2-Bromobenzoyl Chloride: A Comprehensive Commentary
Historical Development
In the late nineteenth century, chemists started targeting specific substitutions on aromatic compounds to create versatile intermediates, and that approach sparked the early days of 2-bromobenzoyl chloride. Organic synthesis shifted quickly as research groups around the world discovered chlorination and bromination reactions could control substitution patterns for industrial uses. By the 1960s, pharmaceutical growth and fine chemical manufacturing pushed 2-bromobenzoyl chloride from a laboratory curiosity to a core building block in research and drug synthesis. Today, global manufacturers stick with this chlorinated acyl compound because it offers reliable reactivity, and those roots go back to the golden era of bench-top discovery and practical industrial chemistry.
Product Overview
2-Bromobenzoyl chloride presents itself as a transparent to pale yellow oily liquid, free-flowing at room temperature. Users, especially in pharmaceutical and agricultural labs, reach for this compound to forge bonds between brominated aromatic rings and a wide palette of nucleophiles—just ask a process chemist how often they open a bottle for small-scale batch runs. With a molecular formula of C7H4BrClO, its unique structure unlocks brominated derivatives essential in active pharmaceutical ingredients, agrochemicals, and specialty chemical sectors, where purity remains a tough but critical concern.
Physical & Chemical Properties
2-Bromobenzoyl chloride boils around 272°C under atmospheric pressure and melts in the chillier range of 22–25°C. In the lab, the faint, acrid suffocation of hydrogen chloride gas reminds anyone working with it not to stay casual about their fume hood etiquette. Hydroscopic tendencies mean a tightly capped vial lives longer, as moisture kicks off a furious release of HCl fumes. Solubility lands neatly in the non-polar spectrum—organic solvents such as dichloromethane, toluene, and chloroform handle it well, but water decomposes it on contact. Its shelf life owes a lot to cool, dry storage, as traces of water cause hydrolysis, reducing both yield and purity during syntheses.
Technical Specifications & Labeling
Producers frequently provide 2-bromobenzoyl chloride with purity exceeding 98%, and often note trace impurities like benzoyl chloride or bromobenzoic acid. Regulatory labeling includes GHS pictograms and hazard statements such as H314 (causes severe skin burns and eye damage), along with UN identification codes. Batch numbers, manufacturing dates, and supplier contact details follow closely behind, as traceability backs up every certificate of analysis. These may sound like paperwork, but a missing or unclear traceability chain means wasted time for both quality control analysts and end users.
Preparation Method
Most syntheses rely on direct chlorination of 2-bromobenzoic acid with thionyl chloride (SOCl2) or oxalyl chloride, often in the presence of catalytic DMF to boost reaction speed. As the thionyl chloride does its job, the set-up hisses with liberated sulfur dioxide and HCl, a reminder that venting and personal protection gear are life-savers, not optional add-ons. Thionyl chloride remains popular for scale-up, offering clean conversion, while oxalyl chloride’s gentle nature draws the favor of research groups focused on minimizing side reactions and byproduct headaches. Some green chemistry approaches try to limit hazardous reagents, with phosphorus trichloride and chlorinating agents seeing occasional use, yet SOCl2 dominates commercial batches thanks to its reliability.
Chemical Reactions & Modifications
As an acyl chloride, 2-bromobenzoyl chloride finds itself at the nexus of nucleophilic acyl substitution chemistry. It reacts briskly with amines to yield brominated benzamides in high yield, which can later morph into heterocyclic frameworks common in high-value drugs. Alcohols give esters when paired with the right base scavenger, and the reaction with water should be avoided unless hydrolysis is actually the goal. Grignard reagents and organolithium compounds react at the acyl carbon, opening doors to new carbon–carbon bonds and transformations leading into multi-step syntheses. Few acyl chlorides offer the same strategic balance between bromination and the reactive carbonyl, so it continues to punch above its weight for both seasoned synthetic chemists and early-career researchers.
Synonyms & Product Names
2-Bromobenzoyl chloride appears on chemical supplier lists under several names: ortho-bromobenzoyl chloride, 2-bromo-benzoyl chloride, o-bromobenzoyl chloride, or as the more systematic benzoic acid, 2-bromo-, chloride. Major suppliers may use catalogue numbers or abbreviations like BBC or BBCCl, though standardized nomenclature in regulatory filings keeps confusion to a minimum. Accurate labeling and naming aren't just a matter of bureaucracy—labs rely on proper identity to avoid mix-ups with close relatives like 4-bromobenzoyl chloride, where a simple error could kill a week’s work.
Safety & Operational Standards
Few chemicals in a synthetic lab require more respect than this one. Splashing causes chemical burns and breathing vapors assaults the respiratory system. I learned fast to keep gloves, goggles, and a sturdy face shield on when handling acyl chlorides. A reliable fume hood and immediate neutralization supplies, such as sodium bicarbonate or crushed ice, need space on the bench too. Regulatory standards from OSHA and REACH demand clear hazard labeling, thorough safety data sheets, and spill control plans in both shipping and workplace handling. Local hazardous waste management firms won’t pick it up unless those requirements are met, adding another layer of vigilance to daily practice.
Application Area
Synthetic chemists and process engineers keep 2-bromobenzoyl chloride on their inventory for its role building more advanced molecules. It pops up in the preparation of substituted benzamides and esters found in pharmaceutical candidates, dye precursors, agrochemical intermediates, and performance polymers. Academic labs value it for method development and mechanistic studies, while industrial R&D labs push for new active pharmaceutical ingredients using its reactive profile. Dermatology treatments, anti-inflammatory drugs, and antifungal agents—many synthesis routes pass through this handy brominated acyl chloride. In crop science, the molecule kickstarts design of herbicides and fungicides targeting plant pathogens. Each sector expects a high-purity, consistent material to minimize product recalls and costly re-analyses.
Research & Development
Research around 2-bromobenzoyl chloride circles around greener syntheses, new catalytic routes, and safer operational chemistry. Some groups look to avoid SOCl2 altogether, trialing less toxic chlorinating agents or flow-chemistry approaches to limit operator exposure. Structure–activity relationship studies focus on its benzamide derivatives, hoping for new antiproliferative agents or enzyme inhibitors with better selectivity. In one project, I found minor tweaks to substituent position enormously affected biological activity, reminding me that the aromatic halogen position can’t be swapped casually. Material scientists experiment with functionalized polyaromatics built from brominated benzoyl units, chasing high-performance coatings and resins. Juggling improved atom economy, worker safety, and benign waste—these goals keep research teams busy as markets evolve.
Toxicity Research
Toxicological data for 2-bromobenzoyl chloride flags it as a strong irritant and a corrosive agent. Skin contact causes burns, and eye contact can mean permanent damage. Inhalation induces respiratory distress and may worsen underlying lung disease. I’ve seen firsthand how a lax attitude about chemical hoods can land an intern in the health center with chemical exposure symptoms, so training remains a daily priority in any lab housing acyl chlorides. Safety data emphasize not only direct toxicity, but also risks from byproduct hydrogen chloride and from improper disposal. Animal studies, although limited for this particular reagent, demonstrate acute toxicity similar to analogues. Long-term exposure risks haven’t fully come to light, but the combination of reactivity and fume toxicity means monitoring protocols can't be skipped.
Future Prospects
Looking ahead, 2-bromobenzoyl chloride continues to draw attention from researchers seeking new chemical transformations and safer, scalable production schemes. Trends in green chemistry push for alternatives to hazardous chlorinating agents and batchwise handling, with continuous flow processing and solid-supported chlorination at the research frontier. Automated synthesis platforms need reagents with reliability and clear hazard profiles, and 2-bromobenzoyl chloride fits that mold if safety procedures do not falter. As pharmaceutical pipelines demand more halogenated candidates, I expect this molecule will remain relevant, nudging process chemists to improve purity, safety, and product stewardship. Markets change, but every time the need arises for a brominated carbonyl moiety, most chemists reach for this trusted reagent, blending history, hazard, and promise in a single bottle.
What is the chemical formula of 2-Bromobenzoyl Chloride?
Getting to Know 2-Bromobenzoyl Chloride
2-Bromobenzoyl chloride may seem like a mouthful, but its identity gets clearer if you imagine working in a chemistry lab at university, fumbling through reagent bottles during organic synthesis. The name already reveals a lot. You’re dealing with a benzoyl group, a chlorine atom attached where a carboxylic acid once lived, and a bromine atom planted on the aromatic ring.
Chemical Structure and Formula
Let’s untangle the formula piece by piece. At its core, benzoyl chloride looks like this: C6H5COCl. Swap out one of the hydrogens for a bromine atom in the ortho position, and the structure shifts, but the pattern holds. The final formula becomes C7H4BrClO.
Why 2-Bromobenzoyl Chloride Matters
In academic research and industrial chemistry, this compound lands in a special category—reactive intermediates. I’ve seen it put to work in the creation of pharmaceuticals and agricultural chemicals. Researchers value it for its ability to introduce both bromine and carbonyl chloride functional groups in a single step, which opens doors for further transformations.
Handling such chemicals brings real responsibility. 2-Bromobenzoyl chloride, like many acyl chlorides, stings your nose with pungent fumes and irritates unprotected skin. Early in my lab days, I learned (the hard way) that skipping gloves isn't worth saving time. Lab manuals and safety data sheets hammer the point home with good reason: exposure can cause real harm.
Practical Challenges in Handling and Use
Working with 2-Bromobenzoyl chloride means dealing with reactivity. Acyl chlorides hate water—touch a drop, and you get a burst of hydrogen chloride gas. Fume hoods become friends, not afterthoughts. Ventilation, goggles, thick gloves all play a part.
Waste disposal challenges also arise. Sending this stuff down the drain isn’t an option due to the environmental risks. Most labs employ strict protocols, neutralizing leftovers carefully and sending waste for professional processing. Each step reflects not just regulation but a genuine respect for public health and the environment.
The Road Ahead: Safer and Greener Alternatives
Many researchers look for gentler routes. In my experience, academic journals fill up with studies on alternative reagents and greener processes. Some chemists swap acyl chlorides for acid anhydrides when possible. It’s not about ditching effective reactions, but about weighing usefulness against hazards.
Better training for students and newcomers also makes a difference. Years ago, chemical safety often meant a five-minute overview that left plenty unsaid. Now, clear safety coaching and updated protocols take center stage. This shift keeps dangerous incidents down and confidence up.
Final Thoughts on Its Chemical Identity
2-Bromobenzoyl chloride—C7H4BrClO—sits at the intersection of practical utility and real-world risks. Anyone who spends time with it gets an early introduction to the demands of chemical responsibility, both inside the lab and beyond its doors. Understanding this chemical goes beyond formulas; it’s about managing risks, respecting its power, and always seeking a better, safer way forward.
What are the common uses of 2-Bromobenzoyl Chloride?
Key Applications Shaping Industry and Research
Walk into any active chemistry lab, and you’re bound to see a lineup of bottles with chemical names that only sound familiar to experts. Among these is 2-Bromobenzoyl chloride, a compound that shows up in the hands of people working on everything from pharmaceuticals to agricultural research. Its uses aren’t tied to gigantic factories alone; small research teams count on it for some of their most promising work.
Helping Synthesize Life-Changing Molecules
Medicinal chemistry teams lean on 2-Bromobenzoyl chloride to piece together complex molecules. The chemical structure—a benzoyl chloride with a bromine at the ortho position—lets it act as an eager participant in making amide and ester bonds. For those of us who have stood over a lab bench on late nights, this means building blocks for new antibiotics, cancer medicines, and several experimental therapies trace their origins back to transformations involving this exact compound.
Some well-regarded pharmaceutical studies single out 2-Bromobenzoyl chloride in the synthesis of nonsteroidal anti-inflammatory drugs and certain local anesthetics. The compound’s reliability and reactivity bring consistency to processes where a bad batch can hold up life-saving treatments. I’ve heard firsthand from drug researchers who praise how this chemical cuts down synthesis steps or offers yields that leave other reagents in the dust.
A Vital Tool in Advanced Material Science
2-Bromobenzoyl chloride doesn’t just solve medical puzzles; it’s crucial for those working on advanced polymers and specialty materials. Materials scientists like using it to tweak the properties of polymers, giving them better thermal stability or unlocking new electrical behaviors. Want to build a new flexible circuit or a coating that resists crazy weather conditions? This compound helps create monomers and crosslinkers that set those properties in motion.
Researchers also add this compound to organic synthesis strategies where a halogen or benzene ring is needed along with a reactive carbonyl function. The bromine offers a point for further modifications—making it a bit like putting a handle on a complex part, so it can attach to something even more useful down the line. For newcomers, it’s not just a piece of the puzzle; it shapes how the whole picture develops.
Environmental and Safety Concerns
Every chemist using 2-Bromobenzoyl chloride knows about its hazards. It fumes in moist air, bites your nose with its sharp smell, and eats through skin and clothing if spilled. This isn’t something handled casually. Strict protocols keep people safe, and the industry watches disposal practices because any slip-up can lead to environmental headaches. Teams need to wear full PPE, work under strong ventilation, and follow disposal regulations designed for corrosive, reactive waste. It’s a reminder that while chemistry brings breakthroughs, safety always anchors progress.
Moving Toward Greener Alternatives
Some emerging companies and university labs now hunt for greener synthesis routes. The aim is to slash the reliance on hazardous reagents without stalling progress in drug discovery or advanced materials. People turn to strategies like flow chemistry and microwaves to cut down waste and exposure. While nothing matches 2-Bromobenzoyl chloride’s versatility for certain tasks, modern innovation pushes toward safer, more sustainable solutions that protect workers and the environment.
Chemical science relies on practical building blocks, and 2-Bromobenzoyl chloride stands out through a combination of reactivity and reliability. The work continues, driven by researchers who know that with every use comes a responsibility to improve safety and look for even better answers.
How should 2-Bromobenzoyl Chloride be stored and handled safely?
Understanding What’s at Stake
Working with chemicals like 2-Bromobenzoyl Chloride calls for real caution. This isn’t the sort of compound found in a home cleaning cabinet. Its sharp, pungent odor and the way it reacts with water or moisture create hazards that can’t be shrugged off. The damage from accidental splashes or leaks lingers. From years in the lab, I’ve seen how even minor spills require a scramble in protective gear and a scrubbing marathon no one enjoys.
Setting Up the Right Storage
Storing this chemical means more than tossing it on a shelf. It reacts badly to dampness, and contact with air can lead to fume release that stings the eyes and nose. Glass containers with tight PTFE-lined caps work best because they resist corrosion and keep vapors where they belong. Years ago, I saw the chaos from a plastic cap that crumbled – the mess took half a day to clean, and left a harsh smell for weeks.
A flammable chemicals cabinet with clear labeling helps. Keeping 2-Bromobenzoyl Chloride far from sources of water, acids, and bases matters more than most realize. These cabinets often contain spill trays. If a bottle fails, the tray catches the leak and gives precious minutes to suit up and neutralize the spill. Room temperature is fine, but sudden heat causes pressure to build up, so never store this chemical near heat vents or direct sunlight.
Safe Handling: People Come First
Everything starts with education. No one should touch 2-Bromobenzoyl Chloride unless they understand what it does and how to treat exposure. I remember my first safety briefing. The instructor hammered home that splash goggles, gloves, and lab coats aren’t optional. Once, a collar button popped while I was pouring. The sleeve of my coat caught a drop. Quick washing kept a painful rash from turning into a scar.
A fume hood changes everything. The fumes irritate throats and eyes fast. Using a fume hood keeps vapors away from faces and lungs, and safe disposal methods prevent the buildup of hazardous waste. Working in pairs lets one person work and the other keep an eye out for trouble.
Prepping for the Unexpected
Spill kits for acid chlorides belong in every chemical storage area that houses 2-Bromobenzoyl Chloride. That means absorbent pads, neutralizers like sodium bicarbonate, and thick nitrile gloves ready to grab. I can’t count the times that kit gave me peace of mind. Fire extinguishers rated for chemical fires should never sit too far away, since organic chlorides can feed flames under the right conditions.
Contingency plans matter. Everyone in a lab should know the closest eyewash station and safety shower by heart. Posting clear signage and talking through emergency procedures takes a few extra minutes, but makes a huge difference during real emergencies.
Solutions for a Safer Workplace
Chemists and facilities managers can reduce risk by limiting the amount of 2-Bromobenzoyl Chloride kept on hand. Just-in-time inventory keeps storage volumes low. Training programs should include regular drills and refreshers, not just annual checkboxes. Manufacturers might help by offering smaller package sizes or safer alternatives when possible.
Technology offers hope, too. Sensors that alert users to leaks or spills in storage cabinets cost a bit more, but I’ve seen one catch a slow vapor leak before anyone fell ill.
Storing and handling chemicals like 2-Bromobenzoyl Chloride safely is not optional or just a list of rules to follow. It’s a responsibility every worker shoulders for everyone around them.
What are the potential health hazards of 2-Bromobenzoyl Chloride?
What This Chemical Actually Does To The Body
2-Bromobenzoyl chloride sounds like something out of a high school chemistry test, yet it often enters workplaces through pharmaceutical or chemical synthesis jobs. Inhaling, touching, or accidentally ingesting this compound isn’t something to brush off. This stuff packs a punch, and research shows it can hurt health across a range of exposure routes.
Physical Contact: More Than Just Minor Irritation
Guessing a chemical’s danger by its smell or appearance doesn’t work here. Even a small splash of 2-Bromobenzoyl chloride on the skin sets off burning, redness, and sometimes ulcers. The compound reacts with the moisture on skin and releases hydrochloric acid. That acid is what starts chewing away at the outer layers—resulting in severe burns with enough exposure. Eyes take a tougher hit. Exposure triggers intense pain, inflammation, tearing, and—if not washed out swiftly—long-term vision problems. Lab workers know this all too well; even goggles sometimes don’t keep out the fine aerosol or vapor forms.
Breathing Fumes: Trouble For Airways And Lungs
Workers report tight chest, coughing, and choking after breathing vapors of 2-Bromobenzoyl chloride for even a short time. This chemical creates fumes made up of hydrochloric acid gas and other byproducts, causing respiratory tract injury. Medical journals document rapid swelling in the throat, as well as potential for bronchospasm. Asthma and chronic bronchitis can develop if exposure stays unchecked. No one wants to cough up blood or struggle for breath simply doing their job, so this is a big concern.
Long-Term and Chronic Exposure Risks
Acute symptoms may get attention, but drawing attention to possible long-term effects proves just as important. Regular, low-level exposure often flies under the radar. The International Agency for Research on Cancer assesses similar aromatic acid chlorides for links to cancer. Acid chlorides like 2-Bromobenzoyl chloride break down and create reactive molecules, which can damage DNA and cells over time. Some studies point to changes in the liver, kidneys, or immune function in workers with repeated exposure. Not every country spells this out with crystal clarity, but worrying signals exist.
Prevention Beats Scrambling Later
Dealing with these hazards calls for common-sense solutions—better safety gear, engineering controls, and strict training. Lab coats, gloves, and tight-sealing eye protection should come standard. Fume hoods and exhaust systems keep vapors away from breathing zones. Safety data sheets for 2-Bromobenzoyl chloride make clear: no half-measures on ventilation. Regular air monitoring also matters. No employee wants to find out about exposure too late. Routine medical checkups for chemical workers give early warning if trouble brews internally.
Building Smarter, Safer Workplaces
Frequent honest discussions make the biggest difference. Management and staff can build safety cultures where nobody feels embarrassed to speak up if PPE breaks down or storage slips. In my time consulting with chemical manufacturing sites, those that created a climate for straightforward reporting tackled incidents faster than those buried in paper trails. Making safety convenient—like glove dispensers next to every bench and labeled washing stations—keeps accidents from becoming medical emergencies.
Final Thought
For every headline about workplace chemicals, real lives stand behind the statistics. Staying alert to the hazards of 2-Bromobenzoyl chloride, learning the warning signs of exposure, and using every tool for prevention keeps those lives safer. It simply comes down to respecting the risks—and making sure nobody pays a health price just for doing their job.
What is the purity and packaging information for 2-Bromobenzoyl Chloride?
Why Purity Actually Matters in the Lab
Plenty of lab techs have spent extra hours tracing weird results back to a batch of chemicals that carried a hidden hitch. In pharmaceutical, agrochemical, or material science work, purity often draws the line between a project that succeeds and one that wastes valuable time. For 2-Bromobenzoyl chloride, research-grade batches reliably show a purity over 98%, sometimes even nudging closer to 99%. Industrial stock might settle a fraction lower, but anything under 95% throws quality control off the rails.
Labs keep their eyes on certificates of analysis, making sure no stray halides, unreacted precursors, or leftover solvents squeeze into that reported purity. Chromatography or NMR reveals trouble faster than wishful thinking. One surprising fact for the unfamiliar: even tiny, invisible contaminants can derail synthesis downstream, scaling up from milligram errors to kilo-level failures.
A detail many folks overlook—water content. 2-Bromobenzoyl chloride practically grabs moisture from the air, and moisture eats away at both purity and reactivity. If the bottle spend more time open than needed, it won’t deliver what’s expected at the bench or in industrial reactors.
Packaging Choices: Not All Bottles Are Equal
Nobody receives specialty chemicals in unlabeled jars. For 2-Bromobenzoyl chloride, glass bottles with airtight Teflon-lined caps win out, especially for analytical use. Plastic leaches, glass doesn’t. It’s not just about keeping the light out or preventing spills—this compound gives off acid fumes and reacts furiously with humidity, so a poor seal invites corrosion and spoiled material.
Standard packaging runs from 25-gram to 100-gram glass bottles for research, stretching up to several kilograms for bulk or manufacturing. Vendors who care about their customers ship these containers in double-bagged secondary containment, sometimes packed with desiccant to siphon off stray moisture if the shipment crosses humid territory.
Labels give more than just a name or batch number. Good suppliers print hazard pictograms up front because this chemical’s fumes irritate eyes, skin, and lungs. The packaging needs to warn users with an expiration date, storage instructions (usually cool and dry, away from acids and bases), and a lot code for quick tracking if a recall or quality concern crops up. I’ve seen plenty of scientists wrestle with faded generic labels, straining to figure out if their bottle still qualifies for accurate results.
Better Transparency and Safer Labs
It’s one thing to talk specs, another to see documentation. Responsible chemical manufacturers upload recent purity test data—actual chromatograms, not just summary numbers—and list out every detectable impurity over 0.1%. That transparency saves companies from repeat errors and makes traceability simple if issues show up in scaled-up production.
For safer storage, separate corrosive chemicals like 2-Bromobenzoyl chloride from amines and water sources, since leaks can start fast if those paths cross. Sharpening SOPs and repacking into smaller bottles right before use keeps the gear fresh for longer. Regulatory guidance in developed markets now pushes for tamper-evident seals and hazard-specific data sheets with every purchase.
In the end, rigor around purity and packaging does more than just satisfy auditors. It keeps both results reproducible and people safe, especially with powerful reagents on the workbench.
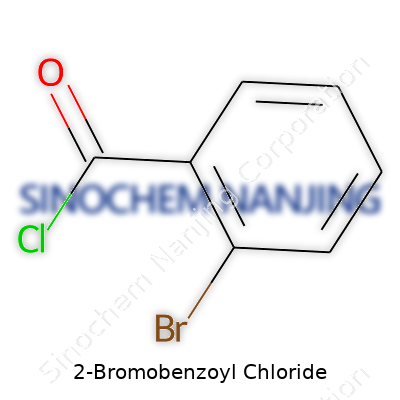
| Names | |
| Preferred IUPAC name | 2-bromobenzoyl chloride |
| Other names |
2-Bromobenzoyl chloride o-Bromobenzoyl chloride α-Bromobenzoyl chloride Benzoyl chloride, 2-bromo- 2-Bromobenzoic acid chloride |
| Pronunciation | /tuː ˌbroʊmoʊˈbɛnzoʊɪl ˈklɔːraɪd/ |
| Identifiers | |
| CAS Number | 4219-71-8 |
| Beilstein Reference | 1209034 |
| ChEBI | CHEBI:27820 |
| ChEMBL | CHEMBL16600 |
| ChemSpider | 2663320 |
| DrugBank | DB07816 |
| ECHA InfoCard | 100.011.010 |
| EC Number | 211-574-2 |
| Gmelin Reference | 14329 |
| KEGG | C19136 |
| MeSH | D015675 |
| PubChem CID | 73809 |
| RTECS number | CG3675000 |
| UNII | Q08N2JDW3V |
| UN number | UN1737 |
| CompTox Dashboard (EPA) | DJ7I6ZJ6HT |
| Properties | |
| Chemical formula | C7H4BrClO |
| Molar mass | 231.45 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | Pungent |
| Density | 1.626 g/mL at 25 °C |
| Solubility in water | Reacts with water |
| log P | 2.9 |
| Vapor pressure | 0.4 mmHg (20°C) |
| Acidity (pKa) | 1.04 |
| Basicity (pKb) | Basicity (pKb): -3.3 |
| Magnetic susceptibility (χ) | -72.0e-6 cm^3/mol |
| Refractive index (nD) | 1.6100 |
| Viscosity | 1.601 cP (20°C) |
| Dipole moment | 2.72 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 339.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | –37.0 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -365.6 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | There is no ATC code assigned to "2-Bromobenzoyl Chloride". |
| Hazards | |
| Main hazards | Harmful if swallowed, causes severe skin burns and eye damage, may cause an allergic skin reaction, toxic if inhaled. |
| GHS labelling | GHS05, GHS06 |
| Pictograms | GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | H302, H314, H315, H319, H335, H400 |
| Precautionary statements | P261, P280, P305+P351+P338, P304+P340, P309+P311 |
| NFPA 704 (fire diamond) | 3-1-0 |
| Flash point | 90°C |
| Lethal dose or concentration | LD₅₀ (oral, rat): 1800 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 1240 mg/kg |
| NIOSH | CY3675000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 1 gram |

