2,6-Dinitroaniline: A Deep Dive Into Its Role, Risks, and Possibilities
Tracing the Beginnings: Historical Development
Looking back at the evolution of 2,6-dinitroaniline, you find its story woven into the larger tapestry of synthetic chemistry that swept through the 19th and 20th centuries. Early work with nitroaromatic compounds helped spark advances in dye and explosives production, but compounds like 2,6-dinitroaniline brought their own specific strengths. Chemists searching for ways to modify aromatic amines found that nitration gave aniline a wide array of properties, many of which fit emerging needs in industry. Over decades, a deeper understanding of both risks and benefits shaped its uses—sometimes first driving up demand, then inviting scrutiny as knowledge of safety impacts grew.
What Is 2,6-Dinitroaniline?
Ask anyone in the chemical manufacturing world about 2,6-dinitroaniline, and you’ll tend to get a reaction filled with respect for its potency and caution for its hazards. It shows up as a yellow to orange solid powder, faintly aromatic, with enough chemical punch to draw both industrial interest and regulatory attention. Used as a building block, it has value in synthesizing dyes, pigments, and chemical intermediates. Its dual nitro groups put it in a special class—energy-rich, yet relatively stable under controlled conditions.
Physical and Chemical Qualities
No point dancing around the facts: 2,6-dinitroaniline packs density and reactivity into a compact form. It doesn’t dissolve well in water and tends to resist mixing—fitting for a compound with both bulk and explosive potential. It melts close to 100°C, which makes it tricky at best for processing, since elevated temperatures can trigger dangerous reactions if safety slips. The molecule’s nitro groups at 2 and 6 positions change its reactivity compared to mono-nitro analogues, and this influences both its industrial handling and the properties of derivatives made from it. Handling dust or fine particles deserves real caution—these can create ignition hazards if mixed with air in the right concentration.
Technical Details and Labeling Requirements
You won’t find 2,6-dinitroaniline sold without a barrage of labels and warnings. Every drum or container leaving a facility comes stamped with hazard icons, handling instructions, and clear chemical names. International shipping rules tie down methods for both packaging and declaring nitro compounds, given the dual threats of toxicity and explosiveness. I’ve seen regulatory teams pore over labeling standards for weeks just to tweak a single clause, because the fallout from mislabeling isn’t hypothetical. Everyone in a production line, from engineer to warehouse worker, faces strict standards for documentation and training. Slips often lead to real consequences—regulatory fines, angry auditors, or worse.
How Chemists Make It: Preparation Method
Making 2,6-dinitroaniline starts with aniline or a suitable precursor, followed by electrophilic aromatic substitution—usually nitration with a mixture of nitric and sulfuric acids. From experience, it’s a stressful mix of heat control and timing. Too cool, and the reaction won’t finish. Too hot, and you face runaway reactions capable of wrecking equipment. Nitrogen oxides and acid mists demand carefully designed ventilation; even the cleanup solids need neutralization before shipment off-site. Teams have to account for both yield and safety, often erring on the side of caution when scaling up an experiment to plant-level production.
Chemical Reactions and Functional Adjustments
The dual nitro groups on the aniline ring give chemists a reactive scaffold for further modification. Reduction reactions turn the nitro groups into amines, which open doors to new products. Diazotization enables coupling with various aromatic partners—key for dye synthesis. Oxidation, halogenation, and even some transition metal-catalyzed reactions expand its conversion pathways, though each comes with a list of what-not-to-do, since the parent structure is unforgiving of mistakes.
What Else Do Folks Call This Compound?
Depending on where you work or what your field looks like, you might see it labeled as 2,6-dinitrobenzenamine or simply DINA. Some catalogs toss out more exotic names, but sticking with the essentials usually avoids confusion. Knowing the alternate names isn’t mere trivia; shipments and documentation hinge on every letter matching up when crossing regulatory borders or dealing with foreign partners.
Staying Safe: Operational Standards and Industry Practices
Factories that handle 2,6-dinitroaniline don’t leave safety to chance. Operators focus on equipment integrity, site training, and near-constant environmental monitoring. Veteran workers often share stories about what happens if procedures slip—from minor accidents that set off evacuation alarms to major incidents that shut down entire facilities for weeks. Proper gear, atmospheric monitoring, grounding of all metal surfaces, and strict temperature control aren’t optional—they’re habits drilled into every technician. National and international agencies publish best practices, and compliance audits come with real muscle behind them.
Industries Tapping 2,6-Dinitroaniline
The dye sector leans on this molecule for azo dye preparation, exploiting its reactivity for vibrant colorants in textiles, inks, and plastics. Pigment factories rely on it for fast, brilliant colors where lightfastness and weather resistance count more than cost. In research, 2,6-dinitroaniline still finds occasional use as a stepping stone in organic synthesis—sometimes carving out space in the design of specialty agrochemicals or advanced polymer materials.
Research and Development Efforts
Labs continue to probe more efficient synthesis options, cut down on acid waste, and trim energy costs during manufacturing. Across academia and industry, you’ll find teams focused on reactivity studies, new pathways for ring modification, and green chemistry tweaks to make production less polluting. Patents sometimes drop hints about new formulations or downstream products made with 2,6-dinitroaniline, pointing to ongoing interest. Researchers also keep screening for safer analogues and alternatives, especially for companies eager to lower their toxic footprint.
Toxic Effects and How They’re Studied
The health risks from exposure to 2,6-dinitroaniline deserve real attention. Labs run animal studies to probe both acute and chronic toxicity, often reporting impacts on blood, liver, and central nervous system. Inhalation or skin contact leads to rapid absorption, while the compound's persistence stirs concern over environmental accumulation and effects on aquatic life. Regulatory toxicologists weigh each new finding against existing limits and sometimes ratchet down allowable workplace concentrations accordingly. My own take after reading stacks of reports: those who work daily around dinitro compounds can never rely on out-of-date safety rules. Vigilance is a must, not a formality.
Where Do We Go From Here? Prospects for Tomorrow
Facing headwinds from tighter chemical controls and shifting market demands, producers and researchers push for safer, greener methods and a possible overhaul of products that rely heavily on compounds like 2,6-dinitroaniline. The pressure to switch to renewable feedstocks, close the loop on chemical waste, and minimize risk to workers grows every year. If history teaches anything, it’s that chemistry evolves alongside regulation and public expectations—so staying flexible, transparent, and focused on both safety and innovation matters more than ever.
What is 2,6-Dinitroaniline used for?
Understanding Its Place in Industry
2,6-Dinitroaniline doesn’t make headlines, but its impact reaches households every day. I’ve walked past the lawns in my neighborhood, bright green and free of weeds, thanks to compounds derived from substances like this one. This chemical provides the backbone for some agricultural solutions that keep crops healthy and productive.
Weed Control: A Big Driver
Walk through any hardware store, and you’ll see shelves lined with weed control products. The backbone for many of these, especially pre-emergent herbicides, comes from the dinitroaniline family. 2,6-Dinitroaniline stands out as a starting material to produce dinitroaniline herbicides, such as pendimethalin and trifluralin. Farmers, landscapers, even backyard gardeners depend on these to protect crops from being overrun with weeds, meaning higher yields and more food produced on the same patch of soil. For farmers in regions where labor for weeding is scarce, these chemicals often make the difference between a harvest and a loss.
Safety and Environmental Questions
I’ve spoken with environmentalists and read my share of data sheets. Synthetic chemicals can raise valid concerns, and dinitroaniline derivatives have attracted attention over water contamination, persistence in soil, and effects on non-target plants. Real risks exist. California, for example, maintains strict monitoring to minimize impact on water supplies. Regulatory bodies step in to set usage limits, buffer zones, and application timing to lower risk to humans and wildlife. Without such checks, these herbicides could spread into waterways or affect crops that were meant to stay chemical-free. Local news stories about groundwater contamination trigger community responses for better oversight.
Research in Synthesis and Manufacturing
Scientists continue to find safer or more efficient ways to produce and use dinitroanilines. Improvements in manufacturing reduce waste and help chemists develop products that target specific weeds rather than casting a wide net that might harm more than intended. Chemical engineers I have met always look for ways to cut down on byproducts, both to improve worker safety and to cut costs. Companies spend big money on developing time-release formulations that keep active ingredients locked away until needed, lowering the need for reapplication and further reducing environmental risks.
Regulation and Consumer Choices
Governments monitor herbicide ingredients closely. In places where regulations aren’t as strong, farmers and their customers lose confidence in food safety. Public scrutiny pressures companies to reach for higher standards and find replacements wherever possible. At the same time, the demand for organic produce pushes research forward, promoting alternatives that help the environment and satisfy consumers who want chemical-free food.
Opportunities for Smarter Farming
It’s clear that the story of 2,6-Dinitroaniline is more than chemistry. It’s about finding a balance between the needs of modern agriculture and the health of our communities and natural systems. Success means better technology, careful regulation, and keeping our eyes open for the next step in responsible weed control. By learning from the past, investing in innovation, and insisting on transparency, those growing our food can manage weeds effectively while staying accountable to the rest of us.
What are the safety precautions for handling 2,6-Dinitroaniline?
Understanding What You’re Handling
2,6-Dinitroaniline brings with it a fair amount of baggage if you work in chemistry or certain manufacturing jobs. The yellow-orange powder grabs attention partly because it’s part of the nitroaromatics family. From experience working in labs, I’ve learned these compounds deserve more than a quick glance at the safety label. They get used in research, chemical synthesis, and can sometimes show up as an intermediate in dye or pesticide production. The kind of hazards tied to this chemical don’t just stay in books.
Hazards Lurk in the Details
2,6-Dinitroaniline can irritate the eyes, skin, and lungs. Its dust may go airborne easily. There’s enough reason to think about respiratory protection, and always use gloves that aren’t just standard latex—opt for nitrile or neoprene, which I’ve found handle organic solvents and aromatic nitro compounds well. Nitrile gloves don’t break down quickly like cheap vinyl. Goggles with a snug seal prevent accidental splashes from getting in your eyes, which can lead to burning and long-term irritation.
Accidental inhalation happens more often than you think, particularly if you’re pouring powder instead of dissolving it. Anyone who’s ever gotten a lungful of dust knows it stings and lingers. Respiratory protection goes beyond the basic surgical mask. A half-mask respirator fitted with organic vapor cartridges and dust filters makes a real difference. Older colleagues tell me they rely on this combo around nitro compounds, especially during cleanup.
Choosing the Right Space and Storage Habits
You can’t ignore ventilation. Fume hoods offer a safe space for weighing and transferring 2,6-Dinitroaniline. Good airflow sweeps away stray dust that settles on clothes, benchtops, and shelves. In shared workplaces, using a fume hood earns nods of approval and sets a standard that others are quick to follow.
Storage determines whether you wake up to a safe lab or a worried call from security. Store the chemical in tightly-sealed glass or high-quality plastic containers, kept cool and dry. Always mark the date it came in, since some nitroaromatics get unstable over time. In my lab, aging stock is always checked during monthly inventory. If you work with small quantities, you minimize the outcome of a spill or accidental exposure.
Spilling and Emergency Response
Nobody plans for a spill, but everyone who works hands-on with chemicals has managed one at some point. Clean up any powder with a vacuum equipped for hazardous dust, not a broom or compressed air. Dumping water straight on a spill can make things worse. Mix with inert absorbent material like vermiculite and seal for disposal. Have an eyewash station and safety shower within a short walk, and keep spill kits visible—both build peace of mind for everyone.
Training for Real-Life Hazards
Experience comes with mishaps, but solid training shortens the learning curve. I’ve watched young technicians learn that reading the SDS once isn’t enough. Chemical safety drills, demonstrations, and open chat about what nearly went wrong have more impact than thick manuals. Keeping the lines open means fewer screw-ups and a stronger safety culture. In places where hazmat training covers specific risks of nitro compounds, you see fewer accidents and more responsible procedures.
Good safety habits aren’t about paranoia—they’re about respecting chemistry. 2,6-Dinitroaniline teaches anyone working with it to pay attention, take things slow, and never get careless. That’s experience speaking, not a warning label.
Is 2,6-Dinitroaniline hazardous to health or the environment?
Getting to Know 2,6-Dinitroaniline
Most people won’t come across 2,6-dinitroaniline unless they work in labs or industries dealing with dyes or chemical synthesis. It doesn’t pop up in daily life like plastics or gasoline, but that shouldn’t mean we ignore what it brings to the table. Before anything lands in industrial formulas, safety ought to sit front and center—and health and environmental impact play serious roles in that.
Health Hazards of 2,6-Dinitroaniline
Researchers know 2,6-dinitroaniline as a substance that releases toxins if mishandled. Breathing in dust from this chemical—or letting it linger on your skin—could lead to problems. Inhaling too much can irritate your lungs, spark headaches, or cause dizziness. Even handling the powder without protection introduces risk, as skin contact may cause redness or burning. There’s not enough solid data to say it causes cancer, but the lack of deep research leaves a gray area that worries many occupational health experts.
What’s striking is that many health effects linked to nitroanilines, in general, include methemoglobinemia—a blood disorder where oxygen delivery drops. Symptoms sometimes go unnoticed at first: fatigue, shortness of breath, blue lips. Over time, these effects pile up. In my own days as a lab assistant, safety data sheets jammed the message home: goggles, gloves, and good ventilation aren’t a suggestion—they’re the rule.
Impact on the Environment
No one wants chemicals escaping into soil or waterways. 2,6-dinitroaniline, thanks to its chemical makeup, doesn’t break down quickly. If it slips into rivers, it sticks around long enough to harm aquatic life. Fish exposed to compounds like these can suffer from growth issues and trouble reproducing. Food chains in rivers and ponds depend on balance, and once you tip the scale, it’s tough to put things right.
Soil contamination raises its own set of problems. Plants, including ones we eat, may absorb these compounds through their roots. That’s not just a worry for wildlife; those substances can end up in food supplies. Farmers, especially those near manufacturing spots, wind up carrying the burden if runoff reaches fields. Regulators in countries like the United States and in the European Union recognize this risk and put rules in place about storage and disposal.
Working Toward Safer Practices
Safer chemical handling starts with good information. Clear labels and training give people the power to make safe choices at work. In plants that manufacture or use 2,6-dinitroaniline, closed systems and local exhaust fans cut exposure. Spill kits and emergency showers back up any slip-ups. Down the line, companies can turn to less hazardous alternatives if possible.
It makes sense to keep pushing for greener alternatives. Governments fund research to find safer replacements for high-risk chemicals used in industry. The market responds, too, when buyers insist on chemicals with better safety profiles. Addressing legacy pollution takes more than rules; it calls for cleanup projects and community transparency. Local voices count, especially for folks living closest to factories.
Looking Ahead
The story of 2,6-dinitroaniline reminds us not to overlook invisible dangers. Protecting health and nature rests on solid information, thoughtful regulation, and clear communication. Talking with people who actually use or live near these substances gives everyone a stronger shot at safety.
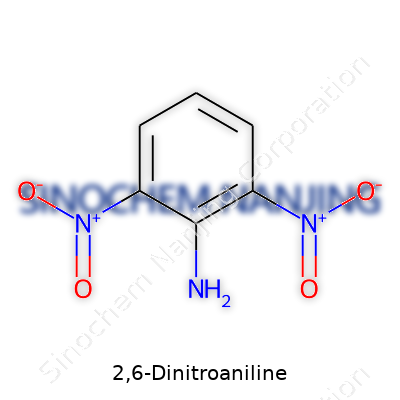
What is the chemical structure and formula of 2,6-Dinitroaniline?
The Basics: What Is 2,6-Dinitroaniline?
2,6-Dinitroaniline is a fairly unique chemical, which shows up in several synthetic routes, especially in making dyes or specialized agricultural chemicals. Structurally, the core comes from aniline—a benzene ring with an amino group (–NH2) attached. In this compound, two nitro groups (–NO2) sit on the ring, precisely at positions 2 and 6. This specific arrangement pushes electron density around the molecule in a way that changes how it reacts with other substances.
Chemical Structure and Formula Decoded
The chemical formula for 2,6-Dinitroaniline is C6H5N3O4. If you sketch it out, you’ll see a benzene ring at the center. One amino group sits on position 1, with nitro groups on positions 2 and 6—count around the ring from the amino group. Those two NO2 groups attach to opposite sides, but both right next to the amino’s location.
This may sound simple, but the arrangement actually makes a major difference compared to its close relatives. For instance, move just one nitro group to another spot, and you end up with a compound that has different chemical and biological behavior. For a chemist, having the right structure rules out costly mistakes in synthesis, so accuracy matters in the lab and everywhere this material gets used.
Real-World Uses, Risks, and Safety
I’ve seen 2,6-Dinitroaniline show up outside the textbook—especially in dye manufacturing. It acts as a key intermediate for azo dyes and sometimes in specialty pigments. Its nitro groups bring a level of reactivity that chemists count on to build complex molecules. Anywhere colorfastness and vibrant shades get prioritized, you might find a process that starts with or passes through a dinitroaniline.
On the other side, safety concerns follow compounds with nitro and amino groups. For example, nitroanilines in general can show toxic effects in people and animals. Mismanagement means environmental spills drift through water tables or linger in soil. So, solid waste protocols and responsible sourcing become more than bureaucratic hurdles—they protect people’s health.
Responsible Handling and Environmental Impact
I remember seeing best practices in a dye lab: gloves, goggles, fume hoods, and clear labeling. Laboratories and production lines build in these controls, from proper ventilation to dedicated waste collection drums. Organic compounds with nitro groups often don’t break down easily, so they demand careful disposal. Green chemistry—a real, growing field—pushes chemists to look for ways to reduce toxic byproducts or even swap out tough-to-handle compounds like dinitroanilines for safer alternatives.
Better monitoring and remediation help after the fact, but prevention works even better. Simple steps like double-checking chemical inventories, using the smallest possible batch size, and careful employee training stop leaks before they start. In my experience, training saves more headaches than any cleanup plan.
Innovation and Alternatives
Research teams keep working toward replacements for legacy chemicals. There’s movement toward renewable feedstocks and benign-by-design molecules, which lower health and environmental costs over the product’s lifecycle. Sometimes, a greener pathway just means swapping out a single intermediate. At other times, the whole synthesis route moves in a new direction. The field doesn’t change overnight, but a steady shift away from persistent chemicals leads to few regrets in the long run.
How should 2,6-Dinitroaniline be stored and disposed of?
A Chemical Worth Respecting
2,6-Dinitroaniline pops up in labs with a clear warning. Its bright yellow color and powdery look often hide the risk. Explosive dust, toxic fumes, and a knack for causing headaches—this compound deserves attention. My own time in university research taught me early on: never treat nitro-aromatics lightly. They demand respect before anything else.
Safe Storage: Setting Up for Success
The main thing with 2,6-Dinitroaniline is to keep it cool, dry, and out of sunlight. Damp rooms or direct heat will ask for trouble. Glass or high-quality plastic containers with airtight lids work best, and every label needs to jump out at you in bold, clear print. It helps the next person avoid guessing games.
Store this stuff in a well-ventilated area away from acids or bases. No need for wild chemical reactions behind closed doors. Flammable materials or anything that burns fast stay as far away as possible. Locked cabinets or chemical storage rooms don’t just follow rules—they protect real people. There’s rarely a good reason for anyone except trusted staff to touch these containers.
Local fire codes often require explosion-proof storage cabinets for nitro compounds. While this might sound extreme, small accidents only happen once before people scramble to fix the gaps. If there’s ever a spill, paper towels and wishful thinking aren’t enough. Absorb with a compatible material, bag it, and find a hazardous waste drum before anything spreads or fumes fill the air.
Disposal: Not a DIY Job
No matter what the internet says, dumping 2,6-Dinitroaniline in the sink or trash does more harm than good. Standard landfill trips let toxins leak into water systems and soil. I’ve seen local news cover chemical leaks into rivers just because someone thought “dilution is the solution.” This mindset damages trust in science and harms the environment.
The right call is turning to trained hazardous waste handlers. Regulations spell out the steps for incineration or chemical treatment—the “burning question” usually has just one answer. A licensed waste company collects the compound, tracks every step, and confirms safe destruction. Governments usually want a paper trail so no polka-dotted powder pops up in places it should never go.
Staying Ahead with Good Training
Handling and storing chemicals like this isn’t about memorizing rules. It starts with strong training, sharp observation, and a culture that encourages double-checking before acting. Supervisors I worked with always stressed preparation, never shortcuts. Companies and schools should run refresher sessions, keep up with changing legal frameworks, and treat old containers as hazards, not hand-me-downs.
More Than Just Compliance
Careful handling of 2,6-Dinitroaniline isn’t just a legal duty—the whole process keeps people healthy and the environment clean. Simple habits—clear labels, tight lids, controlled access—set the standard for protecting labs and communities. The small step of calling professionals for disposal keeps workplaces out of the headlines and groundwater free from toxins.
| Names | |
| Preferred IUPAC name | 2,6-dinitrobenzenamine |
| Other names |
2,6-Dinitrobenzenamine 1-Amino-2,6-dinitrobenzene |
| Pronunciation | /ˌdaɪˌnaɪtroʊˈænɪliːn/ |
| Identifiers | |
| CAS Number | 97-02-9 |
| Beilstein Reference | 1360260 |
| ChEBI | CHEBI:28255 |
| ChEMBL | CHEMBL34018 |
| ChemSpider | 20706 |
| DrugBank | DB08499 |
| ECHA InfoCard | 100.025.080 |
| EC Number | 208-698-6 |
| Gmelin Reference | 7789 |
| KEGG | C14369 |
| MeSH | D017972 |
| PubChem CID | 7286 |
| RTECS number | BU8925000 |
| UNII | Q8T9R6T52J |
| UN number | UN1320 |
| CompTox Dashboard (EPA) | 7B9Z78644E |
| Properties | |
| Chemical formula | C6H5N3O4 |
| Molar mass | 198.13 g/mol |
| Appearance | Yellow to orange powder. |
| Odor | Odorless |
| Density | 1.57 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 1.98 |
| Vapor pressure | 1.58E-4 mmHg at 25 °C |
| Acidity (pKa) | 12.97 |
| Basicity (pKb) | 10.10 |
| Magnetic susceptibility (χ) | -41.5·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.675 |
| Viscosity | 2.07 cP (25°C) |
| Dipole moment | 2.98 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 190.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -71.0 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1062.4 kJ/mol |
| Pharmacology | |
| ATC code | D06AA10 |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and eye irritation, may cause respiratory irritation, possibly explosive if heated. |
| GHS labelling | GHS02, GHS07, GHS08 |
| Pictograms | GHS06, GHS03 |
| Signal word | Warning |
| Hazard statements | H302, H319, H335, H351 |
| Precautionary statements | P261, P280, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 2,2,0,OXP |
| Flash point | 140 °C |
| Autoignition temperature | 520 °C (968 °F; 793 K) |
| Lethal dose or concentration | LD50 oral rat 570 mg/kg |
| LD50 (median dose) | Rat oral LD50: 640 mg/kg |
| NIOSH | JN9625000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.05 mg/m³ |
| IDLH (Immediate danger) | IDLH: 50 mg/m3 |
| Related compounds | |
| Related compounds |
Aniline 2,4-Dinitroaniline Nitroaniline 2,6-Dinitrotoluene Dinitrobenzene |

