Reflecting on 2,6-Dimethylmorpholine: Insight and Impact
Tracing the Path from Origins to Industry
2,6-Dimethylmorpholine stands as a chemical whose journey mirrors the bigger story of specialty amines in modern chemistry. Developed in the haze of post-war chemical innovation, this compound didn’t spring out of nowhere. Researchers in the mid-20th century started shaping substituted morpholines, hunting for greater efficiency in production and broader flexibility across industries. As old-school methods of synthesizing morpholine derivatives relied heavily on trial and error, gradual improvements led to the discovery of more selective routes that curbed byproducts and made scaling up more reliable and less wasteful. This march toward cleaner, more predictable synthesis helped turn 2,6-Dimethylmorpholine from a shelf curiosity into something that quietly powers sectors ranging from pharmaceuticals to specialty coatings.
What the Product Really Offers
From a user’s point of view, the main reason anyone turns to 2,6-Dimethylmorpholine is simple—it brings a unique blend of reactivity and selectivity. The two methyl groups nestling at the 2 and 6 positions shape its solubility and chemical behavior. This molecule resists easy classification as just another intermediate. Chemists appreciate its performance in processes demanding tough solvents or when you search for a precise alteration of chemical scaffolds. Whether working with drug design or tweaking epoxies, you know you’re getting a compound that won’t overreact, but can still lend a hand where fine-tuning is needed most.
Understanding Physical and Chemical Traits
Its physical shape—a small, cyclic amine—sets the tone for 2,6-Dimethylmorpholine’s chemistry. Usually, it appears as a clear to slightly yellow liquid, owing to its fully saturated ring and the two added methyl decorations. With a boiling point higher than simpler morpholines and water-like miscibility for polar solvents, I’ve seen it handle conditions that would unravel many amines. Its basicity runs a touch lower than that of unsubstituted morpholine, an effect that echoes through its reactivity. When handling this chemical, I value the lower volatility, which eases the pressure in manufacturing settings lacking expensive containment gear.
What Technical Specs and Labeling Mean in Practice
Labeling and documentation around 2,6-Dimethylmorpholine hardly exist for show; they protect life and limb. Tightly defined purity ranges and specific handling instructions act as a shield for everyone in the lab. Careless mislabeling can lead to surprise incidents, especially since the chemical’s activity swings depending on impurities. So when regulatory standards call for detailed breakdowns—right down to ppm levels of suspect contaminants—the intent is not to bury users in paperwork, but to reduce real-world risk. Technicians who know their way around these labels not only keep the process running smoothly, but also help sidestep costly shutdowns from health and safety violations.
Getting from Raw Materials to Finished Product
Preparation of 2,6-Dimethylmorpholine rarely happens on a hobbyist’s bench. Industrial production works by reacting 2,6-dimethyl-1,4-dichlorobutane or related precursors with ammonia or secondary amines in the presence of a catalyst. This method, while seemingly simple, demands careful control of temperature and pressure to keep tars and side products in check. Experienced operators pay attention to sequence, since feeding chemicals out of order leads to dark, sticky messes that nobody wants to clean up. Cleanroom techniques may not be essential, but sloppy work brings headaches that escalate as batch sizes increase. It’s not just about yield; it’s about making something people can trust downstream.
Chemical Reactions and Modifications That Matter
2,6-Dimethylmorpholine doesn’t just sit in a bottle. Its core structure makes it a flexible starting point for further chemical adventures. I’ve seen teams modify it at the nitrogen atom, swapping in alkyl or acyl groups, or even oxidizing the ring for added functionality. The methyl groups give a bit of steric hindrance, meaning reactions often happen slower and more selectively than with bare morpholine. Synthetic chemists like this compound when looking to control reaction pathways and suppress unwanted branches. End uses range from introducing quirky substituents to core pharmaceuticals all the way to specialized stabilizers in polymer production.
What to Call It and Why It Matters
It helps to know that 2,6-Dimethylmorpholine goes by more than one name. Synonyms include 2,6-Dimethyl-4-morpholine and its systematic IUPAC name, N,O-Tetramethylethanolamine. In databases or regulatory documents, catching the right synonym prevents expensive mix-ups, especially where safety and compliance are concerned. Skipping due diligence on nomenclature in tech transfer or procurement can stall projects for weeks, or worse, introduce unintended hazards by misidentifying toxic or incompatible materials.
Setting a High Bar: Safety and Operational Standards
Any time I work around 2,6-Dimethylmorpholine, the stakes feel high because the hazards, though manageable, are real. Its low volatility eases inhalation risk compared to other amines, but that doesn’t make safety eyewear or gloves optional. Good lab hygiene, proper ventilation, and strong adherence to permissible exposure limits not only protect workers—they also build cultures of respect and accountability that keep businesses resilient. Waste management for spent solutions or off-spec product demands attention, since organic amines have environmental risks that extend far beyond initial handling. Downplaying safety in this context often leads to incidents and regulatory scrutiny that can ruin reputations or even shutter facilities.
Where 2,6-Dimethylmorpholine Finds Real Use
Application stretches wide for 2,6-Dimethylmorpholine. Its resistance to unwanted side reactions and unique reactivity carve out roles in specialty polymers, epoxy resins, and the fine chemicals sector. Pharmaceutical developers use it as a valuable intermediate, particularly where chirality or functional group control is key. In my own experience with paints and coatings, 2,6-Dimethylmorpholine serves as a catalyst or auxiliary, lending weather-resistance and durability. It also attracts interest from agrochemical producers for synthesis of selective pesticides, balancing efficacy with environmental footprint. Each application draws on the molecule’s stubbornness against unwanted decomposition and ability to customize properties through fine chemical manipulation.
Research and Development: Chasing Better Solutions
Research labs worldwide keep tuning the chemistry and economics around 2,6-Dimethylmorpholine. Current R&D priorities include refining synthesis for lower energy input and waste generation. Tougher environmental regulations press producers to develop closed-loop methods, recapture solvent, and eliminate legacy contaminants. Teams explore greener catalysts and push analytical tech to make quality control sharper and quicker. Broader questions run through pharmaceutical, agricultural, and material innovation: can this core structure unlock new performance in self-healing materials or more selective drug candidates? Industry-academic partnerships look poised to stretch what this morpholine derivative can do far beyond its original scope.
Stopping to Think: Toxicity and Worker Health
Scientific literature tells us 2,6-Dimethylmorpholine can be a skin and eye irritant and shouldn’t be taken lightly. Chronic exposure evidence remains sparse, but I wouldn’t trust long-term safety to hope or tradition. Animal studies point to mild toxicity at substantial doses; lower level risks for reproductive or developmental harm lack confirmation, pushing many employers to err on the side of caution. Standard toxicological probes only scratch the surface, especially in combined exposures, so continuous review of worker health and periodic risk assessments stay crucial in any setting where chemicals like this play a daily part.
Looking Forward: Where Does It Go from Here?
Prospects for 2,6-Dimethylmorpholine keep evolving as industries shift toward tighter safety nets and greener methods. Synthetic routes look set for further streamlining, cutting both costs and the environmental burden tied to legacy manufacturing. Uptake in high-performance resins and functional polymers appears likely to grow, especially as technologies demand materials that hold up in tougher conditions. Patents covering new uses continue to surface, hinting at applications just over the horizon, from improved medical imaging reagents to smart-release agricultural agents. Harnessing the full potential of 2,6-Dimethylmorpholine requires balancing safety, economics, and forward-thinking design—a challenge worth the effort for chemists, manufacturers, and end-users in equal measure.
What is 2,6-Dimethylmorpholine used for?
Hidden Value Behind a Simple Structure
Walk into any modern chemical plant and you'll probably never hear much about 2,6-Dimethylmorpholine by name. That sounds like a minor character in a long list of substances, but for anyone who's worked on large-scale process chemistry or with polyurethane foams, it's a workhorse. The molecule, a cyclic amine with two methyl groups, pulls more weight than its name suggests. It shows up in situations that call for precise, repeatable reactions—think hard coatings, spongy foams, and reliable adhesives.
The Role in Polyurethane Foam Production
Foam seems simple until you try to make millions of sofa cushions or car seats every year. I remember standing in a factory as sheets of white, springy foam emerged, not thinking twice about what made the reaction so controlled. The answer often involves a clever catalyst, and 2,6-Dimethylmorpholine has carved out its place as a polyurethane catalyst. Without that sort of amine-based catalyst in the mix, the reaction between polyols and isocyanates either slows down to a crawl or goes wild—never a happy medium. A steady supply of foam with just-right spring and density keeps factories running and furniture companies happy.
Supporting Solvent Systems
Beyond just foams, specialty coatings, and adhesives benefit from solvents with tailored properties. 2,6-Dimethylmorpholine dissolves well in both water and many organic solvents, making it a handy tool for adjusting paint viscosity or helping ingredients mix evenly. A balanced formula matters in products where too runny or too thick can ruin an entire batch, driving up costs and creating waste. Chemists appreciate any reliable solvent or co-solvent that brings stability to their mixes, and this morpholine derivative wins trust by doing its job quietly in the background.
Importance in Fine Chemicals and Pharmaceuticals
Sometimes the game changes entirely—like in drug development. 2,6-Dimethylmorpholine finds use as a building block in fine chemical synthesis. Considering the pressure and scale involved in making pharmaceutical precursors, even a small shift in yield or purity has a massive impact downstream. Reliable intermediates keep supply chains running, and every chemist who’s ever pushed a reaction one step closer to a needed medicine values molecules that work without fuss.
Handling the Safety Factor
In everyday industry use, practical experience says: treat all chemicals with respect. 2,6-Dimethylmorpholine isn’t particularly infamous for toxicity, but direct exposure in cramped, poorly ventilated spaces can still irritate eyes, skin, or the respiratory tract. On-site training and good ventilation go miles toward keeping people safe. Regular monitoring and up-to-date material safety data sheets keep teams ready. There’s no shortcut to safety, and anyone managing production lines appreciates direct messaging and clear protocols.
Pushing Toward Greener Chemistry
Sustainability now matters more than ever. Customers and regulators want less waste and fewer emissions. While 2,6-Dimethylmorpholine does what it’s always done for polymers and coatings, research around new processes won’t stand still. Scientists look for ways to make traditional molecules from renewable sources, recycle when possible, and improve every step for a lighter footprint. Early-stage projects suggest advances in catalyst efficiency and raw material sourcing. Each improvement chips away at the old problems of waste, expense, and risk, building a smarter way forward in chemical manufacturing.
What is the chemical formula of 2,6-Dimethylmorpholine?
Understanding 2,6-Dimethylmorpholine
2,6-Dimethylmorpholine might not make headlines every day, but it’s one of those niche chemicals that keep science and industry ticking along. Its chemical formula is C6H13NO. Behind those seven letters and numbers, there’s a real structure that influences how the chemical behaves in the lab and on the factory floor. I’ve seen how one misplaced number or letter in a formula could toss a whole batch off course. From formulation errors to safety hazards, small details carry big weight in real-world settings.
Molecular Structure: More Than Just Numbers
The backbone of 2,6-Dimethylmorpholine is a six-membered morpholine ring, which combines oxygen and nitrogen atoms in its structure. The 2 and 6 methyl groups add some bulk, and those extra methyl groups shift its properties in significant ways. People often assume all morpholines act the same way, but subtle differences like this mean different boiling points, different reactivities, and even altered toxicity profiles. Over time, I’ve learned not to gloss over such variations. Checking the structure helps avoid guesswork, especially in projects dealing with drug development or chemical synthesis where purity and specificity matter.
Uses and Practical Implications
Morpholines, and especially methylated derivatives like 2,6-Dimethylmorpholine, pop up in a range of applications. These range from polymers, specialty solvents, to pharmaceuticals and agricultural chemicals. Its basicity and reactivity owe a lot to that formula—C6H13NO packs just the right arrangement for niche reactions. Years ago, pharmacists I worked with leaned on this compound for custom synthesis routes, placing real trust in the accuracy of these formulas. Inconsistent composition meant failed reactions and lost time.
Safety and Handling Concerns
Chemicals with nitrogen and oxygen atoms, like 2,6-Dimethylmorpholine, can raise red flags in terms of safety and environmental impact. Inhalation risks, flammability, and reactivity are just the start. During one stint consulting for a specialty coatings factory, overlooking the formula meant incorrect storage recommendations. It’s simpler to maintain safety when formulas are respected and safety datasheets reflect each detail. Wrong assumptions could drive serious health issues—occupational asthma, eye irritation, and more.
Improving Communication in Science
Precise chemical formulas aren’t just technical details—they are communication tools. I once saw a team flustered over mismatched datasheets across suppliers. A slight deviation in naming could mean a different isomer, leading to wasted money and production delays. Standardized reporting of compounds, including the chemical formula C6H13NO for 2,6-Dimethylmorpholine, makes it easier for everybody from lab techs to policy regulators to stay on the same page.
Possible Solutions and Best Practices
Reliable scientific communication starts with verified data sources. Cross-referencing with resources like PubChem or Sigma-Aldrich catalogs reduces error. Training for young chemists should stress not just memorization but fact-checking. Inside the lab, keeping thorough records and double-checking chemical identification lowers risk. My own routine has always included a couple of minutes to scan over formulas, even on trusted datasheets. Open talk between suppliers, researchers, and end users ensures transparency and shared responsibility for safety and accuracy.
Is 2,6-Dimethylmorpholine hazardous or toxic?
Understanding What 2,6-Dimethylmorpholine Does
The name 2,6-Dimethylmorpholine doesn’t roll off the tongue, yet it ends up in more chemical settings than most people realize. It gets used in synthesizing other chemicals and serves as a building block for coatings, solvents, and even pharmaceuticals. Many chemicals in labs and factories don’t make headlines or raise eyebrows until someone asks, “Is this stuff safe?”
What Science Says About the Risks
Researchers have studied 2,6-Dimethylmorpholine’s impact on people and the environment, but finding detailed toxicity data takes effort. Safety data sheets do show that the substance can cause skin and eye irritation. Lab tests reveal that higher concentrations might harm organs if people breathe the vapors for long or spill them on skin repeatedly. Accidental swallowing definitely raises red flags, based on animal studies. Workers handling the pure compound without gloves or a mask gamble with their health, as the chemical’s fumes can irritate the respiratory tract.
The big picture in chemical safety draws from comparisons. 2,6-Dimethylmorpholine doesn’t seem as notorious as classic industrial toxins. For example, chemicals like benzene and chloroform pose bigger risks, showing clear links to cancer and organ damage. Still, that does not give a free pass to 2,6-Dimethylmorpholine. The lack of headlines often just means fewer studies or lower volumes in use.
Regulation Makes a Difference
Governments watch over chemical safety, but enforcement varies by country and application. In the U.S., the Environmental Protection Agency demands reporting of significant new uses for many industrial compounds, including certain morpholine derivatives. Local workplace regulators require protective gear and proper ventilation around chemicals labeled as irritants or possible health hazards. European agencies label 2,6-Dimethylmorpholine as “harmful if swallowed” and “irritant,” so companies must list it clearly on shipping manifests and storage containers.
Real-life stories from factory shifts and research labs show that ignoring these labels brings trouble. Some labs have seen chemical burns or rashes after someone cut corners. It doesn’t help that 2,6-Dimethylmorpholine looks and smells like a lot of other harmless chemicals on the shelf.
Small Risks Multiply Without Respect
A chemical that irritates skin or lungs may not sound like the villain in a news report, yet small injuries add up. A lost afternoon due to a rash or wheezy lungs means real money gone and morale dropping. If routine spills or lax procedures build up, workers start taking unnecessary chances, thinking nothing bad will happen. This attitude leads to accidents, injuries, and sometimes legal trouble for businesses blind to workplace safety.
Building a Culture of Prevention
Keeping 2,6-Dimethylmorpholine from turning into a health statistic involves more than paperwork. Proper labeling, good ventilation, and consistent use of gloves and goggles keep people safer. Training every worker to treat every bottle seriously pays off, not just by following laws, but by avoiding downtime and medical bills. Veteran chemists I’ve worked with say nothing replaces walking through the safety steps and watching out for colleagues. If a new substance shows up, reading the material safety data sheet and checking for latest updates should come before turning on equipment.
A few years back, I worked in a mid-sized research lab with rotating interns and grad students. The most important lesson taught by our safety officer stuck with me: respect every bottle as if it’s dangerous, because sometimes it is. If in doubt, ask. That habit kept our crew out of the emergency room, and it still shapes my approach to chemical safety today.
How should 2,6-Dimethylmorpholine be stored?
Safety Depends on Awareness
People use 2,6-Dimethylmorpholine for all sorts of specialty reactions in chemistry, so questions about storage crop up more often than many realize. Years ago, I worked in a lab that logged nearly every bottle that came through the door. The real test came not when new materials arrived, but a few months later, when the labels started curling, the boxes got shuffled, and someone remarked, “Has this been stored right?” That’s when good habits show their worth.
Understanding the Risks
Chemists classify 2,6-Dimethylmorpholine as a flammable liquid. Even if your workspace uses only a small amount, careless handling can trigger big problems. This isn’t hypothetical. A friend’s lab had to deal with a fire in a storage cabinet—nothing dramatic, but they lost equipment, research, and sleep. The chemical itself gives off vapors that can form explosive mixtures with air. Investigations show many accidents start in overcrowded storage spaces where incompatible chemicals get stacked together.
Choosing the Right Location
Solid walls, ventilation, and a touch of common sense separate a good storage system from one that invites trouble. Flammable liquids need to stay in OSHA-approved flame-proof cabinets. Never leave bottles out on a benchtop or in direct sunlight. Temperature swings affect both safety and the quality of the product, so a cool, dry spot turns out to be the best bet. You’ll want to avoid areas near oxidizers or acids; mixing these can set the stage for unwanted reactions. Labels help, but a clear, updated chemical inventory system goes further.
Mitigating Accidents with Proper Planning
You can read mountains of policy, but experience shows the practical steps work best. Never store more than you need for current projects. Cap containers tightly, and use secondary containment. If a leak happens, a tray keeps it contained. Regulatory groups like the National Fire Protection Association and local fire codes offer clear guidelines because even small spills lead to expensive cleanups and can expose your team to toxic fumes. If you work in an environment where hundreds of reagents fill cabinets, regular inspections make a difference.
Training Is as Key as Locks
People think about locks, alarms, and secure doors—these all help. But knowledge matters more. Every person in the lab or shop space ought to know exactly where items go and why. In my current lab, new hires shadow experienced staff and learn why each cabinet holds its particular set of chemicals. Mistakes happen most often when folks work alone or try shortcuts. Reviewing safety data sheets together reinforces smart practices, keeps everyone on the same page, and goes a long way to prevent injury and material loss.
Simple Steps, Long-Term Gains
Every time I walk past a properly labeled flammable cabinet, it reminds me: safe storage begins with personal responsibility. Distribute chemicals thoughtfully, minimize excess, and refresh training regularly. These habits shield you from accidents and avoid regulatory headaches. With a sensible approach and a bit of vigilance, tragic stories become relics of the past.
What are the safety precautions when handling 2,6-Dimethylmorpholine?
Thinking Beyond the Lab Manual
Working with chemicals like 2,6-Dimethylmorpholine goes deeper than glancing at a safety sheet. My early years in chemistry taught me that trust in routine breaks down fast the first time a chemical stings your nose or irritates your skin. 2,6-Dimethylmorpholine, often found in polymer work or as a catalyst, can slip past the careless user’s guard. Fumes from this compound cause headaches, dizziness, and lung irritation if they build up. Direct contact brings redness and a burning sensation. Stories abound about folks finding out the hard way, usually because someone skipped simple protections.
Why Good Ventilation Beats Regret
An open window rarely cuts it in a busy lab or workshop. Running a fume hood makes an obvious difference; enough airflow sends those hazardous vapors up and away, not circling near faces. The one time I cut corners—no fun at all. I learned to double-check air flow and keep fresh air moving, not just to avoid trouble, but because tired, foggy heads make mistakes easy. Having carbon filters or exhaust fans ready means not lying awake wondering if a headache is just from overwork.
Hands and Eyes—Keep Them Covered
Even when 2,6-Dimethylmorpholine doesn’t splash, mistakes happen in busy spaces. I use nitrile gloves, skipping latex because some people break out or develop rashes. Lab coats and closed-toed shoes sound obvious until someone forgets and ends up with chemical-soaked socks. Safety goggles never feel optional; they catch fine droplets drifting up or sprays from spilled glassware. I saw a colleague once rub an itchy eye with a glove. He knew better, but those seconds spent rinsing at the eye wash station stretched out long and frightening. Lessons like that drive the point home.
Storage—Not Just Put Away on a Shelf
Throwing 2,6-Dimethylmorpholine into a crowded cabinet never ends well. I always keep this chemical far from acids and oxidizers. Sealed containers, clearly labeled, go in dedicated spots, not just for convenience but to catch mistakes before they happen. A dry, cool area thwarts leaks and fumes. Spillage in storage can be a nightmare. I’ve seen it corrode shelves and trigger cleanup drills because vapors spread so fast. Simple precautions—lined shelves and spill trays—keep headaches small and cleanups fast.
Don’t Skip Training and Emergency Prep
Knowing the emergency plan saves time during spills or accidental exposure. I’ve trained new folks by walking through real what-ifs: someone knocks over a beaker, forgets their gloves, or misses a warning label. Precise instructions on using eyewash stations, showers, and fire extinguishers don’t seem dramatic until you actually need them. Relying on muscle memory means no panic, just action. Keeping a spill kit ready—absorbent pads, neutralizers, and sealed disposal bags—makes the law and the workplace both easier to navigate.
Respect Goes a Long Way
Chemicals like 2,6-Dimethylmorpholine don’t shake your hand before hurting you. Over time, respect for these risks grows from countless reminders that comfort and habit never outweigh preparation. I check labels, trust my training, and handle spills head-on. The more consistently we treat safety gear and procedures as everyday tools, not burdens, the better our labs, shops, and lungs stay.
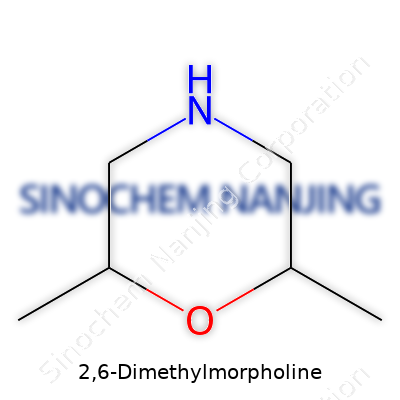
| Names | |
| Preferred IUPAC name | 4,6-Dimethylmorpholin-2-amine |
| Other names |
2,6-Dimethyl-4-morpholine 2,6-Dimethyl-1,4-oxazinane Morpholine, 2,6-dimethyl- |
| Pronunciation | /ˈtuː,sɪks-daɪˈmɛθɪl-mɔːrfəˌliːn/ |
| Identifiers | |
| CAS Number | 6365-45-9 |
| Beilstein Reference | 76594 |
| ChEBI | CHEBI:85347 |
| ChEMBL | CHEMBL3213783 |
| ChemSpider | 157186 |
| DrugBank | DB08372 |
| ECHA InfoCard | ECHA InfoCard: 100.012.339 |
| EC Number | 210-045-3 |
| Gmelin Reference | 9203 |
| KEGG | C19268 |
| MeSH | D016693 |
| PubChem CID | 10937 |
| RTECS number | QJ7875000 |
| UNII | BMT1M10327 |
| UN number | 2264 |
| Properties | |
| Chemical formula | C6H13NO |
| Molar mass | 115.18 g/mol |
| Appearance | Colorless to yellow liquid |
| Odor | amine-like |
| Density | 0.925 g/mL at 25 °C |
| Solubility in water | miscible |
| log P | 0.2 |
| Vapor pressure | 0.9 mmHg (20 °C) |
| Acidity (pKa) | 9.15 |
| Basicity (pKb) | pKb = 5.83 |
| Magnetic susceptibility (χ) | -48.4·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.429 |
| Viscosity | 1.9 mPa·s |
| Dipole moment | 2.75 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 218.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -285.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4037.8 kJ·mol⁻¹ |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | Harmful if swallowed. Causes severe skin burns and eye damage. |
| Precautionary statements | P261, P280, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 1-3-0 |
| Flash point | 46 °C |
| Autoignition temperature | 245 °C |
| Explosive limits | 1.5-9.4% |
| Lethal dose or concentration | LD₅₀ (oral, rat): 910 mg/kg |
| LD50 (median dose) | LD50 (median dose): 820 mg/kg (rat, oral) |
| NIOSH | NQ3675000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 10 ppm |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
Morpholine 2,3-Dimethylmorpholine 2,5-Dimethylmorpholine 2,6-Diethylmorpholine 2-Methylmorpholine 4-Methylmorpholine |

