2-Bornanol: More Than a Simple Terpene Alcohol
Historical Development
Chemistry often moves forward because people stay curious about why things behave the way they do. The story of 2-Bornanol speaks to this spirit. As far back as the 19th century, natural products like camphor and borneol attracted scientists hoping to pin down what gave pine oils and herbal medicines their snap and scent. Bornanol sits close to these compounds on the chemical family tree. When researchers isolated it, often working from plants like Dryobalanops and Artemisia, they noticed its odd combination of mintiness and woody aroma. Its identification and study benefitted from advances in distillation, crystallization, and, later, chromatography. Chemists using glassware rather than today’s high-tech equipment managed to pick apart complex essential oils, forcing us to look closer at the structures of what we smell, use, and ingest. That willingness to experiment, even with what seemed mundane, opened the door to bornanol’s discovery and to an appreciation for the subtleties between one isomer and the next.
Product Overview
2-Bornanol, also known by its other names like isoborneol or norcamphol, carves out a distinct place in both research and industry. As a bicyclic alcohol, it brings a unique structure – part rigid, part flexible – and this makes a big difference in how it behaves. Its crisp scent lands 2-Bornanol in the world of flavorings and perfumery, but technical applications quickly came to the fore. This isn’t just another chemical stacked on a warehouse shelf; it reflects decades of study and evolving standards from how it gets extracted to where it gets used. Its lingering presence in old herbal remedies hints at a broader significance, not only in commerce but also in cultural and scientific heritage.
Physical & Chemical Properties
If you pick up a pure sample of 2-Bornanol, it usually appears as white crystalline flakes or granules, though the form can vary depending on storage conditions. With a melting point hovering close to 212 degrees Fahrenheit, it stays solid unless heated, and its boiling point comes up quickly owing to its volatility. That sharp, somewhat camphoraceous odor that strikes the nose comes from a double ring system that keeps the molecule rigid, a trait that sets it apart from simpler alcohols. Its solubility in water falls on the low side, but it dissolves nicely in ethanol, diethyl ether, and most organic solvents, enabling it to blend into a wide range of products. Chemists keep close tabs on optical activity too, as the molecule’s handedness shapes both its reactivity and sensory profile. These basic facts matter for more than trivia; labs rely on them for everything from quality control to handling precautions.
Technical Specifications & Labeling
In a lab or manufacturing setting, there’s little room for guessing games. Purity remains one of the biggest factors. Most industrial sources aim for purity upwards of 98 percent, using precise measurement protocols, GC-MS analysis, and batch certifications that tie back to lot numbers. Accurate labels not only specify chemical identity but often include melting and boiling points, molecular weight, and detailed hazard statements. Regulatory compliance with standards such as REACH in Europe and TSCA in the United States underscores the push for transparency and safety throughout the handling process. This helps suppliers, buyers, and end-users trace product quality from raw source to finished blend, reducing the risk of unexpected reactions or contamination in downstream applications.
Preparation Method
Making 2-Bornanol generally tracks back to two classic strategies. The first originates from reducing camphor with sodium and alcohol or a similar metal-hydride process, leading to a mixture of borneol and isoborneol, with separation achieved through careful crystallization or distillation. The second, more modern, route often starts with turpentine oil chemistry, which manipulates pinene through rearrangement and successive functionalization to yield 2-Bornanol. Green chemistry approaches enter the picture as environmental scrutiny grows; catalysis using reusable metal complexes, or even engineered microbial pathways, show up in recent research. No single pathway dominates, but the choice hinges on cost, scale, and environmental impact. The variability reminds us that chemical manufacturing, like cooking, depends as much on tradition as on technical innovation.
Chemical Reactions & Modifications
Bornanol’s reactivity owes much to its secondary alcohol group and the locked-in structure of its two rings. Standard oxidation transforms it into camphor, a chemical shunt that’s both academically and commercially valuable. Esterification, where the alcohol function gets swapped out for an ester group, broadens its utility in fragrances. Substitutions and rearrangements can generate a wide palette of derivatives, some of which step into pharmaceutical roles. Hydrogen bonding and steric hindrance play major roles in dictating which transformation works best, highlighting the tight link between physical structure and chemical behavior. Researchers benefit from this flexibility, adapting bornanol’s core for new catalysts, chiral auxiliaries, and synthetic intermediates.
Synonyms & Product Names
The world rarely sticks to one label for a compound that keeps showing up everywhere. Along with “2-Bornanol,” chemical catalogs and academic papers refer to it as “endo-2-bornanol,” “isoborneol,” or sometimes just “norcamphol.” In China, “Isoborneol” often features in both technical and folk medicine circles. Each name brings regional spelling quirks and historical baggage, sometimes leading to confusion. Standardization across international borders isn’t just about semantics; it cuts down on shipping errors, research mishaps, and regulatory headaches. Chemists keep cross-references handy because one missed synonym can derail whole projects or send an order to the wrong lab.
Safety & Operational Standards
Working with 2-Bornanol, safety isn’t just a regulatory buzzword – it grows out of everyday experience. Its volatility and moderate flammability put it in the same handling class as other small organic alcohols. Adequate ventilation, flame-proof storage, and personal protective gear like gloves and goggles keep risks in check. Direct exposure can irritate the skin or mucous membranes, and in rare cases, trigger sensitivities with repeated contact. Industrial settings step up checks with routine fire risk audits and training drills. Cleaning up a spill with the wrong materials or without clear protocols can turn a minor accident into a months-long headache. Following clear standards doesn’t just protect people; it prevents lost inventory, legal snarls, and expensive downtime.
Application Area
2-Bornanol threads through a surprising mix of sectors. Flavors and fragrances lean on its distinctive aroma. In this context, the human nose is the final judge: variations in purity, stereochemistry, or even storage conditions can tip a perfume from pleasant to harsh. Traditional medicine circles, especially across Asia, turn to borneol-type compounds as cooling agents in liniments and herbal remedies, though efficacy claims remain contested in medical literature. Pharmaceutical researchers investigate derivatives for anti-inflammatory and analgesic properties, with some past studies pointing toward limited bioavailability or issues crossing biological barriers. Synthetic chemists value bornanol as a chiral starting material, thanks to its rigid backbone and defined stereochemistry. Lately, its role has expanded to quirky niches, from synthesis of fine chemicals to modifiers in polymer research. Each market segment sets different requirements, underlining the importance of traceability and consistent quality.
Research & Development
Scientists see 2-Bornanol as more than a bench curiosity. Detailed NMR, MS, and chiral chromatography studies dig into its stereochemistry and interaction with biomolecules. Recent innovation focuses on greener synthesis routes, high-yield asymmetric catalysis, and biotransformation using genetically engineered micro-organisms. There’s a push to map out safe production from renewable feedstocks, bringing down waste and reducing hazardous byproducts. Computational chemistry homes in on its molecular recognition capabilities, hoping to design sensors or molecular switches for analytical and diagnostic use. The growing interest springs from the intersection of consumer demand, regulatory environment, and basic scientific novelty. Labs weigh the cost and benefit of scaling up, often bouncing between fundamental discovery and practical commercialization.
Toxicity Research
Most existing literature puts 2-Bornanol in the low-to-moderate toxicity range, though gaps remain in evaluating chronic and low-dose exposure. Acute effects tend to mimic those of related monoterpenoids: skin and mucous membrane irritation, with higher doses depressing the central nervous system in animal studies. The environmental profile stays relatively benign compared to heavy metals or persistent organics, but animal studies suggest possible aquatic toxicity at elevated concentrations. Regulatory bodies call for more targeted human exposure studies and long-term environmental fate research. For those in the industry, the lesson is clear: don’t take safety profiles for granted, and treat even familiar substances with measured respect. Ongoing research anchors future regulation and shapes how companies train their staff or inform customers.
Future Prospects
The outlook for 2-Bornanol seems tied to larger questions about sustainable sourcing, advanced manufacturing, and consumer expectations. Interest in plant-based and green chemistry approaches could tip the scales toward bio-engineered pathways that cut down on environmental impact. Pharmaceutical research, especially on pain relief and cognitive function, might uncover new uses, but this depends on rigorous clinical trials and tighter regulatory oversight. Markets for fine chemicals, specialty flavors, and high-end cosmetics demand ever-more consistent purity and traceability, pushing innovation in analytical protocols and quality assurance. The broader chemical industry’s pivot away from petrochemical feedstocks opens a window for new research on scaling up biogenic synthesis. Long-term, those willing to invest in thoughtful innovation, robust safety protocols, and transparent communication with regulators and customers will shape where bornanol fits in the coming decades.
What is 2-Bornanol used for?
Breaking Down the Role of 2-Bornanol
Plenty of people have never heard of 2-bornanol, even though its story stretches across several industries. Chemists know it for its place among terpenoids, a family of naturally occurring molecules. Most folks would never give it a thought unless they work in a lab, but its impact still reaches further than we might expect.
A Close Companion in Organic Chemistry
I remember early days in the lab, green gloves, and a bench crowded with reagents. We often ran across 2-bornanol while learning about reductions and syntheses. This compound stands out because its structure—a secondary alcohol that’s rigid in shape—makes it a handy model for teaching stereochemistry. It’s not just a demo tool, though. Universities and research outfits count on it for synthesizing new compounds and figuring out reaction pathways.
Perfumes and Fragrances: Not Just for the Lab
Moving away from glassware, 2-bornanol’s crisp, camphor-like scent catches the interest of those in the fragrance world. It’s not as famous as linalool or geraniol, but perfumers value its clean aroma. Companies blend it into colognes and air fresheners because it freshens up the base, often giving body to pine or herbal notes. That has become a bit more important as the market shifts to green chemistry—people want sustainable alternatives, and terpenoids like 2-bornanol come from renewable plant sources.
Pharmaceutical Roots and Health Applications
2-Bornanol doesn’t fill pharmacy shelves on its own. Instead, its importance grows in the background. Pharmaceutical researchers prize it as a building block. They transform it into active molecules for medicine testing and drug development. Its sturdy structure means it works well for making derivatives—small changes can turn it into compounds the body accepts more easily. The rise of green chemistry stresses the need for safer, plant-based starting materials in medicine production. More companies, especially those focused on sustainability, have started to pay attention to compounds like bornanol.
Natural Pesticides and Eco-Friendly Cleaners
Anyone who’s dealt with garden pests or stubborn grime knows the need for effective yet gentle chemicals. 2-Bornanol doesn’t headline household cleaning brands, but formulations sometimes include it because of its antimicrobial traits. Similar terpenoid alcohols show promise as plant-based pesticides and surface disinfectants. During my own research into natural pest control, I saw labs testing bornanol-containing extracts against crop fungi and bacteria. These approaches could lower reliance on petroleum-based chemicals and benefit the shift to greener agriculture.
Hurdles and Hope in Industry Adoption
Despite these uses, not every company adopts 2-bornanol quickly. Its sourcing can get pricey, and large-scale extraction needs careful planning to stay eco-friendly. Research into better synthesis methods, including biotechnological routes, could make access wider and greener. Encouragement from environmental policies might spur more investment in sustainable terpenoid chemistry, which could boost future applications across pharmaceuticals, scents, and eco-products.
Looking Forward
2-Bornanol might never become a household name. Still, its versatility touches many parts of daily life, from the scent on a fresh towel to the base of future medicines. Keeping an eye on sustainable ways to make and use compounds like this can bring safer, more effective products to shelves and labs alike.
What is the chemical structure of 2-Bornanol?
A Closer Look at 2-Bornanol
Standing in a classroom with a model kit in hand, many chemistry students wrestle with organic molecules that blur and blend together. 2-Bornanol comes from a class of compounds known as bicyclic alcohols. From personal experience, visualizing these structures isn’t just an academic exercise—it builds a foundation for grasping more complex biochemical transformations later on.
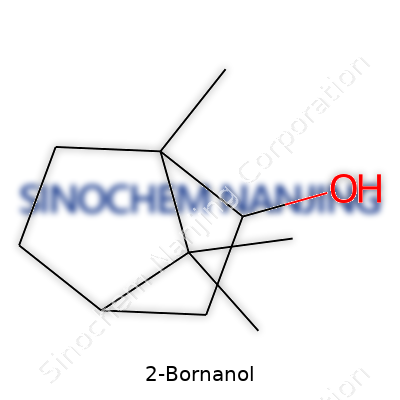
2-Bornanol, with a molecular formula of C10H18O, draws a lot of curiosity thanks to its quirky, camphor-like structure. At the center, you get a fused ring system typical of the bicyclo[2.2.1]heptane (norbornane) motif. Attaching an -OH group at the number two carbon (hence the name) gives this compound its alcohol character.
Why Its Structure Matters
Instead of seeing organic chemistry as a parade of memorization, I've found that recognizing functional groups and backbone structures changes the whole learning process. In 2-Bornanol, there’s something satisfying about tracing the carbon skeleton, seeing both rings wrap around each other, and spotting the hydroxyl group jutting out from the bridgehead carbon. Those bonds and bridgeheads tell us plenty about the molecule’s stability, reactivity, and uses.
Unlike straight-chain alcohols that dissolve willingly in water and mix into everyday solvents, bornanol’s structure keeps it relatively hydrophobic. Industries often use 2-Bornanol in fragrances and flavors, banking on its woody, camphorous smell. That distinctive aroma connects back to the chemical backbone, showing how shape defines function even in our daily experiences.
Putting the Structure to Use
The physical arrangement limits what you can do with the molecule in the lab, making some reactions trickier. For instance, oxidation of 2-Bornanol leads not to an open-chain aldehyde or ketone, but to camphor—a product with real-world usefulness in medicine and aromatics. People have used camphor in balms and ointments for long stretches of human history, and converting between these molecules highlights the value of understanding that skeleton.
Plenty of research dives into how the structure of molecules like 2-Bornanol underpins their antimicrobial or biological properties. One study I came across explored how such compounds interact with cell membranes, noting that their rigidity and bulky shape influence both their mobility and their ability to disrupt microbial life. These findings open doors for safer disinfectants or new approaches in perfumery and medicinal chemistry.
Where Deeper Knowledge Leads
Getting comfortable with these building blocks pays dividends for anyone heading into drug discovery, food science, or environmental chemistry. For students, seeing how a unique carbon frame transforms the properties of a molecule encourages curiosity instead of rote learning. Rather than drifting into chemical jargon, the focus narrows on how structure and smell, structure and solubility, structure and biological action all weave together.
Studying 2-Bornanol connects abstract textbook chemistry to the tangible—whether that means a wintergreen aroma in a lip balm, a note in a fine fragrance, or a starting point for a new medicine. It’s a simple illustration of how paying close attention to molecular detail builds both academic know-how and practical tools for everyday life.
How should 2-Bornanol be stored?
Understanding What You’re Dealing With
2-Bornanol isn’t something most people keep next to their olive oil. It’s a solid alcohol with camphor-like roots, often tucked away by chemists and folks who work with scents, flavors or specialty organics. Its low toxicity draws less worry than some nastier chemicals, but respect remains essential since it can still irritate skin, eyes, and the respiratory tract.
Avoiding Unwanted Surprises
You’ll see 2-Bornanol described as flammable and incompatible with strong acids and oxidizers. Even though these reactions aren’t explosive in a Michael Bay movie kind of way, careless exposure still invites fires or chemical accidents. It’s not about fearmongering; it’s about clear-headed respect for what could happen if things go sideways. Over the years, I’ve seen people stash volatile materials in closets with rags and cleaning fluids—which nearly guarantees trouble unless you’ve got a separate, ventilated spot built for that job.
The Common-Sense Storage Basics
Dry, cool, and well-ventilated conditions protect this material and everyone around it. Moisture in the air can mess with organics, leading to degradation. Warm rooms may seem harmless on a mild day, but a poorly stored chemical next to a radiator, for instance, can shift from safe to dangerous with a few degrees’ change. I once worked with a group that turned a seldom-used cabinet into an accidental sauna, which led to corroded labels and half-sealed jars—no one wants a repeat of that kind of avoidable mess.
Avoid storing it near careless heat sources like windows, radiators, or sunlit car trunks. Locked cabinets with minimal fuss, situated far from anything that sparks, is good practice. Use a dedicated label system—ideally with clear dates of receipt and first use. If you operate in a building with other chemicals, keep strong acids, oxidizers, and anything reactive in separate spots.
Packaging and Containers Matter
I’ve seen the difference a sturdy glass container makes versus a plastic one that warps if solvents wiggle their way in. Glass with tight-fitting lids prevents evaporation. Stopper or tightly capped vials offer decent protection, but double-check that all seals and closures can withstand the stuff inside—bornanol won’t leak through metal but can soften certain plastics over months. Clear labeling on the container is just as important as where you put it, especially once the original packaging disappears after resupply or transfers.
Keeping an Eye on Inventory
Routine checks on storerooms pay off. Labels fade, jars shift. You don’t want “mystery bottles” haunting your shelves long after the person who put them there has moved on. A logbook or spreadsheet helps keep track: I’ve relied on these for years because memory slips and teams change. Spotting an empty or “almost empty” container also cuts down on scrambling for more in the middle of a project.
Personal Protective Equipment Isn’t Optional
Before grabbing a jar, slip on gloves and safety goggles. Even for transfers and weighing, a dust mask or light respirator cuts down on accidental inhalation. I’ve learned not to skip these habits after observing someone once rinse a chemical burn off their hand. The memory sticks with you and reminds you how routine safeguards beat lucky escapes every time.
Seeking Safer, Smarter Storage
If you’re working with larger amounts or shared spaces, invest in flame-retardant cabinets designed for chemicals. These cost far less than medical bills or ruined equipment. For smaller labs or home setups, a lockable metal locker away from high-traffic spots still offers solid protection. Regular audits keep you honest, and a bit of care upfront eliminates most disasters before they start.
Door signs, good lighting, and clear access to Material Safety Data Sheets add extra layers of security. These precautions make you a good neighbor and a more reliable scientist—both outcomes worth aiming for, no matter the size of your project.
Is 2-Bornanol hazardous or toxic?
Understanding 2-Bornanol in the Real World
2-Bornanol comes from the family of secondary alcohols. Anyone who has taken a college chemistry class might remember making it in a lab, or catching a whiff of its pine-like scent. Industries use 2-Bornanol for making flavors, fragrances, and certain medicines. It might sound technical, but it pops up in places you might not expect—think mouthwash or cough syrup.
What Makes a Chemical Hazardous?
People worry about chemicals, sometimes for good reason. Safety questions around 2-Bornanol usually boil down to two things: is it toxic if you touch or breathe in a bit, and will it hurt you long-term? According to the National Center for Biotechnology Information (NCBI) and the PubChem database, 2-Bornanol shows low acute toxicity. That means it doesn’t generally cause immediate harm from short-term exposure. Most of the time, irritations—like itchy eyes, coughing after breathing it in, or a rash—show up from heavier exposure. Over years working in laboratories, I've handled pure 2-Bornanol with basic gloves and goggles, and never saw colleagues get more than the odd skin irritation or headache if someone spilled some on their lab coat.
Looking at the Science
Data from animal studies in the US National Toxicology Program gives more context. Mice and rats exposed to relatively high levels by mouth or inhalation showed mild symptoms—things like minor liver swelling or stomach upset. The quantities used in these studies far outweigh what anyone would usually encounter: a far cry from what’s in a consumer product. Regulators in the US, Europe, and Japan don’t consider 2-Bornanol a major toxic threat at levels people typically experience. It’s not in the same league as lead, benzene, or formaldehyde—common household hazards with much stronger evidence for long-term health risks. For those working with bornanol every day, the risk might be higher, and that’s why chemical handling rules exist.
Everyday Exposure
Concerns sometimes come from unfamiliarity. Shop shelves brim with cleaning products, gum, and cheap perfumes, and consumers scan ingredient lists for anything unpronounceable. If 2-Bornanol is there at all, it usually sits in tiny concentrations. Sitting behind a pharmacy counter, I’d get questions from cautious parents. Here’s the reality: in the small doses found in flavorings and fragrances, 2-Bornanol passes the safety bar set by government agencies. You wouldn’t want to guzzle a jar of the pure compound, but a trace in toothpaste won’t put your health in danger.
Making Things Safer: What’s Worth Watching
Good practice makes the difference. In manufacturing, workers must wear gloves, eye protection, and sometimes vapor masks. If you spill some on your hands, a good wash does the trick. Spills on clothing should prompt a quick change, and ventilation matters in small labs where vapor could build up. For consumers, the risks drop off—there’s more ethanol in mouthwash than 2-Bornanol, and most people don’t sweat over a bit of aftershave.
To cut risk, scientists keep testing even familiar chemicals. Data review, worker education, and transparent labeling help. If someone has a chemical allergy, they should read ingredient lists or ask a pharmacist. For nearly everyone else, 2-Bornanol is more about piney flavors and perfumed soaps than hazard signs or poison labels.
Trust, Transparency, and Health
Public trust comes from honest communication about risks and benefits of everyday chemicals. Regulators, researchers, and producers carry the responsibility—posting data, responding to medical claims, and updating guidelines as new studies appear. Of course, anyone handling concentrated chemicals should keep it safe—nobody wins by cutting corners. At the same time, the weight of evidence shows that, in real-world use, toxic worries about 2-Bornanol miss the mark for ordinary folks.
What is the molecular formula and weight of 2-Bornanol?
Molecular Formula and Weight of 2-Bornanol
Anyone interested in chemistry soon comes across compounds like 2-Bornanol. Not many people give molecular formulas much thought, but that string of letters and numbers is more than just jargon for scientists. For 2-Bornanol, that formula is C10H18O. This detail pinpoints its place in the family tree of organic molecules. Knowing that formula helps chemists, pharmacists, and even folks in the flavor industry recognize what they’re handling. The molecular weight of 2-Bornanol is about 154.25 g/mol. Numbers like these aren’t just trivia—they decide how this substance behaves in your flask or even, potentially, in a consumer product.
Why Structure and Weight Matter in Chemistry
Structurally, 2-Bornanol belongs to the class of bicyclic alcohols, specifically it’s a derivative of camphane. The arrangement of its carbon atoms, hydrogens, and that single oxygen atom isn’t just a technicality. It shapes how 2-Bornanol smells, how it tastes, how it reacts in making other chemicals. A close cousin of borneol, 2-Bornanol also goes by the name "endo-2-Bornanol." Tiny changes in structure push big differences in reaction and use. In my own academic experience, checking the molecular weight was a basic but crucial step before mixing or scaling up a reaction. Getting that wrong means wasted time, resources, and sometimes riskier lab conditions.
Real-World Relevance and Industrial Connections
It’s easy to overlook compounds that don’t make headline news, but 2-Bornanol turns up in more places than you’d guess. In research, it’s used to study stereochemistry and various organic reactions. This molecule has appeared in essential oil studies, and it sparks curiosity for anyone interested in sustainable or bio-based chemistry. With the chemical industry regularly looking for renewable alternatives, compounds like 2-Bornanol—sourced from natural oils or built in a lab—become important benchmarks. In pharmaceutical circles, similar molecules have shown promise as components for medications or active ingredients.
Supporting Facts and Scientific Reliability
Reliable data comes from published articles, safety data sheets, and chemical catalogs. For 2-Bornanol, values for molecular weight and formula remain consistent in trusted sources like PubChem and the Merck Index. These resources give scientists a starting point to check purity, plan syntheses, and ensure safety. Without facts grounded in strong evidence, errors in the lab can become costly or dangerous. My early mistakes in the lab drove home how missing or misusing these raw numbers can derail entire projects or experiments.
Where Issues Appear, Solutions Follow
Sometimes, finding high-purity 2-Bornanol isn’t straightforward, especially outside of specialist suppliers. Researchers can face supply limits or fluctuations in price. One practical approach focuses on improved synthesis methods from camphor or pinene sources. Another looks at bioengineering microorganisms to create specific alcohols, including 2-Bornanol, in controlled settings. Investing more in bio-based pathways might cut costs and environmental footprints. Making this type of chemical accessible at scale often relies on work done by chemists who pay close attention to details—like getting the formula and molecular weight right every time. Mistakes here reverberate through a whole chain of innovation.
| Names | |
| Preferred IUPAC name | endo-1,7,7-Trimethylbicyclo[2.2.1]heptan-2-ol |
| Other names |
endo-2-Norbornanol exo-2-Hydroxybicyclo[2.2.1]heptane endo-Norborneol |
| Pronunciation | /ˈbɔːr.nə.nɒl/ |
| Identifiers | |
| CAS Number | 464-43-7 |
| Beilstein Reference | 1209287 |
| ChEBI | CHEBI:15754 |
| ChEMBL | CHEMBL504441 |
| ChemSpider | 12319 |
| DrugBank | DB01982 |
| ECHA InfoCard | 100.005.095 |
| EC Number | 1.1.1.198 |
| Gmelin Reference | 6991 |
| KEGG | C06536 |
| MeSH | D016617 |
| PubChem CID | 6446 |
| RTECS number | DD3325000 |
| UNII | YN1X2M58A2 |
| UN number | “1993” |
| Properties | |
| Chemical formula | C10H18O |
| Molar mass | 154.25 g/mol |
| Appearance | white crystalline solid |
| Odor | camphoraceous |
| Density | 0.962 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 1.84 |
| Vapor pressure | 0.02 mmHg (25 °C) |
| Acidity (pKa) | 16.3 |
| Basicity (pKb) | 7.92 |
| Magnetic susceptibility (χ) | -78.9×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.4840 |
| Viscosity | 7.62 cP (20°C) |
| Dipole moment | 1.66 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 203.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | –345.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3205 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | C1C(C)CC(C)(C1)O |
| Signal word | Warning |
| Hazard statements | H319: Causes serious eye irritation. |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P264, P271, P280, P301+P312, P303+P361+P353, P304+P340, P305+P351+P338, P312, P337+P313, P370+P378, P403+P235, P405, P501 |
| NFPA 704 (fire diamond) | 1,2,0 |
| Flash point | 93 °C |
| Autoignition temperature | 286 °C |
| Explosive limits | Upper: 5.5%, Lower: 1.2% |
| Lethal dose or concentration | LD50 (rat, oral): 2500 mg/kg |
| LD50 (median dose) | 1860 mg/kg (rat, oral) |
| NIOSH | DF1400000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 200-223-5 |
| IDLH (Immediate danger) | Not established |
| Related compounds | |
| Related compounds |
Borneol Camphor Isoborneol Norbornane |

