Looking Deeper into 2,6-Xylenol: More than Just a Chemical
Historical Roots and the Road to Today
Chemical breakthroughs shape all sorts of industries, but a lot of people don’t know the backstories behind substances like 2,6-xylenol, also called 2,6-dimethylphenol. The roots of xylenol chemistry go back to the late 19th century, when advances in coal tar distillation and synthetic organic chemistry produced a wave of new molecules. Synthetic phenols spun off as more than curiosities—they found uses in everything from medicine to plastics. 2,6-xylenol grew out of this family, riding the momentum of global industrialization. By the middle of the 20th century, it carved out a place in electrical insulation, resin manufacture, and specialty polymers. The historical path sharpened as demand for reliable insulating materials increased, and companies sought chemicals that delivered the right mix of thermal stability and flexibility. The development wasn’t just about raw scientific curiosity—it was also about practical needs in manufacturing, energy, and electronics.
Product Overview: Understanding What Sets 2,6-Xylenol Apart
2,6-Xylenol stands out because of its straightforward structure—a benzene ring with two methyl groups at the 2 and 6 positions, and a hydroxyl replacing one of the hydrogens. This small adjustment leads to physical and chemical properties that stack up differently from other phenol derivatives. If you’ve ever watched lab techs debate over which phenol is right for a job, you’ll notice how much mileage comes from these side groups. 2,6-xylenol draws attention for how it manages heat and its role in creating polymers that don’t break down so easily under stress. It’s no huge mystery why people in resin and insulation industries keep it near the top of their shop lists.
Physical & Chemical Properties: The Building Blocks of Application
From a straight scientific point of view, 2,6-xylenol presents itself as a white or off-white crystalline solid at room temperature, melting down at a bit more than 110°C and boiling suspiciously close to 210°C. It brings a distinct phenolic smell that no trained nose mistakes. Where regular phenol dissolves pretty handily in water, 2,6-xylenol doesn’t—its methyl groups crowd around the ring and resist breaking loose. You get better luck with organic solvents like alcohol, ether, or benzene. If you care about stability, this chemical teaches you something: those methyl groups block access, making the molecule less likely to overload itself with unwanted side reactions. This trait turns out to be gold for a chemist working with high-temperature processes or long-life materials. The technical specification sheets usually focus on purity, moisture levels, and melting point, but real-world batches sometimes reveal small impurities from the production process—a reminder that chemistry doesn’t always dance to our rules.
Technical Details and Labeling: Management in the Lab and Industry
In the wild world of labeling and shipping, keeping track of chemicals like 2,6-xylenol means reading Material Safety Data Sheets and regulatory guidance closely. Labels mention the substance as 2,6-dimethylphenol, with clear warnings about possible hazards. Industry veterans learn to check lot purity, especially if the end use tolerates only a slim margin for error, like in electronics or pharmaceuticals. In shipping and handling, tight standards apply—a compound that drifts close to phenol in structure won’t get treated like sugar. You see strict guidelines on container selection, temperature regulation, and storage away from acids, oxidizers, or strong bases.
Preparation Method: From Raw Material to Useful Product
Synthesis of 2,6-xylenol doesn’t happen by accident. The classic path starts from m-xylene, running it through hydroxylation reactions or using catalytic processes that encourage methyl groups to take root in just the right spots. Industrial practice moves away from the old harsh conditions and looks for safer, atom-efficient techniques to cut waste and energy use. Companies lean toward metal-catalyzed oxidative hydroxylation, where you tweak reaction temperature and pressure to squeeze out the best yield. Chemists who lived through production scale-ups will tell you: nothing goes exactly as the textbooks claim. System tweaks, optimized purification, and real-time quality monitoring often draw the line between financial success and an expensive headache.
Chemical Reactions and Modifications: Expanding the Toolset
2,6-Xylenol isn’t just a dead end compound. Chemists value its active hydroxyl position and those resilient methyl groups—traits that open doors for further chemical transformations. In industry, the most famous route takes 2,6-xylenol as the key feedstock for making polyphenylene oxide (PPO), a polymer that revolutionized high-end electrical insulation thanks to its resistance to heat and corrosion. Nitration, alkylation, and halogenation reactions allow for a spread of derivatives, each carving out different roles in things like dyes, agricultural chemicals, pharmaceuticals, and advanced materials. These follow-on reactions excite researchers who see possibilities for materials that do more than just stand up under pressure—they last longer, resist flame, and have tailor-made electrical properties.
Synonyms and Everyday Product Names: Speaking the Language
Scientists know 2,6-xylenol by a spread of names depending on the context: 2,6-dimethylphenol, 2-hydroxy-m-xylene, and even DMA phenol slip into papers and supplier lists. For anyone trying to order a drum or interpret a regulatory filing, catching all these synonymous terms becomes key. The multiplicity of names can trip up less-experienced staff, risking mix-ups or exposure to the wrong compound.
Safety and Operational Standards: Balancing Utility and Risk
Personal experience working with phenolic compounds brings a healthy respect for the safety sheet. 2,6-Xylenol, like its relatives, can wreak havoc if handled carelessly—it irritates skin, damages mucous membranes, and brings toxicity worries with chronic exposure. Lab teams take no shortcuts on gloves, shields, and ventilation. Facilities invest in containment, first-aid procedures, and regular staff briefings, recognizing the chemical's acute risks. In manufacturing, automated dosing and closed-loop transfer systems reduce direct contact, which lowers incident rates compared to open-handling practices years ago. Legislation and international standards like REACH in Europe drive producers to document every step and reassess risks periodically.
Where 2,6-Xylenol Makes a Mark: Application Areas
Most people may never see 2,6-xylenol in raw form, but they interact with products based on its derivatives every day. Electrical and electronic parts built with polyphenylene oxide insulation break the cycle of overheating and short-circuiting. Industrial resin manufacturers rely on it for composite laminates that reinforce wind turbines or automotive components. Certain specialty adhesives owe their stickiness and strength to variants made from this base molecule. In agriculture, it’s sometimes a crucial intermediate for pesticides that need to last in the field, but not persist in harmful ways. The cosmetics and pharmaceutical sectors run more tests before using phenolic chemicals, but research crops up linking newly engineered derivatives to antimicrobial and preservative properties.
Innovation and the Push Forward: Research and Development
The conversation around 2,6-xylenol never stands still. Research groups look for greener, safer production methods. Up-and-coming approaches highlight renewable feedstocks and metal-free catalytic cycles for sustainability. On the product side, tweaking the molecule’s basic framework may lead to high-performance polymers that survive even tougher environments, like spacecraft or offshore platforms. Some R&D circles hunt for medical applications—think antibacterial surfaces for hospitals or smarter preservative systems in personal care. People often talk about “next-generation” phenolic resins that capture the downsides of the first wave—lower toxicity, less environmental baggage, greater recycling potential.
Toxicity Research: A Responsible Approach
Toxicology keeps the industry honest. Decades of testing show that 2,6-xylenol brings moderate acute toxicity in animals, particularly through inhalation or ingestion. Chronic exposure reveals risks to liver and kidney function, and researchers flag possible reproductive effects after long-term experiments. Environmentalists worry about its persistence in water sources if discharge isn’t tightly controlled, which can disrupt aquatic life cycles. Modern toxicological work aims for deeper understanding and more accurate risk prediction, pointing toward stricter workplace exposure limits and advances in waste treatment. For industries committed to social responsibility, running tighter controls becomes part of earning public trust—and regulatory leeway.
The Road Ahead: Future Prospects
Looking beyond the basic science and factory floor, it’s clear that 2,6-xylenol will play a role in the next era of advanced materials. As the world asks for cleaner technologies, researchers are nudging phenolic chemistry toward lower emissions, safer intermediates, and more recyclable end-products. The march toward a circular economy pressures old-school chemical practices to adapt—no more turning a blind eye to hazardous by-products or energy waste. Industry-watchers expect changes in process design, tighter integration of waste management, and sharper incentives for companies that go beyond compliance. For people on the ground, that means a chemistry that keeps pace not just with market demand, but with society’s deeper questions about safety, health, and long-term impact.
What is 2,6-Xylenol used for?
Life Behind the Lab Door
Walk through a chemical plant with enough time, and you’ll catch a curious smell—the faint, sharp tang of 2,6-Xylenol. Fewer folks talk about it in their everyday lives, yet this chemical shapes plenty of products people touch or use without thinking. I spent some hot summers in a factory setting, watching workers blend this compound in stainless tanks, safety goggles fogged up from the steam. It’s not a glamorous world, but it’s honest work, and 2,6-Xylenol’s value goes far beyond the chemistry books.
Building Blocks for Industry
2,6-Xylenol, also called 2,6-dimethylphenol, doesn’t show up on grocery store shelves. Instead, it acts as a kind of utility player in manufacturing. One of its main jobs lies in helping make phenolic resins—those tough, heat-resistant plastics you spot in circuit boards, adhesives, and brake linings. Next time your car stops smoothly on a rainy day, some credit goes back to this chemistry, even if nobody gets a parade for it.
It goes into engineering plastics like polyphenylene oxide (PPO) as well. These plastics are workhorses in automotive parts, electronics casings, and even water filtration tools. Polyphenylene oxide doesn’t warp under heat, nor does it soak up water like a kitchen sponge. The process that turns 2,6-Xylenol into this kind of tough plastic looks simple in textbooks, but reality means layers of safety gear, temperature checks, and constant monitoring. The skills behind this make chemical plant workers the unsung backbone for everyday devices that never get a second thought until something fails.
Beyond the Factory Floor
Daily use isn’t where the magic of 2,6-Xylenol stops. It lands in smaller but essential jobs—preservatives for wood treatments, rubber chemicals, dyes, and specialty coatings that protect metal from rust. Think about the fence posts that don’t splinter or rot or the vivid colors that mark out street lines year after year; this chemical lingers behind the scenes.
Fungicides for crops can depend on chemical relatives of 2,6-Xylenol. Farms with powdery mildew or other tough blights use these molecules to turn fields from hopeless to harvest-ready. It’s easy to forget the hands behind the harvest—farmer, chemist, and those in between—helping food reach the table.
Risks, Safety, and Smarter Solutions
Some dangers ride along with value. 2,6-Xylenol’s dust and fumes cause skin and respiratory irritation, so protective gloves and vented masks matter. My time in industry drilled this lesson in—no shortcuts. Small errors risk health, and I remember a coworker developing rashes after skipping gloves during a spill. Long-term exposure research tells us more about the need to limit contact, improve ventilation, and substitute less hazardous materials where possible.
Renewable and greener chemistry stands as a solution, offering an escape from harsh processing and risky byproducts. Biobased feedstocks and advanced reactors cut emissions and waste. Investment in better filtration and closed-loop containment lowers chemical hazards. Industry leaders with hands-on shop floor backgrounds tend to push for these changes, knowing the difference between textbook theory and daily practice.
2,6-Xylenol won’t grab headlines, yet workers in safety goggles and the engineers behind the formulas know its real impact. Roads stay safer, food lines stay secure, and technology keeps moving, thanks to this unsung hero of chemical manufacturing.
Is 2,6-Xylenol hazardous or toxic?
Looking at the Risks of 2,6-Xylenol
2,6-Xylenol pops up in places you might not expect. It’s a chemical used in disinfectants, plastics, and even dyes. Most people never see the stuff up close, but there’s no denying the stakes for anyone working near it or using products made with it. Talking about how 2,6-xylenol can affect people and the world isn’t just good science—it’s part of making sure everyone stays safe and healthy.
What Science Says About Toxicity
2,6-Xylenol can cause some trouble for your skin, eyes, and lungs. Get it on your hands and irritation follows. Breathe in those vapors and you might notice a scratchy throat or headaches. These are short-term problems, but repeated exposure has a way of adding up. Over time, chronic health issues like liver or kidney damage can show up when safety rules go ignored in workplaces handling large amounts. Data from the National Institute for Occupational Safety and Health highlights how people working with phenols like this have higher risks of both acute and long-term symptoms, especially without protective gear.
Folks who work in labs or factories know that smell—sweet and pungent. That strong scent hints at its potency. It’s not the sort of compound you shrug off after one whiff. I’ve read incident reports where spills lead to evacuations and medical treatments. That experience sticks, showing just how powerful the effects can be without proper controls.
Environmental Side of the Story
What escapes the lab or the plant doesn’t vanish. 2,6-Xylenol gets into wastewater. Bacteria typically break phenols down, but large spills or dumps overwhelm these systems. Fish and other aquatic life take the hit first, with chemical stress messing up delicate systems and food chains. The U.S. Environmental Protection Agency’s monitoring of phenols turns up repeated warnings about their buildup and the risks to local ecosystems. Slow breakdown in water means danger lingers longer than most people expect.
Protecting People and Nature
Workers and managers have no shortage of safety requirements to meet. Gloves, goggles, and masks form the first line of defense. Training everyone in safe handling and emergency response lowers odds of accidents. Regular checks on equipment and ventilation systems matter just as much as the rules on paper. Anyone skimping on these steps risks more than just regulatory fines—people’s health hangs in the balance.
Communities near chemical plants benefit from clear reporting about local emissions and accidents. Some places already use public data dashboards, so neighbors know what’s in their air and water. Faster and more transparent updates go a long way toward building trust. Clean-up programs backed by real money and oversight help restore what’s been damaged, but prevention always beats after-the-fact repairs.
Safe alternatives deserve more attention. Industries have started phasing out some older phenols for less toxic substitutes where possible. Governments and researchers can speed up the shift by funding safer chemistry and pushing for stricter environmental rules. If you ask anyone who’s handled hazardous chemicals day in and day out, they’ll tell you—no shortcut compares to real investment in safety and cleaner options.
What are the storage requirements for 2,6-Xylenol?
Why Storage Matters for 2,6-Xylenol
2,6-Xylenol shows up in plenty of industries, especially around paints, resins, and some disinfectants. You can find it in a white to pinkish solid form with a distinct, sharp odor. If you've ever worked in a warehouse that handles industrial chemicals, you already know how quickly things go wrong if storage isn’t up to standard. Since health, safety, and even the environment sit on the line, treating this compound with respect in storage spaces makes a real difference.
Proper Conditions Keep Problems Away
Heat and 2,6-Xylenol don’t mix well. Elevating room temperatures causes the material to degrade and increases the risk of fire. I’ve watched crews hustle because a storage area didn’t have working air conditioning. Given that this compound flashes at 116°C (240°F), there’s no sense in testing fate. Cool, dry setups make all the difference.
Around moisture, 2,6-Xylenol clumps and sometimes reacts. Dampness often invites corrosion, which can eat through container seals. Once I saw a small drum with rusted lids, leaving a sharp reminder: dehumidifiers should run, and leaks need immediate repairs.
Container Choices and Location
Metal drumming provides sturdy storage as long as things aren’t corroding, but plastic drums and lined steel barrels also do the job. Any container ought to seal tight—no cracks or worn-out gaskets. Once someone skipped annual inspections, and vapors started seeping out. The whole warehouse reeked, and the safety team landed in hot water during their next audit.
Good storage facilities keep chemical drums out of sunlight. UV exposure degrades containers and contents alike. Warehouses that install blinds or use solid walls avoid these problems, because sunlight has a sneaky way of raising temperature and weakening packaging.
Fire Safety and Chemical Segregation
Many smaller facilities try to save space by stacking everything side by side, but with 2,6-Xylenol, fire prevention calls for keeping oxidizers, acids, and open flames far away. Foam extinguishers and sprinklers stay close at hand. I’ve been through a fire drill where every second mattered. The ability to grab the right extinguisher, without searching, changed everything.
Proper labeling means no guesswork. Clear hazard signage, legible container tags, and updated storage logs keep mistakes from happening when rotating stock or responding to emergencies.
Personal and Environmental Impact
Fumes from 2,6-Xylenol bring headaches and, over longer exposure, cause serious trouble. You never want those vapors in the air where people come to work. Decent ventilation turns out to be just as important as temperature. Using PPE like gloves and masks protects anyone handling the material directly. Spills have a way of finding floor drains, so secondary containment, like spill pallets and lip trays, adds a basic but critical layer of security for workers and the environment outside.
Getting It Right
Managing chemical storage means more than checking off a list. In my experience, safe setups lean on regular training, good signage, working HVAC, and staff who keep their eyes open for trouble. The right steps today keep headaches and disasters out of tomorrow’s news.
How should 2,6-Xylenol be handled safely?
Understanding 2,6-Xylenol’s Risks
2,6-Xylenol shows up in chemical plants, labs, and industrial workshops as a building block for many products like resins and antioxidants. But this white crystalline powder carries some punch. Direct contact with skin or eyes burns and irritates fast, and inhaling its dust doesn’t do lungs any favors—high levels lead to dizziness, headaches, and sore throats. From a safety angle, 2,6-Xylenol’s toxicity makes it a chemical to handle with real care.
Personal Protective Equipment: The Basics Don’t Let You Down
From personal experience in small-scale production labs, skipping gloves or cheaping out on goggles just leads to trouble. Nitrile or butyl rubber gloves hold up better than latex, stopping that harsh stinging you get if this stuff sits on bare skin for more than a few seconds. Splash goggles wrap around to keep stray crystals or liquid from finding an eye. Pulling on a lab coat helps even if you think you’re just weighing out a spoonful. Those steps slow you down a bit, but skin rashes can sideline a worker all week.
Ventilation and Dust Control
Breathing in xylenol dust is less dramatic than a splash, but the long-term effects creep up. Dust chambers and fume hoods aren’t just decoration—even in a small setup, that powerful smell hints at irritation lurking. Fans and local exhaust keep the workspace clear. I’ve seen good ventilation make all the difference: headaches disappear, work speeds up, and nobody leaves feeling sick. Coughing all afternoon makes you wish for better airflow, every time.
Storage: Simple Mistakes, Big Trouble
2,6-Xylenol goes unstable if you mix it with strong oxidizers, acid, or even direct sunlight for too long. Keeping it in brown glass bottles in a cool, shaded spot reduces accidental reactions. Every broken jar and every spill means wasted time scrambling with absorbent pads, so it pays to double-check lids and keep only what you need on the bench. Labeling seems tedious but the moment confusion breaks out, labels save hours of sorting and avoid customers ending up with the wrong compound.
Emergency Response is the Hidden Backbone
No training exercise prepares you for the rush of a real spill—but muscle memory counts. Easy access to eyewash stations and safety showers, plus clear, unblocked exits, save real people from lasting harm. Calling out for help, having spill kits ready, and knowing whom to contact after an exposure make the difference. Every company I’ve worked for set up drills. In chaos, that split-second recall matters more than any poster on the wall.
Procedures Don’t Write Themselves
Keeping usage logs and running quarterly audits catch problems before they spread. Mixing up paperwork, skipping over leak checks, or letting PPE stocks run low often lead to near-misses. Workers buy in more when safety rules feel practical and when management actually uses them. Meetings spent on the why, not just the how, help everyone feel accountable.
Solutions: Culture Over Checklists
Long-term, safety depends on everyone in the room. Incentives help—companies that celebrate accident-free shifts build trust and reduce shortcuts. Training sessions refresh even veteran chemists, and giving workers a voice in procedure reviews brings out the real pain points. Partnering with health professionals, keeping up with changing regulations, and using modern monitoring tech stop most disasters before they get close. In the end, safe handling moves beyond compliance—healthy workers and clean operations pay dividends nobody can deny.
What is the chemical formula of 2,6-Xylenol?
Getting to Know 2,6-Xylenol
Most of us don’t spend our afternoons thinking about chemical formulas. Still, every bottle cleaner, every whiff of disinfectant, owes its punch to a network of molecules. 2,6-Xylenol crops up a lot in these everyday moments, and its chemical formula is C8H10O. Breaking it down, this means the molecule carries eight carbons, ten hydrogens, and a single oxygen—arranged in a way that makes it a powerful ingredient in places we might least expect.
Why the Formula C8H10O Matters
Look past the jumble of letters and numbers; this tiny formula forms the backbone of countless industrial processes. C8H10O structures two methyl groups and a hydroxyl group wrapped around a benzene ring. This layout gives 2,6-Xylenol solid antimicrobial power, making it pop up in cleaners, resins, and even some specialized polymers. Some folks in manufacturing count on 2,6-Xylenol for its stability and reactivity. It lines up well for synthesizing disinfectants, antioxidants, and even certain pharmaceuticals.
From the Lab to the Store Shelf
Turning chemicals like 2,6-Xylenol into finished products is where things get interesting. I’ve watched chemists combine xylenol with other agents, coax new molecules from those stable methyl and hydroxyl points. This isn’t just science on a chalkboard. These interactions affect how safely a product can be washed down the drain or sprayed around kids and pets. Analytical data shows that the specific arrangement of methyl and hydroxyl groups makes 2,6-Xylenol less likely to break down unexpectedly when exposed to heat or light, avoiding nasty surprises in quality control down the line.
Health and Safety Concerns
Being around chemicals in the lab brings a strong respect for safe handling. 2,6-Xylenol can irritate the eyes and skin, and if it enters the body in large enough doses, it becomes toxic. Keeping tabs on exposure is more than red tape. My own experience in quality control hammered in the importance of protective gear and tight storage. Industry studies back this up, and regulatory agencies in both the US and Europe set exposure limits based on robust toxicological reviews.
Environmental Footprint
Waste byproducts of 2,6-Xylenol can find their way into waterways. Evidence from environmental monitoring efforts shows certain aquatic organisms get hurt at concentrations that barely register in human terms. I’ve seen treatment teams work long hours to strip phenolic compounds out of wastewater before discharge. Their success supports clear, data-driven policies for plant operations. Advanced filtration systems and closed-loop recycling processes cut down on the risks, protecting both ecosystems and public health.
Practical Routes Forward
We don't escape chemical formulas—C8H10O will keep turning up on product sheets and data logs. Industry veterans point to better on-site recycling and greener catalysts that lower the environmental toll. Solid education, good data, and clear communication with the public bridge science and safety. Through strong oversight, focused research, and transparency, 2,6-Xylenol fits into a balanced approach where chemistry improves lives without creating fresh hazards.
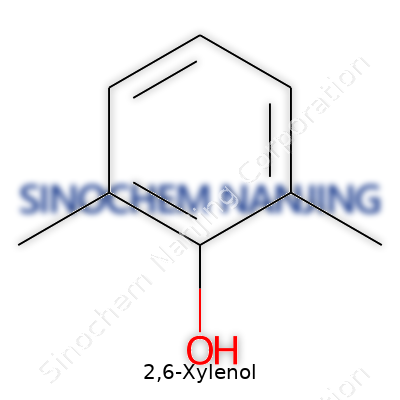
| Names | |
| Preferred IUPAC name | 2,6-dimethylphenol |
| Other names |
2,6-Dimethylphenol 2,6-Xylene-ol |
| Pronunciation | /tuː,sɪks-zaɪˈliːnɒl/ |
| Identifiers | |
| CAS Number | 576-26-1 |
| Beilstein Reference | 1361110 |
| ChEBI | CHEBI:16812 |
| ChEMBL | CHEMBL14122 |
| ChemSpider | 12316 |
| DrugBank | DB03744 |
| ECHA InfoCard | 100.007.768 |
| EC Number | 1.10.3.2 |
| Gmelin Reference | 83277 |
| KEGG | C01918 |
| MeSH | D015555 |
| PubChem CID | 13850 |
| RTECS number | ZE4500000 |
| UNII | 8OQ2X9QZV4 |
| UN number | UN2261 |
| Properties | |
| Chemical formula | C8H10O |
| Molar mass | 122.17 g/mol |
| Appearance | White to faintly yellow crystalline solid |
| Odor | phenolic |
| Density | 1.03 g/cm³ |
| Solubility in water | 7.95 g/L (20 °C) |
| log P | 1.98 |
| Vapor pressure | 0.11 mmHg (25°C) |
| Acidity (pKa) | 9.94 |
| Basicity (pKb) | 9.98 |
| Magnetic susceptibility (χ) | -67.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.531 |
| Viscosity | 8.4 mPa·s (25 °C) |
| Dipole moment | 1.69 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 196.2 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -326.5 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3685.7 kJ/mol |
| Pharmacology | |
| ATC code | D02AE02 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H315, H318, H335 |
| Precautionary statements | P280, P302+P352, P305+P351+P338, P310 |
| NFPA 704 (fire diamond) | 2,3,0,溶剂 |
| Flash point | 85 °C (closed cup) |
| Autoignition temperature | 530 °C |
| Explosive limits | 1.6–9.3% |
| Lethal dose or concentration | LD50 oral rat 795 mg/kg |
| LD50 (median dose) | LD50 (median dose): 795 mg/kg (rat, oral) |
| NIOSH | ZE2450000 |
| PEL (Permissible) | PEL (Permissible) of 2,6-Xylenol: 5 ppm (19 mg/m³) |
| REL (Recommended) | Air Quality Guideline: 240 µg/m³ |
| IDLH (Immediate danger) | 50 ppm |
| Related compounds | |
| Related compounds |
Phenol o-Cresol m-Cresol p-Cresol 4-Ethylphenol Pyrocatechol |

