2,6-Dichlorobenzaldehyde Oxime: Insight, Application, and Future Directions
Historical Development
Chemists first synthesized 2,6-dichlorobenzaldehyde oxime as research around chlorinated aromatics heated up in the mid-20th century. Folks sought better synthetic options after World War II, looking for more selective reagents and intermediates as boom times for fine chemicals, dyes, and agrochemicals arrived. Organic chemistry labs across Europe and North America made solid progress, rooting their work both in the theoretical studies of functional group transformations and in hands-on experimentation with chlorinated benzenes. Early literature describes persistent problems isolating pure products from complex reaction mixtures, but as technology—such as column chromatography and reliable melting point equipment—landed in academic and industrial labs, confidence and yields improved. Patent filings from that era show competing companies pushing for novel applications of dichlorinated aromatic molecules, laying real groundwork for the specialty chemical industry we see now.
Product Overview
2,6-Dichlorobenzaldehyde oxime carries deep relevance in organic synthesis. Its unique oxime group attached to a dichlorinated aromatic ring unlocks selective reactivity. Firms in the chemical supply business usually handle it in solid form for lab and pilot plant use, offering packing in heavy-duty containers to cut down on moisture exposure. The product gives chemists an entry point to diverse transformation reactions thanks to the electron-withdrawing effect from the chlorines, which modulates both the stability and nucleophilicity of the oxime group. Beyond research, technical specialists in agrochemical, pigment, and even plastics industries consider this material for its building-block value and, sometimes, its stabilizing effects on intermediates.
Physical & Chemical Properties
This compound appears as an off-white to light tan solid in most lab shipments. Slight yellowing sometimes signals minor impurities or storage outside ideal conditions. By experience, it dissolves sparingly in cold water but more readily in mixed organic solvents, such as dichloromethane, acetonitrile, or ethanol. The chlorinated aromatic ring delivers marked chemical stability, but under heated or strongly acidic environments the oxime bond can fall prey to hydrolysis or rearrangement. Melting point checks typically fall in the 126–130°C range, so careful temperature control pays off for those collecting pure fractions. It emanates a faint, recognizable odor reminding many chemists of other aldehyde derivatives.
Technical Specifications & Labeling
In regulated chemical commerce, labels detail purity—often measured at ≥98% by HPLC or GC-MS—moisture levels, and possible isomer content. Detailed batch reports support traceability, while the UN number and hazard pictograms signal to operators the potential for harmful exposure. Lot numbers serve double duty—facilitating recalls and anchoring ongoing research projects in reproducible reality. Those handling bulk quantities adhere to labeling protocols reflecting regional rules, such as GHS standards, to streamline safe transfer between supply chain partners. I’ve seen more than one project veer off track due to unordered or mislabeled stock—routine documentation can make or break reliability in the lab.
Preparation Method
Most commercial syntheses tap into the electrophilic chlorination pathway, starting from 2,6-dichlorobenzaldehyde. Oximation follows—typically involving hydroxylamine hydrochloride in a buffered aqueous or alcoholic solution. Small-scale procedures rely on maintaining pH control to prevent unwanted side reactions, such as aldehyde reduction or over-oxidation, while scaling up requires attention to reaction vessel compatibility and efficient separation of by-products. The resulting solid is isolated by filtration, washed with cold ethanol, and recrystallized where purity above research grade is required. This pathway keeps maintenance costs predictable for manufacturers and lets R&D teams tweak reaction parameters for specialized needs.
Chemical Reactions & Modifications
2,6-Dichlorobenzaldehyde oxime opens the door to a range of transformations. Reduction under catalytic hydrogenation or with selective metal hydrides yields primary amines, which form the backbone of further synthetic applications. N-O bond cleavages generate benzaldehyde derivatives that chemists pursue for aroma, pharmaceutical, or polymer uses. The oximino group can be further functionalized—alkylated, acylated, or converted to heterocycles—pushing innovation in dye and pigment chemistry where colorfastness and thermal stability matter. The chlorine atoms guide substitution reactions across the aromatic ring, especially under nucleophilic aromatic substitution conditions favored in electronic and functional materials industries. Tactical modifications to the core structure let scientists tailor material properties, all while maintaining a foothold in reproducible, scalable synthetic methods.
Synonyms & Product Names
The compound crops up in catalogs and research literature under a handful of names. “2,6-Dichlorobenzaldehyde oxime” is the most direct, but you’ll also see “2,6-dichloro-phenylaldoxime” and “2,6-dichlorobenzaloxime.” Occasionally, shorter designations like “DCBA oxime” come into play in lab logs or internal reports. On the manufacturing and distributor end, SKU-linked names tend to drop in catalog or supply codes for logistical efficiency. Despite varied nomenclature, experienced chemists read the chlorinated ring pattern and functional group, keeping synthesis strategies predictable and robust.
Safety & Operational Standards
Anyone handling 2,6-dichlorobenzaldehyde oxime spends time reviewing its safety data sheet (SDS). Direct contact can irritate skin, eyes, and the respiratory tract; inhalation of dust or inappropriate handling can prompt allergic reactions or, in rare situations, more severe toxicological consequences. I’ve seen both new hires and seasoned professionals underestimate the risks—lab coats, eye protection, and gloves, coupled with effective ventilation, anchor most safe practices. Storage away from strong oxidizers and acids prevents unwanted decomposition. Waste disposal follows hazardous organic material routines, anchored by local environmental legislation and institutional protocols. Routine safety drills and up-to-date first aid stations keep labs on the right side of operational integrity.
Application Area
This compound fills real gaps across research and industry. Small- and medium-sized enterprises in dye manufacturing lean on it for its role as a precursor in azo and indole pigment synthesis. Agrochemical developers push its structure into preclinical screens, hoping new derivatives block fungal or insect threats more efficiently. Electronics manufacturers test related oxime compounds for thermal and chemical resistance in polymers used in circuit boards and specialty coatings. Analytical chemistry sets value its presence in test reactions, benchmarking selective transformations or developing calibration standards. All these uses stress the need for robust supply lines, regulatory approval pipelines, and the willingness to adapt reaction conditions on the fly to suit changing project scopes.
Research & Development
Teams in academia and industry devote real time to understanding what can be coaxed from 2,6-dichlorobenzaldehyde oxime. Researchers dive deep into the compound’s tautomerism, electron density distribution, and influence on subsequent reactivity—hoping to uncover better catalysts or smarter functionalization strategies. I’ve joined projects testing new substitution patterns, probing how different buffer systems or catalyst loads impact product distribution. Industrial groups often drop it into high-throughput synthetic screens, betting on novel pharmacophores or improved performance coatings. Lab managers keep analytic equipment—HPLC, NMR, MS—in constant rotation to pin down impurities and degradation pathways. Outside the lab, patent filings and start-up launches signal that commercial interest in specialized derivatives hasn’t faded anytime soon.
Toxicity Research
Toxicologists drilled into the acute and chronic effects of dichlorinated aromatic oximes as part of broader environmental monitoring protocols. Initial screens showed low acute toxicity at typical laboratory exposure levels—but as with many aromatic compounds, chronic effects such as sensitization or metabolic breakdown products warranted real concern. Environmental chemists flagged possible persistence in soil and water, drawing attention to degradation products under UV and microbial activity. Animal studies inform safety margins but consistent, independent data on human exposure, especially at scale, remains limited. Industry and academia both see pressure to keep digging for a fuller picture, especially as new application areas emerge and regulatory scrutiny expands.
Future Prospects
Demand for 2,6-dichlorobenzaldehyde oxime sits at the intersection of specialty organic synthesis and sustainable development. Companies active in green chemistry want to tweak synthesis pathways for less waste and better atom economy. Researchers aim for sharper selectivity in follow-up reactions, reducing purification burdens and energy use in plant settings. Regulatory shifts toward tighter safety limits and traceability challenge both suppliers and end users to invest in better monitoring and compliance tools. As the digital marketplace for chemicals expands, traceable, authenticated batches tied to robust analytical support will win out. Academics and industrial scientists press for comprehensive studies on both long-term exposure and pathways to bio-based analogs, with an eye on building safer and more sustainable processes for decades to come.
What is the chemical formula of 2,6-Dichlorobenzaldehyde Oxime?
The Story Behind the Formula
I first came across 2,6-dichlorobenzaldehyde oxime during a stint in an undergraduate research lab. Chemical formulas can feel like secret codes, but this one—C7H5Cl2NO—reveals a lot once you dig in. The structure starts with a benzene ring, two chlorine atoms at the 2- and 6-positions, and an oxime group replacing the standard aldehyde. Not just a random arrangement—these details determine how this compound acts in the world, from the lab bench to the field.
Why the Structure Matters to Us
Every functional group on that ring brings a property along. Swap in the oxime group and suddenly this is a tool for capturing and converting molecules, whether you're exploring pesticides, trying to guard crops from mold, or building out new materials. The two chlorines pack extra punch—turning this chemical into something more resistant to breakdown in the environment compared to an unhalogenated cousin. That means both promise and concern: it sticks around, and that can help or hurt.
My own work with related compounds showed how small changes in chemistry shape what happens next. That formula, C7H5Cl2NO, isn’t just a string of letters and numbers. It represents the backbone of a family of molecules with a big presence in organic synthesis. Lab teams reach for these deviously simple chemicals when they need a selective reaction or plan to create a bigger, more complex molecule.
What We Should Watch Out For
Chlorinated aromatics like this one raise more than a passing question about environmental safety. Nature doesn’t break down chlorine bonds quickly; these stick around, finding their way into water, soil, and air. Researchers have found traces of similar molecules even in places far from any factory or farm. That persistence earns regulatory attention. No surprise: the science on long-term impact has gaps, and public trust calls out for transparency here.
Some oximes connect to bigger questions about human health and safety. Exposure risks depend on how and where chemicals are released, so handling and disposal need solid protocols. If a drum leaks in a plant, or a spill runs off into local rivers, families living nearby could face unwanted exposure. That means looking at not just formulas on paper, but real consequences for communities who often feel overlooked by decision-makers.
Toward Safer and Smarter Chemistry
Synthetic chemists know the appeal of a molecule that does its job. I get it—those tools build new medicines and materials. Still, the next step asks how we can innovate with more care. That might mean adjusting formulas so molecules break down faster in nature, picking substituents that offer performance but less persistence, or using green chemistry methods for synthesis and processing. At industry scale, these choices can feel expensive up front, but longer-term savings and public health benefits build trust and goodwill.
Every time I run into another dense chemical formula, I remember the first lesson from my old PI: Always look past the label. Ask what this chemical will do, and what trail it leaves behind. 2,6-dichlorobenzaldehyde oxime, with its formula C7H5Cl2NO, is more than a textbook example—it’s a reminder that we’re still learning how to balance what’s possible in chemistry with what’s wise. Pushing toward solutions means chemists, communities, and policy voices all share a seat at the table.
What are the common applications or uses of 2,6-Dichlorobenzaldehyde Oxime?
What Makes This Compound So Noteworthy?
2,6-Dichlorobenzaldehyde Oxime stands out for anyone who's ever worked with organic synthesis or chemical manufacturing. Its name catches the eye on ingredient lists, but real-world uses show why scientists and manufacturers value it. As someone who's tinkered with both research and hands-on chemistry, I've seen this molecule help shape specialty products far beyond the lab.
Building Better Crop Protection
Modern farming often depends on chemicals that target weeds and safeguard harvests. Companies blend 2,6-Dichlorobenzaldehyde Oxime into the mix to help create herbicides and fungicides with increased activity and just the right level of selectivity. This one compound helps bind to weed and fungus enzymes, boosting how well a formula tackles tough plant invaders while sparing the crops. Sustainability and food production go hand-in-hand with reliable crop yields, and chemical advances like this make a real impact.
Pharmaceutical Starting Point
Chemists searching for the next life-saving medicine need complex building blocks — not every innovation starts from scratch. This oxime turns up as a handy intermediate for certain antihypertensive and anti-inflammatory drugs. Medicinal chemists find reliable reactions and a predictable profile when using this molecule as a scaffold, which takes some of the guesswork out of developing new treatments. The need for precise and consistent starting materials becomes clear after a few failed syntheses or costly reruns in the lab.
Industrial Benefits in Chemical Synthesis
Its structure offers flexibility for custom manufacturing. During specialty polymer creation or fine chemical production, manufacturers add 2,6-Dichlorobenzaldehyde Oxime to steer the direction of a reaction or to anchor a key fragment. Its presence can mean better yields and cleaner end-products, which anyone working with tight budgets and regulatory oversight can appreciate. Every improved step in an industrial process reduces waste and keeps both costs and environmental footprints in check.
Specialty Analytical Applications
Laboratories lean on this oxime to detect and separate metals in environmental and industrial samples. The oxime reacts with trace elements, giving chemists clearer readings in techniques such as spectrophotometry or chromatography. If you've ever handled water quality tests or soil analysis, you know the headache of uncertainty in measurements. Reliable reagents like this remove doubt and provide results people, businesses, and regulators can trust.
Safety, Regulation, and Room for Improvement
Production and use do not come without scrutiny. Handling this compound requires well-ventilated space and protective gear, as with any reactive organic chemical. Producers focus on minimizing risks throughout the production chain and final product application. Recent regulatory changes ask companies to disclose more data about chemical properties and persistence, helping keep workers and consumers safer. Finding greener pathways to make or degrade this compound has become a focus in ongoing research. Partnerships between academic labs and industry groups show promise in reducing unwanted byproducts and improving overall safety.
Potential For Future Innovation
Any compound with so many uses draws attention from both innovators and watchdogs. Breakthroughs in bio-based synthesis or advances in formulation could open up additional applications. Researchers continue to search for ways to reduce environmental impact and maximize the benefits offered by 2,6-Dichlorobenzaldehyde Oxime. As knowledge grows and regulations reflect new realities, safer handling and smarter design will shape its path forward.
What is the recommended storage condition for 2,6-Dichlorobenzaldehyde Oxime?
Overview
2,6-Dichlorobenzaldehyde oxime shows up in labs, research projects, and chemical manufacturing for a reason. It carries strong reactivity and makes its way into the synthesis of more complex organic compounds—sometimes pharmaceuticals, sometimes specialty materials. These qualities make proper storage more than a checklist item. Safe, stable storage keeps this compound in its best working shape and blocks hazards from cropping up where they are not welcome.
What Keeps 2,6-Dichlorobenzaldehyde Oxime Safe and Effective
From my time working around chemical storerooms, I’ve learned that containment and clarity matter most. 2,6-Dichlorobenzaldehyde oxime benefits from a cool, dry, and well-ventilated spot away from bright light and heat sources. Humidity tends to creep in and encourages clumping or degradation, ruining your sample. Once, I watched a batch go lumpy in a damp cabinet, and the loss set back a project by days. Storing chemicals like this one inside a tightly sealed amber bottle blocks those pesky UV rays and keeps out moisture in the air.
The temperature target sits below 25°C, which usually points to a climate-controlled storeroom. The idea is to avoid fluctuations because chemical stability can decline with temperature swings. A sudden spike in heat, maybe from a sun-facing window or malfunctioning AC, bumps decomposition risk and can spark off-gassing or fumes—a real headache in terms of both safety and sample reliability.
Why Storage Location Matters
Placement inside a chemical store makes real-world sense. It should always rest away from acids, bases, and especially strong oxidizers. Cross-reactivity creates more than a mess—it could start a chain reaction, fire, or release hazardous vapors. In one industrial setting, I saw a poorly separated shelf allow a spill from a strong acid to mix with a nearby organic. The resulting cleanup and ventilation showed just how fast things escalate when storage plans get sloppy.
Labels play a big role here. Every bottle deserves a clear, visible label with the full name plus hazard info, storage precautions, and date of receipt. These labels are a lifesaver during audits and emergencies. In the rush of routine, a mislabeled or faded bottle can lead to accidental misuse or unsafe mixing, putting staff and processes at risk. Dating reagents also avoids accidental use of degraded samples, which can skew data and delay projects.
Physical Storage Practices
Solid compounds like 2,6-Dichlorobenzaldehyde oxime should keep to their own containers. Avoid scooping from communal jars; cross-contamination builds up over time. Personal experience taught me that using clean, dry tools for each transfer makes a difference in purity levels. Cardboard boxes or paper wrappings never cut it—chemical-grade glass or compatible plastics prevent leaching or accidental reactions with packaging. Stored on non-reactive shelving, preferably metal with spill trays, each vial or jar finds a steady spot in a well-ventilated zone with no risk of flooding or excess damp.
Potential Solutions for Better Chemical Management
Many labs turn to digital inventory systems and regular audits. These make tracking batches and expiration dates easier, so reagents like 2,6-Dichlorobenzaldehyde oxime won’t be used past their prime. Installing humidity and temperature loggers gives real-time alerts before conditions stray from the safe zone. Staff training, not just annual refreshers, but practical locker-room conversations keep risks top of mind. I’ve found that proactive communication and visible reminders around storage areas do more for safety than rules buried in a training guide ever could.
Smart, careful storage not only protects people, but also guarantees quality outcomes in any scientific or industrial project that counts on 2,6-Dichlorobenzaldehyde oxime. Each step in storage, labeled well and controlled closely, builds trust in the sample and the science that follows.
Is 2,6-Dichlorobenzaldehyde Oxime hazardous or does it have any safety precautions?
Looking Closely at 2,6-Dichlorobenzaldehyde Oxime
Many chemicals used in labs or industry have long, hard-to-pronounce names. 2,6-Dichlorobenzaldehyde oxime is one of those. On the surface, it sounds unfamiliar, but chemicals like this play roles in research or synthesis, sometimes acting as building blocks for bigger, more widely used compounds.
The Risks: What Workers and Users Need to Know
Safety never gets old, especially when handling things that can affect health. With 2,6-Dichlorobenzaldehyde oxime, real-world experience says to approach with care. Like other similar oxime-based compounds, skin and eye contact can irritate, and breathing in dust or fumes can leave a tickle in the throat or worse — coughing, headache, trouble breathing. The chlorinated parts of this molecule often show up in chemicals that do more than irritate. Chlorinated benzenes, in particular, sometimes linger in the environment and resist breaking down.
Health agencies flag related oximes as having toxic tremors, especially after repeated exposure. That means if you spend each day around them without solid safety gear, your body remembers. Headaches or dizziness can pop up. Nobody wants to end a shift with eyes watering and skin itching.
Looking at the Numbers and Guidelines
The basics rarely shift: gloves, goggles, and coats protect skin and eyes from accidental sloshes or splashes. In brick-and-mortar labs and at companies serious about compliance, fume hoods vent away anything airborne before workers can breathe it. Looking up the Safety Data Sheet (SDS) for 2,6-Dichlorobenzaldehyde oxime tells a clear story — dispose of waste carefully, avoid open flames, and keep the chemical away from food and drink.
Most national agencies haven’t set big, bold limits specifically for this chemical. But similar substances prompt the use of caution. The American Conference of Governmental Industrial Hygienists (ACGIH) sets exposure guidelines for many oximes and chlorinated aromatics. Not all of these rules land in law, but good workplaces treat them as de facto rules: always assume some risk, minimize exposure, plan for accidents.
Preventing Trouble: What Works on the Floor
In my early lab days, I learned lessons the hard way: a splash of a simple oxime mix left my hands tingling for hours. After that, I doubled up on gloves and made sure the eye wash station wasn’t just for show. Ventilation dropped the chemical’s strong, sharp smell within minutes. After cleanup, all contaminated waste got sealed and labeled — nobody wants to mistake it for regular trash.
It’s common sense to avoid eating and drinking anywhere near chemicals that can irritate or poison. Workers who go home with lingering chemical scents on hands or clothes usually skipped basic hygiene — something most supervisors watch closely. Good routines, clear labels, and regular safety drills keep the shock factor down if spills do happen.
Building a Safer Routine
Ready access to gloves, face shields, and lab coats is more than just a policy. Spotting small spills and cleaning them up before lunch break keeps everyone safer. Every chemical container should get a clear label and tight seal, especially for compounds with tough names and tougher health effects. Open bottles under a vent. Don’t let curiosity tempt you to sniff or touch unknown powders or liquids.
New hires or even seasoned chemists benefit from short, stand-up meetings where last week’s safety goof-ups get reviewed. Hearing about close calls sticks much longer than reading them off a distant poster. In short, 2,6-Dichlorobenzaldehyde oxime isn’t the stuff of movies, but it won’t forgive carelessness either. Anyone who respects the chemical and follows the guidelines will head home healthy at the end of the day.
What is the purity level or specification offered for 2,6-Dichlorobenzaldehyde Oxime?
Purity: More Than Just a Number
I’ve spent years talking with chemists, engineers, and folks in the lab coat crowd. Every time the topic shifts to specialty chemicals, purity turns out to be a big deal, especially with compounds like 2,6-Dichlorobenzaldehyde Oxime. Most suppliers point to a standard purity level—98% or higher—and that’s often written right onto the certificate of analysis. That 2% might sound insignificant, but in practice, it can spell the difference between a result you can trust and wasted hours in the lab.
Why High Purity Isn’t Just for Show
Anyone who’s worked with synthesis projects knows impurities have a sneaky way of interfering. One contaminant can throw off a reaction or taint a finished product. If you use a batch with 95% purity instead of the industry-typical 98% or more, expect headaches down the road. Impurities can introduce side reactions or affect the stability of downstream products. This is especially clear in fields like pharmaceuticals, crop protection, or advanced materials where quality or safety standards are strict. Regulators focus on these details, and so should every buyer or researcher.
Specification Sheets: What They Really Tell You
Suppliers hand over specification sheets dense with data—melting point, moisture content, visible appearance, and so on. Out in the real world, people focus on the purity percentage above almost everything else. A reputable supplier provides a breakdown of possible impurities, and sometimes lists allowable heavy metals, residual solvents, or other chlorinated compounds. These numbers matter. Even a tiny bit of excess chlorinated impurity could cause incompatibility with sensitive catalytic reactions or skew biological testing results. These sheets also often include testing methods; you’ll find thin-layer chromatography (TLC), gas chromatography (GC), and high-performance liquid chromatography (HPLC) cited for analysis. Purity figures mean more when you know how they were measured.
Traceability and Trust
Purity alone doesn’t establish credibility. I remember a case where a researcher struggled with inconsistent analytical outcomes, only to discover two batches of the “same” chemical varied in key impurity profiles. Reliable suppliers back their purity claims with traceable batch records, full documentation, and even the names and signatures of their quality team. Labels and paperwork should match the sample—they serve as the backbone for trust. Most universities and big companies now refuse shipments without this level of transparency.
What Impacts Those Purity Specs?
Technical grade products generally fall in the 95-97% range, often destined for industrial processes. Research and pharma sectors require 98%, 99%, or even 99.5%. That difference seems small, but consider how hard manufacturers work to hit those numbers—multiple recrystallizations, advanced purification equipment, and rigorous filtration. These steps ramp up production costs. Lower price tags tend to signal lower grades, and sometimes less rigorous control during manufacturing.
Raising the Bar for Quality
Seeing so many industries depending on this single compound, it’s clear high purity does more than check a regulatory box. The better the starting material, the fewer surprises in experiments and the greater confidence scientists have in what they’re building. Some labs have even started running their own quality checks post-delivery, insisting on a second round of analysis to confirm claims. Long-term, this level of diligence saves time, prevents accidents, and helps unlock new discoveries by removing nagging doubt about chemical quality.
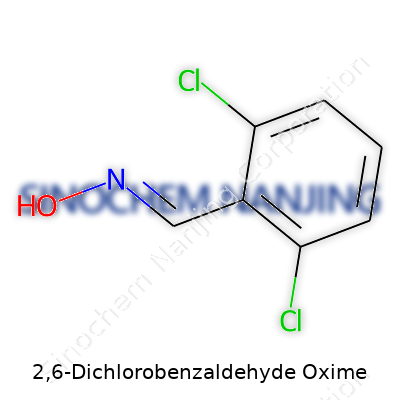
| Names | |
| Preferred IUPAC name | (E)-2,6-dichlorobenzaldehyde oxime |
| Other names |
2,6-Dichlorobenzaldehyde, oxime 2,6-Dichlorobenzaloxime |
| Pronunciation | /ˈtuː,sɪks daɪˌklɔːroʊˌbɛnˈzæl.dɪ.haɪd ˈɒkˌsiːm/ |
| Identifiers | |
| CAS Number | [17747-87-6] |
| 3D model (JSmol) | `'NC(=N/O)C1=C(Cl)C=CC(Cl)=C1'` |
| Beilstein Reference | 87325 |
| ChEBI | CHEBI:84617 |
| ChEMBL | CHEMBL140964 |
| ChemSpider | 8217605 |
| DrugBank | DB08390 |
| ECHA InfoCard | ECHA InfoCard: 100.046.691 |
| EC Number | 611-381-7 |
| Gmelin Reference | 103076 |
| KEGG | C19529 |
| MeSH | D000075433 |
| PubChem CID | 3430307 |
| RTECS number | DH6650000 |
| UNII | OL2NY0894X |
| UN number | UN3439 |
| Properties | |
| Chemical formula | C7H5Cl2NO |
| Molar mass | 204.04 g/mol |
| Appearance | White to light yellow crystalline powder |
| Odor | Odorless |
| Density | 1.35 g/cm³ |
| Solubility in water | Insoluble in water |
| log P | 1.98 |
| Vapor pressure | 6.2 × 10⁻⁵ mmHg (25°C) |
| Acidity (pKa) | 11.04 |
| Magnetic susceptibility (χ) | -65.0 · 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.621 |
| Dipole moment | 2.7626 Debye |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 253.8 J·mol⁻¹·K⁻¹ |
| Pharmacology | |
| ATC code | N01AX06 |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and eye irritation, may cause respiratory irritation. |
| GHS labelling | GHS07, GHS09 |
| Pictograms | GHS05,GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P261, P264, P271, P272, P280, P302+P352, P321, P363, P333+P313, P362+P364 |
| NFPA 704 (fire diamond) | 2,6-Dichlorobenzaldehyde Oxime: NFPA 704: 2-2-0 |
| Flash point | > 112°C |
| Autoignition temperature | > 200 °C |
| Lethal dose or concentration | LD50 oral rat 1800 mg/kg |
| LD50 (median dose) | LD50 (median dose): 640 mg/kg (rat, oral) |
| NIOSH | SN9840000 |
| PEL (Permissible) | No OSHA PEL established |
| REL (Recommended) | 10 mg/m3 |
| IDLH (Immediate danger) | No IDLH established. |
| Related compounds | |
| Related compounds |
Benzaldehyde oxime 2,6-Dichlorobenzaldehyde 2,6-Dichlorobenzonitrile 2,6-Dichlorobenzyl alcohol 2,6-Dichloroaniline |

