2-Aminopyridine: Knowledge, Impact, and Future Potential
Following the Footsteps of 2-Aminopyridine: A Chemical’s Path Through Time
Chemistry often turns humble molecules into powerhouses of science. 2-Aminopyridine belongs to that select group. In the late 1800s, researchers exploring coal tar derivatives first pulled pyridine compounds from thick, black fractions meant for waste. Curious minds started tinkering with these new finds; once someone swapped out a hydrogen atom on pyridine’s six-membered ring, dropping in an amino group at the second position, 2-Aminopyridine emerged. Over the 20th century, interest never faded. Its simplicity soon showed a knack for adaptability, making chemists realize that small changes unlocked new properties or set the stage for even more complex molecules. My own first encounter with 2-Aminopyridine happened in an undergraduate organic synthesis lab, where I watched its reactivity unfold under conditions that would stop other molecules cold.
What Makes 2-Aminopyridine Stand Out?
2-Aminopyridine may sound unassuming, but its structure—a six-membered aromatic ring with both a nitrogen atom in the ring and an amino group sticking off one carbon—delivers a punch in both versatility and reactivity. Pure 2-Aminopyridine appears as a colorless to light yellow crystalline powder. You’ll notice a faint, fishy odor, something anyone working in an organic lab won’t forget soon. Its melting point sits near 59–61°C, reasonable enough for handling without elaborate precautions. Solubility in water runs moderate, which helps during application or isolation steps, and it dissolves well in organic solvents like ethanol and ether. As for pH, it registers basic due to both the pyridine nitrogen and the amino group, which can make purification trickier but offers more levers for chemical manipulations. The chemistry community doesn't keep such details secret; they get cited repeatedly in handbooks and research papers.
More Than a Name: Deciphering Identity and Synonyms
Chemists use more names for 2-Aminopyridine than most folks use for their pets. The logical system gives C5H6N2 as its formula, or “2-pyridinylamine” when following strict rules. Trade and lab slang often call it “2-AP,” “Pyridin-2-amine," or simply “aminopyridine” in informal settings. Each label reveals who’s in the conversation: synthetic chemists, pharmaceutical scientists, or regulatory folks. Recognizing all these terms isn’t a matter of rote memorization; it lets people track down critical safety and research details hidden under alternate monikers.
Peering Into Technical Specs and Labeling
Strict guidelines shape how 2-Aminopyridine gets handled or sold, driven by both safety and the demands of downstream users. Chemists care about purity. Trace metal levels and sulfonate impurities should fall under strict tolerances. Technical sheets spell out these numbers, listing melting points, solubility ranges, and spectral data such as NMR and IR frequencies. Packaging labels typically identify the chemical unequivocally, providing hazard pictograms and risk statements. Those labels aren’t mere formalities—one mix-up in a supply room or on a delivery truck can cause both safety and research disasters.
How Chemists Prepare and Modify 2-Aminopyridine
The drive to make 2-Aminopyridine starts with pyridine itself. Nitration at the second position, followed by catalytic reduction, transforms pyridine’s ring into the sought-after amino derivative. Some industrial setups prefer direct amination routes, swapping halogen atoms on 2-halopyridines with ammonia under pressure. Laboratory preparation gets a boost from modern transition metal catalysts. I recall navigating batch synthesis with palladium-on-carbon, watching the color shift as reduction completed. University labs often teach these methods not because they’re the only ways in town, but because they set the stage for understanding broader concepts of aromatic substitution and reduction.
Reactive Behavior Unleashes Utility
Handing 2-Aminopyridine a reagent almost always triggers drama. The amino group, eager to donate electrons, encourages coupling reactions and ring modifications. Medicinal chemists seize on this for heterocyclic scaffold construction. Multicomponent reactions allow chemists to assemble more complex molecules faster, saving both effort and material costs. Its ring nitrogen exerts a weaker, more subtle influence—sometimes quietly coordinating with metal catalysts or shifting the electron density to nudge selectivity in favor of specific products. Cross-coupling approaches, like Suzuki and Buchwald–Hartwig reactions, extend what can be built from a single batch of 2-Aminopyridine. This isn’t just academic fun; it lets pharma researchers generate drug-like molecules at scales useful for testing—and eventual production.
Staying Safe: Hazard Awareness and Operational Standards
You can’t talk about amines in the workplace without touching on their safety profiles, and 2-Aminopyridine walks a narrow line. Acute toxicity levels raise concern, especially if inhaled or ingested. It gets classified as harmful; chronic exposure may affect the nervous system. Personal protective equipment like gloves and lab coats aren’t optional—they form the baseline. Fume hoods become mandatory, not luxury, during open transfers or synthesis runs. Waste must follow regulated disposal pipelines, preventing both human and environmental exposure. Experienced technicians treat every open container with the respect usually reserved for much larger hazards, precisely because years of incident reports have hammered home the dangers that arise from casual shortcuts.
From Synthesis Bench to Industry Applications
2-Aminopyridine carves out major roles in pharmaceuticals, agriculture, and specialty chemical manufacture. Drug developers find it irreplaceable in creating angina medications and older antitubercular agents. The agrochemical arena uses it to generate herbicides and fungicides targeting specific pests without heavy collateral damage. Material scientists tweak its structure to fashion corrosion inhibitors and complexing agents, boosting performance in water-treatment or oil-extraction fields. In my own work, it often serves as a linchpin for assembling research tools: fluorescent probes, enzyme inhibitors, or even sensor elements. Not every molecule manages such a wide range, yet 2-Aminopyridine adapts with enviable flexibility.
Frontiers and Current Work: Research and Development Push Forward
Research enthusiasm shows no sign of cooling. Scientists keep challenging boundaries by modifying both the amino group and the ring system. Modern synthesis teams, wielding both computational tools and experimental setups, rapidly design and test novel derivatives for potential as new drugs or functional materials. Studies focusing on pharmacokinetics and receptor interactions uncover possible uses against increasingly tough microbial strains. Green chemistry advocates explore more sustainable preparation routes, either recycling catalysts or switching to renewable feedstocks. In multidisciplinary teams, 2-Aminopyridine helps bridge gaps between medicinal chemistry, biochemistry, and environmental engineering. Every research leap forward often springs from the same core molecular scaffold, proving there’s life in even the oldest of organic molecules.
Toxicity Studies: Understanding and Mitigating Risks
Toxicologists don’t take shortcuts evaluating 2-Aminopyridine’s dangers. Animal studies have driven regulatory changes, flagging risks tied to neurological symptoms after sustained exposure. Occupational monitoring highlights the need for air controls and emergency protocols; exposure beyond safety limits can prompt swift headaches, dizziness, or worse. Chronic effects data, while not as deep as some other compounds, nonetheless suggest caution for prolonged contact. Recent years have pushed for more rigorous environmental fate studies, as runoff and waste streams from factories could introduce 2-Aminopyridine or its byproducts into waterways. Advances in detection—high-performance liquid chromatography and mass spectrometry in particular—let labs rapidly catch contamination before it spreads. Regulatory bodies tend to listen when multiple streams of data point to real, rather than theoretical, hazards.
Looking Ahead: Where 2-Aminopyridine Goes Next
Scientific curiosity tends not to let useful, reactive molecules gather dust. Researchers eye new frontiers for 2-Aminopyridine that move far beyond its early days. Pharmaceutical innovation depends on fresh analogues to counter drug resistance and reach new therapeutic targets. Advanced materials research harnesses the molecule in crafting smart polymers or new generations of electronic components. Synthetic teams experiment with miniaturized flow reactors, knocking down both waste and costs for complex derivatizations. Challenges stay on the horizon—the need for safer handling, greener production, and tighter toxicity profiles remains—yet each decade peels back more untapped opportunity from this small, aromatic amine. If history’s any guide, the next innovations will build on foundations already long established in both the lab and the field.
What is 2-Aminopyridine used for?
Understanding 2-Aminopyridine in Everyday Terms
2-Aminopyridine sounds like something only scientists would care about, but it pops up in ways that touch all of us. This compound sits at the crossroads of medicine, agriculture, and even personal safety. Each of these areas reveals the practical reasons people keep working with it, despite the notorious sharp smell and tricky handling.
Pharmaceutical Value: Beyond the Basics
2-Aminopyridine serves as a building block for medicines. Drug research teams keep reaching for it because the structure unlocks so many possibilities. Pharmaceutical chemists use it to design treatments for a whole range of neurological disorders. A drug called dalfampridine, developed for multiple sclerosis, uses this compound as a core ingredient. Dalfampridine helps patients improve their walking ability. This offers hope and independence to people who often face daily struggles simply moving around.
Other medicines in the anti-viral and anti-cancer worlds draw from 2-Aminopyridine too. Research journals highlight this compound in newer generations of painkillers as well. The reason goes back to the way its atoms fit together. By swapping one group for another, chemists can “tune” its activity inside the body. Unlike some precursors, this ingredient gives researchers flexibility to create compounds that battle viruses, stubborn infections, or even tumors.
Agricultural and Chemical Uses: More Than Meets the Eye
Farms and gardens get an indirect boost from this molecule. It shows up in the blueprints for certain herbicides, which farmers use to fight weeds that sap crops’ strength. Insecticides, which keep pests in check, owe some of their punch to 2-Aminopyridine. This keeps foods growing strong and healthy, which every shopper depends on—even if most of us never learn the chemistry.
Industrial chemists make use of 2-Aminopyridine as an “intermediate”—a handy stop on the way to much bigger molecules. It helps form dyes, corrosion inhibitors, and specialty chemicals found in processes from rubber manufacturing to circuit board assembly. Its chemical behavior allows quick attachment to other ingredients, making it a favorite in labs that demand speed and precision.
Safety and Environmental Questions
Handling this compound calls for skill and caution. Workers who deal with it up close wear gloves and eye protection. Prolonged exposure or careless handling can cause headaches, skin irritation, or worse. Regulations keep tight control on its manufacture and shipping. From experience working with chemical inventories, I always noticed every bottle came labeled with prominent warnings—and for good reason. Out in the world, small slip-ups can ripple out and cause harm.
On the environmental side, people working with it limit waste and water runoff. Modern safety data sheets urge companies to contain, process, and dispose of any leftovers responsibly. Industry experts design handling plans to avoid neighborhood or wildlife exposure.
What Keeps the Industry Moving Forward
The challenge remains: how can we gain all these benefits while cutting out the risks? Researchers look for safer derivatives or improved production methods every year. Some labs have already adopted greener chemistry recipes, looking to cut down on toxic byproducts without sacrificing the efficiency that 2-Aminopyridine provides.
With new medical breakthroughs ahead and a growing focus on environmental care, 2-Aminopyridine’s story keeps evolving. Chemists and manufacturers push ahead—not just for the sake of chemistry, but to make sure progress lines up with safety and everyday need.
What is the CAS number of 2-Aminopyridine?
Why Chemical Identification Matters
Every time I walk down the aisle of a laboratory supply store, I see a parade of bottles labeled with complicated names and mysterious numbers. That number isn’t just a code. It’s the key to tracking safety data, sourcing high-quality material, and avoiding expensive mistakes. For 2-Aminopyridine, that number is 504-29-0, and it’s worth knowing why this simple string of digits can make or break a research project or industrial process.
CAS Numbers: Not Just a Form of Bureaucracy
The Chemical Abstracts Service (CAS) created a registry that sorts substances with a unique identifier. Every compound, including 2-Aminopyridine, gets its own CAS number. I remember hunting through catalogs, searching for a reliable supplier. Without a CAS number, I ended up with products that didn’t match what I needed. The CAS number protects buyers, chemists, and manufacturers. Mistaking an isomer or a similar compound isn’t only inconvenient; sometimes it’s dangerous.
2-Aminopyridine in the Lab and the Real World
2-Aminopyridine isn’t just a name you’d find in a dusty textbook. It’s a building block for pharmaceuticals, dyes, and pesticides. Its correct identification is vital in medical research, where a small mix-up could alter test results and affect patient safety. I’ve talked with synthetic chemists who rely on the certainty that a bottle labeled CAS 504-29-0 will behave as expected. Real money, time, and sometimes lives ride on that confidence.
Addressing Common Issues
One issue I’ve run across is suppliers substituting different grades or even different chemicals when orders ignore CAS numbers. It’s tempting to shop by price or brand, but that opens the door for errors. Fake products are a growing concern as globally sourced materials flood the market. Last year, an incident involving contaminated 2-Aminopyridine caused delays at a contract lab. An in-depth investigation traced the cause back to a missing CAS number in the procurement paperwork.
Supporting Transparency and Trust
The European Union, United States, and many other countries hold strict rules around chemical traceability. CAS numbers make this possible. In my own experience, quality documentation only means something if it can be checked and verified. A missing or mixed-up number leads to paperwork disasters when inspections come around. Clear labeling and procurement standards build trust not only in regulatory agencies, but with business partners and customers as well.
Looking for Solutions
Training helps. Teams who learn to check CAS numbers from the start avoid major headaches later. Digital inventory systems that link every bottle to its unique CAS entry cut down on human error. Companies placing QR codes on containers tied to reputable databases offer another layer of defense against counterfeits.
More suppliers joining programs that verify product identity, purity, and chain-of-custody means fewer problems reaching end users. It’s not just about compliance. For everyone who works with compounds like 2-Aminopyridine, paying attention to that identification number is about making science and business a little safer, a little more certain, and a lot more trustworthy.
Is 2-Aminopyridine hazardous or toxic?
What is 2-Aminopyridine?
2-Aminopyridine shows up as a chemical with some pretty far-reaching uses. It acts as a building block for medicines, dyes, and even pesticides. In many labs, folks use it almost every day. The thing about chemicals that work this hard—they often come with some hazards.
Hazards of 2-Aminopyridine
Anyone who’s spent time around chemicals knows the cardinal rule—read the safety data. With 2-Aminopyridine, it’s more than red tape. Exposure can harm your skin, eyes, or lungs. Inhaling the dust or vapors can trigger coughing, sore throat, or even headaches. If the skin or eyes meet this stuff, irritation doesn’t take long. At higher doses, the problems get bigger. Serious cases might bring on nausea, confusion, or convulsions. The risks aren’t just from breathing it in; swallowing just a bit could land you in the emergency room.
Long-Term Effects and Evidence
The science here deserves attention. Animal studies raise red flags around nerve symptoms and potentially harmful effects to organs when exposure gets out of hand. There’s no large body of evidence tying 2-Aminopyridine to cancer in humans, but there’s not much to guarantee it’s harmless, either. Medical workers and chemical handlers need to keep that in mind.
Handling: Precautions Matter
Old hands in the chemical trade will always slap on the gloves, goggles, and a lab coat before opening a bottle of 2-Aminopyridine. Good ventilation cuts down on fumes. Spills call for immediate cleanup, no shortcuts. Some companies keep eyewash stations and emergency showers close by for a reason.
Labeling and storage count for a lot. Tight lids, sturdy containers, and clear warnings keep workplace accidents from becoming news stories. I’ve seen what happens when someone gets careless with a toxic chemical—at the very least, they lose a day at work, sometimes more. Chronic exposure often flies under the radar; that’s where the real danger lingers.
The Bigger Picture
In industrial settings, risk comes with the territory, but education draws the line between caution and disaster. Workers given proper training on 2-Aminopyridine stay healthier. I remember a coworker who once ignored the safety sheet—he ended up with a nasty rash and months of paperwork. Companies paying attention to worker health also reduce costly accidents.
Laws in the U.S., Europe, and Asia treat 2-Aminopyridine as hazardous. Agencies like OSHA insist on protective equipment. It isn’t just about ticking boxes—these standards grew out of real injuries. Following them never feels like overkill, especially after I’ve watched the after-effects of chemical mishaps.
Looking Ahead
Science finds safer options all the time, but 2-Aminopyridine remains necessary for many processes. Until replacements prove their worth, tight rules and strong habits offer the best defense. A bit of respect, a lot of common sense, and updated information make a difference. That’s how you keep a useful chemical from turning into a real problem—for you and everyone down the line.
What is the molecular formula of 2-Aminopyridine?
What Makes 2-Aminopyridine Special?
2-Aminopyridine grabs the attention of scientists and industry workers alike because this little molecule packs more punch than its size suggests. Found in laboratories and specialty chemical plants, 2-Aminopyridine serves as a building block for things you rarely notice but use every day—think pharmaceuticals, dyes, and corrosion inhibitors. Those who work in analytical chemistry will remember the first time they studied this compound; for me, it was during a late-night prep for a practical exam, where recognizing the structure proved more valuable than cramming hundreds of obscure compounds. The structure is straightforward—imagine a six-membered ring with one nitrogen atom and an amino group sitting next to it. That's why the molecular formula comes up so often in basic organic chemistry quizzes.
The Simple Formula Behind Many Complex Processes
The molecular formula of 2-Aminopyridine is C5H6N2. Each molecule contains five carbon atoms, six hydrogens, and two nitrogens. You can see each element’s role in a typical skeletal formula: that ring structure doesn’t just stay on the notepad—it determines how the molecule reacts with others or how easily it dissolves. In medicinal chemistry, small changes to a molecular skeleton like this one can change a headache pill into an antihistamine. I’ve seen researchers lean on the reactivity of that amino group, tweaking conditions to make new compounds for screening. With C5H6N2, the formula itself opens doors to a cascade of applications.
Why Getting These Details Right Matters
People might shrug off memorizing molecular formulas until the day a single atom’s error throws off an entire batch’s yield. Out in industry, no one wants to see product recalls or, worse, a missed discovery because technicians misidentified a reagent. Chemical regulations call for consistency—labels must match what’s inside, everything down to molecular formulas. My own early days on the pilot plant floor taught me how fast a misplaced digit turns a routine reaction into an investigation, wasting time and money. For 2-Aminopyridine, clarity and accuracy keep everyone, from researchers to regulators, moving in the right direction.
Addressing Common Challenges
Many who handle 2-Aminopyridine will tell you the same story: even small mistakes in naming or formula entry can lead to monumental headaches. Chemists need software checks, peer reviews, and good note-keeping habits, because no process is too basic to mess up. One regular practice that helped my team involved double-checking inventory during shift changes—not glamorous, but a real time saver. Documenting lab procedures in a way that makes sense—not just for compliance, but for the next person picking up the project—reduces error rates and ramps up productivity.
Moving Beyond the Formula
Not every compound with C5H6N2 etched into its datasheet will stand out on the shelf. Knowing its properties and uses still depends on understanding the core structure. Focusing on practical steps—like investing in hands-on training, reviewing safety protocols, and keeping lab documentation clear—safeguards both research and industrial operations. Chemical knowledge always circles back to the basics: the right structure, the right formula. And once you’ve seen how often accuracy pays off, you start looking at molecules with a whole different kind of respect.
How should 2-Aminopyridine be stored?
Pushing Safety to the Top of the List
2-Aminopyridine doesn’t take up much space in a storage room, but ignoring its quirks can spell trouble. It’s tempting to toss bottles on a high shelf and move on, but certain chemicals play by their own rules. This particular one sits in many research labs and pharmaceutical facilities. In practice, one lesson stands out — rely on strict organization or pay the price. Lab safety manuals drill this point home for a reason: even a few grams of a reactive compound can turn a workday upside down.
Understanding Its Personality
Experience teaches that 2-Aminopyridine loves dry, cool environments. This stuff doesn’t appreciate moisture, and it gets along poorly with heat or exposed light. Left out near a radiator in a humid room, you risk changes to purity, or, even worse, a slow build-up of chemical changes that can creep up undetected. A neighbor once cut corners by leaving bottles uncapped for “just a few minutes.” Two weeks later, the label was unreadable and crystals formed on the threads. Small mistakes compound quickly.
Coming Face-to-Face with Hazards
Getting familiar with potential dangers helps put things into perspective. 2-Aminopyridine isn’t volatile like some solvents, but repeated exposure can cause trouble. Skin irritation, breathing issues, and headaches aren’t just lines in a catalog—they can show up with poor handling. Medical journals document why it requires attention: once you inhale its dust or let it seep through gloves, you’re left with long-term health risks.
Stories from the industrial side show careless storage can lead to accidental mixing with oxidizers or acids. A university near my hometown faced a costly evacuation from a mislabeled shelf: 2-Aminopyridine crossed paths with a mild acid, and cleanup crews spent hours ventilating the lab. Every safety data sheet highlights the need to keep incompatible materials apart for a reason.
Steps that Actually Make a Difference
Storing 2-Aminopyridine properly isn’t about fancy technology. Workers and students alike learn that a tight-sealing amber glass bottle blocks out light and moisture. Keeping containers locked up—preferably in a dedicated chemical cabinet, away from heating pipes or open sunlight—goes a long way. Desiccants inside the cabinet can work wonders, especially in humid climates or older buildings. In my own workplace, a simple habit of labeling dates and rotating stock keeps the oldest bottles up front, avoiding mystery residues or forgotten spills at the back.
Practical Solutions and Lasting Habits
Building strong habits means fewer accidents, less waste, and smoother inspections. Regular inventory checks and logging storage practices help everyone stay on the same page. Emergency supplies, like spill kits and extra gloves, offer backup when things don’t go as planned. For folks overseeing high-volume labs, investing in online training and quick-access safety charts sharpens awareness. Even at a small scale, a weekly walk-through with eyes on proper sealing and separation makes a bigger difference than any rulebook alone.
Taking these basic steps signals commitment—both to health and to the integrity of research. Experience, not just rules, shapes safe workspaces. People grow more confident around chemicals when safety routines become second nature. No one likes the sting of preventable mistakes when the answer sat one cabinet away the whole time.
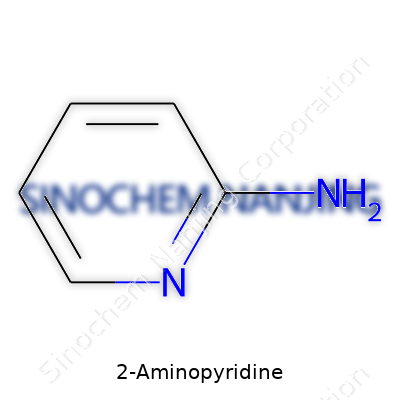
| Names | |
| Preferred IUPAC name | Pyridin-2-amine |
| Other names |
2-Pyridinamine 2-Pyridylamine |
| Pronunciation | /tuː əˌmiːnoʊ pɪˈrɪdiːn/ |
| Identifiers | |
| CAS Number | 504-29-0 |
| Beilstein Reference | 98617 |
| ChEBI | CHEBI:18035 |
| ChEMBL | CHEMBL1406 |
| ChemSpider | 597 |
| DrugBank | DB00206 |
| ECHA InfoCard | 0353-002-00-2 |
| EC Number | 205-503-0 |
| Gmelin Reference | 7878 |
| KEGG | C00366 |
| MeSH | D010375 |
| PubChem CID | 7811 |
| RTECS number | UG3675000 |
| UNII | 5JW8554L3P |
| UN number | UN2671 |
| Properties | |
| Chemical formula | C5H6N2 |
| Molar mass | 94.12 g/mol |
| Appearance | White to pale yellow solid |
| Odor | amine-like |
| Density | 1.09 g/cm³ |
| Solubility in water | Very soluble |
| log P | 0.57 |
| Vapor pressure | 0.065 mmHg (25°C) |
| Acidity (pKa) | 6.86 |
| Basicity (pKb) | 7.08 |
| Magnetic susceptibility (χ) | -46.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.571 |
| Viscosity | 3.05 mPa·s (20°C) |
| Dipole moment | 1.57 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 119.8 J∙mol⁻¹∙K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 56.2 kJ mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -2510 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | N07XX04 |
| Hazards | |
| GHS labelling | GHS07, GHS08 |
| Pictograms | GHS07 |
| Signal word | Danger |
| Hazard statements | H301: Toxic if swallowed. H311: Toxic in contact with skin. H331: Toxic if inhaled. H302: Harmful if swallowed. H315: Causes skin irritation. H319: Causes serious eye irritation. H335: May cause respiratory irritation. |
| Precautionary statements | P260, P261, P264, P270, P271, P273, P301+P312, P302+P352, P304+P340, P305+P351+P338, P311, P330, P362+P364, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-0 Health=3, Flammability=2, Instability=0 |
| Flash point | 107°C |
| Autoignition temperature | 480°C |
| Explosive limits | 1.7% - 10.2% |
| Lethal dose or concentration | LD50 oral rat 200 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 200 mg/kg |
| NIOSH | UY5600000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of 2-Aminopyridine: 0.5 ppm (2 mg/m³) |
| REL (Recommended) | 0.05 mg/m³ |
| IDLH (Immediate danger) | 150 ppm |
| Related compounds | |
| Related compounds |
2,6-Diaminopyridine 3-Aminopyridine 4-Aminopyridine Pyridine 2-Hydroxypyridine |

