing folk decant into a "clean" soda bottle, I never trusted any relabeled jug again. Vapor can escape around loose lids, so screwing caps tightly eliminates unnecessary exposure. It makes sense to keep fresh labels showing the risks and review them with new team members, especially those used to handling less volatile acids. No one gets used to the sickly odor, and complacency always leads to mistakes.
Keep Cool, Stay Dry: Controlling the Chemical’s Mood Swings
Temperature matters for more than comfort. Elevated heat speeds up decomposition and evaporation, pressurizing containers until they bulge and burst. A cool room—below 25°C—keeps things stable. Moisture draws in through leaky caps or packaging, sometimes triggering hydrolysis that generates dangerous byproducts. I never store this acid near the sink or on the bottom shelf, where drips are common. If you use a walk-in chemical storage room, make sure trays sit well above any floor drains. This avoids solvents and acids mixing if spills occur, limiting cross-reactions that can lead to fires or toxic releases.
Never put this acid alongside bases, amines, or oxidizers. All it takes is one broken jar or a lazy contractor moving things around to cause a chain reaction. A catalogue picture isn’t worth risking a whole building, and the chemical inventory should flag items stored near incompatibles for swift correction.
Training and Vigilance: The Human Piece
Training is the only real insurance. Nobody should open or store 2-bromopropionic acid without gloves, eye protection, and a fume hood nearby. This acid sneaks past the nose before it burns, so never rely on smell as your alarm system. Common sense and repeated drills make sure the safety routine turns into muscle memory rather than an afterthought. Regular inventory checks stop the old, forgotten bottles from turning into surprise hazards. Leaky containers and faded labels show up only if people look for them.
Practical Steps Build Trust
Storage rules support safety, productivity, and environmental care. Following real-world advice, not just regulatory text, turns risk management from paperwork into workplace culture. Years surrounded by acids, both benign and mean, have shown me that every careful step earns trust—from colleagues, regulators, and the wider world. Protecting people and property is never optional, especially when one careless moment can leave lasting harm.
What are the physical and chemical properties of 2-Bromopropionic Acid?
What You Notice First: Physical Traits
2-Bromopropionic acid doesn’t exactly stand out at a first glance. Scoop a bit and you’ll find a colorless to pale yellow liquid, sometimes appearing as a crystalline solid if kept cold. Its boiling point sits close to 189 °C, which means you won’t watch this stuff turn to steam on a stovetop. Pour some out, and the strong, pungent smell sticks around. The acid dissolves pretty well in water, too, so you don’t end up fighting stubborn residue in the lab sink. Its melting point lands somewhere around 45 °C, depending on purity, so it shifts from solid to liquid just above room temperature. Not all chemicals are so generous when it comes to mixing; drop 2-bromopropionic acid into ethanol or diethyl ether, and it’ll blend right in.
Chemical Behavior: Not Just Sitting Around
Chemists tend to remember this molecule for its mix of a carboxylic acid group and a bromine atom hanging off a propionic acid skeleton. That small tweak—swapping a hydrogen for bromine on the alpha carbon—makes a huge difference. During reactions, this bromine serves as a reliable leaving group, so the compound fits right into organic synthesis, especially if someone wants to build up other complex molecules. If you toss it into a reaction with a strong base, the acid readily sheds a proton to turn into its conjugate base for further chemistry mischief.
Not all substances with halogen content behave well, but this acid keeps a lower profile compared to many heavier halogen cousins. On my own benches, dealing with 2-bromopropionic acid never felt as risky as handling free-floating bromine. Nobody expects to see clouds form unless something’s gone very wrong.
Why Does This All Matter?
I’ve seen this acid act as a hidden workhorse in pharmaceutical labs. The bromo group gives researchers a chance to create chiral centers with more control, which often finds its way into the hunt for new drugs. Factories need molecules they can rely on for consistency, and 2-bromopropionic acid comes through with high shelf-stability when kept cool and sealed tight. Too many chemicals degrade too fast, but this one holds up unless exposed to strong light or heat.
Is It Safe? What’s the Risk?
Staying safe matters more than scoring a reaction yield. My glovebox came out every time I worked with any bromine compound, and not just from habit. Inhalation can cause irritation, and the eyes won’t thank you if a drop splashes in. The acid can corrode metal if spilled, so stainless steel sinks and quick cleanup are your friends. It doesn’t take huge volumes before environmental issues stack up either. Persistent brominated compounds deserve respect, so disposal follows strict guidelines, never down the drain. Data from toxicology studies point to moderate acute effects, but most labs work in well-ventilated areas with proper storage.
Practical Solutions for Cleaner Handling
Working with 2-bromopropionic acid means leaning on safety goggles, fume hoods, and double-checking every label. Labs should have spill kits on hand and hazardous waste bins clearly marked. Training goes a long way; younger chemists learn through hands-on practice and seeing how seasoned pros handle every step. Using smaller batches and vented storage lowers risk and cuts down surplus disposal.
Moving Forward with Confidence
Knowing the ins and outs of 2-bromopropionic acid brings safer, smarter research. Experience in the lab teaches the real weight behind these facts—not just reading a safety sheet at a desk. Staying informed and careful opens doors for discovery, without letting health or safety become afterthoughts.
| Names | |
| Preferred IUPAC name | 2-bromopropanoic acid |
| Other names |
2-Bromopropanoic acid alpha-Bromopropionic acid DL-2-Bromopropionic acid Propanoic acid, 2-bromo- 2-Bromopropionsäure |
| Pronunciation | /tuː-broʊˌmoʊ-proʊˈpiːɒnɪk ˈæsɪd/ |
| Identifiers | |
| CAS Number | 598-72-1 |
| Beilstein Reference | 1720721 |
| ChEBI | CHEBI:64070 |
| ChEMBL | CHEMBL16260 |
| ChemSpider | 82107 |
| DrugBank | DB02170 |
| ECHA InfoCard | 14f80975-3777-4eb5-85e9-b977e29c82c5 |
| EC Number | 209-795-0 |
| Gmelin Reference | 6077 |
| KEGG | C01737 |
| MeSH | D017792 |
| PubChem CID | 65886 |
| RTECS number | UF0850000 |
| UNII | 1W4K38F57C |
| UN number | UN2527 |
| Properties | |
| Chemical formula | C3H5BrO2 |
| Molar mass | 150.01 g/mol |
| Appearance | Colorless to yellow liquid |
| Odor | Unpleasant |
| Density | 1.584 g/mL at 25 °C |
| Solubility in water | Soluble in water |
| log P | -0.38 |
| Vapor pressure | 1 mmHg (25 °C) |
| Acidity (pKa) | 2.59 |
| Basicity (pKb) | 1.33 |
| Magnetic susceptibility (χ) | -45.8e-6 cm³/mol |
| Refractive index (nD) | 1.4390 |
| Viscosity | 1.095 cP (20°C) |
| Dipole moment | 2.48 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 190.3 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -516.5 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1260.7 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS07 |
| Signal word | Danger |
| Hazard statements | H302, H315, H319 |
| Precautionary statements | Precautionary statements: "P261, P264, P270, P271, P301+P312, P302+P352, P304+P340, P305+P351+P338, P312, P330, P332+P313, P337+P313, P362+P364, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 2-3-0-W |
| Flash point | 138°C |
| Autoignition temperature | Autoignition temperature: 650 °C (1202 °F; 923 K) |
| Lethal dose or concentration | LD50 (oral, rat): 2520 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat LD50: 310 mg/kg |
| NIOSH | Not listed |
| PEL (Permissible) | Not established |
| REL (Recommended) | 20 - 25°C |
| IDLH (Immediate danger) | IDLH: 200 mg/m³ |
| Related compounds | |
| Related compounds |
2-Chloropropionic acid Lactic acid 2-Hydroxypropionic acid 3-Bromopropionic acid Propionic acid |

