2-Amino-5-Nitro-2'-Chlorobenzophenone: A Deep Dive Into Its Journey, Properties, and Possibilities
Historical Development
Decades ago, chemists searching for new building blocks stumbled across benzophenone derivatives with just the right tweaks—nitro, amino, and chloro substitutions in strategic spots on the molecule. 2-Amino-5-Nitro-2'-Chlorobenzophenone got its foothold in chemical research after organonitrogen compounds demonstrated extraordinary ability to serve both as intermediates and as endpoints in pharmaceuticals and dyes. Patent records and early publications suggest that by the late 20th century, researchers in Europe and Asia worked out routes to this compound for specialized dyes and antimicrobial agents, using early chromatography to identify and purify what was sometimes only a trace byproduct, but sometimes a main event. These early efforts set the stage for refinements in both laboratory and industrial production.
Product Overview
Stepping into the world of specialty chemicals, this compound usually shows up as an off-white or light yellow powder. Chemists working with it immediately notice its solid, crystalline appearance and a subtle but distinct odor. Requiring careful handling, the presence of the amino, nitro, and chloro groups gives it more than one avenue for reaction, with the benzophenone backbone allowing deep integration in synthetic strategies. Its naming conventions reflect this versatility—people in the lab know it as 2-Amino-5-Nitro-2'-Chlorobenzophenone, but chemical catalogs sometimes offer alternative names that reflect its structure or synthetic origins.
Physical & Chemical Properties
This compound brings an intriguing physical profile to the bench. With a melting point usually in the 140-150°C range, lab workers find it easy to collect and purify by recrystallization. Solubility stands out—while water shuns the powder, organic solvents like acetone and ethyl acetate dissolve it well, making it flexible for extractions and column work. The molecular weight falls just above 270 g/mol—a moderate heft for a mid-sized aromatic compound. The nitro group on the five-position adds electron withdrawal, shifting the reactivity around the benzene rings and lending a golden hue to solutions. The solid's stability under cool, dry conditions means storage isn't complicated, though it asks for protection from light and moisture to prevent slow degradation.
Technical Specifications & Labeling
Standard analytical values, backed by NMR, IR, mass spectrometry, and elemental analysis, give manufacturers and users a clear portrait. Chemists keep a close eye on chemical purity—often topping 98% for laboratory-grade material, with impurities like other chlorinated benzophenones held below 0.5%. Product codes reflect small batch manufacturing runs from specialty suppliers. Labels on drums or vials display GHS hazard symbols, signal words alerting to potential health impacts, and clear batch tracking codes for recall integrity.
Preparation Method
The main synthetic route starts with a Friedel-Crafts acylation—often beginning with chloro-nitrobenzene and aniline or related compounds, mediated by aluminum chloride under cool conditions. After initial coupling, nitration with a mixed-acid process introduces the nitro group without overreacting the aromatic system. Chemists use careful controls to direct substitution to the five-position, monitoring temperature, acid concentration, and reaction time. Workup involves neutralizing acids, solvent extractions, and repeated crystallizations to isolate pure product. Scale-up processes for industry sometimes substitute microwave assistance or continuous flow technology, trimming down waste and speeding up throughput.
Chemical Reactions & Modifications
This molecule’s life doesn’t end at synthesis. The combination of nitro and amino groups opens up reduction possibilities—yielding diamino or hydroxy derivatives when needed for specific targets in medicinal or dye chemistry. The chloro substituent offers a reliable point for nucleophilic aromatic substitution, so researchers design routes to more elaborate phenyl ethers and amines. Oxidation or further nitration can bring out new color properties for dye industries, while coupling reactions put it at the heart of bridging molecules in polymer and material science.
Synonyms & Product Names
Across catalogs and journals, alternative names pop up and sometimes confuse: 2'-Chloro-2-amino-5-nitrobenzophenone, 5-Nitro-2-aminobenzophenone-2'-chloride, and in some texts, simple codes or numbers reflecting in-house tracking at pharmaceutical companies. Repository databases keep all variants indexed so researchers can track literature and health records without missing data.
Safety & Operational Standards
Lab safety matters. The presence of a nitro group often means possible skin and respiratory irritation. Gloved hands, safety eyewear, and fume hoods matter as much as clean glassware. Dust control steps in, since the powder tends to become airborne if handled roughly. Material Safety Data Sheets alert users about minimizing ingestion and inhalation risks. Disposal follows local hazardous waste regulations—never down the drain. In workplaces, training programs reinforce spillage response strategies and first-aid know-how. Facilities emphasize tracking exposure duration and concentrations, ensuring operators avoid cumulative risk often linked with aromatic amines and chlorinated compounds.
Application Area
Look inside modern dye chemistry and you’ll spot this benzophenone as a key intermediate—its electron-rich and electron-poor sites anchoring new chromophores for vibrant pigments and inks. Medicinal chemists recognize its backbone in candidate molecules for antimicrobial and anti-inflammatory drug research, sometimes as a target, sometimes as a stepping stone on the synthesis road. Polymer manufacturers value it as a specialty additive, exploiting its chemical reactivity to enhance cross-linking performance in engineering plastics. Some niche uses even show it in liquid crystals and specialty coatings for advanced optics.
Research & Development
R&D programs continue to dig deeper. Recent years brought out new application angles in photonics, where chemists leverage the interaction between the benzophenone core and various substituents to build tunable photo-initiators. Teams across Asia and Europe report progress in green chemistry approaches—switching away from crude acids and chlorinated solvents in favor of milder, more sustainable reagents. A few collaborative university-industrial consortia focus on unlocking biological properties. They tailor derivatives aiming for antimicrobial potency with lower mammalian toxicity. Databases of high-throughput screening reports keep filling, underscoring its status as a pivot molecule for many discovery projects.
Toxicity Research
Toxicologists approach this class of compounds warily, given the history of both nitro and chloro-aromatics showing complications in mammals. Animal studies point to limited acute toxicity at routine laboratory doses, but long-term effects—often overlooked until recently—raise red flags for carcinogenicity. Chronic inhalation or skin absorption studies highlight risks tied to repeated occupational contact. Environmental researchers probe its persistence, noting it resists rapid biodegradation in water and soil, and warn about unintended accumulation in aquifers. Government agencies call for detailed reporting of effluent streams, especially from manufacturing plants. Ongoing work in molecular modeling seeks to predict breakdown pathways and pinpoint less hazardous analogs.
Future Prospects
The compound continues to attract attention from both academia and industry. Teams work to engineer less toxic, more biodegradable analogues for broader use in pharmaceutical and agricultural chemistry. Process optimization aims to synthesize this compound with lower environmental impact, tapping into continuous flow reactors and biocatalysis. Regulatory agencies pressure manufacturers to explore reusable solvent systems and closed-loop water handling. On the drug discovery front, medicinal chemists invest in SAR (structure-activity relationship) mapping to identify new leads in anti-infective fields without old-style toxicity baggage. As collaborative data-sharing projects expand, the hope remains to chart out safe, effective uses and push the bounds of what this benzophenone structure makes possible.
What is the chemical structure of 2-Amino-5-Nitro-2'-Chlorobenzophenone?
Chemical Structure and Features
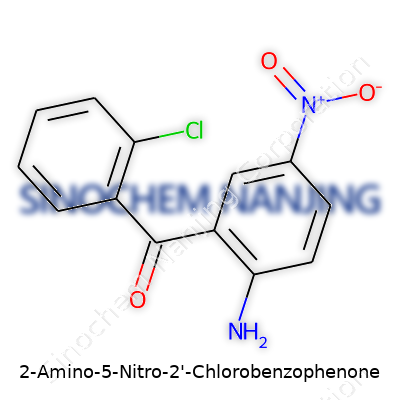
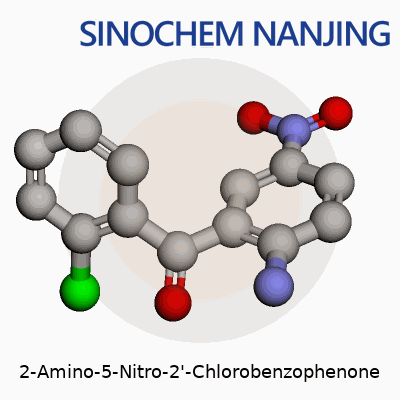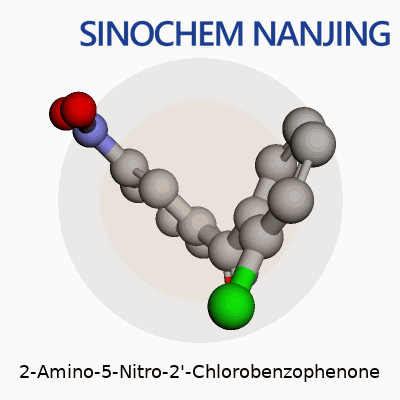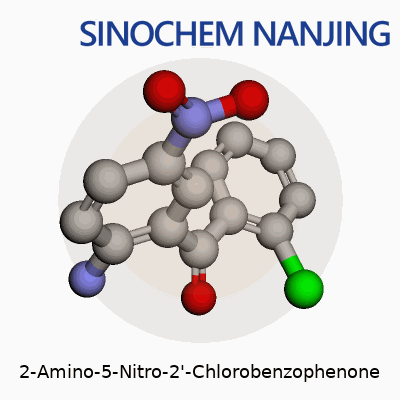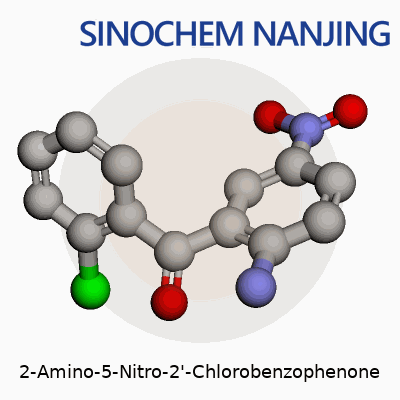
Sometimes, a chemical name reads like a code. In the case of 2-Amino-5-Nitro-2'-Chlorobenzophenone, it pays off to break that code down. At the core sits benzophenone, with two benzene rings joined by a single carbonyl (C=O) group. Adding functional groups brings important changes. One benzene ring carries an amino group (-NH2) at the second carbon position and a nitro group (–NO2) at the fifth. Across the molecular bridge, the other ring has chlorine at the second position. This leads to a molecular formula of C13H9ClN2O3.
What draws attention is the combination of electron-donating (amino) and electron-withdrawing (nitro, chloro) groups gathered around the benzophenone core. Modifications like these shift the chemical character, impacting everything from melting point to solubility and reactivity. In basic organic chemistry labs, exploring such tweaks drives home how a single atom can nudge a molecule’s entire behavior.
Practical Role in Synthesis
Organic synthesis relies on compounds like this to open new possibilities. When building bridges between drug candidates or dyes and pigments, chemists often reach for benzophenone derivatives. Structures with both amino and nitro groups have proven handy for creating advanced molecules for pharmaceuticals, agricultural chemicals, and specialty materials. Chlorine serves as a common leaving group or a site for further substitution, making chlorinated aromatics flexible tools for research and industry alike.
I’ve seen how a structure like 2-Amino-5-Nitro-2'-Chlorobenzophenone fits in a wider toolkit. In undergraduate synthesis experiments, students use similar molecules as intermediates for more complex reactions or in coupling chemistry, learning that a well-placed nitro group not only pulls electrons away, but sets up selective reactions under milder conditions—cutting down on the harsh chemicals some older methods depend on. This links smart chemistry with safer, more sustainable lab practices.
Applications and Concerns
Dye manufacturing draws on the color-influencing properties of nitro and amino substitutions on aromatic rings. It amazes me how small tweaks can shift absorption of visible light and produce intense hues. Laboratories and pilot plants depend on structures like this to help tailor-make pigments for fibers or inks. In research settings, the reactivity controlled by these groups lets chemists create a library of targeted compounds, hunting for the next breakthrough molecule in diagnostics or electronics.
Challenges crop up, though. Nitroaromatic compounds demand respect for their toxicity and environmental persistence. Some nitro-benzophenones have flagged safety concerns due to their potential for forming hazardous byproducts. Chlorinated aromatics raise their own issues: chlorination can boost biological resistance, but persistent residues can threaten water and soil. Addressing these risks matters as much as chasing performance. Process safety calls for good ventilation, robust waste streams, and substitution with greener alternatives where possible.
Building Toward Safer, More Useful Chemistry
The chemistry community remains active in designing new benzophenone derivatives with fewer environmental concerns, better safety profiles, and strong technical properties. Stepwise innovation often starts with basic building blocks like 2-Amino-5-Nitro-2'-Chlorobenzophenone. With smart choices—like using catalytic rather than stoichiometric reagents or recovering solvents—chemists can keep pushing molecular boundaries without overshooting on safety or sustainability. Deep knowledge of structure-function relationships and practical handling goes hand in hand with new discoveries, keeping the feedback loop between lab insights and real-world responsibility healthy and strong.
What are the main applications or uses of 2-Amino-5-Nitro-2'-Chlorobenzophenone?
Real Chemistry at Work
Plenty of people have never heard the name 2-Amino-5-Nitro-2'-Chlorobenzophenone. It doesn’t show up on TV commercials, and it certainly hasn’t sparked any Twitter debates. Still, labs and factories are using this compound almost every day, mostly because it helps solve problems nobody wants to talk about but everyone expects to be handled.
Pharmaceuticals: Turning Small Steps Into Big Cures
In drug development, tiny changes can push a science project all the way into a working medicine. This compound brings an extra nitrogen, a nitro group, and a chlorine atom together on a benzophenone backbone. Those features make it a popular “building block” for pharmaceutical researchers. Synthetic chemists often start with molecules like these to work their way toward more complex structures—think of them as the raw material for blending up antibiotics, antivirals, anticonvulsants, and other life-improving drugs.
Some sources say ketone-based intermediates like 2-Amino-5-Nitro-2'-Chlorobenzophenone end up in anti-inflammatory or antibacterial agents. Getting new compounds across the finish line isn’t easy. Undesirable side effects, resistance, or stability problems throw up walls in every direction. Starting with a versatile intermediate gives the flexibility to tweak the molecule over and over again. It’s not glamorous, but it’s the backbone of progress in medicinal chemistry.
Dyes and Pigment Creation
Industries always push for colors that resist fading, manage higher temperatures, and survive rigorous cleaning. That’s where nitro and chloro groups earn their cost in industrial dyes. This compound’s mix of electron-withdrawing groups makes it valuable in synthesizing a range of bright and long-lasting colors, especially those needed for synthetic fabrics. Nitro-containing benzophenones play a background role in ensuring your T-shirt doesn’t turn gray after three washes.
I’ve met textile chemists who swear by a handful of chemical precursors, and derivatives of 2-Amino-5-Nitro-2'-Chlorobenzophenone show up in their “must-have” lists. Most consumers won’t know which molecule kept their new couch cover bright through a year of use, but real money gets spent on these behind-the-scenes molecules.
Specialty Polymers and Materials Science
Not every application makes headlines, but specialty plastics and coatings lean on this compound in their recipes. Adding or swapping out chemical groups on the benzophenone skeleton helps control how tough the plastic feels or how it handles sunlight. UV-stable coatings show up everywhere, from medical equipment to electronics casings. Any work that increases scratch resistance or durability has a place for intermediates like this.
Challenges and The Path Forward
Safety and environmental protection have always shaped work in chemistry, and compounds like this don’t slip under the radar. Handling nitro or halogenated aromatics requires strong systems for air quality, waste management, and worker protection. Researchers keep searching for ways to develop greener synthesis methods, including catalytic routes that use less energy and make fewer toxic byproducts.
As the world keeps pushing for better medicines, longer-lasting materials, and responsible production, 2-Amino-5-Nitro-2'-Chlorobenzophenone’s story will keep evolving. Chemists devote real effort to making sure life-saving drugs and everyday items come with fewer environmental strings attached.
What is the CAS number for 2-Amino-5-Nitro-2'-Chlorobenzophenone?
More Than a Number: Clarity in Chemicals
Every chemical has a unique personality, but keeping them straight would be almost impossible without a universal system. Take 2-Amino-5-Nitro-2'-Chlorobenzophenone, which carries the Chemical Abstracts Service (CAS) number 2140-73-8. That string of digits isn't just bureaucracy in action; it brings order to a world of sprawling, sometimes confusing chemical names and structures. Someone can refer to it by CAS and know they're dealing with precisely the same stuff, no matter if they're working in a lab in Tokyo or talking to a supplier in Texas.
Precision Trumps Ambiguity
Naming chemicals can turn into a wild west of synonyms, trade names, and little tweaks in a molecule. In college, I watched researchers squint at two nearly identical bottles, double-checking paperwork, worried a tiny mistake could throw off weeks of work. A CAS number cuts through that fog. It doesn't care about language, misspelling, or marketing rebrands. Down to its core, it points to a single substance—no confusion, just clarity.
Quality and Safety in Manufacturing
Companies blending pharmaceutical ingredients, dyes, or specialty resins can’t afford guesswork. With 2-Amino-5-Nitro-2'-Chlorobenzophenone, that number 2140-73-8 guarantees consistency. Uniform products mean fewer recalls, fewer dangerous mix-ups, and a safety net for workers and consumers. Regulatory agencies lean on the CAS system for tracking and compliance. Laws in the United States, the European Union, and Asia all use CAS numbers to tie together regulation, trade, and environmental monitoring.
The Backbone of Research
Anyone searching for scientific data, patents, or material safety information can zero in on a chemical using its CAS number. It saves time, cuts mistakes, and adds transparency to sharing knowledge. I’ve spent endless hours in digital libraries, where searching by name sent me off in circles, but entering 2140-73-8 offered a direct line to journals, toxicology databases, and supplier catalogs. This speeds up breakthroughs and prevents wasted efforts on duplicated work.
Combatting Counterfeit Chemicals
Counterfeit chemicals remain a problem in today’s global market. Labels spoofed, packaging copied, and impurities sneaking into supply chains—it creates risks for all kinds of industries. Relying on CAS numbers can slow that tide. It's easier to spot mismatched numbers on invoices and test results. That vigilance helps keep unsafe products off the shelves and out of sensitive environments where purity and stability are everything.
CAS Numbers and the Future
Technology keeps moving, but the need for precise tracking grows. Artificial intelligence, advanced manufacturing, and stricter environmental rules all depend on rock-solid chemical identification. Databases feed on CAS numbers to monitor pollutants, predict hazards, and trace ingredients back to their sources. This offers a way forward as we juggle progress with responsibility.
Seeking Solutions
Improving chemical transparency takes more than just handing out numbers. Training for lab techs, clearer supply documentation, and better enforcement build a safer industry. Governments can invest in digital tools that make CAS lookup routine and mandatory in all records. Partnerships between companies and academic researchers bring real-world feedback into refining chemical registries. Each step tightens the loop, keeping science open and honest, and protecting the people and ecosystems in the process.
How should 2-Amino-5-Nitro-2'-Chlorobenzophenone be stored and handled safely?
Getting the Basics Right
In labs and factories, storage and handling of chemicals don’t leave room for guessing games. With compounds like 2-Amino-5-Nitro-2'-Chlorobenzophenone, common sense plays a bigger role than fancy protocols. The compound typically finds use in chemical synthesis and sometimes in pharmaceutical intermediates. It brings risks along with benefits, and ignoring the basics can get people hurt.
Temperature and Moisture Matter
2-Amino-5-Nitro-2'-Chlorobenzophenone prefers a dry, cool place. Think of it like flour in the pantry — heat or moisture clumping things up, except this time, the stakes run much higher. Straight facts: moisture in the air can kick off unwanted reactions. I’ve watched storage rooms where humidity ruined batches of even less reactive chemicals. Chemical safety sheets show decomposition as a real risk, so erring on the side of cool, dry storage isn’t overkill.
Choosing the Right Container
Not every container fits. Glass jars with airtight seals work best, keeping both air and accidental hands at bay. Labels should never peel off or smudge, saving headaches during inventory. Metal may leech or react, and plastic sometimes warps or cracks. Tough materials lower the risk of leaks. I’ve seen poorly sealed substances corrode shelving and threaten techs. Clean, sturdy jars earn their keep in any serious lab.
Avoiding Cross-Contamination
Chemicals get confused for one another far too easily. One bottle out-of-place or mislabelled, and mistakes wait to happen. 2-Amino-5-Nitro-2'-Chlorobenzophenone shouldn’t live anywhere near acids, oxidizers, or flammable solvents. Each shelf ought to tell you clearly what belongs where. Chips and spills draw pests and cause deeper hazards, so regular checks make a real difference.
Safe Handling: Personal Protection Isn’t Optional
Every grown-up chemist knows stories about taking shortcuts around gloves and goggles — mixed with regret. Rashes, respiratory effects, and worse have followed from lazy habits. For this compound, gloves (nitrile or neoprene) and goggles protect against immediate skin and eye problems. Fume hoods keep vapors or dust out of the lungs, which should be standard for anything with nitro or chloro groups. Instructing newcomers with the blunt truth — not just manuals — keeps everyone honest about safety gear.
Spill and Waste Protocols
Spills go from minor annoyances to crises if ignored. After a spill, using absorbents like vermiculite or sand works well, then sealing contaminated material in marked disposal bags. Nothing gets tossed in regular trash or poured down the drain. I’ve learned the hard way that outdated disposal practices lead to costly cleanups, and sometimes legal risks. Double-checking local hazardous waste rules before disposal saves more than just fines.
The Importance of Training and Vigilance
Proper training never gets old. Teams should review handling protocols, run through spill drills, and challenge each other on what to do in an emergency. One careless moment can set a project or even a career back. Having clear signage, accessible material safety data sheets, and open talk about near-misses keeps everyone sharp. Real safety comes from honest conversations, not a checklist approach, especially around tricky chemicals like 2-Amino-5-Nitro-2'-Chlorobenzophenone.
What are the safety precautions and potential hazards associated with 2-Amino-5-Nitro-2'-Chlorobenzophenone?
The Real Risks in the Lab
Working with chemicals every day has a way of sharpening your awareness. Some compounds command extra respect, even from old-timers who have seen it all. 2-Amino-5-nitro-2'-chlorobenzophenone is one of those, especially for research chemists or anyone mixing batches in the pharmaceutical supply chain. This isn’t just another jar of yellow powder. It packs a mix of health and safety risks that call for clear protocols and solid habits.
Why This Compound Matters
Chemists use it in certain syntheses, particularly as a building block in dye or drug development. The benefits are clear—rare functional groups, reliable reaction profiles, useful intermediates. Yet, even these practical perks can’t overshadow the need for safety in every step, from weighing out a sample to cleaning up afterward.
Health Hazards
Personal safety always comes first. Just a whiff or brief skin contact with 2-amino-5-nitro-2'-chlorobenzophenone can trigger irritation. Eyes water, skin breaks out, throats grow raw. As with many nitroaromatic compounds, inhaling dust or fine particles risks more than a minor rash. Chronic exposure isn’t well-studied, but general chemical principles tell us to expect cumulative effects that don’t always go away.
Some related benzophenones have raised alarms due to their toxicity or links to long-term health issues. Until a full toxicological profile gets published—usually long after the first containers arrive—lab workers should treat this compound as both a short- and long-term health risk. No “routine” day around that.
Environmental Impact
Labs rarely dump chemicals down the sink, and this one demands even more care. Its backbone resists breakdown, and the nitro and chloro groups suggest persistence in water or soil. Runoff or accidental release could linger far longer than most expect. Nitrated aromatics have been known to harm aquatic life and disrupt microbial communities. Waste management grows into a pressing issue. Not only for compliance, but out of respect for the place we all share outside the lab.
Precautions That Actually Work
Everyone in the lab—students, techs, senior chemists—should reach for gloves, goggles, and a well-fit lab coat as the bare minimum. Respirators and fume hoods become more than box-ticking exercises when solvents or powders like this one get involved. An open bench or makeshift shield just doesn’t cut it.
Storage needs real discipline, too. Separate this powder from incompatible chemicals such as strong reducing agents, acids, or anything prone to spontaneous reactions. I’ve seen fires start from sloppy storage more than once, and records show that old, forgotten bottles in the wrong spot caused most of them.
Fixing Gaps in Training
Written protocols only go so far if no one reads them. Supervisors must walk new hires through the process, not just hand over an MSDS. Practical drills, clear signage, and checklists that someone actually signs off on—these habits kept me and my colleagues on track when the pace picked up.
For long-term safety, facilities should commit to regular air monitoring and periodic reviews of handling practices. Lessons learned from near-misses or spills belong in shared meetings, not buried in paperwork. No lab can claim perfection, but honest feedback reduces risk better than any poster on the wall.
Smart Disposal and Emergency Response
Disposal means sealed, labeled containers picked up by licensed crews, never the general trash. In case of a spill, containment trumps panic. Clear the area, ventilate if it’s safe, and use proper absorbent—not paper towels—and specialized PPE. Keep emergency contacts up-to-date and visible.
Every step, from weighing to waste, asks for respect and diligence. This isn’t just another chemical name—it represents a responsibility to our colleagues, ourselves, and the environment we all share.
| Names | |
| Other names |
AKOS BBS-00007649 MLS001018304 |
| Pronunciation | /tuː-əˈmiːnoʊ-faɪv-ˈnaɪtroʊ-tuː ˈklɔːroʊ-benˈzoʊfiːˌnoʊn/ |
| Identifiers | |
| CAS Number | 55149-93-8 |
| Beilstein Reference | 1368228 |
| ChEBI | CHEBI:131786 |
| ChEMBL | CHEMBL3326018 |
| ChemSpider | 2246112 |
| DrugBank | DB08274 |
| ECHA InfoCard | ECHA InfoCard: 100.032.895 |
| EC Number | 4.1.1.49 |
| Gmelin Reference | 84186 |
| KEGG | C14322 |
| MeSH | D015777 |
| PubChem CID | 21724123 |
| RTECS number | DJ8753000 |
| UNII | 6CQ84V580M |
| UN number | UN3447 |
| Properties | |
| Chemical formula | C13H9ClN2O3 |
| Molar mass | 306.65 g/mol |
| Appearance | Yellow solid |
| Odor | Odorless |
| Density | 1.48 g/cm3 |
| Solubility in water | Insoluble |
| log P | 1.98 |
| Vapor pressure | 0.0000172 mmHg at 25°C |
| Acidity (pKa) | 13.7 |
| Basicity (pKb) | 2.77 |
| Magnetic susceptibility (χ) | -69.04 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.683 |
| Dipole moment | 3.56 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 343.6 J/(mol·K) |
| Std enthalpy of formation (ΔfH⦵298) | –83.3 kJ/mol |
| Pharmacology | |
| ATC code | N02BE51 |
| Hazards | |
| Main hazards | Harmful if swallowed, causes serious eye irritation, may cause respiratory irritation |
| GHS labelling | GHS07, GHS09 |
| Pictograms | GHS07,GHS09 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | Precautionary statements: P261, P305+P351+P338, P312, P337+P313 |
| NFPA 704 (fire diamond) | 2-3-1-0 |
| Flash point | 158.1 °C |
| Lethal dose or concentration | LD₅₀ (oral, rat) >2000 mg/kg |
| LD50 (median dose) | LD50 (oral, rat) = 640 mg/kg |
| NIOSH | SN1650000 |
| REL (Recommended) | 50 mg |
| IDLH (Immediate danger) | Unknown |

