2-Aminobiphenyl: Charting the Journey and Future of a Controversial Compound
Historical Development
2-Aminobiphenyl holds a distinct spot in the history of organic chemistry. In the early chapters of synthetic dye-making, organic chemists across Europe searched for new routes to generate colorants from coal tar derivatives. During these explorations, 2-Aminobiphenyl appeared as both a byproduct and a targeted result. Discovery came not in isolation but from fierce competition and cross-learning among labs and industries eager to build vibrant, fade-resistant dyes. The substances that emerged from these efforts provided lasting hues for textiles but also introduced new health concerns. Over time, regulatory oversight and modern analytical techniques made identifying and assessing such compounds possible. Years of research eventually linked 2-Aminobiphenyl to adverse health outcomes among workers, especially those handling dye intermediates in poorly ventilated spaces. The resonance of its story remains felt today, not just in chemistry textbooks but in workplace safety protocols shaped by its legacy.
Product Overview
At its core, 2-Aminobiphenyl is an aromatic amine with two benzene rings connected by a single bond and an amino group attached to one ring. Laboratories see it mainly as an intermediate – a necessary stepstone toward more complex molecules. Industry and academic sectors may refer to it as ortho-aminobiphenyl. Interest in 2-Aminobiphenyl stems both from its chemical reactivity and from the caution sparked by its toxicity. Few modern facilities handle this material without strict controls, reflecting lessons learned from its 20th-century misadventures.
Physical & Chemical Properties
This compound appears as off-white or pale yellow crystals at room temperature. Odor does not stand out, but its molecular makeup – formula C12H11N, molar mass just over 169 – echoes the simplicity of its structure. It melts around 52°C, boils above 320°C, and dissolves in a number of organic solvents, especially ethers and alcohols. The molecule’s planar biphenyl backbone supports resonance stabilization, increasing its chemical stability while also favoring specific substitution patterns during reactions.
Technical Specifications & Labeling
Chemists know that handling 2-Aminobiphenyl means close attention to purity and batch consistency. Material often comes specified at levels above 98% purity, with impurity checks for isomers and traces of other biphenyl derivatives. Regulations dictate hazardous labeling—globally harmonized system pictograms, warnings about toxicity and suspected carcinogenicity, and instructions for restricted use. The emphasis on accuracy and transparency in documentation comes from years of experience with both laboratory mishaps and industrial exposure cases.
Preparation Method
Older approaches to synthesizing 2-Aminobiphenyl started from benzidine or biphenyl intermediates and involved reduction or substitution steps, sometimes under harsh or unpredictable conditions. Over decades, chemists optimized these routes, often starting with biphenyl, performing nitration to introduce a nitro group at the ortho position, then using catalytic hydrogenation to reduce it to the amine. Modern processes favor catalysts that minimize byproducts, conserve energy, and reduce environmental impact, but the core chemistry draws from routes sketched out in textbooks from previous generations.
Chemical Reactions & Modifications
2-Aminobiphenyl participates in a variety of reactions. Its amine group allows for acylation, sulfonation, and diazotization, paving the way for azo dye and pigment production. Coupling reactions benefit from the electronic properties of the biphenyl core, offering routes to both small molecules and polymers. Within research settings, 2-Aminobiphenyl often serves as a model substrate for studies in aromaticity, reactivity trends, and catalysis. Chemists also explore its surface modifications for applications in sensors or advanced materials, often leveraging its unique blend of aromaticity and functional group chemistry.
Synonyms & Product Names
Industries and research circles recognize this chemical by several names. Besides 2-Aminobiphenyl and ortho-aminobiphenyl, references appear across older literature as biphenyl-2-ylamine, 2-Phenylaniline, and 2-Biphenylamine. Each title tells something about the mindset of the chemist or manufacturer naming it—sometimes following IUPAC rules, sometimes sticking with colloquial or trade-influenced variants. This variety in names has occasionally led to confusion, especially during regulatory reviews and the cataloging of historical exposure data.
Safety & Operational Standards
Workplace handling of 2-Aminobiphenyl underscores the importance of protective equipment—gloves that resist organic solvents, goggles, sealed fume hoods, and monitored waste disposal. The hazardous reputation this compound carries stems from epidemiological studies that tracked increased cancers among exposed workers. Regulatory bodies such as OSHA and EU REACH impose strict controls on permissible exposures and require detailed recordkeeping of usage and disposal. The compound’s inclusion on lists of known human carcinogens merits regular health surveillance for anyone involved with its synthesis or processing. In my own experience, training with such substances sharpens risk awareness, reinforcing the value of procedural discipline and emergency planning.
Application Area
2-Aminobiphenyl found early utility in making dyes—vivid colors that brought everything from fabrics to paper to life. Over time, applications diversified into pharmaceuticals, specialty polymers, and analytical reagents. In recent years, interest shifted away from large-scale use due to safety barriers, but research applications persist. Chemical synthesis often requires intermediate steps that use structures similar to 2-Aminobiphenyl for building up complexity. Occasional uses in developing new ligands, small-molecule inhibitors, or advanced materials highlight the molecule’s enduring appeal despite its risks.
Research & Development
Researchers map the reactivity and structure of 2-Aminobiphenyl not just to create new substances but to probe fundamental questions about aromatic chemistry and molecular electronics. The molecule serves as a testing ground for new catalysts—especially those aiming for selectivity or energy efficiency. Chemists also investigate derivatives that offer less toxicity or different electronic properties, aiming to harness the strengths of the biphenyl structure while moderating its hazards. Recent advances use computational models to predict outcomes and guide the choice of reaction partners. The continuing efforts to refine, replace, or rehabilitate this compound’s applications reveal chemistry’s ongoing negotiation with its own legacy.
Toxicity Research
Few compounds cover as much ground in industrial hygiene textbooks as 2-Aminobiphenyl. Researchers spent decades unraveling how exposure leads to increased cancer risks, especially in the bladder. Animal studies and human cohort reviews align in showing damage from both long-term inhalation and direct skin contact. Metabolites generated in the liver become ultimately responsible for DNA damage, which can then spark the chain reaction leading to cancer. This concern drove many countries to all but ban the substance outside of carefully controlled environments. Ongoing research now probes the finer details—identifying the most dangerous metabolic intermediates and plotting how genetics can increase or decrease susceptibility. In contemporary labs, studies switch focus toward more sensitive detection methods and biological monitoring for early warning of overexposure.
Future Prospects
Looking ahead, 2-Aminobiphenyl faces a narrowed scope for widespread industrial use, haunted by its carcinogenic profile. But its value as a research tool remains strong. Scientists still pick apart its reactions hoping to develop analogs that hold promise for safer, greener chemistry. Regulatory advances look to close any loopholes and push for even tighter controls in handling and waste management. In specialized research, the seasoned wisdom learned from 2-Aminobiphenyl may keep shaping how new chemicals enter the market, especially regarding early toxicology and proactive safety design. With holistic assessment and relentless innovation, the lessons carried by this once-common dye intermediate still reshape the balance between progress and protection.
What is 2-Aminobiphenyl used for?
2-Aminobiphenyl’s Main Role in Industry
Factories and research labs have a history with 2-Aminobiphenyl, mostly because it helps create dyes and pigments that bring color to everything from textiles to plastics. This compound gets involved in the early stages of manufacturing, where chemists combine and build larger, more complex molecules. Over the years, as a chemical building block, it has supported the production of various specialty chemicals. This versatility makes it useful for coloring formulas, rubber processing, and even in developing certain pharmaceuticals in the past.
Health Risks Tied to Exposure
2-Aminobiphenyl doesn’t get a friendly reception from health experts. Once people learned about its dangerous effects, particularly its link to bladder cancer, tough regulations followed. Inhalation or direct contact with this substance over time can severely harm workers. Studies published by the National Cancer Institute and other public health organizations have linked it to increased cancer risk among employees in dye and rubber manufacturing. Personally, knowing what workers faced before these risks became widely known gives me more reason to support clear labeling and strict workplace safety standards.
Regulation and Worker Safety
Governments in the United States, Europe, and other regions now strictly regulate 2-Aminobiphenyl. The Occupational Safety and Health Administration (OSHA) and similar agencies around the world monitor factory air and protect workers with rules that limit contact. Protective gear, specialized ventilation, and closed-system processing became the norm. Any gaps in training or equipment maintenance put lives at risk.
Alternatives and Safer Practices
The health dangers led many manufacturers to drop 2-Aminobiphenyl whenever possible. Some found replacements that work just as well for creating dyes and rubber chemicals, with far lower health risks. These swaps often cost more or take time to perfect, but the trade-off comes with longer, healthier lives for people who might otherwise be exposed to a dangerous chemical.
Looking at the Big Picture
Public awareness has grown, but many older facilities and products could still contain traces of hazardous compounds created using 2-Aminobiphenyl. Environmental cleanup and screening efforts remain necessary, especially in regions where regulations arrived late. In these cases, catching risks early with regular blood monitoring and air quality testing might prevent tragedies before they start.
What Can Be Done?
Workers need honest information, ongoing training, and the right tools to minimize contact. Companies that phase out high-risk chemicals—wherever possible—show respect for people on the production line. Waste treatment improvements, tighter supply chain checks, and adopting greener chemistry approaches go a long way toward reducing harm. Researchers keep pushing for safer substitutes, but until every last dangerous process is replaced, constant vigilance stays crucial.
Why This Matters
The story of 2-Aminobiphenyl offers a sharp reminder about the cost of progress and where responsibility lies. Each stained pair of blue jeans or shiny plastic bottle holds a backstory—and sometimes that means workers faced real danger while making them. I’ve seen how fast change can happen when people speak out, demand safer jobs, and make science part of everyday decision-making. The safer future for chemistry depends on listening to those lessons from the past, keeping health at the center of industry, and never taking shortcuts where human lives come into play.
Is 2-Aminobiphenyl hazardous or carcinogenic?
Understanding 2-Aminobiphenyl’s Risks
2-Aminobiphenyl doesn’t show up in daily conversation, yet it sparked my attention the first time I crossed its path in a lab safety class years ago. Few people outside industrial circles know its story, but workers and researchers have tackled its toxic legacy for decades. The facts on the ground are solid: 2-Aminobiphenyl, a byproduct of manufacturing dyes and rubber, carries a heavy health warning. Studies highlight a clear link between this chemical and higher rates of bladder cancer. Sources like the International Agency for Research on Cancer put it in the same risk zone as known dangers like asbestos and benzidine.
Real Experiences from the Factory Floor
I spent time talking to factory workers who operated in dye plants through the late 20th century. They describe harsh smells clinging to their clothes and cautionary tales repeated in locker rooms. Back then, the conversation was more rumor than science, but suspicions grew as friends and coworkers started battling bladder trouble. Medical literature later caught up, confirming that workers exposed for years faced much higher odds of developing cancer. For families and communities linked to these industries, the repercussions still echo.
Why the Science Matters
Peer-reviewed studies, not just anecdotes and workplace stories, back up these fears. The chemical slips into the body through skin or lungs, then breaks down into even more reactive byproducts that damage DNA. The evidence comes from both animal experiments and observations of people exposed on the job. Review panels found cancer cases lining up in clusters near factories using 2-Aminobiphenyl. You don’t need a chemistry degree to see what that means for health risks.
Regulatory Pushback and Community Awareness
Regulators in North America, Europe, and Asia moved to restrict and even ban the use of 2-Aminobiphenyl as soon as its dangers became clear. Limits on workplace exposure, better protective gear, and health screenings stopped thousands of cases before they started. Even so, older facilities, contaminated sites, and imported goods sometimes keep this threat alive. Active monitoring and enforced clean-up orders keep pressure on businesses.
Finding Safer Paths
Substitutes proved tricky to develop at first. Some chemicals that replaced 2-Aminobiphenyl brought hazards of their own. After years of testing, researchers identified dyes and manufacturing routes that work without unleashing these dangerous byproducts. International cooperation and shared research drove down cancer rates among industrial workers, offering hope that hazards like this don’t have to stick around forever. Workers, unions, and advocates taught regulators a hard lesson: nothing beats vigilance, early warning signs, and giving people the right to walk away from unsafe work.
Reflecting on Change
The story of 2-Aminobiphenyl remains important. It shows how industry, science, and real-world experience combine to identify invisible dangers. It also highlights why staying informed and fighting for transparency in manufacturing protects more than just the bottom line. In a fast-changing world of chemicals and technology, the echoes of past mistakes give steady guidance toward safer workplaces and healthier lives.
What are the physical and chemical properties of 2-Aminobiphenyl?
Getting to Know the Substance
2-Aminobiphenyl crops up in plenty of industrial processes, and chemists have known about it for decades. This compound forms from two benzene rings linked with an amine group, showing up as a light tan crystalline solid. It barely dissolves in water, stubbornly clinging together in solid form, but it gives in to most organic solvents. The way it holds up physically shapes how safely it can be handled or removed from waste streams.
Physical Nature Shapes Its Risks
Anyone in a lab who’s worked with 2-Aminobiphenyl recognizes the faint but unmistakable odor. Its melting point sits near 110°C, which puts it on the lower end for aromatic amines. The boiling point runs around 340°C, so open flames or extreme heat ramp up the dangers. Vapor isn’t a major worry at room temperature, but dust or fine crystals present real risks. Prolonged contact with skin or inhalation poses serious health hazards—this isn’t just academic: OSHA lists 2-Aminobiphenyl as a confirmed human carcinogen, linked to bladder cancer in workers over long exposure.
Chemical Behavior Decides Use and Danger
This compound owes much of its reactivity to the amine group attached to the biphenyl core. The nitrogen picks up protons in acidic environments, forming water-soluble salts, but that isn’t enough to tame it in wastewater. It undergoes slow degradation in soil and water. Most people outside chemistry labs don’t see these reactions, but wastewater treatment engineers sweat over them. Even at small concentrations, 2-Aminobiphenyl sticks around, and regular treatment processes struggle to break it up.
Stability matters for storage and use. It stays put under normal conditions, not giving off heat or decomposing, yet it reacts with strong oxidizers. Factory safety crews look closely at storage compatibility charts before deciding where to put it. Accidental mixing with bleach or peroxides creates real risk, not just for ruined product but for fires or toxic emissions.
Looking for Smarter Solutions
Preventing environmental escape of 2-Aminobiphenyl starts with proper handling. Good chemical hygiene saves lives—closed systems, protective gear, and rigorous air filtering shield workers from unintentional exposure. Workers in dye making or rubber processing industries earn better health outcomes with safer production lines and solid personal protective equipment, not hand-waving about “best practices.”
Disposing of even trace amounts can be thorny. High-temperature incineration stands out as the best bet for total destruction, though it costs more. In labs, locking up residues for regulated waste disposal becomes the cautious route. Some researchers study microbial degradation, nudging bacteria to smash up aromatic amines, but this promise needs more testing before replacing the incinerators.
Why It Matters: Health and Responsibility
People want to trust the air and water near their homes and workplaces. Knowing the stubborn, harmful nature of 2-Aminobiphenyl gives weight to strict regulatory limits. Europe and the United States both restrict its use, especially in consumer goods. My own experience tracking chemical inventories shows that open communication and reporting keep accidents down and help communities stay informed. Sharing accurate data and enforcing real controls keep everyone—from seasoned chemists to nearby families—safer.
How should 2-Aminobiphenyl be safely handled and stored?
Understanding the Risks
Anyone working in laboratories or industries comes across chemicals like 2-aminobiphenyl. This compound has a serious reputation: research links it to cancer, especially bladder cancer. Without exaggeration, even a little bit of careless exposure can snowball into health issues. Decades ago, factory workers got sick because safety measures lagged behind. Modern science has shown exactly how dangerous this aromatic amine can be, and regulations today reflect those lessons the hard way.
Practical Ways to Stay Safe
Handling 2-aminobiphenyl safely means going beyond dust masks and gloves. Every bottle needs a clear label, and every user should know exactly what they’re working with. Wear disposable nitrile gloves, long sleeves, and safety goggles. Don’t eat, drink, or smoke in the same room. Wash up thoroughly after handling, and never leave your work clothing mixed with your street clothes.
This isn’t paranoia. Research from the National Institute for Occupational Safety and Health and similar bodies spells out how even minor spills or skin contact can seep into the bloodstream. A small lapse—skipping goggles, not tying back hair, or working with an open window—puts not just yourself but everyone near you at risk.
Storage That Protects Both People and Product
Storing this compound takes planning. 2-aminobiphenyl sits best in tight-sealing, chemical-resistant glass containers. Stash them on shelves made of materials that won’t react with the chemical. Lock storage rooms. Keep the temperature steady and the area dry. Good airflow matters: a chemical fume hood isn’t overkill; it’s a necessity. Signs should scream caution, not whisper it, with clear wording that spells out both the hazard and emergency procedures.
Don’t keep this stuff near any food, acids, or open flames. Once, I saw someone place a tightly capped vial next to a strong oxidizer, brushing off concerns. Later, vapors corroded nearby metal shelves. It’s not enough to rely on instinct—a chemical’s incompatibility chart should stay taped somewhere visible in any lab.
Training Stops Accidents Before They Start
Throwing someone into a lab without proper training creates accidents waiting to happen. Annual safety refreshers go further than basic onboarding. In labs I’ve worked in, we ran emergency spill and eyewash drills every few months. If a bottle tips, everyone knows where the shower and eyewash stations sit. Smart teams make sure new hires get hands-on training, not just handbooks.
Good recordkeeping also keeps surprises away. Log which chemicals arrive, note the batch numbers, and track usage. It’s tempting to skip these steps, but finding an expired or unaccounted-for bottle during a routine check beats discovering a leaking mess months later.
Disposal Follows Strict Rules
Don’t pour leftover 2-aminobiphenyl down the drain or toss it in the trash. Contact hazardous waste disposal experts. In my experience, the extra paperwork and fees sting less than the risk of environmental harm or hefty regulatory fines. Keep small spills in mind, too: absorb with appropriate material and bag it up according to chemical waste protocols.
Solutions for Safer Workplaces
Culture matters as much as gear. If management values safety, investments in good storage cabinets, ventilation upgrades, and continuing education become routine. A suggestion box or safety committee helps catch problems early. No one should fear speaking up just because a procedure takes a few extra minutes.
Protecting people from 2-aminobiphenyl doesn’t make lab work slower or pricier in the long run—it keeps the workplace healthy and lets science move forward without sidelining safety.
What is the molecular formula and weight of 2-Aminobiphenyl?
Digging Into the Chemistry
2-Aminobiphenyl holds a key spot in labs and industry, largely because of its foundation in organic chemistry. Its molecular formula, C12H11N, ensures this aromatic amine stands out. If you look at the blueprint, it comes from two joined benzene rings, with an amino group at the “2” position. This creates both its functionality and hazardous legacy. The molecular weight, 169.23 grams per mole, remains critical for chemists managing purity, calculating doses, or tracking residues in environmental testing.
Real Impact on People and the Environment
It goes beyond numbers. Working in chemical analysis, I’ve seen firsthand the tight regulations surrounding 2-Aminobiphenyl. This substance earned notoriety in the 20th century, mainly as a precursor in dye production and sometimes as an impurity in certain plastics. The core problem—its potent carcinogenic effects, particularly in occupational settings. Many workers, including those in textile and dye manufacturing, faced significant health risks before strict exposure limits arrived.
The risks aren’t just in the workplace. Groundwater and soil contamination have surfaced in areas around older industrial sites. Understanding the formula lets environmental scientists predict how it might break down, persist, and move through ecosystems. It serves as a reminder that real-world chemistry never stops at the lab door.
Facts Everyone Should Know
Let’s look at some truths. The IARC lists 2-Aminobiphenyl as a Group 1 carcinogen, meaning there’s strong evidence it causes cancer in people. Years back, bladder cancer cases in factory workers caught public attention, sparking legal action and reforms. Even exposure at very low levels can add up over time. Once inside the body, the compound interacts with DNA, making monitoring and mitigation a serious priority.
Laboratories often use advanced chromatography and mass spectrometry to detect even tiny amounts in biological or environmental samples. Knowing its molecular weight (169.23 g/mol) allows technicians to set up their equipment accurately, catch the compound precisely, and avoid false negatives. Public health work benefits directly from these solid details, as timely detection triggers clean-up or medical action.
Tackling the Risks, Improving Future Outcomes
Modern regulations prevent scenarios our predecessors couldn’t avoid. Occupational limits, personal protective equipment, and closed-system processes all come from a grounded understanding of what this molecule can do. We see success stories in how industries have shifted away from using or producing high-risk chemicals like 2-Aminobiphenyl or have tightly controlled their storage and disposal.
There’s also room for continuous improvement. Supporting green chemistry initiatives lowers the odds of harmful byproducts during synthesis, sometimes eliminating hazardous intermediates outright. Environmental remediation uses microbes capable of breaking down biphenyls, which further eases the legacy of previous decades.
Why the Details Matter
Knowing the formula C12H11N or the exact weight isn’t just trivia. For those running water tests after reports of industrial spills, checking fish health downstream from old factories, or ensuring food packaging is safe, this knowledge stands as frontline defense. Today’s transparency and data-driven decisions have roots in careful chemical basics, and that’s a win for everyone who counts on safe workplaces or clean environments.
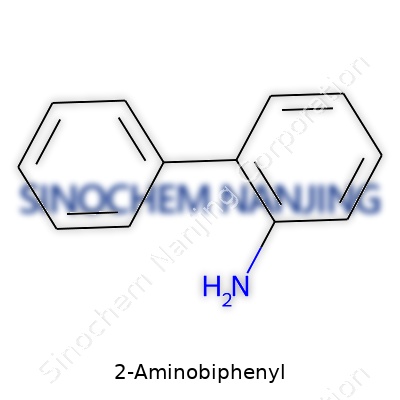
| Names | |
| Preferred IUPAC name | [1,1'-Biphenyl]-2-amine |
| Other names |
o-Aminobiphenyl 2-Phenylaniline 2-Biphenylamine 2-Aminodiphenyl |
| Pronunciation | /tuː əˈmiːnəʊˈbaɪfiːnɪl/ |
| Identifiers | |
| CAS Number | 90-41-5 |
| Beilstein Reference | 1209283 |
| ChEBI | CHEBI:28264 |
| ChEMBL | CHEMBL14011 |
| ChemSpider | 8896 |
| DrugBank | DB13184 |
| ECHA InfoCard | 100.008.241 |
| EC Number | 202-177-1 |
| Gmelin Reference | 61938 |
| KEGG | C06585 |
| MeSH | D010375 |
| PubChem CID | 5800 |
| RTECS number | CY1400000 |
| UNII | 9LU6D9AQ2V |
| UN number | UN2565 |
| Properties | |
| Chemical formula | C12H11N |
| Molar mass | 169.22 g/mol |
| Appearance | White to light yellow crystals |
| Odor | amine-like |
| Density | 1.186 g/cm³ |
| Solubility in water | Insoluble |
| log P | 1.8 |
| Vapor pressure | 0.0025 mmHg (25°C) |
| Acidity (pKa) | 4.86 |
| Basicity (pKb) | 9.17 |
| Magnetic susceptibility (χ) | -62.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.670 |
| Viscosity | 3.38 mPa·s (25 °C) |
| Dipole moment | 1.5 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 163.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 106.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -231 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | D01AE02 |
| Hazards | |
| Main hazards | Carcinogenic; harmful if swallowed, inhaled, or absorbed through the skin; causes skin and eye irritation. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS07, GHS08 |
| Signal word | Danger |
| Hazard statements | H350: May cause cancer. |
| Precautionary statements | P280, P302+P352, P308+P313, P332+P313, P362+P364, P501 |
| Flash point | 104°C |
| Autoignition temperature | 650°C |
| Lethal dose or concentration | LD50 oral rat 200 mg/kg |
| LD50 (median dose) | 373 mg/kg (rat, oral) |
| NIOSH | UX0700000 |
| PEL (Permissible) | PEL: 0.1 mg/m³ |
| REL (Recommended) | 0.02 |
| IDLH (Immediate danger) | 15 mg/m3 |
| Related compounds | |
| Related compounds |
Biphenyl 3-Aminobiphenyl 4-Aminobiphenyl N-Phenylpolyaniline |

