2,5-Dimethyl-2,4-Hexadiene: A Commentary on Progress, Value, and Future Paths
Historical Development
Chemistry evolves by building on the discoveries of those who chase patterns. Take the history of 2,5-Dimethyl-2,4-hexadiene. This unsaturated hydrocarbon attracted attention around the mid-20th century as researchers dug deep into the behaviors of aliphatic dienes. It came out of studies where chemists tried to unlock the structures and synthesis routes of materials with two double bonds, eager for molecules that could stretch the envelope in plastics and coatings. Practical development picked up when industrial challenges asked for building blocks flexible enough to take on chemical modifications. Through lab notebooks and patents, the substance wove itself into research dedicated to plastics, resins, and synthetic rubbers. Rather than being a star on its own, it serves as an important link in the long chain of organic chemical development.
Product Overview: Purpose Drives Innovation
2,5-Dimethyl-2,4-hexadiene does not occupy the limelight, but the people who work in chemical manufacturing know it well. This diene lands on the shelves as a clear liquid, usually stored in amber bottles, often under nitrogen to keep out oxygen that could set off unwanted changes. Chemists often see value in its reactivity and its ability to contribute to tailored synthetic pathways. Whether the end goal is a specialty resin or a neat trick in organometallic catalysis, the substance earns its keep by being a pivot point in reactions asking for double bonds spaced apart. Small differences in molecular structure add up to big differences in reactivity, and this makes the compound a bit of a workhorse in the hands of someone who knows how to coax it along.
Physical & Chemical Properties: More Than Just Numbers
In the lab, numbers might describe it as a colorless liquid with a boiling point that can cross above 130 degrees Celsius and a density hugging close to 0.7-0.8 g/mL. But those numbers translate into behavior: it evaporates quickly if left open, smells faintly reminiscent of other small dienes, and resists dissolving in water. Its two double bonds, separated by a single carbon, lay the groundwork for reactions from simple additions to more nuanced Diels-Alder syntheses. With low polarity, it mixes well with a range of solvents, from alcohols to ethers. Sometimes gloves and eye protection seem like a hassle, but contact with skin burns and irritation can creep up fast, urged on by its tendency to interact with organic tissues. People who treat it casually usually only do so once.
Technical Specifications & Labeling: What Matters Most
Specialists in production and quality assurance measure purity, water content, and color—metrics that make or break a batch for downstream chemistry. It usually arrives labeled with its CAS number and hazard pictograms. The details matter: if the hexadiene contains anything other than trace impurities, costly side reactions can pop up, undercutting the whole process. Specs come from established test methods like GC or NMR. Labels often remind users not just about flammability, but about the strict limits on temperature and light exposure. Reading the fine print pays off—safety, efficiency, and repeatability hang in the balance.
Preparation Method: Getting the Goods
The approach most labs take for making 2,5-dimethyl-2,4-hexadiene is as unglamorous as chemistry gets. Chemists rely on dehydration and dehydrohalogenation steps, starting from a suitable hexane derivative. Getting high yields without dragging over too many side products requires either careful distillation or the right setup for azeotropic removal of water. Straightforward, yet easy to mess up, as anyone who’s fussed with sensitive distillations knows. Purification ranks as one of the thorniest issues because colorless does not always mean pure. Someone who’s been burned by an off-color batch never rushes this stage again.
Chemical Reactions & Modifications: Stretching Its Potential
The double bonds offer up all sorts of possibilities. In synthesis, this diene stands out for its willingness to take part in Diels-Alder reactions, letting chemists snap together rings or graft on new functions. Hydrogenation brings the saturated product; halogenation gives dichloro derivatives; addition reactions let it lock up other elements across those double bonds. More than a few patent applications center around using it to introduce branching into polymers or custom tailoring curable materials. People who appreciate creative freedom in molecular design know these features open doors if you have the patience to optimize conditions. A little insight, a little trial and error, and suddenly that colorless liquid shapes product innovation from coatings to adhesives.
Synonyms & Product Names: Navigating the Maze
Ask for 2,5-dimethyl-2,4-hexadiene and you’ll sometimes hear other names tossed around: "2,5-dimethylhexadiene" or "diisopropyl ethylene." Not everyone sticks with the full IUPAC label, but experienced scientists keep a close eye to avoid confusion. Synonyms sometimes trip up even seasoned staff unless the labeling or documentation stays sharp. Minor naming slip-ups have delayed reactions or mismatched orders in more than one lab. It helps to cross-check against the CAS number or structural formula to stay on track.
Safety & Operational Standards: More Than Just Paperwork
Chemicals like this demand respect. Flammability, eye irritation, and documented toxicity make safety glasses, gloves, and fume hoods non-negotiable. Local fire codes and OSHA standards call for explosion-proof storage and regulated waste disposal. Complacency—forgetting to check a vent line or skipping a step in the disposal log—invites trouble fast. Emergency plans matter because small mistakes compound quickly when volatile organics catch a spark or leak. Everyone who has survived a lab spill or near miss learns to follow these standards not just for formality but because no product justifies an ER visit. Shortcuts in handling almost always cost more than they save.
Application Area: Where the Chemistry Gets Real
People drawn to this diene usually want a reactive intermediate for polymer manufacturing, agrochemical design, or electronic materials. In my time handling resins, the compound stood at the intersection of strength, flexibility, and cost. Its use in specialty adhesives shows up wherever manufacturers need a material that holds under stress but won’t break the bank. It takes its place in formulations for synthetic rubbers, letting engineers craft products with set mechanical properties. Electronic industries check out its features for specialty resins and encapsulants, where electrical and chemical stability matter. The truth is, the invisible labor of intermediates like this creates the foundation for bigger innovations—the kind most people never see but benefit from every day.
Research & Development: The Quiet Pursuit of Better Paths
There’s always someone tweaking the formula, searching for better yields, greener synthesis paths, or novel applications. Research that champions sustainability puts energy into catalytic routes using less toxic feedstocks or renewable starting materials. New discoveries in catalyst design or alternative solvents might soon sidestep some of the hazards or improve atom economy. Teams working on next-gen coatings explore copolymerization techniques, hoping to unlock properties traditional materials can’t deliver. Experience tells us that progress in these areas relies on collaboration—engineers, chemists, and safety specialists working in sync, all driven by genuine curiosity as well as the unmet needs of the industries they support.
Toxicity Research: Lessons Still Unfolding
Evidence from animal models and workplace monitoring signals risks from inhalation or skin contact. Eye and respiratory irritation pop up at low concentrations, while high exposures tie into liver and neurological effects. The gaps in long-term data worry some researchers—there’s reason to keep pushing for broader toxicity screening. Better evaluation methods and epidemiological tracking could catch issues early rather than letting risks slip through the cracks. We owe it to students, operators, and communities to keep the focus on transparency and public health, not just on short-term cost savings or simplified labeling.
Future Prospects: Opportunity in the Details
Looking ahead, this humble diene finds itself in a changing field. Industry trends push for cleaner, safer, and more efficient synthesis routes. Companies with robust safety cultures and agile R&D teams may harness its potential for new polymer structures or advanced electronic materials. Demand grows for specialty intermediates that align with stricter environmental rules. I see opportunity in developing continuous-flow syntheses, integrated recycling, or biodegradable derivatives—areas where innovation brings ethical progress as well as economic return. Listening to the lessons from decades of use, and giving as much weight to environmental and health data as to performance specs, stands as the surest way to ensure this chemical keeps adding value rather than cost or risk.
What is 2,5-Dimethyl-2,4-Hexadiene used for?
Chemical Middleman in Synthesis
2,5-Dimethyl-2,4-hexadiene doesn’t grab attention with a flashy name, but it plays a key role as a raw material in chemical manufacturing. Its double bonds give it life as an intermediate, a foundation for making other chemicals that show up in things like polymers, flavors, agricultural products, or specialty coatings. Those six carbons strung together with two methyl groups and two double bonds bring both reactivity and selectivity, which chemists count on when building more complex compounds. In industrial work, one ingredient flows into another, and hexadiene stands out for the way it links up in various reactions. The presence of conjugated double bonds helps generate cyclic compounds or serve in Diels-Alder reactions. That’s at the roots of what we use in modern plastics and synthetic rubbers—think of everything from garden hoses to sneaker soles.
Building Aromatic Scents and Flavors
Hexadiene doesn’t just belong in big factories or with white lab coats. Break down the molecules, and you find the backbone of fragrances or flavors sneaking into daily life. Some derivatives that start with this diene end up in artificial flavorings used in candy, soft drinks, or processed snacks. Fruit notes and earthy undertones owe a lot to these starting compounds. The people working in the flavor and fragrance sector lean on reliable building blocks. Not every molecule carries a pleasant scent straight out of a beaker, but tweaking and refining lets flavorists and perfumers create something consumers find familiar yet unique.
Anchor Compound in Material Science
Flexible plastics, engineered resins, or adhesives often spring from the work that begins with relatively basic chemicals. 2,5-Dimethyl-2,4-hexadiene acts as a type of scaffolding. For instance, in certain specialty rubbers, this diene’s configuration enables cross-linking—giving the right stretch, bounce, or resistance to wear. It also shows up in research on new polymers for electronics or lightweight components. Every time a researcher runs a test on new coatings or resins, foundational chemicals like this one get their moment. Success stories in product development, from water-resistant jackets to reliable electrical insulation, can trace their origins to thoughtful use of dienes with particular side groups. Even very specialized uses, such as prototype OLED displays or improved solar panel coatings, tie back to these early ingredients.
Safety and Environmental Responsibility
Any chemical with strong double bonds brings promise but also responsibility. Volatile organic chemicals like 2,5-dimethyl-2,4-hexadiene require careful handling. Exposure can irritate skin and mucous membranes, which means proper ventilation, gloves, and eye protection must be standard. Regulators keep a steady eye on such substances—companies handling them follow reporting rules and best practices to avoid accidents or pollution. Waste streams and emissions bring another challenge. Projects in green chemistry aim to swap out fossil-based dienes for ones sourced from renewables, and to minimize the environmental footprint of leftover chemicals. Old stories in manufacturing speak of spills or exposure, but today’s industry works on better containment, training, and monitoring.
Pushing for Safer, Greener Chemistry
Professional pride comes from getting more output with fewer problems. Engineers and chemists innovate in recycling catalysts, tweaking reactivity to lower side products, and developing alternatives from bio-based feedstocks wherever possible. Consumer demand for sustainable choices sparks more research into methods that reduce hazards and waste. Tighter collaboration between industry, academia, and regulators gives everyone more confidence in safety measures and environmental protections, setting the stage for safer and smarter uses of hexadiene derivatives across supply chains.
What is the chemical formula and structure of 2,5-Dimethyl-2,4-Hexadiene?
Chemical Identity and Formula
2,5-Dimethyl-2,4-hexadiene belongs to a group of organic compounds known as dienes, which feature two double bonds. The formula for this molecule is C8H14. In terms of structure, the backbone shows six carbons with two double bonds and two methyl groups hanging off the second and fifth carbons. This gives the full name some real meaning—hexadiene because of the six carbons and two double bonds, and the dimethyl part signals the side groups.
Peeling Back the Structure
Working through the structure, you notice it unfolds as CH3–C(CH3)=CH–CH=CH–CH3. Looking closer, carbon two carries both a hydrogen and a methyl group, and the double bond runs from carbon two to carbon three. Another double bond connects carbon four to carbon five. At carbon five, a methyl branch sticks out as well. Organic chemists draw this configuration out in zig-zag fashion, making sure to highlight those double bonds at the right spots, as they define much of how the molecule behaves.
Why Chemical Structure Matters in Daily Life
From a practical perspective, knowing the structure of a molecule like this helps predict reactivity. Double bonds signal spots where chemical change starts. I’ve seen plenty of reactions where having those methyl groups nearby shifts things—sometimes stabilizing a reaction, sometimes pushing things in a new direction. In fuel chemistry, similar molecules take part in making components for gasoline. The shape of the molecule changes properties like boiling point, which matters a ton if you’re refining fuels.
Talking Safety and Handling
Having spent time in small research labs, I know that many hydrocarbons with double bonds can be unstable or even form peroxides over time. This impacts storage and transport. Safety data always points to keeping these compounds in tightly sealed, light-blocking containers. Ventilation in work spaces stays essential too, as breathing in vapors from hydrocarbons can cause headaches and nausea. It’s a detail that gets missed until someone has to air out the lab. Protective gloves and goggles aren’t just for show when you’re handling unsaturated organics.
Environmental and Health Angles
As air quality standards tighten, awareness about VOCs—volatile organic compounds—rises. Dienes like 2,5-dimethyl-2,4-hexadiene count among these. They contribute to smog formation and react with other chemicals under sunlight. Back when I did fieldwork near industrial zones, we tracked emissions that included a mix of hydrocarbons. Even at low concentrations, persistent exposure can harm lung health and aggravate respiratory issues. Stricter regulations on emissions may help reduce risk to workers and neighbors.
Steps Toward Safe Use and Future Research
People in the industry have started relying on better monitoring equipment and stricter protocols for storing and moving organic compounds. Research into green chemistry hopes to replace more hazardous molecules with safer alternatives. In academia, labs look for catalysts that work under milder conditions to minimize energy costs and waste. Working with compounds like 2,5-dimethyl-2,4-hexadiene becomes less daunting if there’s solid data and transparency about risks. Outreach to students and new lab members makes a big difference in setting good habits early on.
How should 2,5-Dimethyl-2,4-Hexadiene be stored and handled?
What Makes This Chemical Demanding Extra Care?
Experience in any lab or industrial setting shows that certain chemicals turn an ordinary day into a lesson on respect for safety. 2,5-Dimethyl-2,4-Hexadiene definitely lands on that list. As an unsaturated hydrocarbon, it brings a high degree of flammability to the table. Fumes from liquids like these carry genuine risk, not just in the lab but anywhere careless storage creeps in. Stories from maintenance workers and process operators—people with the smell still in their clothes long after work—remind us these substances call for real diligence.
Storage That Works—And Why Some Choices Don’t
Metal drums with tight seals last because vapors find their way out of plastic more quickly, threatening both people and workspaces. Shelving chemicals like this next to open flames or even warm lights creates accidents waiting to happen. Cold, well-ventilated rooms give far fewer headaches. It’s not about locking things away out of sight, but about controlling air exchange and keeping the temperature down. Anyone who’s grabbed a warm solvent jug by mistake remembers the panic and rush to the eyewash.
Manufacturers suggest designated flame-proof storage lockers, typically with automatic enclosures. Keeping diene containers off the floor means less chance of punctures from forklift traffic, and never stacking containers too high keeps everybody’s toes and backs safer. Storing compounds like this, even just one shelf away from oxidizers or acids, opens the door for combinations that send safety managers running.
Personal Protection—Protection That Actually Works
A textbook might say gloves and goggles are enough, but bitter experience tells a different story. Nitrile gloves usually outperform latex when dealing with hydrocarbons. Sealed goggles beat open-sided glasses. In places where spills or splashes happen, a full-face shield becomes standard gear. Even a small skin exposure can mean discomfort, sometimes burns, so keeping sleeves down and hands protected matters for real.
Chemical fume hoods play a crucial role. At most plants and research labs, the sight of motorized sash windows and the hum of fans means someone takes air quality and vapor control seriously. Opening a drum or transferring a liquid with direct ventilation means everyone breathes easier—literally. In smaller setups, exhaust fans mounted near storage spots make a noticeable difference, especially if used every time drums get opened.
Dealing with Spills—Hard Lessons Learned
Mistakes happen—even seasoned technicians run into valves that stick or seals that crack. The right absorbents, like activated carbon or inert clay-based products, soak up these hydrocarbons fast. Open rags or paper towels invite disaster by evaporating vapors into the room. Disposal rules demand strict attention, so bundling up soaked material and labeling it clearly prevents confusion at waste pickup.
Regular drills, reminders on what to do, and honest discussions around recent mistakes keep habits sharp. Nobody likes donning a respirator, but knowing it’s at arm’s reach—tested and working—can save a life or save a project from catastrophic contamination. At sites that handle volatile liquids, nobody’s thankful for shortcuts or dried-up safety protocols.
Pushing Toward Better Practices
People who’ve worked with diene compounds for a decade or more agree: Written procedures hold power only if everyone cares enough to follow them each day. Digital inventory systems flag stock levels, trigger alerts before the expiry dates, and track who used what. This practical tracking cuts down on accidents caused by forgotten containers or mystery spills. The expectation grows that each team member not only follows but improves the protocols, sharing real-world tips on what’s worked. That willingness to adapt stands out as the trait that keeps chemicals like 2,5-Dimethyl-2,4-Hexadiene from turning workdays into emergencies.
What are the safety precautions and hazards associated with 2,5-Dimethyl-2,4-Hexadiene?
What’s the Deal with 2,5-Dimethyl-2,4-Hexadiene?
There’s always some buzz around chemicals like 2,5-Dimethyl-2,4-Hexadiene, and for good reason. It pops up in research labs and some niche manufacturing settings, thanks to its double bonds making it useful for organic synthesis and specialty projects. But this isn’t your everyday household cleaner. This stuff demands respect if you’re working with it, and not just because of a complicated name on the label.
The Hazards on the Table
Several red flags come up with 2,5-Dimethyl-2,4-Hexadiene. Breathing in its vapors doesn’t do your lungs any favors. You might notice irritation in your nose and throat or get headaches if you hang around it without protection. Getting some of it on your skin will probably cause redness or itching, a sign the surface barrier we take for granted needs some backup. Eyes? Even a little exposure can have you wishing you’d doubled up on safety glasses.
This chemical catches fire easily, so a spark or static discharge can kick off a lot of trouble. Liquid running across a bench isn’t just a cleanup hassle—it turns into flammable vapor quickly, and that vapor hangs in the air, looking for an ignition source. In some cases, these kinds of chemicals form peroxides, which turn stable liquids into time bombs, especially in closed containers left on the back of a shelf.
Precautions that Actually Work
Every seasoned chemist I know has stories about the close calls, all because they underestimated just how sneaky volatile liquids can be. Good airflow keeps those vapors from building up, so opening a window in a stuffy lab or, better yet, working under a fume hood gives far better protection. Nitrile gloves and goggles matter—no one wants skin rash or gritty eyes.
Lab coats aren’t just for looking like you know what you’re doing. They put a layer between your skin and unexpected splashes. After years around reactive chemicals, I’ve learned to go for closed shoes, even for a trip across the lab. There’s always a dropped beaker waiting to leak something toxic onto an unsuspecting foot.
Keeping sources of heat and spark away becomes automatic. I once saw a static discharge from a synthetic shirt light up chemicals left uncapped, a reminder to ground yourself and the containers. Storing 2,5-Dimethyl-2,4-Hexadiene in well-sealed containers, kept out of sunlight and far from oxidizers, keeps accidents to a minimum. Old bottles, especially those left sitting for months, deserve a careful look before opening to check for peroxide crystals or odd smells.
Why This Matters
It only takes one incident for a workplace to shift from normal to emergency mode. I remember a spill in a university lab that sent three people to urgent care—just a small bottle tipping over. That pushed the team to tighten up training and enforcement, not because of pressure from the top, but because no project is worth risking health or safety.
Regulators, including OSHA and the EPA, require clear labeling and up-to-date Material Safety Data Sheets, based on real cases and lab mishaps. Training shouldn’t be an afterthought. Making sure everyone in the room knows what that label actually means often makes the difference between a minor mistake and a call to the fire department.
Real Solutions for Staying Out of Trouble
Double-checking safety gear before a project, storing chemicals properly, and keeping clear, honest communication isn’t just bureaucracy. It builds a culture where people look out for each other. Investing in fume hoods, regular training, and proper waste disposal costs time and money, but that’s nothing compared to the fallout from ignoring potential hazards.
The bottom line: 2,5-Dimethyl-2,4-Hexadiene brings opportunity and risk. Respect it, gear up, and keep the environment controlled—your lungs, skin, and lab mates will thank you. That sense of responsibility sticks with you, long after you finish the reaction and turn off the lights.
Is 2,5-Dimethyl-2,4-Hexadiene available in different purity grades or packaging sizes?
Why Purity and Size Actually Matter
I still remember my early days in a university chemistry lab—chasing after pure reagents, always watching budgets, learning the costly difference that just a percent of impurity could make. In practice, whether you’re running a high-stakes synthesis for pharmaceutical development or preparing teaching experiments for undergraduates, the purity of 2,5-Dimethyl-2,4-Hexadiene shapes both results and safety.
Most chemical suppliers sell this compound in several purity grades. Research-grade often clocks in at 97% to 99% purity. Sometimes, technical-grade comes at a lower cost but with less rigorous filtration or distillation. For researchers working on sensitive syntheses—especially in organometallic or advanced polymer applications—access to higher grades reduces the noise in results and removes a layer of troubleshooting. If a single contaminant throws off a test, the next few hours or even days unravel what went wrong. Getting purity right the first time saves both money and patience.
Packing Real-World Orders
For a small lab, one-liter glass bottles or even 100-milliliter vials suffice. Large manufacturing sites lean towards bigger drums or metal cans, stretching into tens or hundreds of liters. It’s about practicality. Frequent small-scale synthesis and analysis become economical with modest packs that reduce waste and storage burdens. On the opposite end, large-scale production is all about cost per unit and supply chain peace of mind—you don’t want to run out in the middle of a process.
Different project sizes in my own experience taught this lesson well. For a semester-long undergraduate project, we barely dented a 250-milliliter bottle of 2,5-Dimethyl-2,4-Hexadiene, and storage requirements loomed in the department’s tiny locked cupboard. In another industry internship, stacks of five-liter cans were delivered on pallets; all the oversight needed to prevent spills or accidents grew as packaging scaled up.
Safety and Storage Concerns
Anyone storing large volumes faces extra hurdles. This compound can be flammable and reactive under certain conditions. Glass bottles work at a bench but become a liability if hit or dropped. Metal containers suit bulk buyers better, lowering the odds of breakage and easier labeling for inventory. Hazard communication and regulatory labels show up—even in simple storage—reminding users of what’s on the line.
Smaller packages might cost more per gram, but minimizing leftover hazardous material pays off. Larger buyers often form agreements with suppliers for not just regular deliveries, but also proper reclamation or disposal of unused product—important since environmental agencies monitor such transactions more closely every year. As an employee years ago, I watched my supervisor negotiate regular pickups for old stock; the savings in accident risk almost outweighed the cost.
The Usage Context Always Defines the Choice
Chemistry never fits a single template. Sometimes, a small but ultra-pure vial saves a research project. Other times, only shipping a drum makes sense for continuous manufacturing. As of 2024, online supplier catalogs and distributor chains make comparing options easier than ever. Reviews, certificates of analysis, and clear customer service channels all help users avoid unpleasant surprises.
Both new users and seasoned chemists benefit from understanding these details up front. Choosing the right purity and pack size for 2,5-Dimethyl-2,4-Hexadiene isn’t just a matter of paperwork—it’s about running a safer, more efficient, and more reliable laboratory or facility.
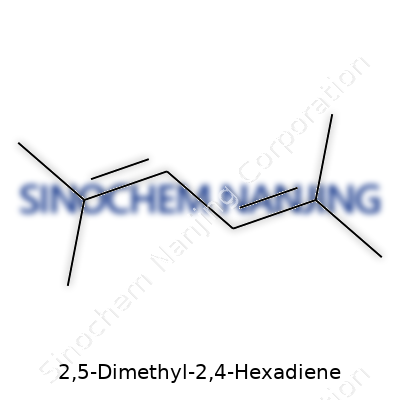
| Names | |
| Preferred IUPAC name | 2,5-dimethylhexa-2,4-diene |
| Other names |
2,5-Dimethyl-2,4-hexadiene 2,5-Dimethylhexadiene NSC 86974 |
| Pronunciation | /tuː,faɪv-daɪˈmɛθəl-tuː,ˈfɔːr-hɛkˈsaɪˌdiːn/ |
| Identifiers | |
| CAS Number | 638-13-9 |
| Beilstein Reference | 635873 |
| ChEBI | CHEBI:53125 |
| ChEMBL | CHEMBL156044 |
| ChemSpider | 12072 |
| DrugBank | DB04243 |
| ECHA InfoCard | 03abf9e0-3b7b-428b-a31d-02fd5b3a8a59 |
| EC Number | 204-130-7 |
| Gmelin Reference | 97051 |
| KEGG | C09678 |
| MeSH | D003605 |
| PubChem CID | 11405 |
| RTECS number | IM4250000 |
| UNII | T8N4988QE3 |
| UN number | UN 2370 |
| Properties | |
| Chemical formula | C8H14 |
| Molar mass | 110.20 g/mol |
| Appearance | Colorless to yellow liquid |
| Odor | sweet odor |
| Density | 0.726 g/mL at 25 °C |
| Solubility in water | Insoluble |
| log P | 2.84 |
| Vapor pressure | 2.1 mmHg (25°C) |
| Acidity (pKa) | 16.7 |
| Magnetic susceptibility (χ) | -9.15 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.448 |
| Viscosity | 1.27 mPa·s (20°C) |
| Dipole moment | 0.44 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 389.4 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 85.4 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -3897.7 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02, GHS07 |
| Signal word | Danger |
| Hazard statements | H226, H315, H319, H335 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P280, P303+P361+P353, P304+P340, P305+P351+P338, P312, P337+P313, P370+P378, P403+P235, P501 |
| NFPA 704 (fire diamond) | 2-2-3- |
| Flash point | 33 °C (91 °F; 306 K) |
| Autoignition temperature | 220 °C |
| Explosive limits | Explosive limits: 1.2–9.7% |
| Lethal dose or concentration | LD50 (oral, rat): 3160 mg/kg |
| LD50 (median dose) | LD50 (median dose): 3160 mg/kg (rat, oral) |
| NIOSH | Not established |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for 2,5-Dimethyl-2,4-Hexadiene: Not established |
| REL (Recommended) | REL (Recommended): NIOSH-TWA 100 ppm (410 mg/m3) |
| IDLH (Immediate danger) | 500 ppm |
| Related compounds | |
| Related compounds |
2,4-Hexadiene 3,4-Dimethyl-2,4-hexadiene 2,3-Dimethyl-1,3-butadiene Isoprene 2,5-Dimethyl-1,5-hexadiene |

