2-Acetylpyrrole: Unpacking a Classic Heterocycle
Historical Development
The story of 2-acetylpyrrole begins in the wave of curiosity and technical skill that marked the early golden age of organic chemistry. Chemists searching for the secrets of bread crusts, roasted coffee, and tobacco smoke eventually noticed this small molecule showing up again and again. The first isolation traces back to the late 19th and early 20th centuries, when researchers tracked down structures behind the flavors that give cooked food its appeal and started applying distillation and extraction to separate these volatile compounds. In university labs, 2-acetylpyrrole served as an example that links Maillard chemistry to flavor and fragrance in daily life, driving the practical side of organic molecule research. These early discoveries gave industry both a new appreciation for food science and access to versatile synthetic building blocks that kept chemical pioneers busy for decades.
Product Overview
2-Acetylpyrrole belongs to the small club of simple pyrrole derivatives with a noticeable impact outside chemistry labs. It shows up as a white to pale yellow crystalline solid, offering a nutty, bready, popcorn-like smell, a profile that plays crucial roles in both flavoring and advanced chemical synthesis. Manufacturers use 2-acetylpyrrole as a key ingredient in food flavorings, tobacco modifications, and even perfumes. Labs regularly reach for it as a chemical precursor—thanks to its reliably reactive structure. Connoisseurs of whisky, coffee, and baked goods can unknowingly thank this compound for some of the comfort and complexity found in familiar tastes.
Physical & Chemical Properties
This compound has a molecular formula of C6H7NO, with a molecular weight of 109.13 g/mol. 2-Acetylpyrrole melts at around 50-53°C and boils at 260-264°C, which gives it suitable versatility from a handling and storage perspective. It dissolves well in ethanol and ether, but water doesn’t do the job as well due to the molecule’s limited hydrogen bonding capacity. The functional group at the second carbon—the acetyl group—flips the molecule’s reactivity compared to pyrrole itself, creating new avenues in chemical manipulation. Its odor threshold is impressively low, explaining why even trace amounts become noticeable in both industrial and food contexts.
Technical Specifications & Labeling
Producers rely on high-purity grades, generally seeking content above 98.5% as measured by gas chromatography. Labeling must include CAS number 1072-83-9, hazard pictograms that note its low flammability, and details on storage away from strong acids and oxidizers. Certificates of analysis routinely specify melting and boiling ranges, residual solvents, and possible impurities such as related pyrroles or acetic acid derivatives. Packaging favors light-resistant, air-tight containers, reflecting both the molecule’s volatility and susceptibility to oxidative changes, which can degrade both aroma and reactivity.
Preparation Method
Synthesizing 2-acetylpyrrole takes several routes, but one robust method stands out: acylation of pyrrole using acetic anhydride in the presence of a Lewis acid like AlCl3. This Friedel–Crafts acylation targets the second position because pyrrole’s electronic structure encourages substitution next to nitrogen, especially under mild control. There’s a fascination in the balance chemists strike—too much heat or too strong a catalyst can generate unwanted byproducts. For large-scale commercial needs, continuous-flow reactors provide better control and yield, while laboratory-scale chemists often favor batch reactions, air cooling, and rapid extraction to minimize side-reactions.
Chemical Reactions & Modifications
The acetyl group at the 2-position makes this molecule a classic launching point for further chemical innovation. Reductive cleavage with agents like lithium aluminum hydride leads to the corresponding ethylpyrrole. Electrophilic halogenation at the 3- or 5-positions offers a path to functionalized intermediates, often used for medicinal or advanced materials research. Oxidation, particularly under strong acid, can break the pyrrole ring, releasing smaller molecules important in flavor chemistry. Condensation reactions with aldehydes or acid chlorides broaden the scope further, enabling synthesis of libraries of heterocyclic compounds with diverse sensory or pharmacological value.
Synonyms & Product Names
Chemists and food scientists might know 2-acetylpyrrole by several other names: 1H-Pyrrol-2-yl methyl ketone, alpha-acetylpyrrole, and methyl 2-pyrrolyl ketone. In various product catalogs, listings might use “pyrrol-2-yl methyl ketone” or even “popcorn ketone,” drawing on its role in flavor design. Regulatory documents and toxicology studies keep to the International Union of Pure and Applied Chemistry (IUPAC) naming convention, with standardized identifiers and trade labels reflecting this strictness.
Safety & Operational Standards
Good manufacturing practice insists on diligent risk labeling and safe handling. Although not particularly toxic in small quantities, 2-acetylpyrrole causes mild irritation to skin and eyes. Proper ventilation remains important during synthesis; the molecule’s volatility can make it easy to inhale vapors, leading to slight respiratory discomfort. Lab staff don gloves and goggles during weighing, dilution, and purification—industry never ignores these precautions, as carelessness can lead to unnecessary exposure. Storage far from open flames or reactive chemicals protects product quality and keeps workspaces hazard-free. Safety Data Sheets (SDS) require routine review every time new handling or packaging methods come into play.
Application Area
Food scientists use 2-acetylpyrrole to inject roasted, baked, or nutty notes into bread, crackers, coffee, chocolate, and tobacco products. It also earns a spot in synthetic fragrances, mimicking the inviting scent of fresh popcorn or bread crust. Pharmaceutical researchers appreciate 2-acetylpyrrole’s easy chemical manipulation; the molecule provides a backbone for antifungal, antibacterial, and anti-inflammatory drug candidates. Materials chemists utilize it for polymer and sensor design, since its aromatic core and reactive sites offer solid versatility for tuning molecular properties. Outside the lab, this molecule sits in many homes, often unrecognized, built into the everyday smells and tastes that define comfort and luxury.
Research & Development
Current research shifts between optimizing synthesis—reducing waste, scaling yields—and mapping new uses in flavor and drug discovery. One promising trend includes exploration of biocatalytic routes, using engineered enzymes to forge and modify the molecule under milder conditions. Computational chemistry now enables teams to predict which derivatives might deliver stronger aroma or improved therapeutic effects, cutting down the costs and risks of trial-and-error benchwork. Collaborative projects across chemical and food science divisions tease apart molecular mechanisms, asking why low doses enhance taste so powerfully or how small tweaks shift pharmacological properties.
Toxicity Research
Studies to date put 2-acetylpyrrole in the “low toxicity” category by most regulatory benchmarks. Rodent trials show modest adverse effects only at high exposures, with no evidence of chronic toxicity or carcinogenicity in standard models. Extensive metabolic profiling finds rapid breakdown by liver enzymes and quick clearance from bloodstreams. Food safety authorities put forth permissible exposure levels based on clear trends from both animal and in vitro studies. Still, ongoing monitoring matters, especially as newer applications reach consumer markets—regulators insist on fresh data tracking inhalation, skin contact, and any possibility of cumulative effects in long-term workplace scenarios.
Future Prospects
2-Acetylpyrrole occupies an unusual strategic spot: reliable for food and fragrance, and flexible enough for specialty chemical innovation. With greater interest in clean-label, natural-sounding ingredients, chemical firms see value in pursuing biosynthetic production, using fungi or engineered bacteria to produce the molecule from simple sugars rather than petrochemical feedstocks. Emerging fields like green chemistry lean into solvent-free or low-energy synthesis, opening up more sustainable business models. Expect future patents to focus on tailored derivatives for designer tastes, more potent bioactive agents, and hybrid materials with engineered sensory or functional characteristics. Local and global regulations will shape adoption rates, but scientific momentum shows no sign of letting up, as the intersection of tradition and innovation keeps 2-acetylpyrrole on everyone’s radar.
What is 2-Acetylpyrrole used for?
Unlocking Flavors in Food Science
Walk into a bakery and breathe deeply. That toasty, slightly nutty aroma floating from the kitchen might have a secret: 2-acetylpyrrole. This small molecule packs a punch in flavor creation. Food scientists prize it for mimicking the smell and taste of freshly baked bread, roasted nuts, and even chocolate. You’ll spot its name on the ingredient list of baked products, coffee flavors, and some brands of cereal. In a world chasing plant-based meats, nothing gets taken for granted about mimicking “roast” or “grill.” 2-acetylpyrrole lends a hand here too, bringing out satisfying finish in meat alternatives.
This isn’t just about creating cravings. People expect specific taste experiences, especially in countries with coffee or bakery traditions running deep. If a substitute product falls short on aroma, shoppers won’t come back. The psychological impact a familiar scent leaves on us—triggering nostalgia, comfort, even joy—drives the food industry to rely on chemicals like 2-acetylpyrrole for that finishing touch.
Enhancing Scents in the Fragrance World
Not only does this compound shine in baking aisles, but it also lends complexity to the perfume world. Perfumers reach for 2-acetylpyrrole when creating “warm” blends. No one likes a flat scent. Notes bringing to mind things like hazelnuts, bread crust, or praline elevate luxury fragrances and high-end candles. This isn’t a new trick, either. Generations of fragrance developers have turned to this molecule for its ability to soften sharp odors and bring out that much-loved “gourmand” trend in perfumes.
Gourmand scents have exploded in popularity over the last decade. If someone’s ever complimented your perfume for smelling like cookies just out of the oven or roasted almonds, there’s a good change 2-acetylpyrrole played a role.
Fine Chemical and Pharmaceutical Roles
Looking at a laboratory shelf, you’ll probably see sample bottles of 2-acetylpyrrole stacked among other niche reagents. Synthetic chemists use it as a building block. Its five-membered ring structure with a nitrogen atom makes it valuable in forming heterocyclic compounds, common ingredients in drug design or new material creation. Research on potential pharmaceuticals, including anti-inflammatory and anti-cancer agents, sometimes starts with this base. Not every scientific path leads directly to medicines, but every new starting point like this opens possibilities.
Linking all these uses are strict quality controls. The chemical industry follows rigid purity standards and tracks the supply chain closely. Buyers expect suppliers to provide detailed analytics for every batch. Trust gets built not only through transparent sourcing but also regular regulatory checks. For companies, repeated investment in documentation and laboratory testing pays off when industries like food or perfume use 2-acetylpyrrole. Bad batches lead to recalls, wasted money, or worse—making headlines for all the wrong reasons.
Future Challenges and Sustainability
Demand for transparency and ethically sourced ingredients grows louder. Some companies have shifted to bio-based versions of 2-acetylpyrrole, using fermentation instead of petrochemical synthesis. The goal: lower carbon footprints, tight traceability, and ingredients free of suspected contaminants. Not all methods of sourcing sit on equal footing—industry insiders debate cost, performance, and whether bio-based always brings a smaller environmental bite. Consumers push this debate forward every time they ask questions about what goes into their food or fragrance.
Aromatic chemicals aren’t household names, but their impact touches our senses every day. Companies and researchers who handle 2-acetylpyrrole shoulder the responsibility to keep their work safe, sustainable, and enjoyable for those savoring every bite and breath.
What is the chemical structure of 2-Acetylpyrrole?
Looking at 2-Acetylpyrrole Up Close
Anyone who’s spent time in a lab, or even around a good bakery, knows the importance of small molecules with big character. 2-Acetylpyrrole proves that point. This compound, just like bread crust or roast coffee, brings a smell people associate with warmth and richness. Tucked into that delicious aroma is a handful of carbon, hydrogen, nitrogen, and oxygen atoms organized in a precise way scientists call C6H7NO.
On paper, 2-acetylpyrrole looks pretty simple: a five-membered ring called pyrrole holds a nitrogen atom at one corner, and an acetyl group latches onto the ring at the second position. That "2" in its name signals exactly where the acetyl group attaches. Chemically, the ring balances both aromatic character and a reactive position where the acetyl group sits. Chemists draw it out as a pentagon with an NH at the top, double bonds humming around the edges, and a CH3CO– group coming off the next carbon beside the nitrogen.
Flavor Chemistry and Real-World Value
I once worked with a flavorist who described how subtle tweaks in a molecule made or broke a new snack release. 2-Acetylpyrrole isn’t just a textbook curiosity—it’s part of life’s daily pleasures. It pops up in roasted foods, baked goods, and a decent cup of chicory coffee. Its structure gives it that roasted-bread-like scent. The acetyl group helps push this aroma forward, while the pyrrole ring roots it, keeping the scent from flying off too quickly. That balance lets chefs and manufacturers use tiny amounts to create big sensations.
Food science builds careers and companies around these molecular details. Research from the Journal of Agricultural and Food Chemistry reports that 2-acetylpyrrole forms when amino acids and sugars meet heat, as in the Maillard reaction—a dance of atoms creating flavor complexity. Understanding the structure unlocks the secret to synthesizing it more efficiently or controlling its production in roasting and baking.
Beyond Taste—Safety and Synthesis
Real-world chemistry always requires a closer look. Safety profiles and regulatory notes matter. The molecular structure of 2-acetylpyrrole means it’s not seen as highly toxic at low concentrations, but everything comes down to exposure. That’s key in flavor labs, where micrograms shape experience but milligrams raise flags.
On the synthetic front, the structure guides industrial production. Chemical suppliers have learned how to make 2-acetylpyrrole from simple building blocks, often using reactions that join an acetyl source with a pyrrole ring under mild conditions. The core structure gives clues for these methods, informing both yields and purity. Efficiency in synthesis limits waste—a big deal for anyone concerned both about cost and environmental impact.
What Researchers and Regulators Can Do
Clear structure maps help researchers track flavor molecules from raw ingredient through final product. Food safety authorities such as the FDA and EFSA use detailed structure data to set guidelines for allowable levels in food. Transparency from manufacturers, combined with strong research on exposure and breakdown products, keeps the public safe without stifling creativity in food design.
Better tools for analyzing chemical structures—faster chromatography, high-resolution mass spectrometry—help companies stick to safe dosages and keep flavors true to tradition. Investment in public research lets small producers understand what the giants already know, helping to keep quality up across the board. Structure isn’t just about atoms on a page; it’s about making daily life just a little richer—or safer—for everyone at the table.
Is 2-Acetylpyrrole safe to handle?
What is 2-Acetylpyrrole?
2-Acetylpyrrole shows up as a useful chemical in labs and industries, sporting a mild roasted, popcorn-like aroma. This molecule pops up in flavor additives and fragrances, making treats smell and taste richer. To someone working outside the lab, it seems harmless, especially given its common uses. Digging into the safety data sheets reveals a different story, and the real question turns out less about how delicious it smells, and more about what kind of care it deserves in the workplace.
Real Hazards in a Small Package
At first glance, 2-Acetylpyrrole lacks red flags seen in nastier industrial chemicals. The substance can irritate skin, eyes, and the respiratory tract, and likely causes more issues if handled frequently without good habits. Over the years working in labs, I’ve seen smart people get complacent with chemicals that don’t bite immediately. Handling powders and liquids like 2-Acetylpyrrole without gloves or eye protection feels like a shortcut until redness, rashes, or burning eyes show up.
According to PubChem and the Sigma-Aldrich safety sheets, direct contact causes irritation. Prolonged or repeated exposure may dry out or crack skin. The vapors, especially in closed areas, bring on headaches, dizziness, or worse. Many smaller labs skip sophisticated ventilation or robust masking because the smell feels “food-like.” That’s a dangerous assumption. People trust their noses too much—even pleasant-smelling chemicals hurt you if inhaled in quantity. Food-grade ingredients follow standards for purity, and not all 2-Acetylpyrrole ends up in consumable goods. Industrial grades sometimes contain other contaminants not suitable for tasting or sniffing.
Gloves and Goggles: Basic Training
No one in my experience ever regretted using gloves, goggles, and a lab coat. If you splash 2-Acetylpyrrole into your eyes, flush with water and seek medical help. Always work with this kind of reagent under a fume hood if it gets warm or stirred so fumes do not build up. Respiratory symptoms sometimes happen after a single encounter in a tight space; a good extraction system makes a difference. My old lab kept simple, sturdy protocols for everything, and skin checks caught problems early. Shortcuts invite issues, and even the “safe” chemicals remind you who’s boss in a poorly ventilated, busy work room.
Training Beats Assumptions
No senior tech lets newcomers fumble with new bottles unsupervised. I trained with folks who made me repeat safety checklists every time until it became automatic. The “just this once” pattern gets people sent to first aid, or occasionally the emergency room. I wish more workplaces took reporting minor symptoms seriously instead of chalking up headaches or rashes to unrelated causes. Solid safety culture grows from clear training and routine reminders, not flashy posters in the staff kitchen.
Solutions That Actually Work
Best results come from consistent practice: gloves for contact, goggles for splashes, and fume hoods for vapor. Only handle what you need, label everything, and keep chemicals sealed when not in use. Stay alert for odd smells or spills, and clean up right away. If symptoms show up, speak up early. Even a mild irritant turns ugly if ignored. People who use 2-Acetylpyrrole smartly rarely find trouble, but arrogance or laziness around chemicals invites it. In a busy workplace, these habits free your mind to focus on the real science, instead of regretting a careless mistake.
Where can I buy 2-Acetylpyrrole?
Purpose Drives the Search
Most people on the hunt for 2-Acetylpyrrole either work in research or food product development. Its toasty, nutty aroma gives chocolates, baked goods, and coffee a punch of roasted flavor. I remember visiting a flavor house in New Jersey years back — the chemists there raved about the delicate complexity a dash of this compound can add. But 2-Acetylpyrrole isn’t stocked at the local pharmacy or grocery store. The chemical sits on the shelves of specialty suppliers who serve industry pros, universities, and sometimes culinary experimenters.
Where to Buy
If you need this molecule, head toward companies like Sigma-Aldrich, TCI America, and Alfa Aesar. These suppliers handle bulk and lab-scale requests. A lot of independent researchers or craft food makers work with smaller distributors such as Oakwood Chemicals or Amadis. Depending on the country, you might see stricter policies — some nations ask for documentation proving legitimate research or manufacturing use.
One quick Google search brings up e-commerce sites and chemical marketplaces. You’ll see plenty of sellers touting purity levels and competitive pricing. Here’s the catch: safety and authenticity should take top priority, no matter how fast or cheap a deal seems. Sketchy suppliers sometimes cut products with contaminants, or sell counterfeit ingredients. Buying direct from established chemical vendors gives peace of mind, especially if the batch lands in food or fragrance products.
Know the Rules
I’ve walked trade shows filled with alarmed conversations about regulatory pressure. Food law varies by country— the US Food and Drug Administration lists 2-Acetylpyrrole as generally recognized as safe (GRAS) for flavor use in small quantities. Europe and many other markets treat it much the same, but always check specific thresholds. Safety sheets matter here. Any professional handling this compound should read the info provided by the seller — from handling tips to exposure limits.
Safety Isn’t Optional
Handling even small amounts, gloves and proper ventilation matter. Working in a commercial kitchen, one learns fast that synthetic flavors need careful respect. Flammable and potentially hazardous under the wrong circumstances, 2-Acetylpyrrole demands basic lab discipline. I’ve seen bright, knowledgeable folks rush through protocols, only to end up with ruined batches and wasted money.
Finding a Reliable Source
Trusted chemical suppliers keep certifications on file. The certificate of analysis gives a snapshot of the lot’s quality. A supplier worth their salt answers questions, whether about ingredient origins or allergens. For small businesses or culinary hobbyists, finding a broker to navigate paperwork and minimum orders often makes life easier. Many universities maintain accounts with larger vendors, streamlining the process for researchers.
If a price seems too good to trust, it probably is. Years ago, a friend ordered what she thought was food-grade vanillin from a gray-market supplier to save a buck; it arrived with odd odors and a mystery powder mixed in. She learned the hard way that cutting corners in sourcing can wreck a project and threaten safety.
Looking Ahead
As more kitchens experiment with authentic flavors, and as product developers chase new sensory experiences, demand for aroma actives like 2-Acetylpyrrole continues. Transparency grows more important as consumers ask where ingredients come from. The future lies in relationships: industry pros working closely with trusted suppliers, food regulators keeping an open dialogue, and consumers willing to learn about what lands in their food.
What are the storage conditions for 2-Acetylpyrrole?
Getting the Basics Right
Anyone who’s spent time around organic chemicals knows how crucial simple, everyday storage habits can be. 2-Acetylpyrrole—used often in food flavoring and fragrance labs for its nutty, almost bready aroma—calls for a bit of respect on the shelf. It’s a small molecule but far from indestructible. You leave it exposed and you may find yourself losing both potency and safety in the blink of an eye.
Keep It Cool, Keep It Dry
Day-to-day, room temperature sometimes seems good enough for so many reagents. Not here. Moisture in the air and a bit of warmth make 2-Acetylpyrrole degrade or oxidize faster. Every experienced hand in the lab knows a refrigerator, set in the 2°C to 8°C range, stretches shelf life and keeps surprises at bay. You pop that bottle in the fridge, away from any source of heat, and you avoid trouble—simple rule, real payoff.
Seal and Shield From Air
Oxygen does more than rust iron. I’ve seen 2-Acetylpyrrole turn less effective just from loose caps and careless handling. If you want to keep each gram potent, reach for airtight containers. Screw tops that seal tight, tamp those down after every use, keep out air. Even better—add a layer of inert gas like nitrogen to the headspace for the bulk stuff, a trick many chemical stores use to slow down any breakdown that starts where liquid meets air.
Light-Proofing Matters
It’s easy for a lab to get busy. Bottles sit under harsh fluorescent lamps, day after day. But 2-Acetylpyrrole doesn’t love the light. Ultraviolet rays especially can start breaking down the structure that makes it valuable. Most reputable suppliers deliver it in amber glass, and there’s a reason for it. If you repackage the compound, keep those dark bottles. If you can’t, stash it in the back of a drawer or closed cabinet. Little acts like these save budget and batch quality, week after week.
Zeroing In on Contamination Risks
Cross-contamination in lab storage gets ignored until something goes wrong. Impurities—dust, bits of earlier samples, even skin oils if you aren’t careful—can end up in the bottle and change both odor and behavior. Use clean spatulas, avoid pouring from the original container whenever possible, and label containers clearly. It looks like overkill, but one ruined experiment or client batch quickly changes your mind.
Putting Safety in Daily Routine
Safety always comes first, and 2-Acetylpyrrole storage is no exception. The compound may not be the most hazardous on the shelf, but skin contact and vapor inhalation cause irritation. Work in a ventilated area, wear gloves, and store it far from oxidizers or acids to avoid unwanted reactions. Standard chemical safety—familiar to any seasoned technician—applies here without shortcuts.
Wrapping Up by Staying Attentive
Proper storage isn’t only about following a set of instructions; it’s everyday diligence. Every time I pick up a bottle, I remind myself that small missteps can cost money and safety for the team. Keeping 2-Acetylpyrrole fresh and safe turns on habits: cool, dry, dark, sealed, and clean. Real-world experience says nobody regrets the extra care, and neither will you.
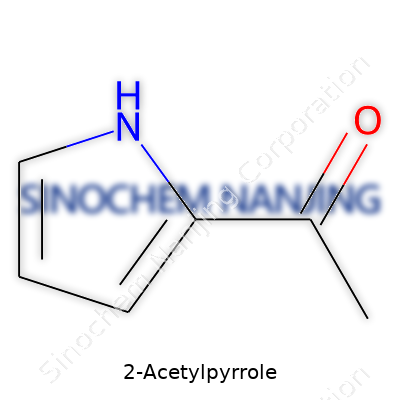
| Names | |
| Preferred IUPAC name | 1-(1H-pyrrol-2-yl)ethan-1-one |
| Pronunciation | /tuː əˈsiːtɪl pɪˈroʊl/ |
| Identifiers | |
| CAS Number | 1072-83-9 |
| Beilstein Reference | 104893 |
| ChEBI | CHEBI:73021 |
| ChEMBL | CHEMBL151610 |
| ChemSpider | 54623 |
| DrugBank | DB03813 |
| ECHA InfoCard | 100.010.263 |
| EC Number | 875-06-3 |
| Gmelin Reference | 7804 |
| KEGG | C02378 |
| MeSH | D010003 |
| PubChem CID | 7030 |
| RTECS number | UJ4375000 |
| UNII | X301W5KQ98 |
| UN number | UN2810 |
| Properties | |
| Chemical formula | C6H7NO |
| Molar mass | 109.13 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | nutty; popcorn; bready |
| Density | 1.082 g/mL at 25 °C |
| Solubility in water | Slightly soluble |
| log P | 0.37 |
| Vapor pressure | 0.0425 mmHg (25 °C) |
| Acidity (pKa) | pKa = 23.5 |
| Basicity (pKb) | 7.0 |
| Magnetic susceptibility (χ) | -7.97 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.541 |
| Viscosity | 2.24 mPa·s (25 °C) |
| Dipole moment | 2.94 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 183.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | –38.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2267 kJ/mol |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and eye irritation, may cause respiratory irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302 + H312 + H332: Harmful if swallowed, in contact with skin or if inhaled. |
| Precautionary statements | P264, P280, P302+P352, P305+P351+P338, P362+P364, P332+P313 |
| NFPA 704 (fire diamond) | 2-1-0 |
| Flash point | 107°C |
| Autoignition temperature | 220 °C |
| Explosive limits | 7.2–14.2% |
| Lethal dose or concentration | LD50 Oral Rat 4930 mg/kg |
| LD50 (median dose) | LD50 (median dose): 2000 mg/kg (oral, rat) |
| NIOSH | UJ4375000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 2 mg/m³ |
| Related compounds | |
| Related compounds |
2-Formylpyrrole 3-Acetylpyridine 2-Acetylthiophene 2-Acetylfuran |

