2-Aminoethanol: A Down-to-Earth Look at an Unsung Chemical Workhorse
Historical Development
Long before the modern chemical industry ramped up, 2-aminoethanol, often called ethanolamine, had already found its way into basic chemistry sets in university labs. Early twentieth-century chemists started isolating it by reacting ammonia with ethylene oxide, quickly realizing that this clear, syrupy liquid was more than just a simple molecule. As chemical manufacturing grew, ethanolamine popped up on the blueprints of factories making everything from rayon to soaps. I remember paging through old chemical engineering textbooks from the 1940s and spotting 2-aminoethanol as a go-to ingredient for gas-scrubbing solutions, a testament to how industries valued its ability to grab acidic gases like carbon dioxide and hydrogen sulfide from crude mixtures. This reliance on the compound spread globally along with the growing need for synthetic cleaners, herbicides, and, later, pharmaceutical intermediates. Its adaptability secured a spot in the backbone of chemical processing—a reputation that hasn’t faded.
Product Overview
Run your finger down the shopping list for bulk chemicals, and you’ll find 2-aminoethanol not far from the top. In its pure form, it flows as a colorless, viscous liquid, smelling faintly of ammonia with a bit of that “clean” chemical edge. It easily mixes into water and alcohol, dissolving without fuss—a welcome trait for plant operators who hate wrestling with finicky compounds. Whether stabilizing emulsions in cosmetics or buffering the pH of textiles, ethanolamine slots into all sorts of roles, always ready to lend its ability to act as both a weak base and a mild solvent.
Physical and Chemical Properties
2-Aminoethanol carries the chemical formula C2H7NO, shining brighter for its dual-action personality; it packs an amine group along with a hydroxyl group. This small combo means it goes to work both as an alcohol and a weak base, making it easy to handle, but strong enough to get real work done. It boils around 170°C, so it handles moderately high heat. It also drags water molecules close, picking up moisture from the air, so storage containers need tight seals. Against acids, it forms salts, while with oxidizers, you’d better keep hands and eyes protected, as reactions can get forceful. Getting your hands on a chemical with such versatility makes me think of a trusty hand tool—simple but always useful, never flashy but indispensable all the same.
Technical Specifications & Labeling
Labels on drums of ethanolamine don’t mince words—they spell out concentration, purity, and the usual industry warnings. Nearly every reputable manufacturer grades ethanolamine for different uses, with purities topping 99% for specialty applications. Safety data reads like a caution sign in a high school lab, urging users to avoid skin contact and splashes, since it can irritate and burn. Regulatory nudges require clear hazard pictograms; the chemical’s corrosive edge means proper splash-proof gear is standard. From a practical standpoint, tight labeling smooths out logistical headaches and helps accident-proof the shop floor, which I’ve learned is a must for safety and compliance crews alike.
Preparation Method
For decades, chemical companies have followed a direct synthesis pathway to make 2-aminoethanol: react ethylene oxide (itself a useful building block) with aqueous ammonia. This reaction runs at raised temperature and pressure to boost yield and reduce waste. Due to the risk factor with ethylene oxide, equipment design focuses heavily on containing leaks and managing heat flow. Processing plants constantly monitor pressure and temperature in real time, since a runaway reaction could mean both lost product and real danger. I recall the wisdom from a veteran plant manager: keep the system cool, catch stray vapors, and the process stays safe and cost-effective.
Chemical Reactions & Modifications
As chemists poked and prodded at ethanolamine, they quickly found its knack for tucking into other molecules. Its amine group cheerfully bonds with acids, creating stable salts—think surfactants or the raw ingredients for detergents. Treat it with fatty acids, and you get ethanolamides, handy in everything from shampoos to industrial lubricants. Mix with chloroacetic acid, and suddenly you’ve made glycine, swinging the molecule into a whole new field of pharmaceutical intrigue. Over years on the production line, I’ve seen how small tweaks—adding a methyl group here, a longer carbon tail there—open up fresh commercial niches, like corrosion inhibitors for mechanical pipework or fungicides for agriculture.
Synonyms & Product Names
Anyone flipping through technical catalogs will spot ethanolamine listed under names like 2-hydroxyethylamine, monoethanolamine, MEA, or simply aminoethanol. Some products boast a splashier trade name, but the core chemistry doesn’t change. Reading old shipping records, I’ve seen the flexibility in labeling causes confusion—buyers sometimes double-check to ensure MEA means ethanolamine and not some other similarly named compound, so clarity in naming remains a point of pride for attentive suppliers.
Safety & Operational Standards
Handling 2-aminoethanol safely draws on a blend of common sense and strict regulatory guidelines. Industrial users train everyone to don goggles and gloves before opening containers, set up splash guards, and install reliable ventilation systems. Regulatory bodies, like OSHA, push for air quality checks and regular skin monitoring to prevent overexposure since repeated contact triggers skin, eye, and respiratory irritation. In smaller doses, the compound lets workers get the job done, but larger spills demand swift action and strict clean-up routines. On one job site, a colleague told me about a poorly ventilated room filling up with vapors so quickly that alarms blared—reminding everyone that even a familiar chemical like this requires a healthy dose of respect.
Application Area
If a sector deals with water treatment, personal care, textiles, or specialty coatings, chances are ethanolamine forms a key part of the mix. Refineries count on its basicity to scrub sour gases, letting operators produce fuels that meet strict emission standards. Detergent makers bank on its emulsifying power, churning out stronger, more stable cleaning products that break down hard residue. In herbicide factories, ethanolamine functions as a neutralizer, lending both stability and solubility to active ingredients for safer application on farmland. Even in cosmetics, tiny splits in its molecular chain let formulators balance pH, create smooth lotions, or disperse dyes in everything from hair products to facial creams. Anyone who’s worked in these sectors can point to MEA as a behind-the-scenes fixer that rarely gets the hero’s spotlight, but quietly keeps big processes moving.
Research & Development
The lab bench remains a hunting ground for improved uses and safer derivatives of 2-aminoethanol. Chemical engineers and toxicologists test new catalysts for sharper, cleaner syntheses—lower temperatures, less byproduct, lower water use. Meanwhile, toxicology teams probe the long-term effects of trace exposures, seeking evidence to guide worker safety standards and public health recommendations. Collaborative teams, drawn from university research and private industry, explore using ethanolamine derivatives in biodegradable surfactants, greener pesticides, or advanced medical imaging agents. Grants flow to projects aiming to recycle MEA-rich waste streams, seeking circular economy models where industrial cleanup pays environmental dividends. From what I’ve seen in recent conference proceedings, governments and NGOs want to track and control every pathway ethanolamine travels—from bulk shipping to landfill run-off—to make the entire chain safer and cleaner.
Toxicity Research
2-Aminoethanol’s potential toxicity gets frequent mention in occupational journals and regulatory notices. Short-term exposure stings skin and eyes; high vapors can irritate the lungs. Repeated exposure can sensitise skin, breaking down its protective layers and increasing chance of infection. Lab studies on rodents and cell cultures flag potential risks for reproductive toxicity or central nervous system effects at very high doses, but workplace data suggests routine handling within set limits rarely crosses into danger. Industry-sponsored studies push for better ventilation, stronger spill response, and focused personal monitoring to dial down any hazards linked to chronic lower-level exposure. My experience with industrial hygienists and medical reviewers shows that regular training and exposure audits help limit both minor accidents and the slower, harder-to-track health impacts.
Future Prospects
Ethanolamine’s story keeps growing as industries reinvent themselves for the twenty-first century. Research into carbon capture and storage tags MEA-based scrubbers as leading candidates to strip CO2 from power plant flue gas, making decarbonized electricity more feasible. Agricultural chemistry circles back for new, less burdensome herbicide bases, while pharmaceutical researchers trace pathways to safer, low-residue medical intermediates. Clean manufacturing initiatives may soon demand bio-based routes to ethanolamine, or greener closing-of-the-loop technologies that reclaim it from post-industrial or post-consumer streams. As conversations about sustainability ramp up, everyone from plant engineers to environmental activists weighs in, determined that old chemicals like 2-aminoethanol get responsibly managed, reimagined, and, where possible, improved for the next generation. No matter how science and society shift, there’s little doubt the versatility of this molecule keeps it a topic of steady debate and active innovation.
What is 2-Aminoethanol used for?
Everyday Uses With Big Impacts
Open up a container of 2-aminoethanol and you’ve got more than just a lab chemical. Most folks working in industries don’t use the actual name; it’s often called ethanolamine. The stuff runs behind the scenes, helping make regular products cleaner, safer, and more reliable. Walk down an aisle in a supermarket, you’ll find soaps, shampoos, and cleaners that rely on it. Adding a bit of ethanolamine lets them dissolve grease and oil that soap alone would leave behind. If a detergent company takes it out, suddenly, stains fight harder to stick around.
Ethanolamine works as an emulsifier and a pH stabilizer. As someone who’s cleaned up plenty after family dinners, I appreciate how dishes sparkle a lot faster with a little help from chemistry. Step into any auto shop, and you’ll see barrels of coolants and oils. Antifreeze in car radiators has 2-aminoethanol added as a corrosion inhibitor. Without it, metal pipes rust and fail much quicker. I’ve seen engines gunked up with buildup just because someone picked the cheap coolant.
Supporting Modern Manufacturing
Cities don’t run on clean water alone. Big factories rely on 2-aminoethanol to “scrub” unwanted gases like carbon dioxide out of their emissions. This substance grabs CO2 molecules in chemical plants and refineries. Instead of dumping fumes raw into the air, factories cut pollution before it leaves the smokestack. Environmental agencies trust this. The science traces back to the early 1930s, when engineers discovered its knack for trapping acid gases.
Pharmaceutical factories count on ethanolamine, too. It helps tweak the pH in everything from aspirin to allergy medication. A medicine you buy at the drugstore often started with this workhorse in a beaker. People hardly ever think about the chemicals that keep these processes running efficiently and safely.
Safety and Responsible Handling in the Workplace
Long hours in a warehouse taught me how chemicals shape lives. I handled drums of ethanolamine before I even knew its full name. A bit on your gloves and you start feeling the slip—a sign it cuts through grime better than most soaps. This solvent brings power, but also requires respect. Breathing in its vapors or letting concentrated forms touch bare skin can cause nausea or burns. Isn’t just an industrial risk; home hobbyists sometimes forget gloves, and wind up regretting it.
Higher demand means more workers risk exposure. I’ve seen safety trainings fall short, with old gloves and missing goggles in break rooms. Companies need to ensure employees get clear instructions, better training, and plenty of protective gear. Regulations already limit exposure, but regular refresher courses and easy access to Material Safety Data Sheets can prevent accidents. No one should get sick just because supervisors skip safety steps.
Looking Forward: Cleaner Ways to Use Chemicals
As the world looks to cut pollution, industries search for greener ways to use chemistry. Companies experimenting with enzyme-based cleaners, bioplastics, or new gas capture technologies still keep an eye on old standbys like 2-aminoethanol. The move toward eco-friendly production doesn’t toss out what works; it finds ways to make proven chemicals safer, easier to recycle, and friendlier to communities. As someone who’s seen how fast things change on a factory floor, one lesson stands out: every improvement starts with honest talk about what’s really inside those barrels.
Is 2-Aminoethanol hazardous or toxic?
The Role of 2-Aminoethanol in Everyday Life
2-Aminoethanol, often called ethanolamine, shows up in a surprising number of products. It helps make soaps, cleaners, polishes, and even some pharmaceuticals. Out on the farm, folks use it in herbicides. Step into a factory, and you’ll spot it in coolant fluids and certain gas treatments. Most people never see it raw, but its use touches pretty much every home and workplace.
Digging Into the Hazards
A chemical with so many uses deserves a close look at its risks. Ethanolamine brings a pretty strong bite if it comes in direct contact with skin or eyes. A splash can burn, and that burn doesn’t always heal fast. Breathing in its vapors leads to irritation in the nose and throat, sometimes a cough. Those working with open drums or pipes full of it, especially in places short on airflow, can end up with worse symptoms—headaches, nausea, or, in rough cases, chemical pneumonia.
Personal experience gives this some color. In a paint factory, a coworker brushed diluted ethanolamine off a tank and didn’t wash it away quickly. The burn stuck around for days. That memory does two things for me: it sticks a real face on chemical safety, and it shows that label warnings truly matter.
Looking at Toxicity and Exposure
Research groups have taken ethanolamine through a battery of animal tests. Rats inhaled it in labs at high concentrations—at those doses, lungs became inflamed, and their airways swelled up. The U.S. Occupational Safety and Health Administration (OSHA) sets the workplace exposure limit at 3 parts per million over a work shift, matching up with the irritation workers can expect above those levels. These rules line up with effects seen over decades of industrial work.
The Environmental Protection Agency (EPA) judges ethanolamine as a moderate hazard. It doesn’t build up in soil or water much, so its environmental footprint drops once it gets diluted. At home, shampoos or soaps containing this chemical use concentrations far below irritation levels, but splash hazards and accidental exposures still turn up in poison control calls, especially with kids.
Prevention and Protection in Everyone’s Hands
For workers, gloves, face shields, and proper ventilation have kept accidental burns and respiratory hits at bay. Factories using even moderate quantities run regular safety training and keep wash stations close by. Small businesses without such resources sometimes rely on outdated guidance. Sharing recent safety info and emphasizing the need for training goes a long way.
At home, nobody expects to need gloves just for household cleaners, though reading labels and locking products away from kids still makes simple sense. Poison control centers report most injuries as minor, with washing and observation as the main response.
Practical Solutions for Safer Use
The answer to risk, in my experience, never comes from panic—it comes from tools and knowledge. Industries that use ethanolamine benefit from regular ventilation checks, spill response drills, and better labeling. People at home don’t gain much from knowing every hazard number, but they get a real boost from clear storage, safe use, and knowing who to call after a mistake.
Staying informed and well-equipped turns a potentially harmful chemical into a controlled tool, without overblown fear or avoidable accident.
What are the storage requirements for 2-Aminoethanol?
Understanding the Risks
2-Aminoethanol, also called ethanolamine, may seem straightforward: a clear liquid, a bit viscous, grabs moisture from the air. The thing is, its risks go further than its appearance. As someone who’s spent time around labs and industrial environments, I can tell you spills and fumes don’t forgive mistakes. The scent—a mix between ammonia and something almost fishy—lingers longer than people expect, and that’s often the first hint you’re dealing with a substance that needs respect. It’s not just an irritant; overexposure leads to headaches, nausea, even chemical burns. That makes good storage a real priority.
Why Containers and Ventilation Make a Difference
Metal drums, HDPE jugs with chemical seals, and glass bottles line many shelves, but not all containers hold up. 2-Aminoethanol reacts with a surprising range of materials—including some plastics and certain metals. Incorrect storage leads to corrosion, leaks, and not-so-pleasant cleanup days. Stainless steel or high-density polyethylene (HDPE) containers work best and don’t warp or disintegrate over time. Never cut corners with lids; a loose cap means vapor drifting into the workspace, and it can grab moisture out of the air, turning storage space into a sticky mess.
Ventilation gets overlooked, especially in cramped storage areas. I’ve seen places tape plastic sheeting over shelves—usually as a workaround for poor airflow. It never works. Fumes build up, and one open container turns a storeroom into a hazard zone. Mechanical ventilation—real fans, not just windows—might seem like overkill, but it stops headaches before they start. Keeping air moving means an accidental spill won’t turn dangerous.
Temperature and Fire Hazards
2-Aminoethanol catches fire at around 85°C—not gasoline-level, but enough to matter. I always check that storerooms run cool and stay shaded, especially in summer. Ambient heat and sunlight speed up evaporation and push up vapor concentrations in the air. Never put a drum next to heat vents or in direct sunlight.
Extra caution makes sense: flame-proof cabinets keep containers safe from accidental ignition, and fume detection systems catch leaks early. Water sprinklers help if things ever go wrong, but never assume they solve everything. A good safety setup combines these pieces so no single mistake brings the system down.
Segregation and Labeling: Lessons Learned
Mixing incompatible chemicals can go sideways in a hurry. Once, I watched a careless contractor stack acids next to bases, and labels fell off in a humid storeroom. It cost everyone a week’s pay in wasted chemicals, and nobody forgets a mess like that. Keep 2-Aminoethanol away from acids, oxidizers, and reactive organic chemicals: these combinations cause violent reactions, gas clouds, or even fires. Bold, color-coded tags make sense—my own prep bench has a rule: if the label isn’t obvious from across the room, fix it.
Training and Checking: Everyday Habits
Safe storage isn’t just one person’s job. Teach everyone how to spot leaks, handle splashes, and sort containers. I remember my first spill—learning to double-check the seal and never, ever set down a container in a puddle. Run a regular inspection, not just a yearly audit. Fume sniffers catch problems that eyes miss, and that habit saves money and time. Maintenance teams thrive on routine: wipe up drips, replace cracked lids, update logs daily.
Solid Habits Keep Everyone Safe
2-Aminoethanol storage doesn’t require high-tech magic. Good containers, dry and cool spaces, strong airflow, and clear labels—stick to those. If a team makes inspection and training routine, accidents stay uncommon. No one wants a ruined storeroom or a trip to the infirmary. Smart storage builds trust and keeps the work moving with fewer surprises.
How should 2-Aminoethanol be handled safely?
Why Respect Matters with This Chemical
Anyone who has worked in a lab or around industrial chemicals meets 2-aminoethanol sooner or later. Sometimes called ethanolamine, it pops up in all sorts of places: detergents, coolants, pharmaceuticals. I remember the first time I cracked open a drum of it in a workshop, and the sharp, fishy smell gave me instant respect. Those vapors shouldn’t get near your lungs, and that liquid doesn’t belong on your bare skin. Respect for the hazards isn’t just good sense; it’s built on what plenty have learned through mistakes.
The Real Hazards
Open a bottle in a cramped room, and the vapors hit fast—burning eyes, irritated sinuses, scratchy throat. Splash a little on the skin, especially with a cut, and you’ll notice a sting that sticks around. Eyes exposed to this stuff take the worst of it, risking serious injury if rinsing gets delayed. Swallowing it? Troubleshooting turns into a medical emergency. The CDC and OSHA both list it as a hazardous chemical, and safety data sheets spell out the danger: burns, respiratory trouble, and long-term risks with repeated exposure.
Learning Good Habits Pays Off
Working with chemicals like this demands habits, not just rules printed on posters. Gloves made from nitrile, well-sealed goggles, and a face shield for bigger tasks stay within arm’s reach. Ventilation turns a risky room into a safer one—I’ve tinkered with setups that range from big fume hoods in university labs to open-window fans at small businesses. Disposable lab coats shield your clothes and reduce the spread of contamination.
Labeling matters, too. A mistake with bottles or containers nearly led to a mess in my early days. Now, containers stay clearly marked, and MSDS sheets sit close by for quick double-checks. Colleagues likely appreciate when everyone follows the same steps, since all it takes is one shortcut for someone nearby to pay the price.
Storing and Disposing Without Guesswork
Leaving 2-aminoethanol next to food or drink always spells trouble. I always set aside a locked cabinet, away from acids, oxidizers, or open flames. The stuff’s flammability makes open flames just plain reckless. Large drums demand both a secure footing and a shield from sunlight or heat.
When it’s time for disposal, dumping it down the drain pollutes water and risks chemical reactions. Local regulations usually call for collection of waste fluids and pickup from chemical disposal companies. I’ve learned to keep empty containers separate and never mix residues—some reactions produce toxic or corrosive gases.
Building Safety into the Culture
I’ve seen great safety programs thrive when everyone buys in. Training for new employees shouldn’t just skim the hazards. Regular drills, eye-wash checks, and shared lessons from near-misses keep people alert. OSHA recommends refresher training once a year, and from personal experience, those sessions build real confidence.
2-aminoethanol means business, and so does chemical safety. With habits rooted in facts, the risks can be managed. The respect for this chemical comes from both experience and vigilance. That’s how you stay healthy, productive, and ready for the next challenge in the lab or on the shop floor.
What is the chemical formula of 2-Aminoethanol?
The Formula and Its Everyday Meaning
Everyone remembers those moments in the chemistry lab, nervously mixing solutions and hoping nothing catches fire. Among those bottles, 2-Aminoethanol often sits overlooked despite its formula, C2H7NO, connecting many dots between industry and daily life. I first saw its impact not in a textbook, but through a relative who ran a cleaning business. Seeing first-hand how much cleaning supplies rely on these chemicals opened my eyes to the odd ways basic chemistry shows up outside the lab.
Behind the Letters: Real-World Chemistry
Give a closer look, and C2H7NO actually stands for more than a structure. One carbon chain, an amine group, an alcohol group—sounds basic. Yet, the mix makes it a backbone in products like liquid soaps, coolants, and pharmaceuticals. Hospitals, machine shops, and homes deal with it, thanks to the perfect blend it brings for balancing acidity and removing grease. Old air conditioning units use it for scrubbing exhaust gases, quietly protecting lungs from polluted air.
Things to Consider with 2-Aminoethanol
Working with it drove home the fact that safety gets overlooked. This substance can irritate skin and eyes—my cousin once suffered a rash that hung around for days because she’d relied on ordinary gloves. Many workers know the pain of a careless splash, and the stuff can also drift off as vapor, causing headaches. Stories like hers make a strong case for better training and clear information about handling chemicals, even those that seem as routine as 2-Aminoethanol.
Why the Formula Matters in Industry
It’s easy to chalk up formulas to just numbers and letters. Yet, every shift at a manufacturing plant rests on reliable knowledge about these building blocks. Thanks to its two-hat structure (amine and alcohol), C2H7NO works like a Swiss army knife in making emulsifiers and detergents that don’t wreck your hands or pipes. It also changes the game for pharmaceuticals, acting as a stabilizer. That versatility brings down costs for businesses and creates jobs—a fact I picked up watching local factories expand operations, powered in part by industries using these chemical tools.
Responsible Use and Looking Forward
The dark side comes from overuse and careless disposal. Waterways downstream from plants sometimes record spikes in nitrogen, a sign of chemical runoff. Eco-conscious approaches count, both for environmental reasons and community health. Investment in closed-loop systems for chemical recovery, recycling efforts by industry players, and transparent reporting marks the way forward. More students might take an interest in green chemistry if companies welcomed apprenticeships and shared knowledge beyond trade secrets.
That simple formula, C2H7NO, punches way above its weight. It’s a link connecting the chemistry class to industry, family businesses, workplace safety, and environmental stewardship—a reminder that even minor compounds shape the stories we live and work by.
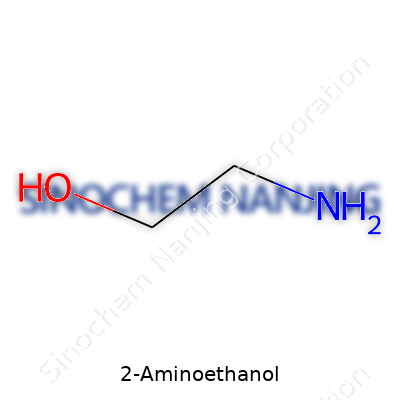
| Names | |
| Preferred IUPAC name | 2-aminoethan-1-ol |
| Other names |
Ethanolamine Monoethanolamine MEA 2-Hydroxyethanamine Glycinol |
| Pronunciation | /tuː əˌmiːnəʊˈɛθənɒl/ |
| Identifiers | |
| CAS Number | 141-43-5 |
| 3D model (JSmol) | `3D structure; JSmol; C(CO)N` |
| Beilstein Reference | 605287 |
| ChEBI | CHEBI:16049 |
| ChEMBL | CHEMBL715 |
| ChemSpider | 8677 |
| DrugBank | DB00160 |
| ECHA InfoCard | DTXSID3029222 |
| EC Number | 205-483-3 |
| Gmelin Reference | 6316 |
| KEGG | C00189 |
| MeSH | D000335 |
| PubChem CID | Mono(2-aminoethyl)amine |
| RTECS number | KK4300000 |
| UNII | 9T8W4FGM35 |
| UN number | UN2491 |
| Properties | |
| Chemical formula | C2H7NO |
| Molar mass | 61.08 g/mol |
| Appearance | Colorless liquid with an ammonia-like odor |
| Odor | Ammonia-like |
| Density | 1.011 g/mL at 25 °C |
| Solubility in water | Completely miscible |
| log P | -1.31 |
| Vapor pressure | 14.7 mmHg (20°C) |
| Acidity (pKa) | 9.5 |
| Basicity (pKb) | 4.75 |
| Magnetic susceptibility (χ) | -11.4 × 10⁻⁶ |
| Refractive index (nD) | 1.454 |
| Viscosity | 24 cP (20 °C) |
| Dipole moment | 1.94 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 108.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -182.10 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1367 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | N07BB03 |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS02,GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | H302, H314, H332 |
| Precautionary statements | P260, P261, P264, P270, P271, P273, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P310, P321, P330, P363, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-0 |
| Flash point | 85 °C (closed cup) |
| Autoignition temperature | 410°C |
| Explosive limits | 3% to 23% |
| Lethal dose or concentration | LD50 Oral Rat 1720 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat 1720 mg/kg |
| NIOSH | B0086 |
| PEL (Permissible) | 3 ppm |
| REL (Recommended) | 200 ppm |
| IDLH (Immediate danger) | 1000 ppm |
| Related compounds | |
| Related compounds |
Ethanolamine hydrochloride Diethanolamine Triethanolamine Aminomethanol Monoethanolamine Methyldiethanolamine |

