2-Bromoaniline: Tracing Its Story, Science, and Impact
Historical Development
The early chemistry of anilines fascinated scientists in the nineteenth century. 2-Bromoaniline emerged from these classic aromatic studies, becoming a reliable building block as researchers explored ways to transform crude coal tar into more useful compounds. Brominated anilines, including the ortho isomer, played a key role in dye development, giving color to textiles, and entered the pharmaceutical sector as new synthetic methods appeared. In the hands of organic chemists, the compound moved from laboratory demonstration to industrial production. Its preparation once depended on skillful handling of elemental bromine and precise control of reaction conditions—something chemists refined across generations in pursuit of better yields and fewer byproducts. The fact that you can now buy it by the drum, complete with tightly defined purity standards, says a lot about its steady demand and the technical progress behind its mass manufacture.
Product Overview
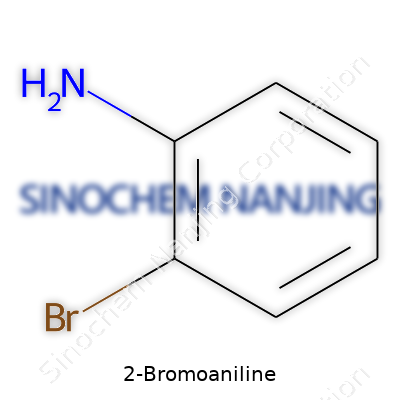
2-Bromoaniline, known by its chemical formula C6H6BrN, looks like a pale brown solid or a faintly colored oil. This aniline derivative groups a bromine atom at the ortho position relative to the amine on the benzene ring. Practical chemists see it as more than a catalog reagent; they reach for it to make agrochemicals, pharmaceuticals, or even functional materials. Its appeal traces back to the way the bromine atom opens up further transformation routes, whether through cross-coupling, halogen-lithium exchange, or palladium-catalyzed reactions. For me, its distinctive, slightly irritating odor triggers memories of crowded undergraduate labs, learning to handle aromatic amines with respect and steady hands.
Physical & Chemical Properties
Physical characteristics of 2-Bromoaniline matter a lot to process engineers and bench chemists. The compound usually melts around 32–36°C and boils above 240°C. Missed melting points in old labs often signaled contamination. Its density approaches 1.6 g/cm³, and the bromine atom accounts for much of the molecule’s heft. Unlike some volatile organics, it won’t float off under standard lab conditions, but its vapor still carries a warning, especially for sensitive noses. On the chemical front, the presence of both a nucleophilic amine and an electrophilic bromine sets the molecule apart. That duality underpins most of its application in organic synthesis. You can watch the rich interplay of aromatic substitution and the electron-donating effects from the NH2 group when working up reactions or planning new routes. Solubility in organic solvents offers flexibility, while water solubility remains low—something that influences choices in workup and waste disposal.
Technical Specifications & Labeling
Rigorous labeling and tight technical specs form the backbone of chemical commerce. Suppliers commonly list CAS number 578-57-4 for 2-Bromoaniline, along with purity grades that reach 98% or higher. Analytical chemists keep an eye out for related anilines, dibromo isomers, and trace water content, all of which can trip up downstream processes. Safety and hazard labels make up an important part of the packaging: skull-and-crossbones motifs, GHS hazard codes, and instructions for storage away from light, moisture, and incompatible oxidizers. The lot number, batch certificate, and material safety data sheet provide accountability. In my hands, success with this compound starts with a glance at that certificate, making sure the amine’s reactivity won’t get lost in impurities or mishandling from the warehouse.
Preparation Method
Most commercial 2-Bromoaniline starts with direct bromination of aniline under controlled conditions, using bromine in an acidic medium to guide substitution toward the ortho position. Controlling temperature, acid concentration, and rate of addition pays off by minimizing overbromination and para isomer formation. Some newer methods swap out elemental bromine for N-bromosuccinimide or even greener alternatives, cutting down sharply on byproducts and waste. In university settings, I saw students struggle with precise isolation—learning why cooling the mixture and managing pH shaped both yield and purity. Accounts from older process chemists recall days of open-flask brominations, but modern practice leans on closed systems and continuous flow approaches for better safety and scale.
Chemical Reactions & Modifications
The story of 2-Bromoaniline turns technical once it enters the synthesis lab. The orthobromo position makes it a strong candidate for Suzuki, Heck, or Buchwald–Hartwig couplings. You can engineer complex biaryls, heterocycles, or extended aromatic systems starting with this single molecule. The amine itself isn’t idle—it gets acylated, diazotized, converted into isocyanates, or built into azobenzenes under the right conditions. Cross-coupling reactions in the pharmaceutical sector depend on brominated anilines, as they merge stability with ease of activation. Chemists take full advantage of the dichotomy: the electronic influence of the amine speeds up certain reactions, cuts down reaction times, and sometimes enables transformations unattractive for other bromoarenes. In my own synthetic work, 2-Bromoaniline’s blend of stability and reactivity has saved me more than once, shortening steps in difficult syntheses or allowing late-stage functionalization when other routes stalled.
Synonyms & Product Names
Names can trip up anyone new to the aromatic amines. 2-Bromoaniline, o-Bromoaniline, ortho-Bromoaniline, Benzenamine, 2-bromo-, and 1-Amino-2-bromobenzene all describe this same molecule. Catalogs list the same compound under these various names as well as its registry numbers. Each supplier prints its own flavor of branding, but the chemistry remains unchanged. In research and development paperwork, consistent naming keeps confusion away, especially with multiple isomers or in regulatory submissions. Seasoned chemists learn to scan alternate names to spot hidden analogs or avoid ordering the wrong material.
Safety & Operational Standards
Practicing good safety with 2-Bromoaniline can’t be an afterthought. Like many aromatic amines and halogenated compounds, it poses acute toxicity by inhalation, skin absorption, or ingestion. Gloves, goggles, and fume hood work shield against direct exposure. Industrial environments often add local exhaust ventilation and automatic shut-offs to tackle larger risks. It catches fire at elevated temperatures, so segregating from oxidizers matters. Chronic exposure leads to concerns around methemoglobinemia, which shows up in literature dating back decades. Emergency protocols require antidotes like methylene blue in rare cases of poisoning. Modern labs run safety drills, track all transfer steps, and store the molecule in labeled, light-resistant bottles—minor oversights have caused headaches and worse. The push for safer handling links directly to better training, smarter engineering controls, and constant hazard review.
Application Area
2-Bromoaniline’s unique combination of functional groups sets it up for diverse use across chemical sectors. In pharmaceuticals, research chemists harness it as a precursor for antihistamines, antibiotics, or antitumor agents built on aromatic scaffolds. Agrochemical developers rely on the compound for herbicides and fungicides targeting pests but sparing crops. Cross-coupling technology has opened up further uses in advanced materials, LEDs, and specialty polymers. In academic research, the compound supports countless method development projects or biological probes. Past supervisors favored it for library synthesis, using the easy transformation of both NH2 and Br groups to populate vast chemical spaces. No matter the application, chemists stay alert for trace impurities from synthesis and make sure it meets the strictest purity standards so bioactivity or material properties don’t get compromised by hidden contaminants.
Research & Development
Innovation with 2-Bromoaniline didn’t stop once it found a place in catalogues. Method development teams target new routes that conserve raw materials, produce less hazardous waste, and offer better selectivity for the ortho isomer. Academic groups build on cross-coupling, experimenting with nickel and copper catalysts in place of precious metals, to keep costs down and minimize resource scarcity. Companies compete to scale up reactions that were once the preserve of gram-scale labs, extending these transformations to metric tons. In recent years, the rise of “green” chemistry has driven up interest in solvent-free processes and electrochemical bromination, wresting even better atom economy and safety from old recipes. Conferences and journals brim with updates on new ligands, catalysts, and flow technologies—signs that even stable molecules like 2-Bromoaniline still serve as testing grounds for chemical ingenuity.
Toxicity Research
Animal studies and mechanistic work show that 2-Bromoaniline brings concerns similar to other halogenated anilines. Acute exposure affects the blood’s oxygen-carrying capacity by interfering with hemoglobin function. Studies observe changes in organ tissues and red blood cell chemistry at higher doses, driving regulatory bodies to assign stringent workplace limits. Human health monitoring in manufacturing plants revealed the need for regular screening for methemoglobinemia among exposed workers. Wastewater treatment research tracks the breakdown of 2-Bromoaniline and highlights risks to aquatic life if not managed carefully. Environmental persistence becomes a focus as more products and byproducts enter water streams. Toxicology labs keep pushing for assays that catch subtle changes in cell cultures and DNA, reflecting growing awareness of the long-term impact. The story told by this research underscores the demand for closed-process manufacturing, personal protective gear, and thorough environmental review.
Future Prospects
2-Bromoaniline’s future looks tied to the larger arc of green chemistry and precision synthesis. Chemists still seek better selectivity, less waste, and more sustainable routes. Computational modeling now helps predict new catalysts or reaction conditions before resources get committed in the lab. Efforts in automation and continuous flow hint at further reductions in reagent consumption and human exposure. Pharmaceutical discovery continues to lean on 2-Bromoaniline’s unique reactivity as a way to scaffold next-generation drugs or materials, while environmental scientists watch for safer alternatives to aniline-based synthons. Education in lab safety and chemical stewardship prevents lapses seen in past decades. Drawing from experience in both research and industrial settings, I see this molecule staying relevant—assuming chemists keep blending creative thinking with responsible innovation, always respecting both what this molecule enables and the hazards it brings along.
What is the molecular formula of 2-Bromoaniline?
Breaking Down 2-Bromoaniline
Anyone who’s ever sat through an organic chemistry lecture probably remembers wrestling with benzene rings, halogen groups, and plenty of memorization. That struggle pays off when you face compounds like 2-Bromoaniline. This molecule keeps popping up in labs and research projects, especially for those interested in making new medicines or specialty materials.
2-Bromoaniline belongs to the aniline family – all of them come from benzene, that simple six-carbon ring often etched into notebooks during study sessions. What sets 2-Bromoaniline apart is the bromine atom, fixed to the second position right beside an amino group on the ring. This relationship gives the molecule unique properties and an unmistakable structural fingerprint.
Molecular Formula: C6H6BrN
Bond by bond, the formula for 2-Bromoaniline works like this: the benzene ring shows up as C6H6. Lose a single hydrogen as the amino group (-NH2) attaches, then swap out another hydrogen for the bromine atom. What’s left is C6H6BrN. It might look simple, yet those four different elements unlock complex chemistry.
It’s worth understanding formulas for more than textbook answers. Industries depend on accuracy here. Chemists who synthesize pharmaceuticals or pesticides can’t afford mistakes with molecule counts. The wrong mixture, even by a tiny margin, may endanger both product and people. Knowing exactly what C6H6BrN means sets those strict safety measures in motion to avoid dangerous byproducts.
Why Formula Knowledge Matters for Non-Chemists
Sometimes, there’s a sense that molecular formulas only matter to folks in white coats or those hunched over lab benches. That isn’t true. As more people care about the chemicals inside their medications, cleaner manufacturing methods or even building better batteries, understanding something like 2-Bromoaniline gives regular folks a chance to ask good questions.
A clear grasp of these details supports everyday decisions, from what goes in our bodies to how communities handle waste. For instance, 2-Bromoaniline holds potential as a building block for dyes or drugs. Waste management protocols lean on accurate formulas to treat runoff safely and avoid health hazards. One forgotten atom in documentation could cause big regulatory headaches — including fines, sick workers, or contaminated water.
Challenges, Solutions, and Responsible Use
Mistakes and mishandling happen. Some of the most damaging chemical spills happened because small errors in formula or structure crept into manufacturing. One solution that works is stronger training for anyone near chemical stocks, not just chemists. Digital tools now support the tracking of formulas and batch creation with computerized checks, reducing human error. For many years, a paper logbook stood as the first and last line of defense; now, these systems bring formulas like C6H6BrN into databases in seconds, so the risks drop sharply.
It’s also wise for everyone—students, workers, and community members—to engage with scientists about how chemicals are handled locally. Public forums, transparent labeling, and open data-sharing about chemicals like 2-Bromoaniline keep pressure on producers to act responsibly.
Whether you run a lab or read the back of a product label, knowing what a formula stands for keeps people safer, sharper, and more empowered. Each molecule—especially those like 2-Bromoaniline—deserves a closer look and a steady hand.
What are the main uses or applications of 2-Bromoaniline?
The Practical Face of an Aromatic Amine
2-Bromoaniline stands out as a building block in organic chemistry. The benzene ring with a bromine atom attached gives this compound a unique spot in synthesis work. Many projects in pharmaceuticals, agrochemicals, and dyes rely on such fine-tuned molecules, and from experience in laboratory settings, I’ve seen how its presence can turn an ordinary starting material into something much more specialized.
The Heart of Pharmaceutical Development
Chemists look to 2-Bromoaniline to forge new pharmaceutical agents. The molecule serves as a launchpad for active drug components. Thanks to its amino group and the reactivity of the bromine, chemists can shape 2-Bromoaniline into pain relievers, anti-cancer drugs, and anti-inflammatories. This transformation process often follows well-known synthetic routes, where the bromine atom helps make further modifications possible. Even though people outside the lab seldom hear about these steps, everything from painkillers to antibiotics might rely on such intermediates at some stage of development.
Making Protective Chemicals for Agriculture
Food production often calls for reliable protection against pests. Companies have turned to 2-Bromoaniline to create herbicides and fungicides, helping farmers manage their fields. Usually, this compound serves as a stepping-stone to those chemicals, offering a way to connect small molecular changes with enormous harvest impacts. In my region, we’ve seen how the right crop treatment means less blight and better yields, and the chemistry behind it often begins with these sorts of tailored intermediates.
The World of Color: Dyes and Pigments
Bright colors and strong pigments mean more than just art—they signal advances in textile and ink technology. 2-Bromoaniline helps dye makers achieve shades that last through sunlight and repeated washing. The bromine atom lets chemists guide reactions to build complex pigment structures. Next time you see a shirt that keeps its color all year, odds are careful chemistry played a role. Working in textile labs, I noticed the demand for vibrant, resistant pigments never fades, and reliable azo dyes often trace their roots back to simple building blocks like this one.
Beyond the Lab Bench: Safety and Responsibility
No talk about chemical intermediates feels complete without looking at safety and health. 2-Bromoaniline can irritate the skin and lungs, so lab workers use gloves, masks, and fume hoods as a matter of habit. Good ventilation and proper disposal keep risks in check, and workplaces regularly update safety training. Companies pay close attention to chemical handling rules since a single accident can set back both people and production. Sharing accurate, up-to-date safety information matters as much as the technical side of the field.
Improving Production: Green Chemistry and Waste Reduction
As demand rises, more producers ask for greener methods. Less waste, lower emissions, and safer reagents improve the whole process. Chemists work on milder reaction conditions, use recyclable solvents, and try to avoid toxic byproducts when handling compounds like 2-Bromoaniline. Some plants recycle leftover chemicals, reducing disposal problems. Embracing these solutions lets companies keep up with safety standards and public trust—an investment that goes far beyond compliance regulations.
What is the CAS number for 2-Bromoaniline?
Why a Simple Number Can Mean So Much in Chemistry
Working with chemicals in any lab, whether a high school classroom or a large pharmaceutical company, means dealing with hundreds of substances that sound frustratingly similar. Want to avoid a mix-up that leads to wasted time or a dangerous reaction? A CAS number cuts through the confusion. For 2-Bromoaniline, that number is 578-57-4. This isn't just a random sequence—the CAS number acts as a universal fingerprint for this molecule, setting it apart from any close sibling.
From Research Bench to Production Floor
I spent plenty of days in a cramped academic lab. Tight budgets, dozens of chemical bottles, and a row of exhausted students left little room for error. The safety sheet on the wall only listed chemicals by their proper CAS number, not their common names. If I needed 2-Bromoaniline for a synthesis, grabbing the wrong isomer because the bottle only had 'bromoaniline' scribbled on it would have thrown off the entire experiment. The CAS system helped us double-check every shipment, every safety sheet, and every label.
2-Bromoaniline’s CAS number, 578-57-4, keeps order. Chemists, regulatory officers, and customs agents use that code. The catalog system recognizes it, regulatory agencies around the globe trust it, and customs officers want to see it on shipping documents. In fact, during one import inspection, an entire shipment of research chemicals nearly got held up simply because the manifest listed trade names instead of CAS numbers—delaying research for weeks. Only after resubmitting proper documentation did the chemicals get released.
Protecting Health and the Environment
It’s easy to forget that a small labeling slip-up could expose someone to dangerous fumes or chemicals. The Occupational Safety and Health Administration (OSHA) and international equivalents refer specifically to chemicals by their CAS numbers on materials safety data sheets. Given that 2-Bromoaniline can be toxic if handled incorrectly, recognizing it by 578-57-4 means clear, traceable records for training, storage, and disposal.
The environmental impact of chemical mistakes also comes into play. If a shipment containing 2-Bromoaniline gets mislabeled and accidentally classed as a less hazardous compound, both transportation workers and ecosystems could pay the price. Agencies like the EPA depend on CAS numbers for tracking reporting and incident responses. Case studies show that waste disposal companies stopped accepting shipments without a matching CAS number, slowing down clean-ups and costing research labs extra money.
Keeping Supply Chains Honest and Secure
Counterfeit and mislabeled chemicals—an issue in the global supply chain—pose serious risks. Honest suppliers display the CAS number clearly, letting their customers cross-check with global databases. Not long ago, an outdated supplier catalog almost tricked a colleague into ordering a different bromoaniline isomer for a crucial step in dye synthesis. Since the supplier provided CAS numbers, she caught the mistake in time and avoided scrapping weeks of work.
Small Number, Big Impact
As technology and chemical research evolve, so does the complexity of regulations and supply networks. Many companies have shifted to digital inventory systems, coding every substance with its CAS number. The chemical industry, with safety and regulatory bodies in step, relies on clear identification to reduce risk. Mistakes happen, but using 578-57-4 for 2-Bromoaniline as a unique keystone makes those mistakes far less likely.
Working Toward Fewer Mistakes
Rolling out straightforward digital tools in labs and teaching solid chemical safety practices from day one can prevent errors. With every student, researcher, and staff member trained to check CAS numbers like 578-57-4, labs get safer, product quality stays consistent, and the environment gets a much-needed break from chemical mishaps.
What safety precautions should be taken when handling 2-Bromoaniline?
What Makes 2-Bromoaniline Risky?
2-Bromoaniline shows up in many labs. From making dyes to pharmaceuticals, plenty of early-career chemists have crossed paths with this compound. The challenges kick in because it brings both toxicity and corrosive qualities. Its vapors aggravate the respiratory system, and prolonged skin exposure can trigger burns or allergic reactions. Many don't realize that 2-Bromoaniline soaks through nitrile gloves over time, and even short contact leaves behind unhealthy residues.
Personal Experience: The First Encounter
The first time I worked with 2-Bromoaniline, the warnings on the bottle seemed intimidating but distant. During a busy session, I missed a drop on the glove. An hour later, my skin turned red and itchy where the glove had stretched. That experience drove home what textbooks always say—this chemical demands respect, not routine.
Protective Gear You Don’t Ignore
Getting lazy about gear puts health on the line. Decent goggles, tight-fitting nitrile gloves with double-gloving during transfers, and a long-sleeved lab coat turn risks into manageable ones. Disposable gloves should be changed at the first sign of wear or contamination. Splash-resistant goggles, not just regular specs, guard against random splatters.
Every lab worker ought to treat their own safety as an investment, not just protocol. The right gloves, fitted goggles and even chemical-resistant sleeves close the gaps where this compound tries to sneak in. A solid pair of shoes, not sandals, matters just as much—floor spills rarely call ahead.
Good Habits Keep the Air Clean
No one should pipette or mix 2-Bromoaniline outside of a working fume hood. Ventilation pulls away invisible fumes that irritate the throat and lungs. Some labs put safety sensors above workbenches to detect vapor releases, a practice that makes sense in crowded spaces.
After handling, hands must get washed with soap and plenty of water, even if gloves never look breached. Any contaminated glassware or pipette tips go straight into marked waste containers, which means no rinsing in the communal sink. Clean-up wipes down surfaces and clears piles of unused tools.
Safe Storage Reduces Surprises
This compound stays happiest inside a tightly-sealed, labeled glass bottle. Keep it stored in a cabinet meant for toxic organics, away from sunlight and far from acids, bases, or oxidizers. An organized chemical shelf sets workers up for fewer mistakes. Spill kits with neutralizing agents belong wherever this bottle gets opened.
Emergency Readiness Can't Wait
A cheap bottle of eyewash or a taped-off shower won't save anyone if the group hasn’t practiced spill drills. Each team member should know which route leads to running water, how to get gloves and jackets off fast, and whom to call for help. Every second saved in an emergency means less damage to skin or lungs.
All these rules feel like overkill until that “what if” moment you wish you’d prepared for. It’s not paranoia—just the calm confidence that today’s good habits mean an uneventful, healthy tomorrow.
What is the physical appearance and solubility of 2-Bromoaniline?
What Does 2-Bromoaniline Look Like?
2-Bromoaniline turns up in the laboratory as a pale, sometimes yellowish solid. It forms needle-shaped or plate-like crystals, not the kind of powder that blows around with a breeze. If you scoop it, you can tell the crystals pick up a bit of light in the right angle. Its color comes from the aromatic ring mixed with a bromine atom, which attaches at the ortho position on the aniline backbone. Chemistry students often notice the faint, sometimes sharp, odor—enough to know you're working with an aromatic amine, but less nose-punching than straight aniline. Physically, the material clumps together and doesn’t create dust clouds like some lighter solids do. This makes spilling on the bench less dramatic, but it’s still wise to handle it in a fume hood due to toxicity risks.
Melting point clocks in around 30°C to 35°C, so it doesn’t take much warmth for it to go from solid to liquid. In a room that's gotten warm, you might notice it softening or even partially melting. The low melting point hints at the kind of molecular interactions involved. This melting behavior makes storage and handling pretty straightforward if the lab temperature stays in check, but heat waves or direct sunlight can turn this crystalline solid into a slippery mess.
Solubility: Where Does 2-Bromoaniline Mix In?
Ask a seasoned chemist about its solubility and you'll get a quick answer: 2-Bromoaniline barely dissolves in water. Aromatic rings resist hanging out with polar water molecules because they like their own crowd—nonpolar solvents. Run a simple experiment and you'll see it separating and floating or sinking, not blending in.
Place it in ethanol, ether, or toluene, and the story changes. In these organic solvents, 2-Bromoaniline dissolves quite well, showing why it gets chosen in organic synthesis and library prep. It goes fully into solution when warmed slightly, which matches the kind of work synthetic chemists do when prepping intermediates. The solubility in alcohols and other organics means researchers don’t waste time filtering undissolved chunks, allowing reactions to run smoother. It doesn’t stay suspended or form colloids in water—it settles, separates, or forms visible layers.
Why These Traits Matter in Practice
Physical state and solubility aren't just trivia for reference tables. These features can make or break a process in any lab setting. For example, in pharmaceutical labs, choosing solvents depends on how quickly compounds dissolve and whether byproducts separate out cleanly. Poor water solubility means 2-Bromoaniline won’t contaminate groundwater easily, yet its preference for organics tells you spills or leaks travel with organic waste streams instead. Its solid form below body temperature keeps accidental exposures down, but improper storage in warmer conditions could raise risks of skin or vapor contact.
Facts bear out the need for good protective measures. According to the European Chemicals Agency, 2-Bromoaniline brings moderate hazards if handled carelessly—especially by inhalation or skin absorption. Good engineering controls and solvent management help keep researchers and the environment safer.
Sorting Out Safe Handling and Disposal
If you handle 2-Bromoaniline, a glovebox or fume hood isn’t overkill—it’s sensible. Airflow sweeps away vapors, and keeping solids away from warm surfaces stops accidental liquefaction. When tasks wrap up, waste goes in organic solvent bins, never down the drain. On the clean-up side, labs using activated carbon or solvent washes see better results than those relying on simple water scrubbing. Relying on what’s known about solubility definitely improves outcomes—both for yields and for people’s health.
| Names | |
| Preferred IUPAC name | 2-bromoaniline |
| Other names |
o-Bromoaniline 2-Bromoaniline 1-Amino-2-bromobenzene 2-Bromophenylamine o-Bromobenzenamine |
| Pronunciation | /tuːˌbroʊmoʊ.əˈnɪliːn/ |
| Identifiers | |
| CAS Number | 615-36-1 |
| Beilstein Reference | 1209226 |
| ChEBI | CHEBI:40506 |
| ChEMBL | CHEMBL205123 |
| ChemSpider | 11917 |
| DrugBank | DB02954 |
| ECHA InfoCard | 05b09cca-97ae-45f6-9b16-abe6d4092cb6 |
| EC Number | 202-125-8 |
| Gmelin Reference | 6036 |
| KEGG | C02554 |
| MeSH | D000319 |
| PubChem CID | 6941 |
| RTECS number | BX1400000 |
| UNII | G0996C096R |
| UN number | 2733 |
| Properties | |
| Chemical formula | C6H6BrN |
| Molar mass | 172.02 g/mol |
| Appearance | light yellow to brown liquid |
| Odor | ammonia-like |
| Density | 1.6 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | 1.96 |
| Vapor pressure | 0.016 mmHg (25°C) |
| Acidity (pKa) | 3.96 |
| Basicity (pKb) | 8.72 |
| Magnetic susceptibility (χ) | -66.0 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.641 |
| Viscosity | 1.474 cP (25°C) |
| Dipole moment | 2.62 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | S⦵298 = ninety-three point four J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 55.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | −3220.6 kJ·mol⁻¹ |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin irritation, causes serious eye irritation, may cause an allergic skin reaction, may cause respiratory irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H301, H311, H331, H315, H319, H335 |
| Precautionary statements | Precautionary statements: P261, P280, P305+P351+P338, P405, P501 |
| NFPA 704 (fire diamond) | 2-2-0 Health=2, Flammability=2, Instability=0 |
| Flash point | 82 °C |
| Autoignition temperature | 703°F (373°C) |
| Lethal dose or concentration | LD50 oral rat 1750 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat 1750 mg/kg |
| NIOSH | BW6650000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 13°C |
| IDLH (Immediate danger) | Not established |
| Related compounds | |
| Related compounds |
Aniline 2-Chloroaniline 4-Bromoaniline 2-Iodoaniline 2-Nitroaniline 2-Aminoanisole |

