The Many Faces of 2,6-Dimethylaniline: More Than Just a Chemical
Historical Development
2,6-Dimethylaniline has been around for more than a century, quietly shaping key industrial transformations. Its roots trace back to the golden era of dye and pigment innovation when chemists began exploring the potential of methyl substitutions on aniline rings. The push for brighter, stabler colors for textiles steered research towards compounds like this one. Industry often looked for ways to improve performance in dyes and intermediates, so this compound naturally found a place in much of that early work. As markets shifted and chemical processes matured, people realized 2,6-Dimethylaniline wasn’t just a pigment precursor but also a springboard for a range of chemical syntheses used in pharmaceuticals and agrochemicals. Once it became more widely available, new routes and uses popped up as researchers tinkered with its characteristics, often seeking to escape reliance on natural sources and instead manufacture these building blocks reliably from coal tar and later petroleum derivatives. Its continued use shows how some early choices in industrial chemistry echo for generations.
Product Overview
Chemists often encounter 2,6-Dimethylaniline in its pure form as a pale yellow to brown liquid, its sharp amine odor unmistakable in labs equipped to handle aromatic amines. Aside from academic interest, large-scale manufacturers see it as a critical intermediate—the real value lies in how the two methyl groups tweak the reactivity of the aniline structure. This subtle change produces clear advantages in downstream reactions: from dye couplings to pharma synthesis, greater control and cleaner outcomes draw continued demand. I’ve worked in labs choosing intermediates for custom molecules, and the presence of those ortho methyls often meant less byproduct and simpler purification, saving time and resources.
Physical & Chemical Properties
Looking at the substance itself, 2,6-Dimethylaniline brings traits that are hard to ignore. It melts just above room temperature and boils far higher, a fact that makes handling easier in most climates. It dissolves in organic solvents like benzene or ether quite readily but refuses to blend with water—no surprise given its aromatic backbone and presence of methyl groups. Its reactivity centers on the amino group, but those methyls at the ortho positions give it a degree of stability compared to plain aniline. That means fewer unwanted reactions, a helpful feature in industrial settings looking for reproducibility. In my time analyzing such compounds, I found these traits allow for predictable processing—good for companies and researchers alike.
Technical Specifications & Labeling
On the labeling and handling front, standards exist for a reason: the compound’s potential hazards, from its volatility to its toxicity, have been documented for decades. Precise purity reporting, detailed chemical identifiers, and clear hazard labels aren’t just red tape; they provide frontline workers with a fighting chance at avoiding unnecessary exposure. The technical side—batch purity, impurity profiles, packaging integrity—comes under meticulous scrutiny not just to meet regulations, but because errors here can compromise entire product lines. I’ve seen projects grind to a halt due to a missed contaminant or mislabeled container. Even small deviations can produce alarms in downstream users.
Preparation Method
Preparing 2,6-Dimethylaniline doesn’t require exotic chemistry but does demand careful process control. The established route starts with 2,6-dimethylnitrobenzene, reduced typically through catalytic hydrogenation or by using iron filings and acid, yielding the target amine in good yield. The nitro precursor itself comes from the selective methylation of nitrobenzene or through controlled nitration of xylene derivatives. Scale-up brings its own headaches—exothermicity, byproduct management, and gas handling all demand robust equipment and vigilance. From personal encounters with scale-up mishaps, I know minor lapses in temperature or pressure control can turn valuable material into a cleanup nightmare.
Chemical Reactions & Modifications
On the laboratory bench, chemists play with 2,6-Dimethylaniline by exploiting that reactive amino group, protected by two methyl groups tucked on each side. Acylations, sulfonations, and couplings form the backbone of dyes, pharmaceuticals, and more complex organics. The methyls slow down unwanted side reactions, steering selectivity in the right direction. One of the most fascinating aspects is how this molecule has become a proving ground for developing new catalysts and synthetic methodologies; subtle tweaks affect rates and selectivity so researchers learn a great deal by starting here. In my own trials with various aromatic substitutions, those two methyls reduce tar formation, which keeps columns cleaner and reactions easier to monitor.
Synonyms & Product Names
Across labs and catalogs, 2,6-Dimethylaniline answers to many names. Chemists often call it 2,6-xylidine, reflecting its xylene origin. It sometimes pops up as xylidene or 2,6-dimethylbenzenamine. These variants circle back to the same structure, which throws off students and even seasoned professionals swapping protocols across borders. I’ve spent more than one day tracking down synonyms just to make sure the right drum reached the right bay; mislabeling can cause dangerous mix-ups when dealing with similar-sounding anilines.
Safety & Operational Standards
Repeated handling of 2,6-Dimethylaniline teaches a kind of respect that only arises after years of working with potent organics. The substance acts as both a skin and respiratory irritant, and animal studies flag up more serious long-term effects. Strict workplace controls—ventilation, gloves, goggles, and rapid access to showers—don’t exist just for show. Emergency response training ensures that workers know how to neutralize spills and avoid accidental exposure. Most modern production follows protocols set by agencies with decades of oversight, not just for the sake of compliance but because accidents with aromatic amines leave lasting marks on both people and property.
Application Area
Users rarely buy 2,6-Dimethylaniline to use directly in a final product but as a springboard in producing something more valuable. Its biggest impact lands in dye and pigment manufacture; its distinctive reactivity pattern means designers can fine-tune hues and performance in synthetic fibers and specialty paints. In the pharmaceutical field, several local anesthetics draw on 2,6-xylidine as a starting block, with modifications leading to drugs like lidocaine. Crop protection researchers utilize it for active ingredient design, seeking improved selectivity and stability in agrochemicals. These applications come from decades of trial, error, and discovery, driven by the molecule’s unique fingerprint.
Research & Development
Academic and industrial R&D both turn often to 2,6-Dimethylaniline when searching for new coupling reactions or cleaner synthetic routes. Researchers experiment with greener reduction methods to improve yields or lessen the environmental burden. More efficient catalysts, safer handling protocols, and analytical methods pick up where early work left off. Some teams use the compound’s structure to explore radical new methodologies in C-H activation or to develop better chromophores for molecular electronics. Across my own collaborations, the ease of modifying the amine group—without sacrificing selectivity—often meant faster screening of candidate reactions before investing in precious catalysts.
Toxicity Research
No discussion about 2,6-Dimethylaniline sidesteps the steady drumbeat of toxicity data gathered over the years. Animal testing points to concerns with methemoglobinemia, a condition where blood loses its ability to carry oxygen efficiently. Occupational data, especially from textile and dye plants during the mid-20th century, highlights long-term risks—including cancer correlations with heavy, chronic exposure. As regulation tightened, industry invested in air monitoring, closed-loop production, and biological monitoring for workers. Robust animal studies, sometimes controversial, still drive the push for alternatives; safer derivatives and better process controls now form core parts of most R&D pipelines. Anyone planning to scale up its use faces mandatory hazard training and disposal protocols shaped by these decades of learning.
Future Prospects
Looking to the future, chemistry moves fast but echoes of the past remain. Synthetic chemists search for less hazardous alternatives with similar utility, but 2,6-Dimethylaniline sticks around thanks to its reliability. Trends in green chemistry put pressure on manufacturers to reduce toxic byproducts in production, sometimes by re-engineering the whole supply chain. New technologies—continuous flow reactors, digitized process control, remote sensing for leaks—promise not just efficiency but better worker safety. On the research front, the push continues for new amines with similar or improved profiles but reduced environmental impact. Despite these changes, the lessons learned from this compound set a template for safer, smarter engagement with all family members in the aromatic amine world.
What are the main uses of 2,6-Dimethylaniline?
2,6-Dimethylaniline: Not Just Another Chemical Name
People often glance at long chemical names and feel their eyes glaze over. 2,6-Dimethylaniline usually gets ignored unless you’ve spent time in a lab or worked in industries that deal with dyes, pharmaceuticals, or rubber. But this molecule packs a punch in the real world, quietly touching everything from books to tires.
Bringing Color to Life: The Dye Industry
Plenty of vibrant textiles and colored print inks trace back to compounds like 2,6-Dimethylaniline. Manufacturers count on it to help create azo dyes and pigments. These aren’t the crayons you find in an office supply store—these dyes show up in denim, uniforms, billboards, and packaging that refuses to fade away in sunlight. As demand for reliable, lasting color keeps climbing in the textile world, 2,6-Dimethylaniline remains a constant workhorse behind the scenes.
Pharmaceuticals and Agrochemicals: Building Blocks for Bigger Things
This compound steps up as an intermediate in the pharmaceutical field. Factories rally to produce molecules like mefenamic acid (a widely used pain reliever and anti-inflammatory agent), and 2,6-Dimethylaniline starts the chain of reactions that brings these finished products to medicine cabinets. If you’ve ever relied on over-the-counter remedies for headaches or cramps, there’s a decent chance the process began with this chemical in an industrial reactor.
Farmers and crop scientists also recognize its usefulness. They lean on it to help synthesize pesticides and herbicides designed to protect yields and recover fields from stubborn weeds. Reliable crop protection means more food on tables, something we can’t afford to take for granted. Without these inputs, the chain from farm to plate would look much different.
Rubber and Plastics: Strength Where It Counts
Modern life wouldn’t run half as smoothly without tough tires and hoses. Manufacturers use 2,6-Dimethylaniline to create antioxidants—chemicals that help rubber withstand heat, pressure, and daily wear. Tires last longer, rubber seals stop leaking so quickly, and industrial belts run months without giving out. These small improvements stretch budgets and keep the world moving, literally and figuratively.
Risks and Responsibility: Safety and the Environment
No conversation about synthetic chemicals works if we skip their hazards. 2,6-Dimethylaniline, like many chemicals in its class, calls for care during handling and disposal. Toxicity to aquatic life and worker health risks have been documented. In my own lab experience, training drills on chemical safety and waste management never felt optional. Anyone using or producing this compound must keep strict protocols in place, from containment to proper personal protective gear.
Looking Ahead: Balancing Innovation With Safety
Sustainable alternatives attract serious interest. Green chemistry research now pushes for safer dye sources and replacement intermediates for pharmaceuticals and crop protection, seeking less risky routes while meeting production needs. Governments and independent auditors track and regulate emissions and usage, nudging industries toward more responsible practices. Continued transparency helps the public trust what comes into their homes and onto their plates.
In short, 2,6-Dimethylaniline plays a bigger part in daily life than most people realize. Whether it’s the colorfast hoodie you love or the painkillers in your desk drawer, the story of this molecule runs much deeper than a tricky name suggests.
Is 2,6-Dimethylaniline hazardous or toxic?
What Is 2,6-Dimethylaniline?
2,6-Dimethylaniline, also called 2,6-xylidine, shows up in various manufacturing steps for dyes, pharmaceuticals, and certain herbicides. With its chemical structure based on aniline, it's part of a larger family known for both utility and risk. My own experience with handling industrial chemicals taught me early that many names don’t signal obvious danger, and that goes for this substance too.
Risks Associated with Exposure
Contact can happen through skin, inhalation, or accidental ingestion. The science behind these risks comes from animal studies and case reports tracking workers dealing with long-term or repeated exposure. Immediate concerns focus on skin and eye irritation. Breathing dust or vapor sends the chemical quickly into the bloodstream, and some people notice headaches, dizziness, or nausea during high exposure events. Chronic exposure creates bigger questions.
One worry among toxicologists centers on methemoglobinemia, a blood disorder that interferes with oxygen’s path around the body. 2,6-Dimethylaniline converts part of the hemoglobin to methemoglobin, which can leave workers feeling blue in the face, short of breath, or worse. This is more than just statistics to me—I remember an incident years ago when a chemical operator didn’t realize her gloves had small tears, only to develop a cluster of these symptoms after regular use. It didn’t take long to piece together the connection to her work station.
Cancer and Long-term Effects
The main cloud hanging over 2,6-dimethylaniline is its cancer risk. Some forms of aniline have already received classification as potential carcinogens. 2,6-dimethylaniline lands in a category where animal experiments show clear evidence of tumors after repeated, high exposure. There’s no plain answer for human cancer risk, but the weight of evidence keeps nudging health agencies to stay cautious.
Beyond cancer, long-term low-level exposure sometimes results in symptoms like weakness, confusion, or coordination trouble. The challenge is that effects can roll in slowly, building up over months or even years before anyone thinks to connect these signs to workplace chemicals. It highlights gaps in monitoring and worker education. Data reported by the U.S. National Toxicology Program and European agencies supports these concerns, which makes me appreciate how regulations have shaped safer workplaces over time.
Minimizing the Hazard
Poor ventilation and limited personal protective equipment always raise the risk. I’ve seen labs run better by simply investing in local exhaust hoods, regular glove checks, and giving everyone a real walkthrough on the Safe Data Sheets. Even outside of big industry, anyone using products made from related chemicals should check for proper labelling and storage instructions. Simple actions—like double-bagging chemical waste and keeping splash goggles at hand—make all the difference. Many of the safety basics cost less than one emergency room visit.
What Is Being Done?
Regulatory agencies across North America and Europe have set clear guidelines for workplace exposure. Limits exist for airborne concentrations, and manufacturers must include clear warnings. Inspectors pay attention, and worker health surveys back up regulatory action. Companies that ignore these rules face heavy fines—and, more importantly, real harm to their teams. Society’s approach puts responsibility in the hands of everyone on a facility’s floor, not just managers or supervisors.
As long as chemicals like 2,6-dimethylaniline stay in use for research or production, dialogue between workers, management, and regulators remains essential. No process replaces vigilance and respect for science-backed safety. For people who spend their days around chemical risks, taking health and safety training seriously isn’t just a check-box on a form. It can be the difference between business as usual and a health scare that changes everything.
How should 2,6-Dimethylaniline be stored and handled safely?
What You Need to Know
2,6-Dimethylaniline shows up often in labs and industrial spaces as a building block for dyes and chemicals. This substance packs a punch: it’s toxic, flammable, and brings some real health hazards to people who don’t know its risks. I remember the feeling of opening a fresh drum in a poorly ventilated warehouse. That sharp, irritating odor told me all I needed to know—skip the shortcuts, carelessness doesn’t end well.
Storage: Respect the Substance
Putting this chemical somewhere that’s dry, cool, and dark does more than check off a rule. It keeps unwanted reactions out of the picture. Humidity or sunlight can mess with the stability of the compound. Direct sunlight speeds up decomposition and nobody wants vapors where folks work or relax.
Sturdy, tightly sealed glass or stainless-steel containers deliver peace of mind. Skip plastics that get soft with solvents. Keep this stuff away from acids, oxidizers, and strong bases. Once I saw an unlabeled jug of this amine stacked next to bleach—bad news waiting to happen.
Labeling doesn’t just help pass inspections. Clear words save lives in emergencies. A bold label shouts out the danger, and no one has to guess what’s inside. Dependable locks on the storage room put up one more barrier to random access. Fewer hands, fewer accidents.
Handling: Everyday Habits Save Skin and Lungs
Face masks and gloves aren’t overkill here. Letting 2,6-Dimethylaniline touch your skin quickly leads to irritation, sometimes burns, and headaches come fast from fumes. Nitrile gloves don’t break down. Goggles catch splashes, and long sleeves keep drips far from skin. One coworker brushed an unprotected wrist and wound up in the nurse’s station. Simple gear would have stopped the whole mess.
Working in a good fume hood is a real game-changer. Even small spills can fill the air with nasty vapors, especially with poor airflow. Everyone in the lab or workshop should know how to turn on exhaust fans. Trust your nose—if something smells strong, step back and check airflow.
Spill kits simplified my cleanup routine. Absorbent pads designed for solvents outwork paper towels. Neutralizing powders make sure nothing stays active on the floor long enough to hurt somebody. Having the emergency eye-wash station within arm’s reach is no accident: it’s required, and you’ll be grateful for it if anything splashes.
Getting the People Piece Right
Training isn’t just a box to check. I remember my first real chemical safety briefing. Stories stuck with me more than slides. Real incidents drive home what’s on the line. Team members need hands-on practice with all the gear and repeat drills for spills, leaks, or accidental contact. Clear step-by-step instructions, posted right next to where this compound gets used, save precious seconds when stress is high. Calling out hazards at every meeting or shift gets routine, but it beats the alternative.
No chemical, no matter its value to research or manufacturing, is worth risking a life. Respect for 2,6-Dimethylaniline—backed by know-how, good gear, and common sense—keeps everyone safe and the workplace running smoothly.
What is the chemical formula and structure of 2,6-Dimethylaniline?
The Basic Structure of 2,6-Dimethylaniline
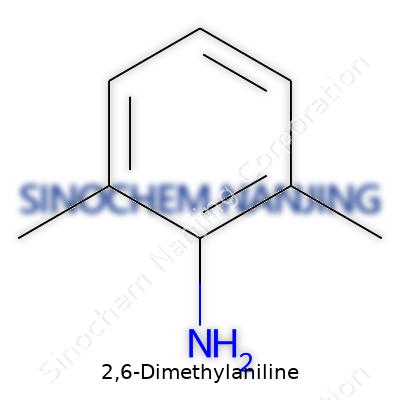
2,6-Dimethylaniline is an organic molecule with a clear-cut identity in the chemical world. The formula stands as C8H11N. You find a benzene ring carrying two methyl groups and an amine group. Picture the benzene ring as the hub; the methyl groups attach to the second and sixth positions around the ring, with an amine group at the first position. The IUPAC name rings true to its structure: 2,6-dimethylaniline. On paper, its skeletal structure draws a hexagonal ring, two –CH3 groups flanking the NH2 end of the ring, creating a specific pattern that sets this compound apart from others in the aniline family.
Why Structure Matters in Everyday Life
Thinking back to my first time working with aromatic amines in the lab, I remember how molecular arrangement guided everything—from how the compound smells to how it reacts. Structural elements shape the chemical’s reactivity, hazards, and uses. In the case of 2,6-dimethylaniline, those methyl groups don’t just hang around as spectators. They slightly hinder the activity of the amine group by their presence, introducing a kind of bulk that makes certain reactions trickier or, in some cases, more selective. That means chemists often pick 2,6-dimethylaniline for specialized synthesis.
Workplaces that manufacture dyes or certain drugs keep it handy. The methyl groups direct how the chemical interacts with other reactants. In one project, the ortho-substitution (attachment at the second and sixth ring positions) prevented unwanted reactions that happened with plain aniline. Aromaticity alone doesn’t explain these patterns; it’s the sum of the structure, electronic effects, and a little hands-on experience to see how small changes shape big outcomes.
Potential Health and Environmental Impact
Not every compound with a familiar ring is safe. This molecule, like other anilines, brings up tough questions about toxicity and handling. Studies from the National Institute for Occupational Safety and Health mention risks of methemoglobinemia and potential carcinogenicity with anilines and their methylated cousins. The methyl groups may make it less or more toxic, depending on metabolic pathways in the body.
Anyone handling this compound works under strict safety rules. I’ve seen research labs and factories use fume hoods, gloves, and disposal guidelines that treat all anilines with respect. The hazards serve as reminders: just because something looks like a close cousin of a common chemical, substitution changes can have far-reaching effects. Regulatory bodies like OSHA and the EPA monitor its use closely. Limits in the workplace and in waste ensure that problems don’t reach the broader environment. Personal vigilance and informed procedures keep everyone safer.
Why the Details Matter Today
A clear picture of 2,6-dimethylaniline—right down to the molecular level—matters because tiny changes affect everything from industrial output to human health. Chemists and industry workers who respect the specifics have more control over processes and outcomes. My time in the lab drove home the value of understanding not just what a molecule is, but how its structure guides its entire life cycle, from synthesis to disposal. Quality science relies on this attention to detail—building safer workplaces, reducing waste, and pushing forward industries that rely on a deeper molecular understanding.
Are there any special disposal requirements for 2,6-Dimethylaniline?
Understanding the Real Hazards
2,6-Dimethylaniline, or just xylidine to some, doesn’t come up in everyday conversation. Yet, in certain labs and industries, it’s common enough that the question of safe disposal shouldn’t be an afterthought. People tend to underestimate its risk. This is a clear example of a chemical with both acute and chronic hazards — inhalation, skin absorption, and yes, a good deal of environmental persistence. The average person outside of a chem lab probably won’t meet it, but for lab technicians and factory workers, it’s a different story.
Why the Danger Feels Personal
A couple years back, a friend of mine tried to clear out some old bottles from a high school storage room. She almost tossed a bottle of xylidine into the regular trash, not realizing that this single act could have turned into a local news story if there had been a spill or worse. Luckily, she caught the warning on the label. This incident drove home for me that some chemicals show their risk at a moment’s inattention.
What We Know from Official Sources
Xylidine comes with the “toxic” and “harmful to aquatic life” labels for a reason. The CDC and others point out it’s toxic if inhaled or swallowed, and it’s notorious for its effects on water and soil. It drifts around in the air, clings to your gloves, and can sneak through a cut in your hand. Ingestion or exposure over time increases risks for problems with blood, kidney, and liver. It’s not a substance anyone can just pour down the drain or send to the landfill without a second thought.
What Makes Proper Disposal So Critical
Lab waste streams rarely get much attention—until something goes wrong. Pouring this chemical or any similar amine down the drain doesn’t make it disappear; it just shifts the problem downstream. Waterworks and municipal filters are not meant to trap these compounds.
Incineration is the only practice that removes the molecule from the environment and neutralizes its toxic effects, which is why government recommendations always come back to this method. In my corner of the world, the local hazardous waste handler collects these types of chemicals in tightly sealed, clearly labeled containers. Their workers never shortcut the paperwork. That tells me the risk is real and that disposal is more about long-term responsibility than checking a box.
Supporting Solutions for People and the Planet
Proper disposal begins with good labeling and storage, and should extend to regular staff training—not just for chemists, but for anyone who handles chemicals. Institutions and businesses could benefit by moving to digital tracking for chemical inventory, which helps avoid that “forgotten bottle in the storeroom” scenario.
Community hazardous waste collection still matters. Most households never see 2,6-Dimethylaniline, but in university towns and industrial zones, everyone benefits if local authorities run frequent and well-publicized takeback events.
Some researchers are also exploring safer chemical alternatives and more environmentally-friendly synthesis techniques in dye and pharmaceutical production. But as long as xylidine is around, strict protocols for hazardous waste must stay enforced.
A Last Word From Experience
From my years working around chemicals, I’ve learned the best policy: treat every unknown as hazardous until proven otherwise. If you ever have a bottle with 2,6-Dimethylaniline, don’t improvise. Call the experts, track the paperwork, and use licensed hazardous waste contractors. The neighborhood, and especially future generations, will thank you.
| Names | |
| Preferred IUPAC name | 2,6-Dimethylaniline |
| Other names |
2,6-Xylidine 2,6-Dimethylbenzenamine 2,6-Dimethylphenylamine |
| Pronunciation | /tuː sɪks daɪˈmɛθɪl əˈnɪliːn/ |
| Identifiers | |
| CAS Number | 87-62-7 |
| Beilstein Reference | 1209241 |
| ChEBI | CHEBI:28698 |
| ChEMBL | CHEMBL13844 |
| ChemSpider | 14325 |
| DrugBank | DB03607 |
| ECHA InfoCard | 100.057.828 |
| EC Number | 205-518-8 |
| Gmelin Reference | 80817 |
| KEGG | C01576 |
| MeSH | D015928 |
| PubChem CID | 7277 |
| RTECS number | SL1575000 |
| UNII | 5U74V0S62S |
| UN number | UN2431 |
| Properties | |
| Chemical formula | C8H11N |
| Molar mass | 121.18 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | aromatic |
| Density | 0.915 g/mL |
| Solubility in water | insoluble |
| log P | 1.90 |
| Vapor pressure | 0.16 mmHg (25 °C) |
| Acidity (pKa) | pKa = 4.34 |
| Basicity (pKb) | 10.67 |
| Magnetic susceptibility (χ) | -8.71 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.553 |
| Viscosity | 2.14 mPa·s (25 °C) |
| Dipole moment | 1.37 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 169.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 83.5 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3787.6 kJ/mol |
| Pharmacology | |
| ATC code | D14AA04 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes skin irritation. Causes serious eye irritation. Suspected of causing cancer. Toxic to aquatic life with long lasting effects. |
| GHS labelling | GHS02, GHS07, GHS08 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H301 + H311 + H331: Toxic if swallowed, in contact with skin or if inhaled. H317: May cause an allergic skin reaction. H373: May cause damage to organs through prolonged or repeated exposure. H400: Very toxic to aquatic life. |
| Precautionary statements | P261, P264, P271, P272, P280, P302+P352, P308+P313, P312, P321, P362+P364, P405, P501 |
| NFPA 704 (fire diamond) | 2,3,2 |
| Flash point | 138°C |
| Autoignition temperature | 540°C |
| Explosive limits | Explosive limits: 1.3–7% |
| Lethal dose or concentration | LD50 oral rat 2040 mg/kg |
| LD50 (median dose) | Rat oral LD50: 1160 mg/kg |
| NIOSH | KN0175000 |
| PEL (Permissible) | PEL: 2 mg/m³ |
| REL (Recommended) | 25 mg/m³ |
| IDLH (Immediate danger) | IDLH: 50 ppm |
| Related compounds | |
| Related compounds |
Aniline 2,6-Dimethylnitrobenzene 2,6-Xylidine 2,6-Dimethylaniline hydrochloride 2,6-Dichloroaniline |

