Exploring 2,6-Dichlorotoluene: More Than a Basic Chemical
Historical Development
Digging into the story of 2,6-dichlorotoluene takes me back to that broader sweep of 20th-century organic chemistry, when aromatic compounds kept moving further into the spotlight. Early on, chemists hunted for ways to tweak molecules from coal tar or petroleum fractions, hoping for something new—often by chlorinating toluene and separating the mixed isomers in inventive ways. The commercial momentum really picked up as folks realized how adaptable chlorotoluenes could be, acting as stepping stones in dye work, drugs, and agrochemicals. From the perspective of industrial chemistry, 2,6-dichlorotoluene rarely grabbed headlines itself, yet quietly supported big leaps in specialty chemicals as demand for tailored molecules expanded. There’s something humbling about how this once-unremarkable byproduct became a must-have ingredient for modern synthesis.
Product Overview and Physical-Chemical Properties
My own first run-in with 2,6-dichlorotoluene in the lab taught me to respect the chemicals that don’t shout for attention. This clear, almost unassuming liquid, immediately gives away its aromatic roots through a strong, distinctive odor that lingers long after you’ve cleaned up. The presence of those two chlorine atoms, snuggled onto the benzene ring, pushes the boiling point up and adds a tangible heft you notice when you pour it. It dissolves in organic solvents like a pro, resisting water with a stubbornness typical of chlorinated toluenes. You pick up on the increased density and the persistent volatility, facts every lab rat respects since even a small spill impacts air quality. Chromatographers and synthetic chemists often seek out this molecule precisely because of how these properties guide reactivity and product isolation—not flashy, but reliable tools in the toolbox.
Technical Specifications and Labeling Nuances
In practice, 2,6-dichlorotoluene usually gets shipped and stored according to purity grades, with careful labeling to nail down isomeric integrity. Mislabeling costs time, and purity matters because trace isomers can wreck later reactions aimed at precise pharmaceutical or agrochemical targets. Documentation reflects this sensitivity: regulatory codes focus on flammability, environmental fate, and chronic exposure risks. In my experience, proper labeling doesn’t just keep inspectors happy—it preserves research budgets and keeps cross-contamination problems out of already unpredictable projects. Quality assurance teams check spectral fingerprints and impurity profiles for good reason: nothing derails benchtop innovation like finding out your “pure” starting material was actually a soup of nearly-right compounds.
Preparation Method
The nitty-gritty of making 2,6-dichlorotoluene sits firmly in a chemist’s comfort zone. Direct chlorination of toluene, nudged along by UV light or ferric chloride, delivers a blend of isomers. Skilled distillation and fractionation draw off the 2,6 isomer, turning what starts as a statistical mixture into a targeted resource. You can see the industrial lesson here—scale brings both opportunity and waste—so much effort goes into cutting down on unwanted isomers and streamlining separation. Whenever new catalytic approaches arrive, they promise smaller environmental footprints and better yields, matching regulatory demands and budget realities. This sort of incremental improvement in isolation shows the sector’s attachment to detail and cost control, as stubborn as the molecule’s own resistance to water.
Chemical Reactions and Modifications
Chemists don’t just stop at making 2,6-dichlorotoluene. Over the years, experts have expanded its playbook, turning it into a base for acylation, nucleophilic substitution, and oxidation. Those two chlorines aren’t just ornamentation—they’re reactive hooks. Maybe you want to swap a chlorine for an amine or add some nitro functionality. Each tweak opens doors to a new fragment used in everything from pigment synthesis to preclinical pharmaceutical campaigns. On bad days, reactivity feels finicky; minor shifts in temperature or solvent can mean the difference between a planned route and a layer of goop in your reaction flask. These frustrations are worth it, though, because downstream products carry real economic and research clout in modern manufacturing.
Synonyms and Product Names
I’ve seen 2,6-dichlorotoluene listed under a jumble of names—none particularly catchy but all essential for research, trade, or regulatory paperwork. Chemical Abstracts Services (CAS) numbers cut through the confusion, but names like 1-methyl-2,6-dichlorobenzene, 2,6-dichloro-1-methylbenzene, or simple DCT pile up from different suppliers and catalogues. Spotting alternate identifiers can be a lifesaver when checking inventories, especially during regulatory audits or when running searches for safety literature. Skipping over these synonyms in a report leaves the door wide open for miscommunication, which always finds a way to complicate even the most careful project.
Safety and Operational Standards
No story about a halogenated aromatic is complete without a straight look at safety. Handling 2,6-dichlorotoluene underlines a wider lesson in lab work: trust safety data more than your nose or familiarity. Inhalation brings headaches and irritation, while long-term exposure calls up questions about chronic effects that modern regulations won’t let folks ignore. Ventilation, gloves, goggles—never negotiable. Disposal channels get extra scrutiny because halogenated waste brings environmental agencies to your doorstep. In my early days, seeing a seasoned colleague handle waste streams with almost ritual care left a mark. With REACH and other regulatory frameworks tightening, companies and universities can’t cut corners anymore. Safety culture grows from routine and experience, but clear rules and incident records have pushed the pace across many sectors.
Application Area
Most people outside chemical synthesis don’t hear about 2,6-dichlorotoluene. Industry insiders use it to launch series of synthetic moves—crafting herbicides, dyestuffs, and precursors that touch almost every part of daily life, even if invisibly. More than once, I’ve tracked project supply chains and watched how a product from the pharmaceutical bench owes much of its shape and function to a well-selected building block like this. In pigment chemistry, selective functionalization of dichlorotoluenes leads to richer colorants and more robust paints. Agrochemical sectors rely on it for efficient preparation of pesticide intermediates, which then drive larger productivity for growers. Sure, the compound doesn’t make headlines, but its prints linger on plenty of end-use products that shape health, color, and crop yield.
Research and Development in Context
Talk to researchers who spend their days inventing new drugs or crop protection tools, and you’ll hear how smaller intermediates make all the difference. The structure of 2,6-dichlorotoluene lets chemists try new coupling strategies, target harder molecules, or improve manufacturing efficiency. Take catalysis work, for example—every year pulls up new tweaks, cutting reaction times or slashing by-product volumes. Current R&D teams want greener, safer processes as industry eyes stricter global standards. The launch of alternative chlorination or selective separation technologies moves the needle both economically and environmentally, and the uptick in sustainable chemistry efforts shows that there’s no slackening in innovation, even for workhorse molecules like this.
Toxicity Research and Human Impact
The question of risk pushes every sector to reckon with what production really costs. Toxicity assessments count for more than just compliance—communities and workers want answers, not just technical assurances. Current animal and cell test results show specific acute and chronic toxicity profiles. Environmental fate studies probe soil, water, and air persistence, reflecting broader concerns about halogenated organics polluting water tables or accumulating in food chains. Over the decades, tighter workplace exposure limits and new analytic methods have brought more reliable answers, supporting better worker protection. The push to develop safer derivatives or alternative routes runs alongside this scrutiny, recognizing that every molecule’s journey includes a social footprint.
Future Prospects and the Road Ahead
As global industry pivots toward safer, smarter manufacturing, compounds like 2,6-dichlorotoluene still find space to influence progress. The push for sustainable and selective chemistry lifts the profile of molecules that offer sturdy building blocks for innovation in fine chemicals and pharmaceuticals. Digital chemistry, process modeling, and advanced automation aim to cut waste and streamline separations, reducing both cost and risk. Interest in precision agriculture and advanced materials means new derivatives and modifications will keep emerging, even as greener synthetic goals shift production models. Expect research around safer alternatives, lower-impact synthesis, and stricter traceability to keep reshaping how—and why—chemists reach for 2,6-dichlorotoluene on the supply shelf.
What is 2,6-Dichlorotoluene used for?
Everyday Chemistry in Action
People pass by chemical plants every day, not thinking twice about what happens inside. Yet, compounds like 2,6-Dichlorotoluene stay busy behind the scenes, shaping daily routines more than most realize. This colorless liquid, with a mild chemical scent, plays a central role across industries that never make headlines.
Building Block for Big Names
Much of 2,6-Dichlorotoluene’s value shows up in how it helps create other chemicals. Manufacturers lean on it to start the process for synthesizing dyes, pigments, and brightening agents woven into clothes, packaging, and even plastic utensils. The chemical gives colorfastness to synthetic fabrics and plastics, keeping colors from washing out or fading. That shines through every time you toss a t-shirt in the laundry or pull out your reusable water bottle.
Beyond color, it serves as a stepping stone for building herbicides and pesticides. In big agriculture, compound purity and reliability mean the difference between spoiled harvests and stable food supplies. Farmers counting on robust yields use weed and pest control products crafted with 2,6-Dichlorotoluene as a skeleton for complex molecules. In this way, crops grow taller and survive longer, easing food insecurity for entire communities.
Pharmaceutical Roots
Pharmaceutical labs also run through gallons of this chemical to create specialized compounds. Antibiotics, antihistamines, and other medicines rely on precise chemical blueprints, and 2,6-Dichlorotoluene lays the groundwork for molecules that help fight infections or control allergic reactions. Experience in pharmaceutical manufacturing has shown that quality control in chemical synthesis reduces the number of failed batches and wasted resources, making treatments more affordable for patients.
Challenges on Safety and the Environment
With industrial use comes responsibility. 2,6-Dichlorotoluene isn’t the kind of thing you want to spill down the drain or release into open air. Inhaling fumes can lead to health problems, and runoff can put stress on rivers and lakes. Long hours spent in industrial settings have brought those risks close to home. Proper training and safety gear mean a lot. Following good handling practices, switching out solvent tanks before leaks start, and scheduling regular inspections create a safer environment for workers and minimize environmental impact.
Some factories now recycle waste streams, pulling out valuable ingredients before disposal. Wastewater treatment systems continue to improve, breaking down dangerous chemicals instead of letting them spread unchecked. Governments keep tight restrictions on emissions and workplace exposure, and that vigilance deserves support, not just from regulators but from everyone involved in the supply chain. Simple steps—walking the floor, double-checking the seals on reactor vessels, calling in maintenance on a questionable pump—can prevent minor issues from turning into emergencies.
Finding Balance
No one expects people outside science and manufacturing to memorize chemical names. Understanding the hidden effort in compounds like 2,6-Dichlorotoluene gives a better sense of what keeps modern life running. Each layer of use, regulation, and safety draws from real-world experience, shaping industries and affecting lives even without ever making the front page.
What is the molecular formula of 2,6-Dichlorotoluene?
Getting Familiar With 2,6-Dichlorotoluene
Chemistry sometimes feels locked away behind complicated names and formulas, though in reality, every compound tells a story worth understanding. 2,6-Dichlorotoluene is one of those names that turns up quietly in lab discussions and technical specifications—something seemingly obscure unless you have needed it in research or industrial work. The molecular formula for 2,6-Dichlorotoluene is C7H6Cl2. This formula means the molecule includes seven carbon atoms, six hydrogen atoms, and two chlorine atoms. Each of those atoms makes a difference not only in structure but also in the way the compound behaves and how people use it around the globe.
Why Molecular Structure Matters
The structure says a lot about how molecules function. In organic chemistry, small changes lead to big differences. Toluene, a simple base molecule, loses its simplicity the moment two chlorine atoms attach at the 2 and 6 positions on the benzene ring. Now, the molecule holds new chemical and physical properties. The two chlorines at these positions don’t just give it a new formula—they make new reactions possible and restrict others. This means manufacturers pick 2,6-Dichlorotoluene for making dyes, herbicides, or certain advanced polymers where the placement of atoms changes the outcome. One small variation can make a solvent or reactant hazardous, effective, or entirely unusable for another task.
Health And Environmental Considerations
Having spent time in both academic and industry labs, I’ve witnessed more than a few chemists grow cautious with compounds like this. Chlorinated organics demand respect. Breathing their vapors, spilling them, or improper disposal all pose real risks. C7H6Cl2 deserves the label of a potential environmental contaminant. Documented cases have shown that improper handling or unchecked emissions can damage aquatic life and even seep into human water supplies. By knowing its formula and structure, professionals develop safer protocols, so harm can be minimized. Personal experience has shown me strict labeling and training—grounded in clear understanding—reduce accidents and long-term exposure.
Real-World Solutions
Replacing older, riskier compounds with safer alternatives makes sense, yet not all processes have ready substitutes. Regulation, like the efforts seen in modern chemical plants, means less discharge enters the environment. It also means more focus lands on pilot projects that recover or destroy waste products completely. Outreach to workers in the chemical field about the dangers posed by C7H6Cl2, highlighting molecular specifics, increases safety. Investments in better filtration and monitoring equipment have paid off, turning theoretical safety guidelines into practiced habits.
Chemists, engineers, safety officers, and environmental scientists all benefit from starting basic: knowing exactly what atoms make up the substance they handle. For 2,6-Dichlorotoluene, its molecular formula isn’t just a technicality. It unlocks a better understanding of potential uses, risks, and safe practices, adding real-world value in every lab and processing facility that encounters it.
Is 2,6-Dichlorotoluene hazardous or toxic?
Getting to Know 2,6-Dichlorotoluene
In the world of specialty chemicals, 2,6-Dichlorotoluene turns up in the production of dyes, agrochemicals, and pharmaceuticals. If you ever worked in a plant or lab, this compound’s sharp “chemical” odor becomes hard to forget. Its structure, a toluene ring with two chlorine atoms, makes it more reactive and sometimes more harmful than plain old toluene.
How Dangerous Is It?
Not every chemical with a scary name causes harm in every situation. But 2,6-Dichlorotoluene raises some red flags, especially for those handling it a lot. The US Environmental Protection Agency lists it as a hazardous substance. Skin contact, breathing in vapors, or accidentally ingesting any can bring on headaches, dizziness, and irritation to the skin or eyes. According to the National Institute for Occupational Safety and Health, high vapor concentrations may knock someone out. The numbers matter—working with small amounts in a well-ventilated space lowers the risk, but breathing in fumes over time or having a spill without gloves brings real trouble.
Impact on the Environment
Spilling large quantities of 2,6-Dichlorotoluene into a river or yard doesn’t go unnoticed. In soil and water, the chemical breaks down slowly. Chlorinated aromatics such as this one can cling to sediments or travel with stormwater. Fish and insects take a hit from long-term exposures, with some studies linking similar compounds to growth changes and lowered survival rates. It doesn’t build up in the food chain the same way PCBs do, but it doesn’t wash away overnight, either.
Protecting Workers and Communities
No chemical safety sheet covers everything that might go wrong, but a few tried-and-true steps make a difference. Wearing nitrile gloves, goggles, and a lab coat keeps most splashes and fumes off your skin and out of your lungs. Chemical fume hoods and good ventilation help workers avoid headaches and worse. If something spills, speedy clean-up using absorbent pads—never letting the stuff run into a drain—limits the spread. Regular air monitoring and training keep those in charge from missing small leaks or signs of overexposure.
Could Safer Alternatives Replace It?
Some companies look for drop-in replacements to lower health or environmental risks, but swapping out 2,6-Dichlorotoluene isn’t easy in every process. Formulators and R&D labs work toward “greener” chemistry with less persistent byproducts. Not every substitute can do the job with the same reactivity or cost, yet industries keep pushing for change, especially as regulations in Europe and North America get tougher.
The Bottom Line on Responsibility
Ignoring the risks around chlorinated aromatics brings harm not just to workers, but to neighborhoods downstream from discharge pipes. Open communication, regular chemical audits, and transparency from manufacturers put everyone on a safer path. Having spent years in labs and production areas, I’ve seen firsthand how slips in safety practices quickly escalate. Real accountability means fixing small mistakes before they lead to emergencies—and listening to concerns from both inside and outside the workplace.
How should 2,6-Dichlorotoluene be stored and handled?
Understanding the Real Risks With 2,6-Dichlorotoluene
Folks who spend their careers around chemicals quickly spot the difference between caution and paranoia. 2,6-Dichlorotoluene might sound technical, but it brings straightforward dangers: it’s flammable, can irritate skin and lungs, and doesn’t mix well with careless routines. I once saw a can left loose near a hot radiator in an older lab — the risk wasn’t about regulations, but about people’s safety. Fire and injury rarely come announced.
Storing it Right: Steps That Save Trouble
Any workplace keeping 2,6-Dichlorotoluene must store it in tightly sealed containers, kept away from heat, sparks, or open flames. I recommend using metal cans with proper seals — plastic sometimes reacts or leaks fumes. A store room with mechanical ventilation helps keep vapors from building up, and any storage spot should stay cool and dry. This isn’t overkill; vapors from solvents like this can drift and find an ignition point faster than most realize.
Clear labeling can’t be skipped. Nobody wants a shelf filled with ambiguous jars or forgotten containers. In my years around shop floors and academic labs, accidents almost always start with a mystery bottle or a hasty, incomplete label. A simple batch number, hazard symbol, and the full product name keep everyone honest.
Personal Protection Isn’t Optional
Some chemicals allow for short sleeves and light gloves — not this one. I’ve relied on chemical goggles, gloves made from nitrile or neoprene, and a lab coat before opening any bottle. Strong ventilation, a working fume hood, or at least a portable exhaust fan not only keeps accidents rarer, it also keeps headaches and lung problems at bay. If 2,6-Dichlorotoluene spills, absorb with sand or a similar material, sweep it into a sealed drum, and use respiratory protection if fumes linger. Don’t improvise with paper towels or old rags, which can catch fire.
Training and Old-Fashioned Accountability
Safety doesn’t come from posters or checklists; it grows from people watching out for each other. On every site I’ve worked, real culture change started once every staff member learned where emergency showers, eyewash stations, and spill kits were stashed — not just the supervisors. A quarterly walkthrough, led by someone who’s actually mixed or transferred chemicals, works better than a thousand memos.
Most serious accidents boil down to three things: unclear labels, cluttered storage, and untrained staff. Training is not a box to tick, but something that should happen face-to-face, hands on, with a chance to ask questions that actually matter. Google’s E-E-A-T outlines the value of real-life experience, and it’s clear here. I trust someone’s advice when they’ve wiped up the spills and handled the containers themselves.
Seeking Smarter Systems
One big change many firms could embrace is a centralized tracking system. In the past, we used paper logbooks, but now digital solutions exist. Simple barcode systems cut out confusion and tell you who opened a drum and when. More data gives a stronger sense of real-world exposure and helps with recalls or audits.
Storing and handling 2,6-Dichlorotoluene safely isn’t a niche topic — it means respecting how much trouble a single slip brings. A solid plan, basic common sense, and some stubborn attention to detail pay off every day.
What are the available packaging sizes for 2,6-Dichlorotoluene?
Understanding Availability
2,6-Dichlorotoluene isn’t something you spot on grocery shelves. Most folks who need it use it in specialty chemical work, pharmaceuticals, dyes, or pesticides. Because of these uses, the packaging sizes largely match what makes sense for labs and factories. If you want to buy some, you’ll notice a handful of package sizes popping up most often: smaller bottles for research and bulk drums for manufacturing.
Packaging Choices for Different Needs
Lab users usually grab 2,6-Dichlorotoluene in glass or HDPE bottles. Typical volumes run from 100 milliliters up to 1 liter. Experienced researchers know this makes lab handling safer and easier to store. It’s easier to keep a small bottle out of the way, keep contamination low, and precisely measure quantities. Larger jugs are less practical for bench work. Suppliers like Sigma-Aldrich or Merck cater to this market, keeping most containers at 500 mL and 1 L for standard orders.
Once you look at pilot plants or full factories, the situation shifts. A small bottle won’t cut it; workers order by the drum. The most common size in industrial supply circles: 200 liters (or 55 gallons). This size takes up a whole drum, which is sturdy enough for shipping by truck or container. That’s a favorite option for mid-scale manufacturers who need safety, cost savings, and fewer refills. Alongside the big drums, it’s not hard to find requests for 25-liter or 50-liter containers to fill the gap, usually in steel or thick-walled polyethylene.
Why Package Size Matters
Anyone who has spent time managing a chemical store room will tell you that packaging impacts both cost and safety. A big drum is cheaper per kilogram, but once it’s open, it can go bad faster or become a spill risk if not managed well. Smaller bottles sit on shelves longer without worry, but the price climbs, and discarding empty packages gets annoying. There’s no one-size-fits-all answer, and the best choice often depends on how fast you’ll use it and how good your safety setup is.
Safe Transport and Storage
2,6-Dichlorotoluene isn’t as notorious as some chemicals for hazard, but it has to be handled with respect. Spills can cause headaches, literally and figuratively. Large containers often come with steel or high-density polyethylene, both proven to reduce leakage and withstand bumpy rides across the country. Smaller bottles use glass for chemical stability and clarity, letting users see what’s left before restocking. Labels always list handling advice and standard UN transport codes for chemical safety.
Improving Access and Reducing Waste
One big lesson from industry and research: flexibility in packaging can cut waste and lower costs. Suppliers who offer both small bottles and drums allow buyers to match purchase size to real need instead of forcing stockpiling or frequent repurchase. Some companies now collect empty drums or offer recycling to keep used containers out of landfills. This kind of thinking not only saves money but also makes chemical handling safer for everyone, from the shipping dock to the lab bench.
Getting What You Need
Whether you’re buying 2,6-Dichlorotoluene for an experiment next week or to run a production line, package size isn’t a small detail. Talk with your supplier about your real usage, ask about return or recycling programs, and check that containers fit your safety and storage plans. The right size ends up saving cash, lowering risk, and keeping the process steady.
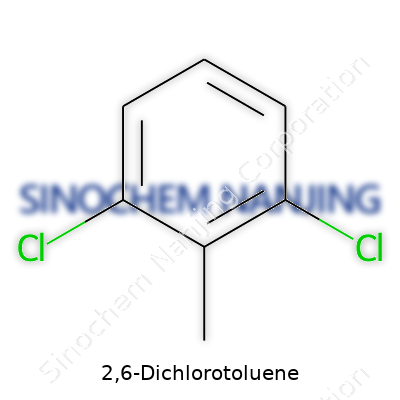
| Names | |
| Preferred IUPAC name | 1,3-Dichloro-2-methylbenzene |
| Other names |
2,6-Dichloro-1-methylbenzene 2,6-Dichloromethylbenzene 2,6-Dichlorotoluol 1-Methyl-2,6-dichlorobenzene |
| Pronunciation | /tuː sɪks daɪˌklɔːroʊ təˈluːiːn/ |
| Identifiers | |
| CAS Number | 83-34-1 |
| Beilstein Reference | 606140 |
| ChEBI | CHEBI:131769 |
| ChEMBL | CHEMBL15455 |
| ChemSpider | 12180 |
| DrugBank | DB02174 |
| ECHA InfoCard | 100.009.020 |
| EC Number | 203-899-7 |
| Gmelin Reference | 136080 |
| KEGG | C19230 |
| MeSH | Dichlorotoluenes |
| PubChem CID | 12163 |
| RTECS number | XS8575000 |
| UNII | V0F4N6E9E7 |
| UN number | UN2321 |
| Properties | |
| Chemical formula | C7H6Cl2 |
| Molar mass | 177.04 g/mol |
| Appearance | Colorless to yellow liquid |
| Odor | Aromatic odor |
| Density | 1.28 g/mL at 25 °C |
| Solubility in water | Insoluble |
| log P | 3.98 |
| Vapor pressure | 0.4 mm Hg (20 °C) |
| Basicity (pKb) | 12.01 |
| Magnetic susceptibility (χ) | -73.0e-6 cm³/mol |
| Refractive index (nD) | 1.552 |
| Viscosity | 0.8 mPa·s (20 °C) |
| Dipole moment | 2.61 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 192.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -83.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4313.6 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H315, H319, H335 |
| Precautionary statements | P210, P261, P273, P280, P301+P312, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 2,1,0 |
| Flash point | 64 °C |
| Autoignition temperature | 605 °C |
| Explosive limits | 1.3–7% |
| Lethal dose or concentration | LD50 (oral, rat): 2600 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 2453 mg/kg |
| NIOSH | *CN 3670000* |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.5 ppm |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
Benzyl chloride Benzal chloride Toluene Benzyl alcohol Benzaldehyde |

