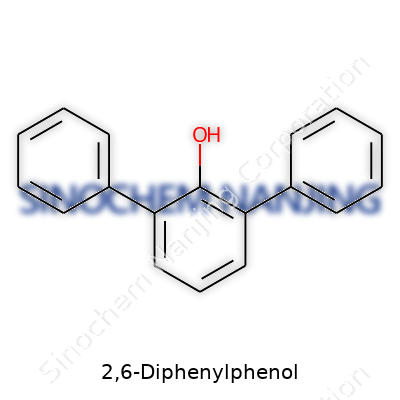
2,6-Diphenylphenol: Pathway of Progress and Prospects
Historical Development
Chemists started looking at phenolic compounds more closely during the late 19th and early 20th centuries, hunting for derivatives that brought some extra muscle to the table. The simple structures of phenol and its sisters didn’t satisfy everybody, so once 2,6-Diphenylphenol showed up, it opened up a fresh set of doors. Experts pushed boundaries of Friedel–Crafts alkylation and Suzuki coupling back in the day, fueling the discovery and refinement of 2,6-Diphenylphenol. The compound’s story weaves through early organic synthesis labs, wartime research aimed at stable antioxidants, and the relentless industrial search for high-performance additives. This backdrop shapes how the chemical found itself not just in bench research, but soon enough in industrial processes ranging from plastics to advanced electronics.
Product Overview
Ask anyone who deals with organic synthesis—2,6-Diphenylphenol tends to show up more often than most folks outside the lab realize. Its three aromatic rings, wired together at specific spots, beef up its stability and ability to play a role in different chemical reactions. Companies label it in various ways, but at its core, it delivers strong antioxidant activity, stands firm against high heat, and won’t shy away from serving as a vital intermediate in complex synthetic chains. The compound’s reach touches pharmaceuticals, specialty polymers, and niche catalysts for industrial reactions.
Physical & Chemical Properties
Solid at room temperature, 2,6-Diphenylphenol brings a white, crystalline appearance—evidence of its clean structure, unless impurities have wormed their way in. The melting point usually hovers just north of 135°C, making it sturdy under most working conditions that technicians face day-to-day. Its solubility leans toward organic solvents; water rarely breaks this molecule apart, so teams working in aqueous systems look elsewhere. The chemical backbone, made up of two phenyls saddled on a central phenol ring at the 2,6-positions, means the molecule stands up to both heat and oxidative stress better than simpler phenols. This resistance matters if you’re looking to build materials engineered to last.
Technical Specifications & Labeling
In most markets, bottles and drums bearing the chemical carry identifiers such as CAS Number 92-06-8. Technical grade material usually arrives at purities above 98%. Routine QA in reputable facilities covers melting point checks, IR and NMR confirmation of structure, and screens for trace metals or other aromatics. MSDS sheets focus on personal protection equipment and safe handling recommendations, addressing dust generation, incompatibility with strong oxidizers, and temperature-controlled storage. Shipping labels flag standard hazard categories, keeping logistics and safety inspectors from missing critical information.
Preparation Method
Industrial-scale production starts with phenol and sets up the right partners for Friedel–Crafts alkylation, introducing phenyl groups at the 2 and 6 spots with the coaxing power of strong acid catalysts—usually AlCl3 or FeCl3. Some routes tap into Grignard chemistry or harness transition metal catalysis to join up the rings, with Suzuki-Miyaura coupling now showing up more as process chemists look for cleaner, higher-yield alternatives. Once you build up the right environment—proper temperature, moisture control, and quench strategy—separation and crystallization yield a stable product fit for downstream use. Waste handling becomes a top priority, as strong acids and byproduct benzene never earned points for environmental friendliness.
Chemical Reactions & Modifications
Labs often modify 2,6-Diphenylphenol to build more complex molecules. The phenolic -OH stands ready for etherification or esterification, routes heavily used in the pharmaceutical sphere. The aromatic framework welcomes halogenation, nitration, or sulfonation, yielding new candidates with altered solubility or reactivity. Chemists often exploit the compound’s stability to assemble oligomers or introduce functional groups that improve compatibility in specialized plastics, resins, and even advanced coatings. Redox transformations also turn it into efficient antioxidant agents, stabilizing reactive intermediates in demanding manufacturing steps.
Synonyms & Product Names
Suppliers and manufacturers list this molecule under a basket of alternative labels, including 2,6-diphenyl-1-hydroxybenzene, DPP, and occasionally old trade names. Supply chain teams looking for reliable sourcing keep an eye out for these aliases, especially when navigating global procurement databases designed with legacy nomenclature in mind. Regulatory filings in various regions may not always match up, calling for sharp attention when comparing compliance documentation before large-scale runs.
Safety & Operational Standards
Handling phenolic compounds remains a high-stakes business—gloves, goggles, and face shields hold a permanent spot at the bench. 2,6-Diphenylphenol’s dust can irritate skin and eyes, and casual contact rarely leads to a good story. Safety officers train workers on spill control, proper ventilation, and secure storage to stop unwanted chemical incidents. Trusted facilities log incident reports and run regular drills, while local and international regulations push for improved worker safeguards. Disposal of spent material and process waste doesn’t get left to last-minute guesswork—strict protocols dictate clear routes for neutralization and off-site processing, reducing negative impacts on local ecosystems.
Application Area
Where performance plastics dictate strict specs, 2,6-Diphenylphenol often becomes the go-to antioxidant, forestalling degradation by heat or UV light. In the adhesive and coatings industries, formulators slot this molecule into blends seeking long-term color stability and resistance to physical stress. Academic groups prize it as a core fragment when exploring new molecular scaffolds, drug candidates, and even smart polymers that respond to external stimuli. Electronic manufacturers dip into this toolbox to engineer materials that need steady dielectric properties across punishing temperature cycles. Food packaging and agricultural films also count on it, though end-use vetting leans heavily on regulatory safety data and trace impurity checks.
Research & Development
R&D teams dig deep into 2,6-Diphenylphenol’s chemistry, pushing limits of what’s possible in both synthetic methodology and functional applications. The molecule’s structure inspires new antioxidant standards, and catalysis experts borrow its framework to anchor innovative ligands for transition metal complexes. Pharmaceutical chemists tweak side chains, searching for leads that pass muster in biological screens. Polymer engineers run iterative tests, hoping to wring out even more toughness and clarity from additive blends. Green chemistry fans look for ways to cut down hazardous waste in its production—reducing catalyst load, recycling spent acids, and dialing up chromatography-free isolation methods in pilot plants. Collaboration between academic labs and process chemists lays down a pipeline for both incremental and game-changing advances.
Toxicity Research
Toxicologists bring a measured focus to phenolic compounds, and 2,6-Diphenylphenol sits under a bright spotlight. Acute exposure may prompt skin, eye, or respiratory issues, pushing worksite managers toward careful exposure limits and regular health screenings. Long-term animal studies set out to map chronic toxicity and carcinogenic potential, with current literature leaning toward low systemic toxicity but not lowering guardrails for unnecessary exposure. Regulatory agencies inspect extensive dossier submissions, demanding both acute and chronic toxicity breakdowns, metabolism data, and evidence of breakdown products in real-life disposal scenarios. Results guide both workplace limits and final product approvals for sectors like packaging or health-related implants.
Future Prospects
The march toward sustainable chemistry puts fresh demands on every building block, and 2,6-Diphenylphenol stands at a crossroads where tradition meets innovation. Green process design and bio-based raw material sourcing demand novel production strategies, driving research into enzyme-assisted synthesis and waste minimization at industrial scale. R&D teams pair computational models with high-throughput screenings to predict new performance additives based on diphenylphenol’s core scaffold, betting on better performance in batteries, wearable electronics, and high-strength composites. Advances in purification and trace detection technology make it possible to detect ever-smaller residues in complex matrices, letting end-users build safer, more reliable products. Lively patent activity signals that nobody feels satisfied just standing still—collaboration between industry, academia, and policymakers keeps raising the bar for performance, safety, and sustainability in all things aromatic and phenolic.
What are the main applications of 2,6-Diphenylphenol?
A Closer Look at a Specialty Chemical
2,6-Diphenylphenol doesn’t turn heads outside the chemistry world, but its real-life impact lands closer to home than most realize. This compound’s structure gives it strong antioxidant power and lets it serve as a key building block in creating specialty materials, especially plastics and polymers. Having worked in the materials sector years ago, I’ve seen companies eye this compound not because it’s flashy, but because it solves core problems in manufacturing and product longevity.
Keeping Plastics Strong and Durable
Plastic products break down over time, mostly because oxygen in the air finds a way to kick off chain reactions that eat away at the material’s structure. 2,6-Diphenylphenol steps in as a defensive shield. Manufacturers blend it into everything from polypropylene to certain high-grade fibers, slowing oxidation and stretching out the life of pipes, containers, automotive interiors, and consumer goods. Data from major chemical suppliers shows strong demand for this type of antioxidant in food packaging and storage, since food safety rules keep getting tighter and shelf life really matters. If you’ve bought groceries in tough, clear plastic tubs, there’s a strong chance this compound helped that packaging resist damage from light and air.
Tackling Polymer Processing Challenges
Sometimes a reaction needs a subtle influencer, something that can help new materials form without knocking everything off balance. Labs lean on 2,6-Diphenylphenol during polymerization as a stabilizer, limiting unwanted side reactions. I remember a project with nylon fibers—adding just a bit during the mixing phase helped us cut waste and avoid yellowing, which mattered a lot for products with strict appearance standards. This careful role makes the compound a staple in advanced fibers and coatings where flawless results are non-negotiable.
A Hidden Helper in Paints and Coatings
Paint and coatings fight a battle against the elements daily, from sun exposure to humidity swings. The phenolic backbone of 2,6-Diphenylphenol gives it a knack for neutralizing molecules that cause fading and breakdown. Suppliers often fold it into protective coatings used on vehicles, appliances, and electronics. This strategy shrinks expenses tied to fading, chalking, or flaking finishes, supporting end users who expect products to look sharp for years.
Fine Chemicals and Pharmaceutical Research
While not a headliner in medicine, 2,6-Diphenylphenol acts as a stepping stone to more complex molecules. Fine chemical labs turn to it for building custom compounds and screening molecules in early-stage drug and agrochemical research. Specialty chemical directories show listings under research reagents—scientists trust it to create analogs or modifications in synthetic pathways. This bridge-building role, though less visible than its work in plastics, speaks to its versatility.
Challenges and Smarter Approaches
From a health and safety point of view, 2,6-Diphenylphenol needs careful handling. Studies indicate low acute toxicity, but as its use broadens, regulatory review has sharpened. Researchers have called for better monitoring of its breakdown products in manufacturing waste. Safer alternatives or greener production methods present a promising direction. Some groups work with plant-based antioxidants or recyclable stabilizers to address these concerns while keeping performance high.
While 2,6-Diphenylphenol doesn’t always get the industry spotlight, its footprint can be found in longer-lasting materials, smoother manufacturing, and innovative lab work. Applying forward-thinking science and responsible oversight can ensure its benefits stretch into the next phase of materials development.
Is 2,6-Diphenylphenol hazardous to health or the environment?
Understanding the Chemical
2,6-Diphenylphenol doesn’t show up in daily life for most people, yet it plays a role behind the scenes in certain manufacturing processes and chemical research. Anyone working around industrial chemistry or materials engineering will see this name pop up from time to time, often linked to specialty polymers or as a stabilizer in plastic production. More recently, increased access to chemical information has led to public conversations about how obscure compounds impact health and the environment.
Is 2,6-Diphenylphenol a Health Hazard?
Looking at the structure, this compound falls under phenolic compounds, which sometimes signal toxicity concerns. Personal experience in the lab has shown me that skin contact with phenolic agents can cause irritation. Most chemical safety data sheets list moderate skin and eye irritation risks for 2,6-Diphenylphenol, so protective gloves and eyewear remain non-negotiable. Toxicological reviews available through databases like PubChem and the ECHA register don’t suggest it is acutely toxic in small, incidental exposures, but always good to treat it with respect. Chronic effects, such as carcinogenicity or reproductive toxicity, haven't been definitively proven, but gaps in long-term data linger. Relying only on the “no evidence found” line sometimes leads to complacency; until there’s more research, caution matters.
Environmental Worries
In an environmental sense, phenolic compounds can upset aquatic systems. Reports from the EPA demonstrate that high concentrations of related phenols disrupt fish and aquatic invertebrate reproduction. Persistence in the environment remains the biggest worry. Many phenolics break down slowly, which means they can accumulate. In lab waste management, we never let phenols enter regular drains—there’s a reason protocols push for secure containment and specialized disposal. Once phenols reach rivers or soil, the consequences often show up years later in subtle but damaging ways. There’s no easy fix for bioaccumulation in the wild; it takes strict regulation and best practice at every level of handling.
Supporting the Claims with Data
Only a handful of studies directly address 2,6-Diphenylphenol, so much of what we know comes from the phenol family as a whole. According to the National Institute for Occupational Safety and Health (NIOSH), phenolic compounds in workplace settings are monitored for both inhalation and skin risks. Acute effects ranging from headaches to nausea rarely make headlines but affect actual workers. Chronic exposure gets less attention, yet small toxic loads add up. Tracking the chemical's lifecycle helps spot any unsafe build-up, and data from manufacturing audits often reveal how quickly a minor leak escalates beyond the factory walls.
Solutions and Mitigation
In practice, simple solutions work best—in the lab and the field. Using effective protective gear reduces most health risks for those handling such chemicals. Ventilation and well-designed extraction systems limit exposure through air. Companies investing in robust waste treatment lower the odds of environmental release. From my own work, partnering with waste management experts yields much better results than makeshift self-disposal tactics. Legislation like the REACH regulation in the EU pressures producers to operate transparently, informing buyers and the public about potential hazards. More funding for independent research will help fill evidence gaps and guide future safety standards. Ultimately, safe handling, transparency, and responsible disposal keep both people and ecosystems healthier, even with unfamiliar compounds like 2,6-Diphenylphenol in the mix.
What is the chemical structure and properties of 2,6-Diphenylphenol?
The Shape of the Molecule
2,6-Diphenylphenol grabs attention because of its distinctive structure. Three benzene rings connect to a central phenol backbone, with the phenolic group sitting in the middle. These rings link at the second and sixth positions, giving the compound a symmetry that influences both reactivity and stability. You end up with a molecular formula of C18H14O. The setup offers a rigid, flat profile, with the oxygen atom of the phenolic group sticking out, ready for chemical action.
What This Structure Means for Properties
The dual phenyl rings on either side serve as guards. They shield the central phenol and slow down reactions that would otherwise attack that site. This is why you see greater stability under heat and less oxidation—a property synthetic chemists prize. These side groups also make the molecule more oil-loving. It won’t mix smoothly with water, but it dissolves well in organic solvents like toluene or chloroform. I once tried to clean a flask with ethanol after a diphenylphenol synthesis and ended up reaching for acetone because standard solvents fizzled out.
Because of its bulky shape, 2,6-Diphenylphenol doesn’t pack tightly. In practice, you get lower melting points compared to plain phenol. It melts at around 90°C, while pure phenol clocks in above 40°C. The extra rings get in the way, acting like campfire logs set crosswise. These “roadblocks” help the molecule resist stacking, and this affects everything from crystallization to handling in industrial settings.
Real-Life Uses Rooted in Chemistry
Scientists lean on 2,6-Diphenylphenol for a reason—it makes a strong building block. The phenolic OH group pulls electrons, making the molecule a better antioxidant than basic phenol. I’ve seen it show up as a stabilizer in plastics, where the triple-ring structure lets it quench free radicals before they run wild and spoil the whole batch. This kind of stabilizer helps plastics stay clear, flexible, and safe even after years of sun and heat exposure.
In the lab, the shape means less trouble with unwanted side reactions. Many phenol derivatives react too quickly, causing side products in pharmaceutical synthesis. Bulky rings slow things down, so chemists target reactions more precisely, cutting waste and trimming costs. Drug researchers take note of this pattern since 2,6-Diphenylphenol can act as a scaffold for developing active molecules in the fight against bacterial or fungal threats. That wide “shoulder” at each end leaves room for attaching more chemical groups, letting scientists tune properties for effectiveness or safety.
Risks and Opportunities Ahead
Compounds based on large, flat structures tend to stick around in the environment. That stickiness raises red flags: not just for the soil and water, but for anyone working in production or cleanup. The hydrophobic profile means careful handling from start to disposal. Regulatory agencies—and anyone who cares about occupational safety—push for better monitoring and containment at every step. My own experience in a chemical safety lab taught me that one dropped beaker creates a mess that doesn’t just mop up with a squirt of bleach. Strong extraction protocols and training matter as much as recipe precision.
Looking forward, new research tries to tame this stubborn stability by tweaking the side groups. By opening doors for easier breakdown, chemists aim to balance lasting protection in finished goods with a lighter touch on the planet. Every innovation here happens because someone dug deep into the structure—because understanding how rings, bonds, and functional groups interact sets the direction for smarter, safer solutions.
How should 2,6-Diphenylphenol be stored and handled safely?
Why Storage Conditions Impact Every Lab
Anyone working with chemicals knows certain compounds ask for focused attention. 2,6-Diphenylphenol falls into that group more than most. I’ve seen labs let their guard down—improper handling turns a day’s work into a week-long mess, with health risks thrown into the mix. This compound can irritate the eyes, skin, and respiratory system. Direct contact brings more risks than most folks realize. It stays stable under the right conditions, but one careless moment opens the door to headaches—literal and figurative.
Keep Containers Sealed and Secure
I’ve spent plenty of time in university stockrooms and industry pilot plants. Leaving a jar half-closed, or with the seal a bit loose, leads to moisture creeping in and chemical degradation. Even worse, potential spills or exposure jump up. Keep containers tightly closed. Glass or high-grade plastic, labeled with content and hazard symbols, stays on my shelf—never improvised jars or re-used bottles. You don’t want cross-contamination or mystery stuff mixing in with your research supply.
Find the Right Environment
2,6-Diphenylphenol asks for cool, dry storage, out of sunlight and away from obvious hazards. Humidity and heat speed up breakdown, turning powders into sticky materials that ruin experiments. My own practice always involves designated chemical storage rooms—ventilated, clearly marked, separated by reactivity class. No shortcuts. I’ve seen folks try shelves in back offices or staff rooms; chemical odor creeps in, creating discomfort and even headaches by the end of a shift.
Personal Protection Matters
Gloves, goggles, and lab coats are non-negotiable. More than once, I’ve felt the sting from accidental splashes while decanting. Even handling sealed containers, I put on nitrile gloves—skin absorption happens easier than most believe. Protective clothes cut down on accidental transfer to surfaces and skin. Over the years, a robust routine replaced shortcuts, because even small exposures add up.
Work with Ventilation—Not Against It
Every decent lab runs some sort of ventilation—either fume hoods or local extraction. I don’t measure or transfer 2,6-Diphenylphenol on an open bench. Performing any chemical work in a closed, ventilated space cuts down exposure, keeps odors at bay, and lets you focus on actual work. It is easy to get complacent with "non-volatile" powders, but airborne particles still pose risks.
Smart Disposal Counts
Disposal brings its own load of responsibilities. Mixing waste streams or sending solvents with 2,6-Diphenylphenol residue straight down the drain breaks rules and harms wastewater systems. I set up dedicated containers for organic residues and label everything clearly. Local hazardous waste pickup solves headaches before they start. Don’t wait for the safety officer to chase you.
Why Organizational Discipline Wins
A culture of discipline shapes safe handling. Document routines, use up-to-date safety data sheets, and keep lines of communication open. New team members in my lab always get a walkthrough, not just a binder. One mistake can cause expensive delays or health problems for colleagues who rely on a shared workspace.
Serious About Solutions
Simple, practical steps protect people and research alike. Secure, dry storage, personal protection, real ventilation, and responsible waste handling save money, health, and time. Promoting well-trained habits outpaces written policies any day—a lesson learned from years of cautious, sometimes hard, experience with 2,6-Diphenylphenol.
What is the typical purity and packaging for 2,6-Diphenylphenol available for purchase?
What Buyers Actually Get
People searching for 2,6-Diphenylphenol don’t just expect any old powder or crystal. They want a product they can trust—something that delivers consistent results in the lab or on the line. In my experience working alongside chemists and procurement teams, purity turns out to be the deal-breaker. The sweet spot sits at 98% purity or higher. Lower than that, and you're rolling the dice on contamination or side-reactions, especially when stakes are high in pharmaceutical or polymer applications. Labs with higher demands lean toward “analytical grade,” which approaches 99%, making it easier to avoid unexpected peaks during testing. That extra margin keeps experiments out of murky territory.
How Purity Impacts Work
Take it from a friend who runs quality control in a materials lab. A single contaminant in 2,6-Diphenylphenol batches can derail a whole day's worth of trials. Traces of water, unknown isomers, or tiny bits of dust show up in the results, leaving people guessing if the equipment’s faulty or the sample’s impure. Chasing down problems wastes time and money. So, it’s not just a preference for high-purity material—it's essential for places that care about repeatable outcomes. Reliable suppliers publish data sheets or certificates of analysis for each batch. Whenever I’ve been part of a project that skipped this step, it ended up with a rushed overnight shipment to replace questionable product.
Packaging Realities: More Than Just a Container
Suppliers know that it’s not enough to toss a compound into any package. This might sound dramatic, but nobody wants chemicals leaching through a flimsy jug. Usually, companies package 2,6-Diphenylphenol in amber glass bottles or tightly sealed plastic containers. Glass wins against reactive chemicals and air, while the amber color blocks light that could alter sensitive compounds. For bulk orders, heavy-duty polyethylene drums or lined steel containers enter the picture. Everything gets sealed tight with tamper-evident lids. In every lab I’ve been in, handling a broken seal kicks off a series of phone calls, report writing, and delays. That’s motivation to get packaging right from the start.
Industry Standards and Your Choices
Across the industry, standard package sizes usually start at 25 grams or 100 grams for small-batch research. For larger applications—maybe a pilot plant or manufacturing run—five-kilogram drums or more get ordered. Labels spell out batch numbers, expiration dates, and handling precautions. Missing labels have led to headaches and, sometimes, wasted batches. Some companies offer customized sizes, but in practice, most end-users stick to the listed options. After a bad experience with untraceable leftovers, a colleague never accepts unmarked containers again.
Looking for Better Solutions
Problems with purity or packaging trace back to skipping supplier vetting or choosing based on price alone. Verifying third-party testing and establishing good communication with reps usually saves trouble. Batch-to-batch consistency matters most, so suppliers with ISO 9001 or similar quality certificates often end up with repeat business. Purchasing teams I’ve worked with rarely switch sources unless consistency drops. If a supplier can help track lots and offers strong documentation, they stand out. This simple approach doesn’t eliminate hiccups, but it lowers the risk of reruns and lab stops.
Staying Ahead in Practice
Seeking high-purity and tough packaging for 2,6-Diphenylphenol allows projects to move forward without guessing at the contents. Sticking with transparent suppliers and reviewing technical documents may seem tedious, but time and again it proves to be the difference between smooth progress and wasted effort.
| Names | |
| Preferred IUPAC name | 2,6-diphenylphenol |
| Other names |
2,6-Diphenyl-1-hydroxybenzene 2,6-Diphenylbenzenol 2,6-Diphenylphenol Phenol, 2,6-diphenyl- |
| Pronunciation | /tuː,sɪks daɪˈfɛn.əlˌfiːn.ɒl/ |
| Identifiers | |
| CAS Number | [1806-26-4] |
| 3D model (JSmol) | `3Dmol.js({'m':'c1ccccc1c2cccc(c2)c3ccccc3O'})` |
| Beilstein Reference | 1208736 |
| ChEBI | CHEBI:34699 |
| ChEMBL | CHEMBL141215 |
| ChemSpider | 140863 |
| DrugBank | DB14641 |
| ECHA InfoCard | 100.047.876 |
| EC Number | 201-002-7 |
| Gmelin Reference | 522719 |
| KEGG | C14112 |
| MeSH | D010997 |
| PubChem CID | 70958 |
| RTECS number | GO2625000 |
| UNII | Z02LA1424V |
| UN number | UN3077 |
| CompTox Dashboard (EPA) | DTXSID2020091 |
| Properties | |
| Chemical formula | C18H14O |
| Molar mass | 282.35 g/mol |
| Appearance | White to light yellow crystalline powder |
| Odor | Odorless |
| Density | 1.14 g/cm3 |
| Solubility in water | Insoluble |
| log P | 4.8 |
| Vapor pressure | 1.6 x 10^-5 mmHg (25°C) |
| Acidity (pKa) | 11.2 |
| Basicity (pKb) | 15.41 |
| Magnetic susceptibility (χ) | -71.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.647 |
| Viscosity | 1.64 cP (150°C) |
| Dipole moment | 2.68 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 250.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -6310 kJ/mol |
| Pharmacology | |
| ATC code | D08AE21 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes skin irritation. Causes serious eye irritation. May cause respiratory irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P261, P264, P270, P271, P304+P340, P312, P403+P233, P501 |
| NFPA 704 (fire diamond) | 1-2-0 |
| Flash point | > 163 °C |
| Autoignition temperature | Autoignition temperature: 600 °C |
| Lethal dose or concentration | LD50 (oral, rat): > 5000 mg/kg |
| LD50 (median dose) | LD50 (median dose): 2100 mg/kg (oral, rat) |
| NIOSH | GZ3150000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 10 mg/m3 |
| Related compounds | |
| Related compounds |
Phenol 2-Phenylphenol 4-Phenylphenol 2,4-Diphenylphenol 2,6-Diphenylaniline |

