2-Amino-1,3,4-Thiadiazole: A Deep Dive Into A Small, Influential Molecule
Historical Development
For most chemists, 2-Amino-1,3,4-thiadiazole represents more than just a building block. Its history traces back to the flurry of postwar organic chemistry, when curiosity about sulfur and nitrogen-containing rings exploded. Researchers in the early twentieth century poked at thiadiazoles, hoping to unlock antibacterial and antitumor agents. 2-Amino-1,3,4-thiadiazole, with its repetitive nitrogen atoms and single sulfur, became a laboratory workhorse. Scientists recognized its potential, not just for its intrinsic structure, but for the modifications it allowed, paving the way for entire families of biologically active compounds and advanced materials.
Product Overview
This molecule, sometimes shorthanded as 2-amino-thiadiazole, slips quietly into both academic and industrial catalogs. It shows up as a fine powder, off-white by most accounts, with a faint smell that hints at its sulfur content. Weight for weight, it packs a hefty punch in synthetic labs, where researchers hunt for pharmaceutical leads or tweak electronic properties in polymers. Many chemists I’ve worked with keep a small jar of it for quick tests that branch into completely unexpected discoveries.
Physical & Chemical Properties
With a molecular formula of C2H3N3S, this compound presents a tidy complexity. Its melting point ranges from 166 to 168°C, allowing for practical purification and handling. The presence of multiple nitrogens and the sulfur atom boost its electron density, offering intriguing reactivity that’s exploited in both medicinal and material chemistry. The molecule dissolves in water only sparingly but responds readily in common organic solvents. Such nuanced solubility pushes chemists to adapt protocols for best results, sometimes mixing solvents or adjusting pH to coax it into solution. Once handled, 2-amino-1,3,4-thiadiazole tends to remain stable in its bottled form, though exposure to strong acids or bases prompts decomposition or ring opening.
Technical Specifications & Labeling
Bottles arrive labeled with concentration data, batch numbers, and purity—generally above 97%. Labels also lay out storage needs, often at room temperature, out of direct sunlight. I’ve noticed labs place an extra warning sticker to flag its reactivity, urging precaution. Documentation on hazardous potential gets shipped with every order. These sheets track compliance with international standards set by agencies like OSHA or REACH, matching real safety with regulatory oversight. Certificates of analysis, attached to each lot, detail results from NMR and HPLC, reassuring researchers about what’s inside.
Preparation Method
Synthesis has come a long way since the first methods involving thiosemicarbazide and hydrazine. Today’s protocols center on cyclization reactions—usually starting with thiosemicarbazide under oxidative conditions, sometimes using ferric chloride or iodine in an alcohol solvent. Yields have crept up as protocols improve purification and waste reduction. I once spent a week trying minor tweaks with different oxidants, only to discover that slower dropwise addition often did more than fancy catalysts. Many labs now stick with classic approaches, handed down and published in organic synthesis texts, but green chemistry edges in, aiming for milder conditions and fewer by-products.
Chemical Reactions & Modifications
The core 1,3,4-thiadiazole ring asks for creative chemistry. Substitution reactions pop up at the 2-amino group, allowing chemists to bolt on aryl, alkyl, or acyl pieces. This spot serves as a launch pad for libraries of new molecules, from simple amides to complex heterocycles. Cross-coupling with halides opens the door to broader chemical spaces. The sulfur atom in the ring does more than bind carbon and nitrogen; it participates in oxidation and reduction cycles, and under certain reagents, the ring itself can expand or contract. Having an amino group ready to react has driven much of its popularity in medicinal chemistry, where researchers hunt for candidates with antimicrobial or anticancer properties.
Synonyms & Product Names
2-Amino-1,3,4-thiadiazole picks up plenty of aliases in the literature—sometimes appearing as 2-Amino-thiadiazole, 1,3,4-Thiadiazol-2-amine, or by registry numbers like CAS: 496-58-0. Some vendors toss in branded ingredient names for blends or related structures. Trade names pop up in pharma and agrochemical databases, adding a veneer of marketing to what is fundamentally the same molecule. No matter the alias, skilled chemists double-check structures to avoid mix-ups in synthesis or analysis.
Safety & Operational Standards
In the lab, practical safety protocols call for gloves, goggles, and good ventilation. Inhalation, ingestion, or skin exposure can cause irritation, although serious toxicity at bench-scale doses remains low. Standard operating procedures demand spill kits and well-marked waste containers, since sulfur- and nitrogen-laced molecules can feed into more reactive chemistries. Waste often gets collected in halogenated waste streams, reflecting potential breakdown products. Material safety data sheets for this compound mirror general guidelines for low-level irritants, but long-term exposure studies raise questions about occupational health. In research facilities, every bottle records the date opened and the name of the researcher, which tightens accountability and sharpens safety culture.
Application Area
Most discoveries in drug development and agrochemicals come through molecules like this. Medicinal chemists use 2-amino-1,3,4-thiadiazole as a launch point to develop antibiotics, CNS drugs, and antitumor leads. Material scientists build off the thiadiazole platform for organic semiconductors, polymers, and advanced friction modifiers for lubricants. In my experience, this molecule has sparked whole series of patent filings, with new derivatives routinely ringing in as inventions. Agrochemical companies test its backbone for fungicides and herbicides, looking for ways to target fungal cell walls or insect nervous systems. Academic labs also keep it close to heart for combinatorial synthesis, protein labeling, and molecular electronics.
Research & Development
Much of the ongoing work chases greater biological activity or more refined selectivity. Researchers build libraries—dozens, sometimes thousands—of derivatives, shuttling compounds through automated screening platforms. Computational chemistry enters the picture, modeling how changes at the amino group or other ring positions affect function. Some groups pivot away from pharma toward semiconductors, where variations in the thiadiazole ring shift electronic properties. In recent years, I’ve watched collaborations spring up between academia and startups, leveraging high-throughput syntheses to uncover unexpected leads for medical or industrial use. A big part of this molecule’s research profile comes from its tractability; it offers enough reactivity to try bold ideas, yet stays stable enough to work with through long assays.
Toxicity Research
Rigorous safety assessments rely on animal models and cell cultures—standard fare for molecules on the path to market. Acute toxicity tends to register low at the small doses typical for pharmaceuticals, but modifying the side chains alters absorption, metabolism, and clearance. Chronic studies highlight potential for DNA interaction, with some derivatives tripping genotoxicity flags. For industrial or agricultural uses, environmental fate studies look at how these chemicals break down in soil and water, aiming to reduce persistence and downstream effects. One major challenge is building a robust public dataset; much of the corporate research stays behind paywalls, so academic toxicologists fill in the gaps by publishing full datasets. Over time, safety standards shift as new data clarify long-term risk.
Future Prospects
Looking ahead, 2-amino-1,3,4-thiadiazole has staying power. Its simple structure offers a foothold for developing new medicines and electronic materials. Efforts to cut the cost and waste of production are already paying off, as green chemistry principles take hold. Advances in automation—from AI-driven molecule design to high-throughput screening—are pushing the field toward faster discovery cycles. As resistance builds to old antibiotics, new scaffolds built off this core could offer solutions in infectious disease. On the technology front, tweaks at the nitrogen or sulfur positions are already fine-tuning conductivity and flexibility in organic electronics. As research broadens, expect this stalwart compound to turn up in an even greater range of applications, reflecting science’s drive to make more from less, while keeping one eye on safety and sustainability.
What are the main applications of 2-Amino-1,3,4-Thiadiazole?
Unpacking the Substance
2-Amino-1,3,4-thiadiazole, with its five-membered ring and rich nitrogen and sulfur content, has grabbed the attention of chemists for decades. Whoever walks through a lab with a focus on organic synthesis might recognize its name scribbled on a glass bottle in tidy marker. Years of research shaped how scientists lean on its unique properties to shape new molecules with real-world impact.
The Medicine Cabinet: Where Chemistry Meets Healing
If you’re flipping through journals about new antibiotics or anti-tumor drugs, odds increase that you’ll spot this molecule in the recipe. It plays a key role in medicinal chemistry, because that thiadiazole ring helps scientists design compounds that interact with complicated biological targets. Some research groups found its derivatives put up a fight against bacteria that resist classic antibiotics. Chemists tweak side chains hanging off the ring to test for anti-inflammatory, anti-seizure, or even anti-tubercular power. For instance, Saxena et al. (2010) described several new molecules based on this core that showed promising results against microbial strains.
Helping Plants and Food Security
Anyone invested in agriculture will notice that plant health has become ground zero for food security. Researchers and big ag companies have flagged 2-amino-1,3,4-thiadiazole as a building block for fungicides and herbicides. Variations give farmers new tools to keep fields clear of harmful fungus or stubborn weeds. These aren't just theoretical ideas—registration filings and product sheets on the market list similar derivatives, so the gap between chemistry and the soil keeps narrowing.
Bridging Gaps in Materials Science
Away from medicine and crops, the molecule's backbone carries value for materials developers. Polymers that resist heat or chemicals draw on thiadiazole units for stability. Some electronics researchers pour time into blending these rings into organic semiconductors and sensors. By controlling electron flow and chemical reactivity, device makers beef up performance or nudge sensitivity for specific signals.
Questions About Safety and Environmental Impact
Friendship with innovation brings responsibility. Chemists who’ve worked with similar nitrogen-sulfur rings understand the red flags around toxicity and long-term environmental fate. Some derivatives break down slowly and can move through water systems or gather in soil. Academic literature points out that while these compounds enable progress, they shouldn't slip into widespread use without deep studies on impacts. Regulatory agencies look for toxicity profiles and demand checks on bioaccumulation before greenlighting new applications.
Charting a Sustainable Path
Sustainable chemistry hinges on knowing what each new tool can do—and where it might trip us up. Firms can invest in greener synthesis methods, focus on biodegradable variants, and set up real-world tracking for residues. Partnerships with academic labs help fill knowledge gaps, so both public health and the planet get a fair deal. Responsible scale-up, lifelong monitoring, and transparent data-sharing build trust, widening the net for safe and practical uses.
What is the molecular formula and structure of 2-Amino-1,3,4-Thiadiazole?
Breaking Down the Molecular Formula
2-Amino-1,3,4-thiadiazole packs a punch into a compact formula: C2H3N3S. The structure includes two carbons, three hydrogens, three nitrogens, and a sulfur atom. That’s not a random collection of atoms. In this type of scaffold, certain elements play unique roles. The thiadiazole ring brings both nitrogen and sulfur together in a five-membered backbone. One of those nitrogens, specifically at position 2, carries an amino group (NH2), and this group often lends interesting reactivity or the chance for biological activity.
The Ring That Binds: Understanding the Skeleton
The thiadiazole ring isn’t just a familiar sight in chemistry, it’s a bit like a sturdy Swiss Army knife for medicinal research. In 2-amino-1,3,4-thiadiazole, you find the sulfur and the nitrogens arranged in a five-membered ring, with the amino group hanging off the nitrogen at the second position. Here’s a quick way to visualize it:
- Five atoms form a ring — one is sulfur, three are nitrogens, one is carbon.
- The amino group attaches at position 2, right on the nitrogen.
- This creates a basic backbone for countless molecular adaptations.
You don’t need computer modeling software to get why chemists find this structure compelling. It’s flat, stable, and ready for modification. That’s why it’s so often used in drug development and pharmaceuticals. Replace that amino, or tack something onto the ring, and you get a whole new batch of properties.
Why Such a Structure Catches Attention
I remember sitting in a lab, watching as a colleague tested derivatives of this scaffold against some tough bacterial strains. 2-Amino-1,3,4-thiadiazole showed intriguing promise. It’s not alone — this ring system pops up in antifungals, anticonvulsants, and even anti-inflammatory signatures. Often, one tweak on the ring changes its function radically. That means, for drug researchers, this structure doesn’t just look good on paper. It opens doors to practical solutions in the clinic.
This kind of backbone checks several boxes: synthetic accessibility, stability, and functional handles for further chemistry. Years working alongside synthetic chemists taught me the value of a structure that can “take a beating” and still perform well in biological tests. In early screening, compounds built off 2-amino-1,3,4-thiadiazole stand out for their chemical versatility. This is how innovation really moves forward in medicinal chemistry — not with a single magic bullet, but robust platforms able to adapt.
Addressing Challenges and Looking Toward Solutions
Synthesizing this molecule in the lab isn’t too complex, but scale-up and purity sometimes pose issues. Factories need reliable access to the right precursors and careful purification steps. Another lane of challenge comes up in drug development. Just because the molecule looks great in flasks, doesn’t mean it automatically behaves in the body. Toxicity, metabolism, and actual therapeutic effect matter. Rigorous testing and thoughtful design based on real-world feedback often separate lab curiosities from working treatments.
Those aiming to unlock the potential of this molecular structure should invest in both robust analytical tools and interdisciplinary collaboration. Chemists, biologists, and clinicians can work from the same playbook if they respect both the molecule and the model systems used to test it. That’s how progress builds — not by treating new scaffolds as generic, but by paying attention to what small changes make possible in real applications.
What safety precautions should be taken when handling 2-Amino-1,3,4-Thiadiazole?
Understanding the Risks
Working in a lab holds a certain excitement, but safety always stands above all else, especially with chemicals like 2-Amino-1,3,4-thiadiazole. Even though this compound pops up in organic synthesis and pharmaceutical development, it’s not the kind of substance to take lightly. A few years ago, I watched a new colleague use insufficient protection handling a similar heterocycle—skin irritation soon followed. Those hard lessons never leave you.
Why Glove Choice Matters
Latex gloves seem handy, but aggressive chemicals like 2-Amino-1,3,4-thiadiazole call for stronger protection. Nitrile or neoprene gloves give better resistance—a necessary step since simple spillages happen even to experienced chemists. Once, in grad school, I trusted a cheap pair of gloves. The unpleasant tingling taught me to double-check glove charts and opt for quality.
Don’t Skip the Fume Hood
Dry powders and dust clouds rarely warn you before it’s too late. Breathing in particles might not cause coughing, but negative effects can unfold much later. Every time I feel tempted to skip the fume hood for “just a quick weigh,” I remember colleagues’ stories about persistent coughs and headaches. Efficient ventilation never feels like overkill; it removes hazards before they reach your lungs. Everyone in my circle agrees—no shortcut justifies bad air.
Eye Protection Isn’t Optional
Splashes from 2-Amino-1,3,4-thiadiazole sting and threaten vision. My mentor used to set his safety goggles out where he could always see them, drumming safety habits into every newcomer. A direct splash in the eye can bring a world of regret. Healthy eyes protect more than sight—they keep careers on course.
Good Practice Starts with Storage
Too many stories start with the wrong bottle on the wrong shelf. These days, every container in my lab gets a proper label, and chemicals stay stashed inside clearly marked cabinets, well away from acids or bases. Mixing incompatible substances courts disaster, both in fumes and fire. Safety, in this case, means reading Material Safety Data Sheets before ever opening a new bottle—facts save fingers and futures alike.
Immediate Action After Exposure
If skin meets compound, no need to wait—flush with running water. If it gets in eyes, a trip to the eyewash station must come before anything else. Call for help if any symptoms show up. Once, a friend delayed after a minor splash, hoping mild redness would fade. Instead, swelling followed, dragging the ordeal longer than any basic treatment would have.
Proper Waste Disposal Makes a Difference
Never pour leftovers down the drain or toss them in regular trash. Hazardous waste bins exist for a reason. Ignoring rules just builds up environmental risks, and the consequences linger long after you’ve locked up the lab for the day. Following local guidelines—no matter how tedious—prevents contamination and fines. A responsible chemist treats these steps as essential, not optional.
Culture of Safety Above All
Experience has shown me that small lapses tend to snowball—missing goggles today, missing gloves tomorrow. Mentoring new lab members means not only teaching protocols, but also sharing stories, both mistakes and close calls. Encouraging everyone to speak up for safety, and taking feedback seriously, protects more than just individuals. It shapes a safer, smarter future for the whole team.
How should 2-Amino-1,3,4-Thiadiazole be stored?
Why Safety Matters with This Chemical
Not every lab job starts with a spark, but many end in the storeroom, wondering what to do with leftover powders. I’ve seen enough to know you can never treat chemicals as just another shelf item—especially won’t with compounds like 2-Amino-1,3,4-Thiadiazole. Its structure is small, but it packs in reactivity that deserves respect. Some researchers use it to build pharmaceuticals, others poke at its biological activity, yet all share the challenge of keeping it safe and sound.
Keep Air, Light, and Moisture Away
Contamination wrecks years of research and could even put colleagues at risk. 2-Amino-1,3,4-Thiadiazole holds up best in a tight amber bottle, tucked away from sunlight. I never trust plastic stoppers for fine chemicals; glass stoppers or Teflon-lined lids build confidence the powder won’t drink up water from the air. Even a brief moment in a damp storeroom triggers clumping—or worse, slow decomposing reactions you won’t see with your naked eye.
Desiccators are not just relics—they’re reliable. Placing your bottle in one, alongside silica gel or molecular sieves, pays off. Take the bottle out, use what you need, seal it, and return it before starting anything else. Lazy habits build up residue and confusion: I label every lid and record opening dates so memory never has to do extra work.
Temperature Isn’t Just a Number
Some treat room temperature like a universal fix, but storage closets swing between hot and chilly all day. It might sound minor, but a few degrees creep up in storage near radiators or windows, cost stability fast. From experience—and guidelines—the sweet spot is a cool, dry environment somewhere around 20°C, well out of sunlight. Avoid refrigerators for chemicals prone to absorb water, unless humidity control steps in.
Some supply houses ship this compound with “store below 25°C” on their labels, and that comes from practical failures and successful runs. Hot summers, old air conditioning systems: I’ve seen labels curl and powders cake, all because storage got overlooked.
Keep Away from Acids and Oxidizers
Cabinets matter as much as bottles. Acids and strong oxidizers never belong next to 2-Amino-1,3,4-Thiadiazole. The risk of cross-contamination turns from headache to hazard if bottles break or vapors mingle. I group chemicals by compatibility—not alphabetical order—so mistakes don’t turn into emergencies. Your fire inspector or lab safety officer agrees.
Handling Spills and Waste
Planning ahead for spills avoids scrambling for gloves or goggles. I keep spill kits nearby, including neutralizing agents and clean scoops, and store waste in well-marked, tightly closed bottles. Never think one-time shortcuts are worth the minor convenience; you don’t get a second chance with chemical safety.
Real Solutions from Daily Routines
Any new chemist benefits from taking a few minutes to walk through storage protocols with experienced colleagues. I learn more from seeing where others set up their cabinets, how they label and organize, than from any manual. A tidy, well-thought-out chemical cabinet prevents accidents and saves time every day. Most accidents I’ve seen started with overlooked bottles, not runaway reactions.
Storing 2-Amino-1,3,4-Thiadiazole correctly isn’t about following boring rules—it’s about protecting your work, your team, and your own peace of mind.
What are the purity specifications and available grades for 2-Amino-1,3,4-Thiadiazole?
Where Purity Actually Matters
People often overlook what goes into sourcing a fine chemical. I’ve seen labs trip over failed reactions, only to discover contaminants in their starting materials. 2-Amino-1,3,4-thiadiazole, a compound showing up in pharmaceutical research, dyes, and agriculture, is a perfect example. Its effectiveness links directly to how pure the material inside that container really is. The playing field isn’t level for everyone—knowing what’s in your vial can make or break your downstream results.
Common Purity Specifications
The highest purity grades for 2-Amino-1,3,4-thiadiazole typically land at or above 98%, with super-clean lots hitting 99% or more. If you open up a certificate of analysis, you’ll usually see “Assay” indicating this percentage. Buyers expect to know the levels of common byproducts—think trace metals, residual solvents, or unreacted starting materials. For research and synthesis in pharma, even half a percent contamination can kill a promising reaction. Labs that settle for lower purity may face ghost peaks in their chromatograms and unexpected toxicity in screens.
Available Grades on the Market
Suppliers sell this compound in several flavors. You’ll see “technical grade” sold for more practical applications, usually checking in around 95% purity. This grade gets scooped up in agricultural or dye stuff use, where exact traceability doesn’t make or break the end product. Folks running synthetic organic processes lean toward the “analytical” or “reagent” grade, with 98% or higher purity that cuts down on side reactions. Then there’s “pharmaceutical grade,” which claims the tightest specs—not just in purity, but in trace heavy metals, moisture, and residual solvent levels. Reputable suppliers give full HPLC and NMR data so chemists can trust what’s in the jar.
Why Purity Affects Everything Downstream
I’ve run syntheses where a cheaper, lower-grade lot seemed like a deal—until we lost weeks cleaning up messes from impurities. A single contaminant can trigger toxicology red flags or mask a real result. Pharmaceutical R&D depends on this level of reliability. Nobody wants to gaze at an NMR spectrum and see surprise peaks with no known origin. More than once, my own projects hit a wall traced back to hidden residuals in the starting material.
Testing and Certification
Trustworthy suppliers don’t just guess at purity—they back it with real analysis. High-pressure liquid chromatography (HPLC), nuclear magnetic resonance (NMR), and even gas chromatography get used to verify what’s inside. I always check for data on appearance, melting point (usually around 198–202°C for the pure version), and detailed impurity profiles. Without a certificate of analysis, ordering bulk is a gamble. Some buyers push for compliance with international standards like ICH Q3A for allowable residual solvents and USP or EP monographs for active pharmaceutical ingredients.
Finding and Demanding Higher Standards
The chemistry world has changed a lot over the last decade. Regulatory crackdowns, especially in pharmaceuticals, now force manufacturers to offer full transparency. End-users can call out red flags and request re-testing. More organizations request audit trails for their chemical sourcing. In my own practice, reaching out to ask for supporting data before a purchase has saved headaches down the line, especially when the compound is bound for an investigational drug batch or critical pilot run.
The Next Step: Smarter Buying Habits
Anyone serious about research quality watches the supply chain closely. Only buying from sources that lay out detailed purity specs and testing reports helps keep projects on track. Screening new suppliers with small test orders uses real-world performance to weed out inconsistencies. Building relationships with suppliers who stay responsive and back up their labels has made a difference in every research team I’ve worked with. That’s how the best labs keep surprises out of their experiments—and out of their data.
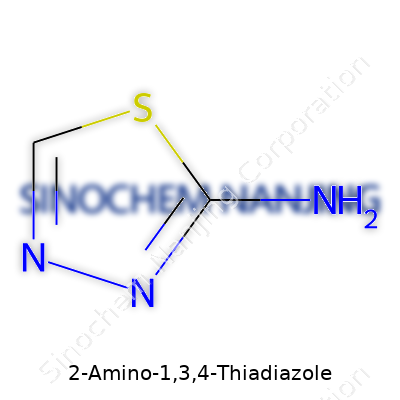
| Names | |
| Preferred IUPAC name | 2-amino-1,3,4-thiadiazole |
| Other names |
2-Aminothiadiazole Aminothiadiazole 1,3,4-Thiadiazol-2-amine |
| Pronunciation | /tuː-əˈmiːnoʊ-wʌn-θaɪ.əˈdaɪ.əzoʊl/ |
| Identifiers | |
| CAS Number | 2710-70-1 |
| 3D model (JSmol) | `3D model (JSmol)` **string** for **2-Amino-1,3,4-Thiadiazole**: ``` CC1=NN=C(S1)N ``` *(This is the SMILES string representation for use in 3D molecular visualization tools like JSmol.)* |
| Beilstein Reference | 120224 |
| ChEBI | CHEBI:18911 |
| ChEMBL | CHEMBL18706 |
| ChemSpider | 25992 |
| DrugBank | DB08375 |
| ECHA InfoCard | 03d1a7cd-3535-4cc9-8d1d-6e83c53ee960 |
| EC Number | 207-712-5 |
| Gmelin Reference | 84159 |
| KEGG | C06565 |
| MeSH | D017905 |
| PubChem CID | 11580 |
| RTECS number | XJ7200000 |
| UNII | 4WB2L39A2K |
| UN number | 2811 |
| Properties | |
| Chemical formula | C2H3N3S |
| Molar mass | 87.12 g/mol |
| Appearance | White to off-white solid |
| Odor | Odorless |
| Density | 1.47 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | -0.35 |
| Vapor pressure | 1.35E-04 mmHg at 25°C |
| Acidity (pKa) | 2.45 |
| Basicity (pKb) | 7.89 |
| Magnetic susceptibility (χ) | -77.0e-6 cm³/mol |
| Refractive index (nD) | 1.708 |
| Viscosity | Viscous liquid |
| Dipole moment | 3.98 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 178.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 87.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4730 kJ mol-1 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes serious eye irritation. Causes skin irritation. May cause respiratory irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS06 |
| Signal word | Warning |
| Hazard statements | Harmful if swallowed. Causes serious eye irritation. Causes skin irritation. May cause respiratory irritation. |
| Precautionary statements | P261, P264, P270, P271, P301+P312, P304+P340, P308+P311, P501 |
| NFPA 704 (fire diamond) | 2-1-0 |
| Flash point | > 168°C |
| Autoignition temperature | 515 °C |
| Lethal dose or concentration | LD50 (oral, rat): 1620 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 1200 mg/kg |
| NIOSH | LT2625000 |
| PEL (Permissible) | Not established |
| Related compounds | |
| Related compounds |
1,3,4-Thiadiazole 2-Mercapto-1,3,4-thiadiazole 2-Amino-5-mercapto-1,3,4-thiadiazole 2-Amino-1,3,4-oxadiazole 2-Amino-1,3,4-thiadiazole-5-carboxylic acid |

