Inside the World of 2,6-Dimethoxybenzoyl Chloride: Reflections on a Modern Chemical Staple
Historical Development
2,6-Dimethoxybenzoyl chloride didn’t spring up out of nowhere. Its origins track back to the broader exploration of substituted benzoyl compounds during the major push in organic synthesis of the mid-20th century. Researchers at the time set out to build more versatile intermediates for pharmaceuticals and dyes, branching off the well-established routes developed for benzoyl chloride, a cornerstone oxidizing and acylating agent. Over the decades, its 2,6-dimethoxy variant started picking up steam, especially in labs where subtle tweaks in aromatic structure translated to big changes in downstream product behavior. This wasn’t just academic curiosity—the compound quickly gained a home in the toolkits of those working to push molecular boundaries, from new drug candidates to improved polymers.
Product Overview
2,6-Dimethoxybenzoyl chloride grabs attention not for being flashy, but for how quietly reliable it proves in the lab. It’s often one of those reagents that chemists keep within arm’s reach, mostly because it brings methoxy substitution to an acyl chloride backbone with little fuss. The introduction of methoxy groups to the aromatic ring shifts its reactivity, often taming the wild reactivity of the chloro group just enough, which can be crucial during multi-step projects where every side reaction means more purification and wasted time. For me, nothing beats knowing that something as simple as structural modification can clear headaches from process scale-up, especially in pharmaceutical work.
Physical & Chemical Properties
Nobody forgets their first encounter with its sharp smell or its tendency to hydrolyze aggressively if you’re careless about moisture. It appears as a colorless or slightly yellow liquid, which starts to make sense once you check the melting and boiling data: it’s still liquid at room temperature, with a boiling point that can climb above 300 °C. That means it sticks around through quite a range of conditions, but water gets it riled up, producing HCl gas and reverting to 2,6-dimethoxybenzoic acid in a hurry. Its reactivity owes a lot to the electron-donating methoxy groups at the ortho positions, which tweak electron density in ways that seasoned organic chemists have learned to exploit.
Technical Specifications & Labeling
Purity makes or breaks a synthesis, and this compound plays no different. You typically see technical or analytical grades rated by GC for organic content, with water and related acids called out to the low single digits in percent. Labeling must be crystal clear here, not just to appease regulators but to keep those handling it safe—“corrosive,” “irritant,” and “reacts violently with water” are more than just warnings; they’re reminders of the stories behind every accidental splash or fume exposure. Labs that work with it daily build routines around dry glassware, tight seals, and access to proper hoods.
Preparation Method
The standard playbook starts with 2,6-dimethoxybenzoic acid—often made by methylating gallic acid or similar precursors—followed by reacting with thionyl chloride or phosphorus pentachloride. I’ve found no trick that smooths this out better than patient addition and watching temperature closely, as over-excess can mean lingering impurities. On a good day, a slow addition with constant stirring in a dry flask, followed by distillation, pops out a relatively clean product ready to take on acylations or custom synthesis jobs. Every practitioner seems to develop a small ritual to ward off hydrolysis, whether it’s purging glassware with argon or simply refusing to rush.
Chemical Reactions & Modifications
You can see the signature of this compound everywhere from peptide chemistry to agricultural science. Its acyl chloride moiety reacts briskly with amines and alcohols, often yielding amides and esters that take on unique properties because of the methoxy groups. My group once leaned on its ability to acylate sensitive substrates that might fall apart in harsher conditions. Beyond classic substitutions, clever folks have branched into introducing this moiety on larger scaffolds, shifting both solubility and activity, layouts that can help with everything from fine-tuning pharmaceutical candidates to generating intermediates for complex natural product synthesis.
Synonyms & Product Names
Depending who you ask, someone might call it 2,6-dimethoxybenzenecarbonyl chloride, o,o-dimethoxybenzoyl chloride, or a shorthand like DBC. This whirlwind of names can trip up even veteran chemists switching suppliers or parsing literature. Standardizing names across research communications matters more than most realize—one poorly labeled vial can derail weeks of planning. These semantic tangles aren’t just nitpicking; clear nomenclature guides safe handling, ordering accuracy, and reproducibility, the basics that science rests upon.
Safety & Operational Standards
Corrosive doesn’t quite cover the story with 2,6-dimethoxybenzoyl chloride. A moment’s slip and you end up with skin burns or lab air thick with irritant fumes. Dry conditions and closed systems aren’t optional. I’ve seen protocols go from smooth to chaos with a single crack in a glove or a missed tightening of a bottle cap. Regular refresher training helps keep these realities at the front of everyone’s mind. Labs build in engineering controls—ventilation, splash shields, and protocols for spill containment—because relying on luck is a poor safety net. Good habits save skin and lungs, and I’ve seen careers upended by ignoring this truth for even a single task.
Application Areas
This compound lives many lives across research and industry. Its acylating abilities make it useful when building up custom pharmaceuticals, linking together molecular fragments that go on to fight disease or act as diagnostic probes. I’ve seen it pop up during scale-up projects where a more reactive acyl chloride would gouge yields or create uncontrolled byproducts. Its structure delivers enough activation without being too hot to handle, a balance more valuable than flashy metrics. Besides the obvious drug applications, it finds work in advancing new dyes, photosensitive materials, and even flavors or fragrances when chemists lean into its subtle aromatic tweaks.
Research & Development
Academic circles often look for new ways to use or tweak its reactivity, exploring derivatizations that change how target molecules interact with their environment or biological systems. Solid-phase synthesis strategies, site-selective acylations, and late-stage modifications all gain from this compound’s temperament. I’ve watched teams shave weeks off their timelines simply by swapping in this reagent for older, less predictable alternatives. Across the world, compounds derived from 2,6-dimethoxybenzoyl chloride keep finding their way into patents and publications, feeding a pace of innovation that rewards creative new linkages and post-functionalizations.
Toxicity Research
Everyone learns quickly that chemical power often comes with a cost—here, that means corrosive and irritant properties that can escalate into chronic health problems without strict safety protocols. Eye and respiratory exposures lead to lasting damage in severe cases. Some toxicity reports suggest organ-specific risks, but most real-world dangers come down to burns from splashes or inhaled fumes from open bottles. This is not a situation for shortcuts or casual attitudes: I’ve seen research paused, and even careers derailed, by underestimating the risks. Standard industrial hygiene, reliable PPE, and up-to-date training keep incidents rare, but complacency never does anyone favors.
Future Prospects
Looking forward, 2,6-dimethoxybenzoyl chloride seems set to remain an unsung hero for complex synthesis. The continued demand for tailored pharmaceuticals, specialty polymers, and fine chemicals grants it steady relevance. As green chemistry pressures grow, the research community has started to hunt for cleaner production routes and less hazardous byproducts—areas where process optimization could tighten its environmental footprint. By integrating this compound more tightly into flow chemistry or solid-supported platforms, groups might lower risk and waste, all while exploring new functional territory. Real change won’t arrive overnight, but innovation has always come down to clever people reworking old standbys for a rapidly changing world.
What is the chemical formula and molecular weight of 2,6-Dimethoxybenzoyl Chloride?
Understanding the Basics
The world of chemicals can sometimes feel overwhelming, with countless names and even more formulas. Take 2,6-Dimethoxybenzoyl chloride, for example. At first glance, its name alone can put off anyone not already knee-deep in organic chemistry. Yet, a simple breakdown helps: its formula is C9H9ClO3. Each cluster of letters and numbers in a formula hints at the building blocks that make the compound tick.
Crunching the Numbers: Molecular Weight
It's easy to forget how much a single molecule weighs, but molecular weight matters in everything from lab work to industrial synthesis. For 2,6-Dimethoxybenzoyl chloride, the answer stands at about 200.62 grams per mole. That number doesn’t just pop out of thin air—it's the sum of all atomic weights in its structure. Knowing this value lets chemists measure out reactants precisely, especially when tiny miscalculations can send a synthesis sideways.
Diving Deeper: Why Structure and Weight Matter
There’s more to 2,6-Dimethoxybenzoyl chloride than numbers on a page. The two methoxy groups at the 2 and 6 spots on the benzene ring shift its reactivity. The presence of a chloride group tacked onto the carbonyl carbon tells chemists this compound can jumpstart acylation reactions—a backbone step for building larger, more complex molecules. Anybody working in the pharma or dye field knows that missing even a single methoxy group or swapping in the wrong halide can throw off a reaction, waste materials, or worse, risk safety.
Safety and Handling: More Than Just Gloves
Handling benzoyl chlorides can’t be done with carelessness. The chloride group can easily react with water, even just the moisture in air, to churn out hydrochloric acid fumes. Anyone who’s ever worked a bench in a chemistry lab remembers the sting from an unexpected whiff of HCl. Good labs always run acyl chloride reactions under a fume hood with gloves and goggles as basic gear. Water and benzoyl chloride belong as far apart as possible. This isn’t just a quiet rule in textbooks—hospital visits from chemical burns stay fresh in a chemist’s memory for years.
Applications and Real-World Impact
On paper, 2,6-Dimethoxybenzoyl chloride looks simple. Bottles on reagent shelves often sit beside older labels and fading hazard stickers, but their reach goes further. These chemicals form the backbone for producing drugs, agricultural chemicals, and specialty materials. Many key pharmaceutical building blocks start with simple-looking benzoyl chlorides, transformed step by step into medicines relied on by millions around the world. Sometimes the difference between a lifesaving drug and a dud boils down to careful tweaks in the starting materials.
Rethinking Chemical Management
No one claims handling reactive compounds is stress-free. Still, minimizing risks starts with clear labeling, sturdy ventilation, and a culture that values safety every bit as much as speed or cost. Regular drills, labeling audits, and up-to-date Material Safety Data Sheets keep everyone grounded. Waste gets tracked and neutralized on the spot, not shoved to the end of the day. Teaching new lab members the personal impact of every gram handled shifts lab culture away from “get it done” to “get it done safely.”
Supporting Smarter Chemistry
Each chemical on the bench tells a story. For 2,6-Dimethoxybenzoyl chloride, knowing the C9H9ClO3 formula and the precise molecular weight lets researchers plan better, waste less, and build smarter. The details matter—every formula and weight, every risk label read and followed, adds up to a smarter, safer lab and, by extension, stronger research results that ripple out to real-world solutions.
What are the common applications or uses of 2,6-Dimethoxybenzoyl Chloride?
Chemistry That Directly Touches Our Lives
2,6-Dimethoxybenzoyl chloride might seem like a tongue-twister, but it has carved out an important niche in several fields. This compound pops up most often in laboratories dedicated to organic chemistry, where researchers look for creative approaches to build new molecules. People sometimes overlook the real-world impact that these specialty chemicals can have, even though they rarely see the spotlight outside scientific circles. My years in the lab have taught me that success often depends on finding just the right building block, and 2,6-Dimethoxybenzoyl chloride shows its worth in these small but crucial details.
Medicinal Chemistry and Drug Discovery
Work on new pharmaceuticals often starts with painstaking trial and error. Medicinal chemists depend on starting materials that are both reliable and adaptable. This is where 2,6-Dimethoxybenzoyl chloride comes into play. Chemists value it for preparing compounds like benzamides and other related structures that lay the groundwork for new medicines. Studies document its role in the synthesis of complex drug candidates, giving researchers a way to modify molecules and tune their activity.
For example, teams looking to develop anti-inflammatory or antimicrobial agents turn to similar core structures. By adding or tweaking side chains using this reagent, they test different versions in the hope of finding molecules with better potency or fewer side effects. There’s nothing flashy about the process. It involves reliable reactions repeated day after day, driven by the hope that one tiny change will spark a discovery.
Building Blocks for Dyes and Pigments
Outside the world of biotechnology and medicine, 2,6-Dimethoxybenzoyl chloride finds work in dye and pigment manufacturing. These products end up in everything from synthetic fabrics to plastics and inks. During my time consulting for a small manufacturer, I saw firsthand how a well-chosen chemical like this one can alter the shades and colorfastness of final products. The methoxy groups on the benzoyl chloride backbone help shape the hue, brightness, and even the durability of certain synthetic dyes.
Researchers interested in creating new shades, or improving stability under sunlight and washing, use this compound to adjust formulas. The chemistry behind it isn’t always obvious to the folks wearing the dyed fabrics, but it makes a noticeable difference in product quality and appeal.
Innovation in Agrochemical Research
Farmers may never read about 2,6-Dimethoxybenzoyl chloride, but it finds a home in pesticide and herbicide discovery. Agrochemical work shares a lot with drug development, in that it involves screening many variants of a molecule. Adjusting molecular features even slightly can produce big changes in how a compound affects weeds or pests. This reagent steps into the process as a way to introduce protective groups or key fragments, letting researchers push closer to safer, more effective agriproducts.
All this work links back to increased crop yields and better food security. Improved formulations benefit farmers and, ultimately, anyone sitting down to a meal.
The Challenges and a Way Forward
With all these applications, there is always a side of caution. Specialty chemicals like 2,6-Dimethoxybenzoyl chloride often demand careful handling and responsible use. Proper storage, protective equipment, and strict protocols keep lab workers safe, but it’s just as important to push for safer alternatives when possible. Green chemistry, focused on less hazardous reagents and procedures, has begun to influence research decisions. I’ve seen teams weigh the environmental footprint against convenience, often favoring sustainability even if it means extra work up front.
Whether in drug design, new colors for our clothes, or safer crops, finding the right tools makes all the difference. Sometimes that tool happens to be a chemical like 2,6-Dimethoxybenzoyl chloride, quietly shaping the products and discoveries we rely on every day.
What are the storage and handling requirements for 2,6-Dimethoxybenzoyl Chloride?
Understanding the Chemical
2,6-Dimethoxybenzoyl chloride isn’t something you’d find sitting next to flour or sugar in a pantry. This chemical, sharper than household cleaners and far more temperamental, puts safety skills to the test in any lab. From my own years working around such compounds, I know keeping things straightforward is key: don’t take shortcuts, and respect the threats these materials can pose.
Temperature Control and Air Exposure
This benzoyl chloride demands cool, dry surroundings. Warmth encourages it to give off corrosive fumes. I’ve seen labs store such chemicals at 2–8°C, away from sunlight and not with standard solvents. Humidity messes with it quickly, causing hydrochloric acid to form. Even a brief moment open to air can send sharp vapors into the room—never pleasant, often dangerous for skin and lungs. Sealing it tight with specialized caps, and double-bagged if possible, isn’t overkill; it’s common sense.
Material Compatibility Matters
Certain plastics buckle, discolor, or leak if exposed. I’ve watched bottles degrade when folks cut corners and used anything but glass or fluoropolymer (like Teflon). Glass resists while keeping contents visible. Corrosion-resistant containers—clean and dry before use—keep accidents at bay.
Separation From Incompatibles
A good rule from the labs I trust: don’t store strong organics and acids together. I once had a “near miss” because oily rags caught a whiff of spilled acid nearby. Mixing up shelves spells trouble when these fumes creep into oxidizers, strong bases, or water. If one isn’t cautious, these small errors add up—fumes drift, reactions kick off, alarms blare, health takes a hit.
Ventilation and Personal Protection
Breathing easy is impossible around benzoyl chlorides without serious airflow. I’ve always insisted on handling these chemicals inside fume hoods. One whiff is enough to make the mistake clear. Sturdy gloves—nitrile or neoprene—keep hands safe, and goggles are more than a guideline. Long sleeves, lab coats, and, sometimes, full mask protection all come into play. Even brief contact leads to painful burns and bad days.
Label, Track, and Respond
Mislabeled bottles cause confusion at best, hospital visits at worst. Every container I’ve handled gets a bold label with the full chemical name, hazards, and date opened. Spill kits with neutralizers and absorbent pads have bailed me out before; quick action stops disaster from growing. Emergencies always run smoother with safety data sheets on hand and staff drilled on steps to take if accidents break routine.
Practical Lessons and Solutions
Safe handling comes down to discipline and respect for the risks. I’ve made it a habit never to open these bottles alone. Double-checking seals, making sure safety showers and eyewash stations work, and keeping a log of use let teams catch mistakes before they turn serious. Secure storage, reliable ventilation, and vigilant personal protection draw a line between safe labs and dangerous ones. Simple routines, when done consistently, keep everyone out of harm’s way.
References and Best Practices
Solid advice comes from the Safety Data Sheet, published research, and well-run chemical supply firms. Following national and local safety regulations stays non-negotiable. These routines have kept me—and many colleagues—safe year after year while handling sharp, unforgiving reagents like 2,6-Dimethoxybenzoyl chloride.
What are the safety hazards and precautions when working with 2,6-Dimethoxybenzoyl Chloride?
The Risks on the Bench
Most chemists working in organic synthesis spot the sharp scent of acid chloride before they see it. 2,6-Dimethoxybenzoyl chloride, with its reactive structure and acyl chloride group, gives off hydrogen chloride fumes. Each time that bottle opens, the risk doesn’t stay only with the direct handler; irritant vapors slip into shared lab air. Eyes start to water, throats itch, and a little spatter on skin may sting right away or grow into a rash hours later.
A bottle of this stuff demands respect. Liquid contact burns. Inhaled vapors reach deep into the respiratory system, not just through the nose, making those with asthma reach for their inhalers. I once saw a postdoc get caught by a single drop while transferring under the hood, and the redness on his wrist lingered into the next week. In labs, small spills quickly turn from mild discomfort to full cleanups—especially since a few milliliters can release enough fume to disrupt a whole small workspace.
Best Practices Learned the Hard Way
Preparing to use 2,6-dimethoxybenzoyl chloride, I never trust just a pair of gloves. Proper nitrile gloves, splash-proof goggles, and a lab coat serve as basic armor. Benzoyl chlorides eat through the wrong glove material, so checking glove compatibility before slotting in my hands is non-negotiable. Contacts and acid chlorides never mix. I've seen chemical splashes underneath a shield while wearing contacts—a mix nobody wants to experience.
Good fume hoods don’t just protect from accidents—they protect long after a reaction has finished. At times, rushed chemists leave open bottles or stoppered flasks outside, and the bench starts smelling sharp and sour. Vapors drift, and anyone entering the room gets exposed. Keeping all transfers, slow or fast, inside the hood always makes the difference. I keep a box of sodium bicarbonate or another neutralizer close by, since spills on the bench need attention right away to avoid turning into dangerous situations.
Teamwork and Environmental Hazards
Working with acid chlorides isn’t a solo job. Nobody on a busy floor wants to find out they got exposed when others knew but didn’t speak up. Training junior researchers and reminding seasoned ones of first-aid routines for splashes and inhalation boosts safety. If you don’t know exactly where eyewash and shower stations are, you aren’t ready to open the bottle.
The waste generated carries its own concerns. Pouring leftover acid chloride into the wrong bottle leads to violent gas release and pressure build-up, risking glass breakage. I’ve seen vents blocked by careless mixing: bottles bulging, stoppers flying. Every ounce goes into dedicated halogenated solvent waste, properly labeled and vented, with regular pickups—otherwise, the risks keep adding up in storage rooms and waste closets.
Building Safer Labs
Many labs now replace old acid chloride routines with alternative acylation reagents when possible. It’s tempting to lean on what feels familiar, but better hazard control starts with this small step. Implementing routine checks on PPE, raising awareness of incompatible glove or bottle materials, and holding regular safety days reinforces habits more than any posted sign.
Nobody forgets their first bad encounter with an acyl chloride. Those lessons—burns, smells, and all—prove why mixing care, teamwork, and up-to-date protocols keeps everyone safer with 2,6-dimethoxybenzoyl chloride and chemicals like it.
Is 2,6-Dimethoxybenzoyl Chloride available in different purities or packaging sizes?
Practical Realities in Chemical Procurement
Chemicals like 2,6-Dimethoxybenzoyl Chloride usually don’t get much limelight outside the research community, but in labs and production facilities, every detail counts—purity, quantity, storage, and even lead time can make or break a project. Many professionals searching for this compound are either in pharmaceuticals, academic labs, or enterprises developing specialty chemicals. The level of purity and size of the package can end up being the deciding factor in moving ahead with purchase or having to look elsewhere.
Concerns About Purity
From years of experience working in a university lab, I’ve seen experiments derail just because a reagent didn’t meet the purity needed. Some projects require something high-end—over 98% purity—because trace impurities cause unwanted reactions, hurt yields, or just muddy up the data. Others chug along fine with a lower grade, maybe something considered “technical” or “lab” and not “analytical.” Sources like Sigma-Aldrich, TCI, or Alfa Aesar usually list multiple grades on their catalogs. You pay more for higher purity, true, but it’s often cheaper than repeating an entire experiment.
Choosing the Right Packaging Size
Most suppliers offer more than one size. Sometimes you get a 5-gram bottle suitable for a few reactions, sometimes a 25-gram jug, or even kilogram-scale for industrial use. There’s a cost calculation with every option: small bottles cost more per gram, but you’re not paying for extra you don’t need. Larger sizes suit ongoing production, but bring up those big questions—storage, stability, and handling. Small labs don’t want excess hazardous waste or expired chemicals gathering dust.
Safety, Shelf Life, and Storage
Chlorides like this tend to hydrolyze in moist air. If you only pop open a bottle now and then, moisture can seep in and degrade your product. Oversized packaging sometimes goes to waste just because the last bits go bad before anyone gets back to it. Short shelf lives aren’t just a problem for the budget; there’s also the issue of safe disposal and regulatory headaches. The Environmental Protection Agency (EPA) and OSHA both require proper handling of these kinds of chemicals, especially when they degrade or turn hazardous.
Supplier Variations and Quality Control
Global sourcing made life easier—and riskier. Not all suppliers hold their products to the same level of quality control. Inconsistent purity, mislabeling, or shipping delays can cause setbacks in both academic and industrial settings. Certificates of Analysis help, but they only tell you so much. Batch-to-batch variability is less common with the big names but can creep in when working with smaller vendors.
Practical Tips and Solutions
Comparing catalog options goes beyond a quick price check. Read through technical datasheets and test reports. Ask vendors for recent certificates and make sure they can guarantee shipment within a specific window. Work out what size and purity your application actually justifies, and don’t tie up budget buying bulk unless there’s real need. For those managing projects or labs, revisit inventory management processes—log received lots, test a portion before critical uses, and train staff on recognizing signs of degradation.
Final Thoughts
Paying attention to purity and packaging isn’t nitpicking—it saves money, time, and sometimes safety down the line. Having a chemical like 2,6-Dimethoxybenzoyl Chloride in the exact right form gets research done and keeps small mistakes from snowballing into expensive problems. Honest conversations with suppliers, diligent storage, and careful planning will always matter, especially with specialty chemicals that drive discovery.
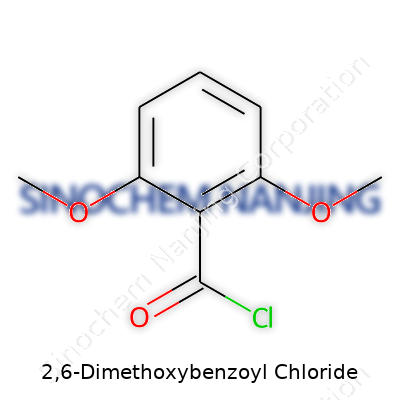
| Names | |
| Preferred IUPAC name | 2,6-dimethoxybenzoyl chloride |
| Other names |
2,6-Dimethoxybenzoyl chloride 2,6-Dimethoxybenzoic acid chloride Benzoyl chloride, 2,6-dimethoxy- NSC 147583 |
| Pronunciation | /ˈtuː,sɪks.daɪˌmɛθ.ɒk.siˈbɛn.zɔɪ ˈklɔːr.aɪd/ |
| Identifiers | |
| CAS Number | [23999-48-6] |
| 3D model (JSmol) | `3D structure; JSmol; string: COc1cccc(OC)c1C(=O)Cl` |
| Beilstein Reference | 607668 |
| ChEBI | CHEBI:91224 |
| ChEMBL | CHEMBL196622 |
| ChemSpider | 10469331 |
| DrugBank | DB04212 |
| ECHA InfoCard | ECHA InfoCard: 100.018.804 |
| EC Number | 204-049-1 |
| Gmelin Reference | 81859 |
| KEGG | C14365 |
| MeSH | D017714 |
| PubChem CID | 76072 |
| RTECS number | DO8900000 |
| UNII | HWF3P48A8W |
| UN number | UN3261 |
| CompTox Dashboard (EPA) | `DTXSID9020651` |
| Properties | |
| Chemical formula | C9H9ClO3 |
| Molar mass | 200.62 g/mol |
| Appearance | White to light yellow crystalline powder |
| Odor | pungent |
| Density | 1.227 g/mL at 25 °C |
| Solubility in water | Insoluble |
| log P | 1.9 |
| Vapor pressure | 0.06 hPa (25 °C) |
| Acidity (pKa) | 13.3 |
| Basicity (pKb) | -2.29 |
| Magnetic susceptibility (χ) | -52.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.5700 |
| Viscosity | 1.429 cP (25°C) |
| Dipole moment | 2.67 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 378.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -393.5 kJ/mol |
| Hazards | |
| Main hazards | Harmful if swallowed, causes severe skin burns and eye damage, causes serious eye irritation, harmful if inhaled. |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS05,GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H314 |
| Precautionary statements | Precautionary statements: P261, P264, P271, P280, P301+P312, P304+P340, P305+P351+P338, P308+P313, P332+P313, P337+P313, P362+P364, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 2,1,0 |
| Flash point | 100 °C |
| Lethal dose or concentration | LD₅₀ oral rat 3250 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 >2000 mg/kg |
| NIOSH | GV5950000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 1 mg/m³ |
| Related compounds | |
| Related compounds |
2,6-Dimethoxybenzoic acid 2,6-Dimethoxybenzaldehyde 2,6-Dimethoxybenzyl chloride 2,6-Dimethoxybenzamide 2,6-Dimethoxybenzonitrile |

