2-Bromoethyl Ethyl Ether: From Discovery to Future Applications
Historical Development
Chemists first stumbled across 2-Bromoethyl ethyl ether in the early days of organic synthesis, a period rich with experimentation and not always much oversight. Researchers in the late 19th and early 20th centuries explored simple ethers and halogenated compounds, often pursuing better solvents, anesthetics, or intermediate chemicals for dyes and agriculture. The development of 2-Bromoethyl ethyl ether speaks to a time when halogenation—introducing a halogen atom to organic molecules—allowed new pathways to more reactive, versatile building blocks. The molecule's birth came from curiosity coupled with the practical drive to create reactive intermediates for more complex reactions, driving pharmaceutical and material advancements not just in laboratories, but in the transformation of everyday life.
Product Overview
In labs and factories, 2-Bromoethyl ethyl ether is a handy reagent. Its structure, with an ethyl group linked to an oxygen atom and a bromoethyl group, makes it much more than a routine organic compound. This simple modification—a bromine replacing a hydrogen atom—radically increases its reactivity, opening doors to alkylation chemistry. It's clear, with a characteristic odor many chemists find instantly recognizable. This isn’t just a textbook chemical; it’s a tool that helps researchers and industrial chemists hook new pieces onto a molecular chain, broadening the spectrum of possible products in pharmaceuticals and agrochemicals.
Physical & Chemical Properties
Look at the liquid—it's colorless, turning a yellowish tint if neglected or allowed to sit in sunlight. It’s heavier than water, with a boiling point much higher than simple ethers. That bromine atom brings density without overwhelming volatility. Despite the extra weight, this molecule remains a vigorous participant in substitution reactions. Its vapor travels fast, and given half a chance, it’ll catch a spark and burn, so good ventilation is essential. Solubility leans toward organic solvents like ether or chloroform, while it avoids mixing into water. This stems straight from its structure—a polar, yet predominantly hydrophobic, profile. Stability is not to be taken for granted; even a trace of acid can trigger decomposition or release of corrosive fumes.
Technical Specifications & Labeling
Bottles arriving in labs carry clear warnings. Purity must reach at least 98%, as impurities can spoil sensitive syntheses or stir up side reactions. Typical labeling includes hazard symbols for flammability, toxicity, and environmental risk. Clear lot numbers and batch records keep tabs on the origin and date, since the shelf life shortens fast if the container isn’t sealed tightly or stored away from light. Chemists rely on gas chromatography or NMR to verify purity, but routine handlers go straight to the label, which lays out storage temperature, safety precautions, and emergency procedures in bold, unmistakable print. Strong odors serve as a deterrent to careless handling, but no label outsmarts human error if the safety training falls short.
Preparation Method
The preparation of 2-Bromoethyl ethyl ether traditionally follows a multi-step process. One route starts with ethyl vinyl ether, which reacts with hydrogen bromide in a controlled reaction yielding the target compound. Alternatively, chemists can employ Williamson ether synthesis, reacting sodium ethoxide with 2-bromoethanol. These methods use routine glassware but precise technique, as deviations in temperature or moisture management alter yields or generate dangerous byproducts. Throughout the process, handlers must respect the volatility of intermediates and the strong reactivity of alkylating agents, ensuring robust fume extraction and real-time monitoring for leaks or runaway reactions.
Chemical Reactions & Modifications
This molecule is a prime candidate for nucleophilic substitution, where a nucleophile attacks the carbon attached to bromine, kicking out the bromide ion. Labs use this characteristic to tack on a vast array of organic groups. These reactions underpin the construction of new pharmaceuticals by building up or modifying base molecules to fine-tune medicinal activity. Academics find it invaluable for generating ethers, amines, or other derivatives, depending on the conditions and the choice of reactant. The structure’s reactivity does have drawbacks. It can alkylate sensitive groups unintentionally, and residual unreacted ether can compromise product purity, so careful purification is key at every step.
Synonyms & Product Names
Catalogs list 2-Bromoethyl ethyl ether under many names, reflecting its different roles and supplier preferences. Some alternatives include Ethyl 2-bromoethyl ether, 1-Bromo-2-ethoxyethane, or simply Bromoethyl ethyl ether. These synonyms show up in regulatory filings, shipping documentation, or SDS sheets. The variety underscores the need for double-checking identifiers, especially as regulatory limits and reporting standards differ from region to region. By cross-referencing the CAS number, chemists nail down the exact substance, sidestepping costly or hazardous mix-ups during procurement or regulatory audits.
Safety & Operational Standards
Safe use starts with infrastructure and training, not just a list of rules. Labs require chemical fume hoods, spark-proof storage, grounded equipment, and PPE including gloves, goggles, and flame-resistant lab coats. Emergency showers and eyewash stations need to sit close by—no exceptions. Every handler reads SDS sheets outlining health risks, which include severe eye and skin irritation, respiratory hazards, and possible effects on the central nervous system after repeated exposure. Any spill, no matter how minor, demands immediate containment and cleanup by trained staff—no shortcuts, because fumes spread rapidly and the risk of ignition hovers over every open bottle. Spill kits designed for halogenated organics, not just generic sorbents, stand ready where this chemical lives and works.
Application Area
Industrial chemists and academic researchers don’t reach for this bottle without a plan. Its main arena is organic synthesis, where it acts as an alkylating agent. This skill never goes out of style—it's central for linking carbon chains and introducing new functional groups. Pharmaceutical companies put it to work creating intermediates for antihistamines, local anesthetics, or cardiovascular drugs. In materials science, some niche polymerization methods use it to build up specialty polymers, thanks to its high reactivity. Agricultural chemists use it to construct precursor molecules for pesticides or herbicides. I’ve seen medicinal chemists rely on its robust reactivity to push forward structure-activity relationship studies, rapidly exploring new variants in the search for better therapeutics. The breadth of applications owes a lot to its willingness to participate in classic organic reactions.
Research & Development
In R&D labs, chemists lean on 2-Bromoethyl ethyl ether to prototype new molecules. Its straightforward modification opens up a wealth of possibilities, and graduate students often cut their teeth optimizing reactions with it thanks to reliable transformation rates. As drug resistance and environmental pressures push innovation, this ether’s role as a foundational building block keeps expanding. Sometimes, a project stumbles not because of the chemistry but due to handling risks—so safety innovation advances along with synthetic creativity. Digital modeling, safer analogs, and continuous-flow systems have entered the picture, further refining its use and the predictability of downstream products. Researchers working across continents collaborate virtually, sharing new tips on reaction conditions or purification hacks that shave weeks off old timelines.
Toxicity Research
With halogenated ethers, toxicity studies must dig deep. Inhalation exposure drives most health warnings, because vapors can depress the nervous system, irritate mucous membranes, or cause headaches. Chronic low-level contact raises flags for liver or kidney effects, though data often lags behind the pace of use. Animal studies highlight dangers, but a lack of long-term human data keeps regulatory groups cautious. Some findings suggest breakdown products could be even more problematic, including bromoacetaldehyde and hydrobromic acid, both of which ramp up risks of tissue damage. Understanding metabolism and breakdown pathways stands central to accurate risk assessment, especially in the face of new environmental or occupational regulations. I’ve watched chemists impose self-discipline on procedures, often doubling up on ventilation even before documentation caught up, simply because a colleague learned the hard way.
Future Prospects
Looking forward, 2-Bromoethyl ethyl ether isn’t losing ground. Pharma and materials chemists constantly need quick, reliable ways to build molecular frameworks, and this ether answers that call. Advances in green chemistry promise safer synthesis methods and cleaner byproducts, pressing manufacturers to deliver more responsibly sourced material. Analytics are likely to tighten up, with international regulators pushing toward even lower exposure thresholds. Digital tools will probably bring predictive toxicology and smarter monitoring, slashing lab accidents and environmental release risks. At the same time, collaborative platforms and open science efforts are likely to spread both best practices and cutting-edge application notes beyond traditional silos, driving both safer handling and smarter uses across the industries that stake their future on molecular innovation.
What is 2-Bromoethyl Ethyl Ether used for?
Practical Uses in the Lab
Sitting in a chemistry storeroom, a bottle labeled “2-Bromoethyl Ethyl Ether” doesn’t draw much attention. Folks outside the field might never come across it, but chemists know this liquid as a key player in organic synthesis. The compound acts as an alkylating agent, helping build more complex molecules. In the toolkit of a research or industrial chemist, this means 2-Bromoethyl Ethyl Ether works as a link to add an ethyl group where one’s needed — and that simple feature paves the way for making medicines, agricultural chemicals, and specialty polymers.
How Chemists Rely on It
Looking at chemical syntheses that build pharmaceuticals or study new materials, having a reactive building block can make or break a project. 2-Bromoethyl Ethyl Ether proves handy for introducing ethoxyethyl groups onto other chemicals, which helps change properties like solubility or bioactivity. In my years of working in research labs, I’ve seen it used to attach protective “coats” to sensitive molecules, keeping them safe through harsh reactions and stripping them off later to get the desired structure.
Factory chemists turn to this compound during scale-up when they can’t find another reagent as cheap and straightforward. Its liquid nature, combined with decent stability, lets operators handle it with ordinary chemical glassware — no need for expensive containment or rarer equipment. Of course, anybody using it knows that safe handling counts. Like most alkylating agents, it can hurt skin or lungs, so gloves, hoods, and careful handling come standard.
Why It Deserves Attention
A lot of major medical advances start small, maybe with someone needing to tack a two-carbon chain onto a complicated molecule. That’s often been the first step in building a new medication or probing what goes wrong in a disease. For example, modifications made with 2-Bromoethyl Ethyl Ether have helped researchers make better drug candidates by improving the way they move through the body or avoid unwanted breakdown.
On the industrial side, specialty chemicals for plastics or coatings get their unique properties because of specific bonds made possible by reagents like 2-Bromoethyl Ethyl Ether. A single improvement in a synthesis can lower costs and make a process less wasteful, benefitting not just manufacturers but the environment. Science rewards tools that can be counted on to do their job again and again — and this compound, despite being less famous than other reagents, often forms the backbone of larger projects.
Challenges and Safer Practices
Each reagent in a laboratory comes with its own hazards. 2-Bromoethyl Ethyl Ether doesn’t buck that trend. Without proper ventilation and protection, it exposes workers to risks of chemical burns and serious health effects. Chemists who’ve worked with it all learn to respect its power to do harm as well as good. Some countries keep rules about its storage, handling, and disposal, and these are in place for good reason. By using less hazardous alternatives when possible, chemists can help keep laboratories safer and waste streams cleaner.
For anyone interested in greener chemistry, the ideal path forward builds on the knowledge we’ve gained with compounds like 2-Bromoethyl Ethyl Ether, using them wisely and striving for safer substitutes, whenever progress allows. New technologies, smarter design, and respect for proven tools will keep innovation moving in the right direction.
What are the safety precautions when handling 2-Bromoethyl Ethyl Ether?
Respect the Risk in the Bottle
2-Bromoethyl ethyl ether brings a dangerous mix of hazards. Not only can it irritate skin and eyes, but inhaling the vapors causes real harm. Anyone who thinks a bit of splashing won’t matter deals with a nasty surprise—the chemical soaks right in and burns. This isn’t just another bottle in the back shelf.
Stories float around research labs of people rushing and splashing dangerous liquids. I remember a classmate forgetting to check gloves for pinholes and winding up in the emergency room because a small drip rolled under the thumb. Even a single drop of 2-Bromoethyl ethyl ether does real damage if it touches your skin. I learned to treat every bottle with the same focus you'd give a lit candle—one careless reach, and you’ll pay for it.
Start With Fresh Air and Closed Doors
Good fume hoods make a difference. I spent a summer internship doing organics work in an old teaching lab, and the difference between open bench top and a working hood couldn’t be clearer. Fume hoods carry off the eye-watering vapors, and the glass sash gives your face some distance. Don’t dive into a project with 2-Bromoethyl ethyl ether in a spotty or broken hood—hold off, get it fixed, or find another lab. It only takes one slip for vapor to build up, and then the headaches and dizzy spells start.
Dress for the Job—Not for Comfort
Every lab tech laughs at the idea of shorts and sandals, but the urge to be comfortable tugs hard in summer. Still, this chemical soaks through jeans before you realize what happened. The right answer stays simple every time: closed shoes, long sleeves, gloves made for organohalides—no latex break-through allowed. Lab coats resist splashes, and sometimes a face shield turns a close call into a shrug rather than a scar. Take off rings, watches, and bracelets—they trap liquids and you don’t want to take chances.
Know How to React, Not Just Prevent
Chemical burns and eye injuries don’t wait for you to check a manual. Emergency showers and eyewash stations sit near every station for good reason. In a real event, seconds count. Practice the steps: If you splash 2-Bromoethyl ethyl ether on skin, drop everything, call for help, and flush with water. Peel clothes away, no matter the cost, since fabric traps chemical against the body. If it hits the eyes, keep them open under running water as long as possible and get straight to a doctor.
Disposal and Storage Save More Than Space
Old solvent bottles crowding the cabinet may not seem urgent, but aging 2-Bromoethyl ethyl ether can build up pressure and even explode. Store only what you need, use vented caps, and label everything with dates. Keep this chemical far from acids, bases, and anything prone to a spark. Incompatible mixing in waste bottles causes fires few people forget. Waste streams deserve strict attention—follow local hazardous waste disposal rules every time. Shortcutting on this front leads to accidents that affect folks down the line, not just the person pouring the waste.
Culture of Safety, Not Just Compliance
Safety rules work best when everyone takes them seriously, not just when an auditor walks through. Share stories, review close calls, and watch out for small mistakes before they become disasters. Training new team members helps the whole group avoid repeating painful lessons. Respect, not fear, guides the hand that handles 2-Bromoethyl ethyl ether—and keeps everyone going home healthy.
What is the chemical formula and structure of 2-Bromoethyl Ethyl Ether?
The Basics of 2-Bromoethyl Ethyl Ether
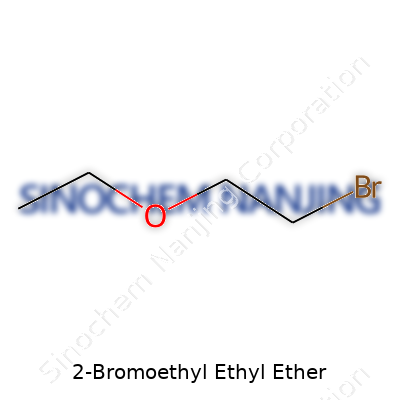
2-Bromoethyl ethyl ether draws attention mostly because of its practical uses as an intermediate in organic synthesis and occasional appearance in lab storerooms. The chemical formula for this ether reads as C4H9BrO. At a glance, it might look like just another string of letters and numbers, but there's quite a bit to unpack for anyone who's mixed chemicals or worked through a synthesis late at night.
Structure: More Than a String of Atoms
The real story of a compound lies in its structure. With 2-Bromoethyl ethyl ether, structure influences everything—solubility, reactivity, even storage precautions. The molecule arranges itself with a two-carbon chain carrying a bromine atom (the bromoethyl part) and an oxygen atom that bridges the bromoethyl to an ethyl group. If we draw it out, the structure appears as CH3CH2OCH2CH2Br.
That oxygen atom at the center acts as a connecting bridge, linking separate worlds—the reactive bromine tail and the stable ethyl. I remember struggling to visualize these sorts of structures in early chemistry lessons, but hands-on lab work drove home just how much difference a tiny shift makes.
Importance of Structure in Chemistry and Daily Work
In the lab, no one simply asks for 2-Bromoethyl ethyl ether and trusts a label. Chemists sketch out the molecule, predicting where reactions might happen. The bromine atom throws a wrench into the otherwise unremarkable ether backbone, making this compound a handy tool in forming carbon–carbon bonds. That reactivity means safety goggles and gloves are a must—bromoalkanes tend to be toxic and sometimes irritating to the skin.
Ethyl ethers like this one can act as building blocks in pharmaceuticals or in the synthesis of specialty chemicals. Its structure, combining a halide with an ether group, makes it more than a shelf-stock compound—its design lets it step into many roles, as long as the user understands what they're handling.
Handling Hazards and Practical Solutions
Anyone spending time in research or manufacturing knows the most beautiful-looking reaction on paper can crash—sometimes because a chemist skipped paying attention to the structure, other times due to overlooked safety risks. 2-Bromoethyl ethyl ether, being an alkylating agent, doesn't just sit around benignly; it can react with DNA or proteins, posing health hazards.
To cut down on risk, users rely on fume hoods, personal protective gear, and careful labeling. I’ve watched colleagues share short checklists next to chemical bottles, not just to meet procedures but to protect the team. Good habits, reinforced with clear structure labeling, can prevent mishaps.
Improving Understanding and Workplace Safety
University courses, research jobs, and industry training could do more to stress the relationship between formula, structure, and behavior. It always feels easier to trust a supplier’s label, but making time to sketch structures, talk through reaction mechanisms, and question unknowns makes a difference. Specialty compounds like 2-Bromoethyl ethyl ether demand respect and attention to detail, especially when novices and experts work side by side.
A solid grasp of structural differences may sound basic—yet, in practice, it can mark the line between a successful synthesis and a trip to the safety shower. Every working chemist stands to gain from thinking beyond formulas and focusing on the true structure, especially for reactive molecules.
How should 2-Bromoethyl Ethyl Ether be stored?
Safe Handling Starts With Respect for the Substance
2-Bromoethyl ethyl ether always reminded me of how chemistry blends caution with curiosity. This chemical doesn’t just demand care because it’s part of some standard procedure; it’s about real risks. Anyone who’s spent time around ethers can tell you, these compounds tend to let their volatility speak for themselves. Here, there’s a special story, since bromine compounds can add another layer to the danger. In my own lab days, I watched more than a few labels go unread or warnings ignored, and those moments turn learning curves into close calls.
Avoiding Fire Hazards and Toxic Fumes
Fire risk isn’t the kind of thing you want to gamble with—especially since ethers, including 2-bromoethyl ethyl ether, love to form peroxides. These can ignite without warning, often during a regular pour or even just sitting on the shelf. So, a good rule of thumb here is to keep storage locations cool and far from anything that sparks, like open flames or running electric motors. I learned the importance of vented flammables cabinets in a cramped academic lab, after years of hearing “just keep it on the bottom shelf.” Not good enough. Fire resistant storage isn’t optional.
Keep It Dry, Keep It Dark
Humidity and sunlight act as villains for this chemical. With time, exposure to light can make ethers degrade, sometimes releasing even nastier byproducts. Water gets into the mix you might face container corrosion or some unpleasant reactions. In practice, this looks like sealing containers completely after each use. Tinted or opaque bottles remove another variable, and for bigger stocks, a dedicated cold room goes a long way. The small extra step of logging opening dates pays off, too. Knowing how old a bottle is saves headaches deciding “Can I still use this?”
Labeling: No Place for Guesswork
I once worked next to a shelf covered in faded masking tape with marker scrawls. Guessing which bottle holds what becomes a dangerous habit. Clear labeling turns a regular safety measure into something personal; it may even save a life. Add key warnings: flammable, toxic, peroxide-former. If staff or students swap shifts, nobody wastes time second-guessing contents. That honesty with labeling builds trust long before the audit or inspection happens.
Containment for Accidents, Planning for People
No matter how careful we act, spills and bottle breaks remain possible. Secondary containment trays catch drips and messes. Nearby spill kits with compatible absorbents let you take fast action. Fume hoods become more than lab showpieces—they mean breathing less of anything that gets loose. Knowing who to call or what steps to follow, posted in plain language on the wall, beats flipping through a 300-page binder. Regular checks of those kits make sure they haven’t expired or walked off with someone’s personal project.
Good Storage Helps Health and Research
Controlling 2-bromoethyl ethyl ether doesn’t just keep people safe; it preserves the chemical itself. Degrading stock leads to ruined experiments, wasted money, and frustration. By treating the substance with the care outlined above, labs and workplaces keep research on track and everyone breathing a little easier. Little details—secure shelving, fresh air, correct containers—might not get the spotlight in glossy lab tours, but they make the biggest difference in the long run.
What are the potential health hazards of 2-Bromoethyl Ethyl Ether?
Looking at Exposure and Health
2-Bromoethyl ethyl ether rarely makes the evening news, but this chemical crops up in some research labs and industrial settings. I’ve stood in a few labs where half the bottles carried warnings about being flammable or toxic. Chemicals like this can slip under the radar until someone starts coughing or develops a strange rash, so it's worth talking straight about the risks.
How Contact Happens
Most folks won’t find this chemical at the local store, but those working in chemical engineering, pharmaceutical research, or specialty manufacturing might run into it. The stuff can go from a liquid in a bottle to vapors in the air without much warning. Typical trouble comes from skin contact, breathing in vapors, or even accidental spills in poorly ventilated rooms.
Health Hazards, Plain and Simple
2-Bromoethyl ethyl ether acts as an irritant. Get some on your hands and expect redness, blistering, or burning. Eyes sting and even a whiff can hit your lungs hard—coughing, sore throat, maybe worse for those with asthma or fragile lungs. Prolonged contact or high doses make things worse: headaches, dizziness, maybe nausea. That uneasy feeling in the gut after spilling some? That's not just nerves. It's your body reacting to poison trying to find a way in.
Studies show chemicals like this do more than irritate—they can harm organs if people have repeated exposure. Your liver, kidneys, and nervous system, those all bear the brunt if you handle these chemicals without gloves or proper ventilation, day after day. Worse, some chemicals in this family have shown up in research with links to cancer after long-term exposures. The full risk for this exact ether isn’t settled yet, but staying cautious makes sense, especially since toxic data lags behind for less common lab chemicals.
Real Stories from the Lab
I’ve seen what sloppy safety habits can do—one spill, and someone lands in the emergency room with chemical burns. I worked with a guy who used to brag about how he could “smell the ethers a mile away.” He coughed for days after one bad fume cloud. If you've spent any time in science labs, you learn quick: skimping on safety shortens careers and sometimes lives.
Solutions and Preventing Hazards
So how do people stay safer? Gloves rated for chemicals, eye protection, and good ventilation top the list. Not just any gloves, either; nitrile or neoprene, because common latex gloves break down quick with solvents. Fume hoods must get checked and maintained, since a dead fan just recycles poison back into the air. Training matters too. No one should walk into a lab without knowing what to do with spills or splashes. Eating or drinking where these chemicals are stored turns small mistakes into big emergency room visits.
Disposal matters as much as handling—pouring chemicals like this down the drain can send toxins into water supplies. There are strict rules for this, and they need to be enforced. Labs and companies must keep records and follow laws about storage, labeling, and disposal. That’s not bureaucracy—it’s about keeping harm out of your body and out of your home.
Final Word
Nobody grows up hoping to get chemical burns or organ damage from work. Care and respect for dangerous chemicals like 2-Bromoethyl ethyl ether keep people healthy. The best solution? Don’t work alone, never cut corners with protective gear, ask questions if you’re unsure, and remember that no job gets done faster by risking your health.
| Names | |
| Preferred IUPAC name | 1-Bromo-2-ethoxyethane |
| Other names |
Ethyl 2-bromoethyl ether 2-Bromoethoxyethane EINECS 203-721-1 Bromoethyl ethyl ether Ethyl bromoethyl ether |
| Pronunciation | /tuː ˈbroʊ.moʊˌɛθ.ɪl ˈiː.θɪl ˈiː.θɚ/ |
| Identifiers | |
| CAS Number | 111-96-6 |
| Beilstein Reference | 3580734 |
| ChEBI | CHEBI:39775 |
| ChEMBL | CHEMBL154600 |
| ChemSpider | 18416 |
| DrugBank | DB14174 |
| ECHA InfoCard | ECHA InfoCard: 100.006.469 |
| EC Number | 205-862-1 |
| Gmelin Reference | 6222 |
| KEGG | C19602 |
| MeSH | D017145 |
| PubChem CID | 79010 |
| RTECS number | KH8225000 |
| UNII | VI5W3H430Y |
| UN number | UN2546 |
| CompTox Dashboard (EPA) | DTXSID5020707 |
| Properties | |
| Chemical formula | C4H9BrO |
| Molar mass | 137.04 g/mol |
| Appearance | Colorless to light yellow liquid |
| Odor | sweet odor |
| Density | 1.38 g/mL at 25 °C (lit.) |
| Solubility in water | Slightly soluble |
| log P | 0.8 |
| Vapor pressure | 4.8 mmHg (20 °C) |
| Acidity (pKa) | 15.9 |
| Basicity (pKb) | 2.15 |
| Magnetic susceptibility (χ) | -74.5·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.438 |
| Viscosity | 1.285 mPa·s (20°C) |
| Dipole moment | 2.06 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 367.4 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -66.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3732.7 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS06 |
| Signal word | Danger |
| Hazard statements | H225, H302, H312, H315, H319, H332, H335 |
| Precautionary statements | P210, P261, P280, P305+P351+P338, P309+P310 |
| NFPA 704 (fire diamond) | 2-2-2-W |
| Flash point | 69 °F (20.6 °C) |
| Autoignition temperature | 215 °C (419 °F; 488 K) |
| Explosive limits | 3.6–15% |
| Lethal dose or concentration | LD50 (oral, rat): 740 mg/kg |
| LD50 (median dose) | LD50: 535 mg/kg (oral, rat) |
| NIOSH | BZ9275000 |
| PEL (Permissible) | PEL: Not established |
| REL (Recommended) | REL: NIOSH considers this substance to be a potential occupational carcinogen. |
| IDLH (Immediate danger) | IDLH: 100 ppm |
| Related compounds | |
| Related compounds |
2-Chloroethyl ethyl ether 2-Iodoethyl ethyl ether Ethylene dibromide 2-Bromoethanol Diethyl ether 2-Bromoethyl methyl ether |

