2,6-Dichloroaniline: A Chemical with Roots, Challenges, and Unseen Impact
Historical Development
Digging into the background of 2,6-dichloroaniline takes us back to the growth of the dye and pesticide industries in the late nineteenth and early twentieth centuries. Chemists hunted for molecules that could open up new colors and new economic opportunities, and chlorine-substituted anilines became a favorite. The rapid industrialization meant that demand for intermediates like 2,6-dichloroaniline grew fast, riding on the back of advances in synthetic chemistry. Factories popped up near trade centers, and researchers drew out new methods, changed reaction conditions, and tweaked old recipes to make production faster, cheaper, and more reliable. Watching the market swell and fragment over decades, anyone with even passing experience in the chemical sector could see how intermediates, once just tools for dye works or pharmaceutical routes, began anchoring entirely new businesses.
Product Overview and Physical & Chemical Properties
Holding a sample of 2,6-dichloroaniline, you see a pale solid, not especially remarkable in appearance but potent in reactivity and usefulness. Aromatic amines like this one share some basic tricks: a sharp smell, low solubility in water, and a tendency to react briskly with acids and other electrophiles. Yet those two chlorine atoms, snug on the benzene ring, matter a lot. They shape the reactivity, shift the melting and boiling points, and open pathways that plain aniline does not reach. The density feels about right for this class—solid but not hard to break up for lab work.
Technical Specifications & Labeling
Industry regulations demand clear labeling to guard against mishap, and most seasoned workers in the sector learn to scan for purity, trace contaminant levels, and specific handling instructions. The label stands as both a shield and a flag. Those who work with this chemical know they must leverage that information for every batch, especially since product specifications can signal what applications are possible or out of bounds. Even a minor fluctuation in purity can change reaction outcomes, so savvy operators treat technical sheets not as fine print but as a vital tool.
Preparation Method
Making 2,6-dichloroaniline takes a chemist through a carefully mapped journey, usually starting with 2,6-dichloronitrobenzene. That nitro group gets reduced, often by iron filings in acid, to produce the aniline. This route echoes the classic reductions that pepper organic lab courses, though scaled for industry with an eye on yield and waste control. While newer, greener methods tempt some, the older iron reduction has hung on because it works well and stays cost-effective unless strict environmental clauses push firms to switch.
Chemical Reactions & Modifications
Once in your hands, 2,6-dichloroaniline opens up a web of possibilities. The amino group lets chemists push forward into diazotization, coupling, or complex formation; the chlorines block some paths but steer reactions toward others. That’s key in specialty dye synthesis or prepping building blocks for active pharmaceutical ingredients. The molecule’s quirks mean that one reaction might roll smoothly while another stalls, reminding every chemist that starting materials matter as much as skill.
Synonyms & Product Names
The same chemical hides behind a thicket of names across continents and catalogs: 2,6-dichlorobenzenamine, 2,6-DCAn, and a handful of trade labels that vary by manufacturer. Supply chains and global trade routes often confuse newcomers with this naming jungle, but veterans in dye manufacture, agrochemicals, and pharmaceutical intermediates develop an ear for the synonyms that actually matter. It pays off to double check, as a mismatched name means wasted time or far worse.
Safety & Operational Standards
Every worker who weighs, grinds, or dissolves this solid knows the risks don’t come just from the dust or fumes but from years of repeated exposure. This goes way beyond the label’s warning icon: the industry trains people rigorously on the use of ventilated hoods, splash goggles, and gloves, because an aromatic amine with halogen substitution does not forgive carelessness. Regulatory agencies set tight exposure limits for a reason. Nowadays, no responsible operator treats such standards as mere red tape, not after cases of chronic illness and workplace incidents have become all too real across factories and labs worldwide.
Application Area
The true reach of 2,6-dichloroaniline shows in its quiet support of bigger stories. It powers the synthesis of herbicides like diuron, colors in textile dyes, photographic chemicals, and rare specialty pigments. Anyone involved in crop protection, dyeing denim, or making photochemicals has probably depended, unknowingly, on this molecule’s structured stubbornness. Changes in pesticide trends, textile demand, or imaging technology ripple straight back to the demand for this intermediate. Economists and supply chain planners track those ripples, even if the product itself rarely makes headlines.
Research & Development
Chemists and chemical engineers keep hunting for better ways to use, modify, and produce 2,6-dichloroaniline. In my own experience on small-scale synthesis projects, companies crave new derivatives or cleaner reaction paths, either to reduce costs or to head off tightening environmental rules. Researchers look into catalytic hydrogenations, enzymatic reductions, or tweaks to storage and stability, chasing efficiency but not at the expense of safety or regulatory headaches. Scholarly journals have laid out a slow march toward more sustainable production, but industry change still happens one line or one regulation at a time.
Toxicity Research
This compound is not gentle to living systems. Decades of toxicological studies warn about its hazardous effects on skin, lungs, and organs with longer-term exposure. Animal studies keep adding fresh caution. For the chemical sector—especially those working in developing economies where safeguards lag behind—those research findings turn into urgent reminders more than dry facts. Staff training, improved personal protection, and medical surveillance can lower the risk, though experts debate how much exposure truly counts as “safe.” Waste management brings its own challenge because residues can contaminate water tables or air, leading to real risks for entire communities.
Future Prospects
Looking ahead, the fate of 2,6-dichloroaniline winds through evolving regulations, advances in green chemistry, and swings in downstream markets. Manufacturers face mounting pressure to cut emissions and tighten waste handling, especially since consumers—armed with a little more knowledge than before—demand proof of responsible sourcing. Chemists try new catalysts and energy-saving routes, hinting at a move away from batch chemistry into continuous or even bio-based processes. At the same time, substitutes nudge closer as companies fear regulatory bans or tighter restrictions, but so far, demand for robust and cheap aromatics keeps the old molecules relevant. The pace of change feels uneven; progress happens in fits and starts, shaped not just by technical breakthroughs but by political will, grassroots campaigns, and shifting consumer culture. Anyone invested in the future of synthetic chemistry pays close attention, because the shape of the next generation of intermediates often predicts the products—and the risks—on tomorrow’s shelves.
What is 2,6-Dichloroaniline used for?
Behind the Chemistry: Where 2,6-Dichloroaniline Goes
Working in the manufacturing sector years ago, I saw chemicals like 2,6-Dichloroaniline show up repeatedly on delivery slips. Most folks outside of labs or factories might not recognize the name, but its role travels much farther than plant gates. This compound often finds a home in pesticide production. Farmers count on crop protection to make sure fields stay productive, and 2,6-Dichloroaniline serves as a stepping stone in making herbicides and fungicides. If you eat bread, rice, or vegetables packed in bulk bins, you’ve likely benefited from the boost these chemicals give crop yields and food security.
Pharmaceuticals rely on this compound too. Drug developers frequently build new medicines from known starting points, and 2,6-Dichloroaniline forms part of several drug-making recipes. Some antihistamines, which help with allergies and seasonal sniffles, begin with this little molecule. Antibiotics also use it while doctors wrestle with resistant infections and search for treatments that work when other options fail.
Staying Safe in Production
While the benefits keep manufacturers coming back to 2,6-Dichloroaniline, safety isn’t automatic. In my early days, I saw clear signs reminding crews to wear gloves and masks. It irritates skin and eyes, and inhaling dust stings. Those handling this chemical can’t just wing it—regulations lay out chemical hygiene plans, disposal methods, and protective gear standards. The U.S. Occupational Safety and Health Administration (OSHA) and similar agencies worldwide publish exposure limits and conduct spot checks. Every worker stepping onto a chemical site sees posters about decontamination and spot-test kits. This oversight stems from accidents of the past and protects people who depend on a paycheck from risky jobs.
Environmental Questions: Now and the Future
Environmental groups keep their eye on anything traveling through soil or water, and 2,6-Dichloroaniline often attracts scrutiny. Herbicide runoff and industrial byproducts bring it into rivers and fields where it doesn’t belong. I once helped at a site where a minor leak ended up costing millions after clean-up teams spent months restoring groundwater. Scientists still study what chronic, low-level exposure does to waterways or fish. The Environmental Protection Agency (EPA) tracks releases and sets reporting guidelines. Better containment systems, real-time leak detection, and replacement materials could cut down on accidents. Companies face hard questions from communities living near plants or downstream.
Finding a Way Forward
Manufacturing brings progress and problems in equal measure. No easy trade-off exists for the comfort of modern crops or medicine. People who work with chemicals want pay that protects their health. Consumers want food and drugs they can trust. Neighbors expect factory operators to treat water and land like it’s their own backyard. Technology helps by improving detection and clean-up, but oversight and public watchdogs matter, too. If industry slows down and looks for safer alternatives—or engineers better processes—it won’t just dodge fines. It keeps the system fair for everybody relying on it, now and tomorrow.
What are the safety hazards associated with 2,6-Dichloroaniline?
The Chemistry of Risk
Most people haven’t heard of 2,6-Dichloroaniline, though it shows up in more places than you'd guess. This chemical helps produce dyes, agrochemicals, and pharmaceuticals. Produced in bulk, it’s a favorite for technical applications. That doesn’t mean it’s safe. Its chemical make-up creates real hazards—hazards some workers face every day.
What Makes 2,6-Dichloroaniline Risky?
One thing that stands out: this compound gives off harmful vapor. Anyone inhaling the dust or fumes complains of throat irritation and trouble breathing. Swallowing even a bit or getting the powder on skin can trigger toxic effects. Skin contact can bring swelling, blisters, and red patches. The compound's color—usually off-white or brownish—develops as it oxidizes, which increases the risk for accidental exposure since dust particles escape easily if containers aren't sealed.
Real-World Accidents: Lessons on the Floor
More than a few workers have ended up in the hospital after breathing in 2,6-Dichloroaniline dust. Reactions include headaches, dizziness, nausea, and rapid heartbeat. Hospitals have reported methemoglobinemia—a blood disorder where the chemical disrupts oxygen flow through the body. The medical record tells us how just a brief slip-up can harm. The U.S. National Library of Medicine has flagged this compound as a potential threat to human organs like the liver and kidneys after repeated or high-dose exposure. Long-term effects raise fears about cancer risks based on animal studies, even though human data still falls short.
Workplace Practices: What Doesn’t Work, What Might
In my experience, quick fixes don’t cut it with chemicals like this. Years ago, I saw folks at a dye production facility working with minimal gear—cloth masks, open windows, maybe a box fan. After a chemical leak, cleanup crews suffered rashes and wheezing. Management had ignored proper extraction hoods and chemical gloves. Simple steps could have spared those workers real harm.
Strict handling guidelines make a difference. High-quality respirators, chemical-resistant gloves, and full-body suits keep exposures lower. Fume hoods and good ventilation matter even more; you can’t trust an open window. Employers must train everyone on the dangers—posting charts and holding drills makes sure knowledge spreads. Employers also need to run regular air quality checks in spaces where the dust could build up.
Lasting Impact: Protecting Health, Protecting Work
Respect for this chemical’s dangers starts with company culture. Workers who feel comfortable talking about safety—not scared to speak up—spot problems faster. Medical surveillance, as required by OSHA and similar regulators, can catch early signs of harm before things get worse. Companies should support anyone reporting symptoms and address small spills or leaks with urgency, not delay. These steps don’t take high-tech solutions; they take commitment to people’s health.
The hazards around 2,6-Dichloroaniline remind us that shortcuts at the bench or the production line can lead to setbacks nobody wants. Prevention works best before anyone lands in the ER. That truth stays with anyone who’s seen chemical exposure up close.
What is the chemical formula and structure of 2,6-Dichloroaniline?
Chemical Formula and Structure
The chemical formula for 2,6-Dichloroaniline is C6H5Cl2N. The structure stands out with a benzene ring, two chlorine atoms sitting at the 2 and 6 positions, and an amino group attached at position 1. This arrangement isn’t just something you’d find on a chemistry quiz. It’s a practical tool, carving out its niche in dye-making and pharmaceutical labs. The pattern of these groups on the benzene ring creates a distinct line between it and other aniline derivatives. Even small changes on the ring force differences in reactivity and toxicity, which chemists pay attention to daily in the lab.
Why Structure Matters
Personal experience in the lab always circles back to how minor changes on a molecule’s skeleton can flip the entire story. 2,6-Dichloroaniline’s setup — chlorine atoms on either side of the amino group — shifts its electronic properties. These shifts influence how the molecule grabs onto other chemicals. In practice, it means 2,6-Dichloroaniline behaves a bit differently from 2,4-Dichloroaniline, even if formulas look crowded with the same letters.
These differences count. Chemical manufacturers eye this molecule because chlorine atoms pull electrons, making the benzene ring less reactive towards some additions. You won’t find side reactions running wild as you would with plain aniline. For dye makers, this increases consistency. For anyone working in crop protection, tweaking the ring builds chemicals that stand up to breakdown in the wild — a trait with serious implications for both pest control and environmental safety.
Industrial and Real-World Applications
Through years of working alongside teams in chemical plants, you start to see the role 2,6-Dichloroaniline has carved out. The dye industry pulls from its stability, pushing for more reliable colors that don’t wash away or fade. Medicine leans on it, building blocks for everything from simple antiseptics to advanced drug compounds. Each time an engineer hunts for a new molecule to fight bacteria, the backbone of 2,6-Dichloroaniline often shows up somewhere on the drawing board.
Chlorinated anilines also turn up in the making of pesticides and herbicides. Chemists value the double chlorine protective shield — it slows the breakdown process, helping products stick around just long enough to do their job. But there’s a catch. Increased durability comes hand-in-hand with persistent compounds in soil and water. This drives the ongoing discussion around environmental safety and public health.
Balancing Use and Risk
Working with chlorinated aromatics, you’re taught early on not to brush off toxicity. Studies link dichloroanilines — particularly at high exposures — to risks for aquatic life and workers handling bulk quantities. Legacy incidents with persistent organic pollutants show what happens when industry ignores safe disposal and containment. It’s a lesson still in play. Regulatory checks now demand close monitoring during production, transport, and waste handling. Modern manufacturing designs improved filtration and neutralization steps, cutting accidental releases before they cause harm.
Alternatives have slowly come into play, as the hunt continues for active compounds with shorter afterlives in water and soil. Green chemistry encourages labs to design for breakdown, seeking functional molecules that leave fewer problems behind. Crafting these next-generation chemicals relies on deep knowledge of structure-activity relationships — something built from decades of hands-on work with molecules like 2,6-Dichloroaniline.
How should 2,6-Dichloroaniline be stored and handled?
The Nature of 2,6-Dichloroaniline
Working in a research lab, I’ve handled my share of hazardous chemicals, and 2,6-Dichloroaniline stands out among them. A chlorinated aromatic amine, it smells a bit like musty oil. If you’ve ever opened a long-forgotten cabinet in the chemical store, you know the scent. It’s used for dyes, pharmaceuticals, and agrochemical production. No surprise, then, that it finds its way into a wide range of workplaces.
Hazards Up Close
Exposure to this compound isn’t something to take lightly. Contact with skin or inhalation can irritate airways, eyes, and cause dizziness. Research from the National Institute for Occupational Safety and Health (NIOSH) flags potential risks of methemoglobinemia — a condition where the blood loses its ability to carry oxygen efficiently. It’s nasty, and I’ve seen firsthand how quickly a simple splash or whiff can turn a regular day into a dash for medical attention.
Long-term exposure may have links to more severe health outcomes. The International Agency for Research on Cancer (IARC) categorizes some anilines as possible carcinogens. Personal protective equipment stops becoming optional and turns into a daily shield.
Storing 2,6-Dichloroaniline the Right Way
Chemicals like this one demand respect and discipline. A locked, well-ventilated chemical storeroom works best. I always make sure bottles stay in tightly sealed glass containers. Metal or plastic can sometimes react, leading to leaks or contamination. Keeping it cool, out of direct sunlight, and away from heat sources prevents unexpected degradation. No one wants a puff of hazardous vapor because of a hot summer day.
Shelf segregation matters. I have seen people cram incompatible chemicals on a single shelf. That’s a recipe for disaster. 2,6-Dichloroaniline pairs especially badly with oxidizers and acids. One careless spill or leak, and you could end up with toxic gases or even fire. Labels have to be clear, not just for regulations, but for the sake of the next person who opens that door. There’s nothing worse than fumbling to read a faded label right before handling a dangerous compound.
Practical Handling Tips
I always wear gloves—nitrile, not latex—along with lab coats and goggles. If I need to move or decant this chemical, it’s in a fume hood, no exceptions. Ventilation is everything. Chemical-resistant trays catch the inevitable drops, and spill kits sit within arm’s reach. Working alone is out of the question; a buddy system isn’t just for swimming pools. Eyes wash stations get tested weekly because you never know who will need them next.
Spilled even a small amount? Proper containment and cleanup become urgent. Activated charcoal or absorbent pads work well for puddles, but contaminated clothing and gloves head straight to hazardous waste, never regular trash. Waste bottles for organics have their own spot, and every drop gets logged. Regulations from OSHA (Occupational Safety and Health Administration) demand it, but it’s common-sense for anyone who values health.
Solving the Knowledge Gap
Lab safety doesn’t come from a single training session. I encourage everyone in my lab to treat safety data sheets as living documents, not just paperwork to sign. Regular drills, refreshers, and honest discussion about near-misses keep the risks front and center in people’s minds. Less-experienced staff can spot mistakes if given the chance to speak up.
Mistakes often stem from rushing or skipping steps. Building a culture where questions and double-checks are welcomed helps prevent accidents. Sharing what’s gone wrong—and what’s gone right—creates a safer workplace for everyone.
What is the purity and specification of the 2,6-Dichloroaniline product?
The Real-World Importance of Purity
2,6-Dichloroaniline often pops up in labs and in industry, especially for making dyes, pigments, and some pharmaceutical building blocks. A batch of this chemical is only as good as its purity. In my years talking with lab technicians and folks in manufacturing, the message is clear: slight impurities can throw entire processes off track. Color goes off, side reactions cost time and money, or health risks jump. That’s not just theory—it’s been seen plenty of times in dye works that ended up with product batches that didn’t pass QC or, worse, put workers at risk because of unexpected by-products.
Purity kicks off with a percentage, often 98% or higher for 2,6-Dichloroaniline in technical uses. This isn’t just a number to slap on a certificate. Sourcing raw material from a reliable supplier can mean the difference between a smooth production run and a full day spent troubleshooting. A recent analysis I read showed that products with just a half percent more impurity than spec caused dye reactions to stall. Sticking close to standard specs—purity over 99%—cuts down on headaches and lets chemists focus on the outcome, not patching problems.
Specification Basics: More than Just a Number
Beyond a simple purity figure, specs tell the whole story. Usually, buyers expect a white to slightly beige crystalline solid with a melting point in the 70°C to 74°C range and a real-world boiling point above 270°C. Water solubility clocks in low, but this fits the process needs. Appearance matters as well; visible specks or discoloration often cue underlying issues—residual metal or organic contaminants, which have their own downstream risks. Most suppliers list specific upper limits for things like iron content (usually below 10 ppm) or moisture (under 0.5%).
Lab test methods matter. Gas chromatography and high-performance liquid chromatography sort out small differences invisible to the eye. I once sat in with a QC crew where that extra decimal place on a GC reading flagged a batch destined to cause trouble. It’s these days, and the consistent tracking, that allow plants to meet product guarantees.
Safety and Storage—The Side of Specification That Gets Forgotten
Packing and storage wrap around purity. Even a top-notch batch can lose value if it’s exposed to the wrong environment. Keeping 2,6-Dichloroaniline sealed, shielded from light and moisture, means specs are in line with the certificate by the time it reaches the reactor. I’ve seen older stocks lose color and spike in impurity tests just from poor storage, showing that attention to these details holds value right up to the moment the drum’s opened.
Ways Forward—How to Keep the Bar High
Tight process controls, trusted suppliers, and honest spec sheets make a difference. Auditing suppliers, checking for full traceability, and running incoming quality checks catch surprises early. Over the years, companies that invest in lab staff and updated testing gear spend less time patching mistakes and more time building new molecules. Reporting issues and feedback to suppliers keeps the bar moving up, not just for one plant but for the industry as a whole. That sort of continuous checking, paired with tough-but-fair specifications, builds real confidence on both sides of the supply chain.
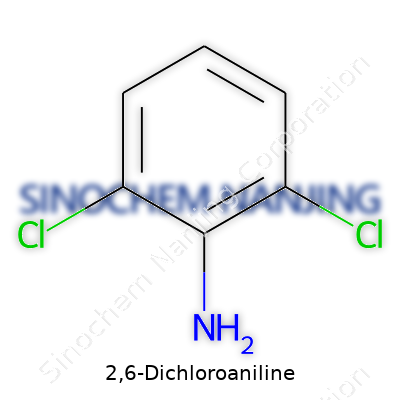
| Names | |
| Preferred IUPAC name | 2,6-dichlorobenzenamine |
| Other names |
2,6-Dichlorobenzenamine 2,6-Dichloro-1-aminobenzene 2,6-Dichloroanilin |
| Pronunciation | /ˌtuː.sɪksˌdaɪˌklɔːrəʊ.əˈnɪliːn/ |
| Identifiers | |
| CAS Number | 87-65-0 |
| Beilstein Reference | 1209224 |
| ChEBI | CHEBI:28312 |
| ChEMBL | CHEMBL15218 |
| ChemSpider | 8478 |
| DrugBank | DB08357 |
| ECHA InfoCard | 03c4009d-cc2c-41d5-a74b-94449c624183 |
| EC Number | 202-479-6 |
| Gmelin Reference | 68276 |
| KEGG | C06561 |
| MeSH | D016954 |
| PubChem CID | 6922 |
| RTECS number | BX8575000 |
| UNII | 6R3859I68F |
| UN number | 2811 |
| Properties | |
| Chemical formula | C6H5Cl2N |
| Molar mass | 162.05 g/mol |
| Appearance | White to grayish crystalline solid |
| Odor | Aromatic odor |
| Density | 1.41 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 2.85 |
| Vapor pressure | 0.02 mmHg (25°C) |
| Acidity (pKa) | 2.56 |
| Basicity (pKb) | 7.34 |
| Magnetic susceptibility (χ) | -71.2×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.611 |
| Viscosity | 2.4 mPa·s (25 °C) |
| Dipole moment | 2.95 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 146.3 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -37.0 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -5737 kJ/mol |
| Hazards | |
| Main hazards | Harmful if swallowed, toxic in contact with skin, toxic if inhaled, causes skin irritation, causes serious eye irritation, may cause an allergic skin reaction. |
| GHS labelling | GHS02, GHS07, GHS08 |
| Pictograms | GHS06,GHS09 |
| Signal word | Warning |
| Hazard statements | H302, H312, H315, H319, H332, H335, H410 |
| Precautionary statements | P261, P264, P270, P272, P273, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P312, P330, P337+P313, P362+P364, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 2,2,0 |
| Flash point | 158 °C |
| Autoignition temperature | 425°C |
| Lethal dose or concentration | LD₅₀ (oral, rat): 995 mg/kg |
| LD50 (median dose) | LD50 (median dose): 1770 mg/kg (oral, rat) |
| NIOSH | PS0350000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.1 mg/m3 |
| IDLH (Immediate danger) | IDLH: 100 mg/m3 |
| Related compounds | |
| Related compounds |
2,4-Dichloroaniline 2,5-Dichloroaniline 3,4-Dichloroaniline 4-Chloroaniline 2-Chloroaniline Aniline 2,6-Dichlorotoluene 2,6-Dichlorophenol |

