Talking About 2,5-Dimethyl-1,5-Hexadiene: Where Science Meets Daily Life
Looking Back: A Quick Glance at the Roots
The story of 2,5-Dimethyl-1,5-hexadiene stretches back to the earlier days of organic chemistry, once curiosity shifted beyond the basics and researchers started taking a closer look at dienes—hydrocarbons with two double bonds. Those working in the early and mid-twentieth century saw compounds like this as a playground for ideas about synthetic routes, reaction mechanisms, and what you might call the architecture of molecules. As industrial chemistry moved past dyes and explosives, scientists realized that flexible molecules with conjugated or isolated double bonds could show some neat behavior, both in bulk reactions and under the microscope.
2,5-Dimethyl-1,5-hexadiene caught attention for more than just having a handle on the periodic table. Chemists sorting through variations on the theme of alkenes quickly learned that the way carbon atoms and their methyl branches arranged themselves changed how a molecule like this reacted—and even how it smelled or looked under normal conditions. It's not about discovering new elements, but about squeezing surprising properties from familiar building blocks. That simple interest in structure and outcome, more than any rush for the patent office, drove a lot of the basic work with this compound before it found a steady place in select applications.
Getting to Know 2,5-Dimethyl-1,5-Hexadiene
It helps to start with a clear picture. 2,5-Dimethyl-1,5-Hexadiene falls under the family of aliphatic dienes—six carbon atoms stretched into a chain, double bonds at the 1 and 5 spots, and methyl groups branching from the 2 and 5 carbons. That setup influences nearly everything it touches. By comparison, some relatives show different behavior, but the twin methyl groups give this molecule a degree of stability and bulk you won’t find everywhere.
As for appearance, you’re usually dealing with a colorless to yellowish liquid. It’s got a boiling point that gives you a fair margin for catch-and-hold types of experiments, sitting higher than a lot of light alkenes but nowhere near trouble for standard glassware. The scent lands with a hint of the “hydrocarbon” label—a reminder of its organic roots. Its solubility profile fits what most organic labs expect: it plays nicely with other nonpolar liquids and keeps its distance from polar solvents like water.
Labeling It Right: Technical Details and Common Synonyms
In the world of technical jargon, it’s best not to lose focus in the jungle of names. This molecule may show up as 2,5-dimethylhexadiene or sometimes just 2,5-DMH. Australian and European labs often use identical designations. Looking at the label, you’ll often see the molecular formula C8H14, which underscores the two methyl branches in a six-carbon backbone. On a shelf, safety panels flag it as flammable and recommend keeping it away from oxidizers. Its standard purity grades usually start at over 95%, targeting research or component blending instead of consumer use.
How to Make It: The Hands-On Approach
The synthesis of 2,5-Dimethyl-1,5-hexadiene mainly relies on dehydration reactions or controlled coupling of shorter alkene chains. In practice, most chemists rely on catalytic dehydrogenation of hexadecane derivatives, sometimes with acid catalysts stirred into the mix. The choice of route often comes down to availability, local regulations, and budget, along with minimizing side products. Large-scale producers usually use temperature-controlled, multi-step operations, since the target compound can escape as vapor or react with air without the right setups. Experienced bench chemists focus just as much on separation and purification as the reaction itself; distillation or chromatography often sorts the product from a soup of similar molecules.
Getting Down to Chemistry: Reactions and Tinkering
Double bonds in 2,5-Dimethyl-1,5-hexadiene open up a world of reactivity. The compound fits into several classic organic transformations. Addition reactions, such as halogenation, mark up the carbon backbone with heavier atoms and produce a library of spin-off chemicals. Cycloaddition stands out, especially Diels-Alder reactions, where this molecule can link up with dienophiles to give rings, which is hugely helpful for building complex structures in pharmaceutical or polymer research.
Controlled hydrogenation brings the molecule to a saturated hexane derivative. Under the right settings, the location of the methyl groups influences reaction speed and outcome far more than you might assume from textbooks. That reactivity also means researchers must pay attention to storage, avoiding not only heat and open flames but strong light and oxidizing agents.
Synonyms & Naming Customs
Besides the official handle, terms like 2,5-DMH and 1,5-hexadiene, 2,5-dimethyl- pop up in scientific articles or product catalogs. The central point is correctness; in regulatory filings, names trace back to IUPAC rules. In everyday communication, shortening or anglicizing the name often makes life easier for bench scientists and engineers juggling several compounds at once.
Why Safety and Smart Handling Matter
From my experience in organic labs, treating every minor hydrocarbon with care beats learning lessons the hard way. For 2,5-Dimethyl-1,5-hexadiene, fire risk is not some distant concern. Its vapor lights up under the right conditions, which means grounded containers and spark-free zones are not just theoretical best practices—they are hard requirements. Eye protection, gloves, and good ventilation block most everyday hazards. Spills call for attention to vapors and a watchful eye for sources of ignition.
On the regulatory side, chemical safety sheets walk users through dangers and recommend swift cleanup with absorbents and proper disposal. Experience tells me that a good lab culture sees these precautions not as obstacles but as tools for keeping the experimentation and discovery process healthy and on track.
Where Does It Go? Application Landscapes
Few people outside advanced labs or specialty plants bump into 2,5-Dimethyl-1,5-hexadiene. It lives a quiet life as a precursor in polymer science, where those double bonds allow it to take part in chain-building chemistry. If you’ve dealt with creating advanced plastics or films, you might bump up against specialty copolymers or block materials built from hexadiene-based units. Beyond that, this compound sometimes plays a supporting role in the assembly of fragrances or more intricate molecules meant for pharmaceutical trials or analysis.
Advanced materials research, including those working on organically modified ceramics, sometimes gives this compound a supporting role. In electronic and optoelectronic work, researchers have used this class of dienes to tweak physical properties like flexibility, response to light, or chemical adhesion, although it rarely features in consumer-facing products.
Where the Cutting Edge Sits: Research and Development
Academic labs have dug into 2,5-Dimethyl-1,5-hexadiene as a test case for examining reaction pathways and kinetics. That means a lot of graduate students have tried running the same reaction under different conditions, learning firsthand how real molecules react differently than the neat diagrams in a textbook. Its well-defined reactivity profile—stable enough for manipulation, lively enough for interesting chemistry—makes it a classic choice for method development. You also see new reaction types, including radical and catalysis-driven changes, using this diene as a benchmark compound.
Polymer scientists, especially those after new elastomers or specialty coatings, see the value in small tweaks to diene structures. Making reaction networks that balance flexibility, chemical resistance, and stretch comes down to the fine details of what molecules like this do in long chains. Industry might not reference it in marketing, but in technical journals, 2,5-Dimethyl-1,5-hexadiene still has a presence as a building block worth understanding and sometimes refining.
Watching for Hazards: Toxicity and Human Impact
The toxicity profile of 2,5-Dimethyl-1,5-hexadiene sits roughly with its hydrocarbon cousins. At high concentrations, vapors can irritate mucus membranes and respiratory tracts. Long-term studies have leaned on animal models and cell assays, and so far, there’s little sign of acute toxicity at the trace levels present during routine lab handling. Still, inhaling any hydrocarbon is a ticket to discomfort and possible long-term issues, so fume hoods aren’t optional luxuries.
Incidents involving this compound tend to come back to lax storage or spills, which speaks to the importance of teaching and enforcing good handling. Data on chronic exposure in formal studies is still patchy, so erring on the side of caution remains the norm, even for seasoned chemists.
Glancing Ahead: Future Prospects
Looking to tomorrow, the story of 2,5-Dimethyl-1,5-hexadiene doesn’t hinge on major breakthroughs so much as steady refinement. Improvements in green chemistry may yield ways to make and modify this molecule with lower waste and less environmental impact. Polymer specialists continue probing new uses, especially in fields where chemical stability and tailored reactivity matter for electronics or advanced materials. Synthetic routes could also change, turning away from hazardous reagents in favor of biosourced raw materials or simpler, more selective catalysts.
In most scenarios, this compound won’t replace common solvents or bulk polymers, but it could give future researchers a sharper tool for building molecules that need a precise arrangement of double bonds and methyl branches. Its ongoing role as a flexible test case for new methods stands as a reminder that everyday chemistry—done right and with care—still lays the foundation for progress in science and industry.
What is 2,5-Dimethyl-1,5-Hexadiene used for?
Diving Into Industrial Uses
Walking through the world of specialty chemicals opens your eyes to compounds you rarely hear in daily conversation. 2,5-Dimethyl-1,5-hexadiene fits right in. At first glance, the name might sound overwhelming. In reality, this little molecule has been making waves across manufacturing and research for decades. The structure, marked by its two double bonds and methyl groups, gives it a unique twist: high reactivity. Chemists go after it because it opens doors to creative synthesis work.
Getting Creative with Polymer Chemistry
One of the biggest playgrounds for this compound sits in polymer science. Chemists rely on 2,5-Dimethyl-1,5-hexadiene to whip up specialty plastics and rubber. These materials don’t end up in every toy or bottle, but they matter—especially for fine-tuned seals, gaskets, or hoses that need to endure serious stress without breaking down too fast. From my time working with a local plastics supplier, I saw teams blend this diene into custom elastomers. Car makers and aerospace designers chase performance, not just looks; getting materials that handle high temperatures and exposure matters to them. This diene gives the right flexibility and stability, making demanding jobs possible.
Sparking New Molecules in the Lab
Research labs treat 2,5-Dimethyl-1,5-hexadiene as a tool for building blocks. It serves as a starting point for more complex organic molecules. In the world of pharmaceuticals, even slightly different chemical backbones can unlock new potential. Scientists have taken this diene through cycloaddition reactions, forming rings and structures that feed into medicines, pesticides, and specialty materials. It’s not about making headlines, but the magic happens behind the scenes, one reaction at a time.
Improving Electronics and Performance Materials
The shift toward better electronics brought this compound into new territory. Additives based on similar dienes, including this one, have found homes in specialty coatings and adhesives for electronics. These coatings protect tiny circuits against moisture and environmental stress. Having worked with an electronics assembly group, I watched engineers select materials by their long-term reliability—no one wants a gadget failing after six months. Using high-grade dienes helps keep moisture out and wires stable.
Weighing Safety and Seeking Sustainable Options
Chemicals like 2,5-Dimethyl-1,5-hexadiene demand respect in the workplace. The sharp odor and volatility set off alarm bells for safe handling. The health impacts from long-term exposure concern both managers and workers. Regulations from OSHA and local agencies demand tight controls: ventilation, closed handling, and constant monitoring. In my experience, teams that invest in training and safety gear see fewer accidents and better retention.
Sustainability keeps nudging every industry. Making this diene often traces back to petrochemical processes, which link to emissions. There’s a push for greener routes; biobased alternatives could shape the future. Creative startups and established chemical giants alike have jumped into research, looking for catalysts that work at lower temperatures or feedstocks that spare the environment more harm.
Moving Forward with Purpose
Anyone who's spent time around manufacturing floors or in chemical labs sees the mosaic that each ingredient creates. 2,5-Dimethyl-1,5-hexadiene doesn’t show up in consumer advertising, but it smooths the path for innovation—whether in a car’s engine block, a medical device, or a rugged smartphone case. Balance between innovation, safety, and sustainability shapes every decision around its use. As regulations tighten and alternative technologies grow, the chemical world will keep evolving, but the core lesson remains: thoughtful sourcing and handling make all the difference.
What is the chemical formula of 2,5-Dimethyl-1,5-Hexadiene?
Everyday Impact of Chemical Formulas
People deal with chemicals almost every day, often without noticing. Simple products, from skin creams to fuels, come from a background of chemical reactions and molecular design. The formula for a compound might look like a set of numbers and letters, but getting it right matters for safe, effective use and further innovation. That accuracy connects with trust in products and scientific progress.
Breaking Down 2,5-Dimethyl-1,5-Hexadiene
Understanding the structure helps. 2,5-Dimethyl-1,5-hexadiene is an organic compound. The name tells a story. The “hexadiene” part points to a six-carbon chain with two double bonds. The numbers “1,5” show where those double bonds appear — between carbons 1 and 2, and carbons 5 and 6. “Dimethyl” lets us know there’s a methyl group attached to carbon 2, and another to carbon 5.
If you draw out the structure, you’d count six carbon atoms in the chain, with a CH3 (methyl group) on carbon 2 and the same group on carbon 5. This gives C8H14 as the chemical formula. The longest part of this process doesn’t sit with counting atoms, but with understanding how different attachments and multiple bonds change both the chemistry and use of the molecule.
Why Accuracy in Chemical Formulas Matters
People trust formulas to be accurate because even a simple error can mean big safety risks or failed experiments. In the past, I’ve worked with research groups that learned this the hard way. A single carbon skipped on a drawing sent people back to the start, costing time, money, and supplies. Students in classrooms feel this sting too. Chemical education relies on getting formulas right, so students can develop an intuition for structure and function.
Accurate formulas also shape regulation. Chemical safety guidelines, environmental monitoring, and industrial standards refer directly to these formulas. If there’s a hazard label on a drum or a data sheet, you want to know it matches what’s inside. The real-world impact shows up any time quality slips — think contaminated products, mislabeled containers, or missed warning signs.
Bridging the Gap Between Textbooks and the Real World
Once you start using chemicals to make new materials or medicines, a correct formula makes or breaks the project. I’ve seen researchers scramble when compounds didn’t react as planned, puzzled by a mistake in an initial structure. People with experience start to spot patterns — similar names mean similar risks, storage styles, or regulations. Textbook knowledge moves into the practical world and sets the ground for discovery and reliability.
Towards Better Chemical Communication
Two things support good outcomes: clear teaching and open sharing of verified information. Teachers use examples like 2,5-dimethyl-1,5-hexadiene to help young chemists see how naming and structure line up. Professionals consult reference materials, updated databases, and trusted tools that help avoid confusion.
For new learners or people working with chemicals, simple resources and diagrams help a lot. There’s a need for digital platforms that don't just spit out answers but explain reasoning, show pitfalls, and connect molecules to their broader uses. The right tool, or the right mentor, steers work in a safer, more creative direction.
Building Confidence in Chemistry
The formula for 2,5-dimethyl-1,5-hexadiene is C8H14. It may look like a basic fact, but its reliable use means safer labs, better products, and more trust in the science. Every time someone double-checks the formula, reads a chemical name closely, or confirms a molecular drawing, they raise the standard for their field and their community.
Is 2,5-Dimethyl-1,5-Hexadiene hazardous or toxic?
Understanding What’s on the Label
Ask anyone who has spent time with industrial chemicals about 2,5-dimethyl-1,5-hexadiene, and you’ll likely hear a familiar story. The name alone doesn’t say much for those outside a lab. For those dealing with chemical stocks, it’s just another bottle in a storeroom with a label carrying hazards. Yet working with it means facing a real need for clear safety information.
Diving into the Risks
The safety data sheet for 2,5-dimethyl-1,5-hexadiene doesn’t pull its punches. A flammable liquid, it can catch fire from open flames, sparks, even a static charge. The vapors spread easily in air, which means a small spill or leak could stretch well beyond where you notice the source. Having spent enough hours overseeing ventilation and fire safety, keeping containers sealed and away from hot surfaces turns into a matter of habit rather than an afterthought.
On the toxicity front, the biggest issue is inhalation and skin exposure. Many aliphatic hydrocarbons in this category don’t just make the nose wrinkle—they can cause dizziness, drowsiness, or headaches after just a short exposure. Take off your gloves for a moment or work in a cramped, unventilated space, the consequences follow quickly. Animal studies with similar diene compounds show effects on the nervous system with significant exposure. The irritation from breathing it or spilling it on your skin is real, not a theoretical problem.
Long-Term and Environmental Impact
2,5-dimethyl-1,5-hexadiene isn’t a household product, but the risk still carries over beyond the lab. Repeated exposure often leads to dermatitis, which lab workers spot by the stubborn rash that lingers longer than expected. There isn’t a long record of cancer risk with this specific molecule, but chemicals with reactive double bonds sometimes have mutagenic properties. Without well-published, large-scale human data, leaning on caution stays smart practice.
Another angle is what happens when it leaves the workplace. Volatile organic compounds don’t stick around; they evaporate quickly and add to local air pollution. Chemicals like this play into ground-level ozone formation. Any environmental release needs control, not just for the folks inside the building but people breathing the air outside.
Protecting Yourself and Others
If you ask most industrial hygienists, they’ll point straight to local exhaust ventilation and closed systems. Personal protection follows next—chemical-resistant gloves, safety goggles, and lab coats. Fire extinguishers rated for chemical fires belong in easy reach. Good storage means keeping containers in a cool spot, tightly closed, away from sources of ignition.
Staying up-to-date on safety training matters more here than with everyday solvents. Accidents rarely wait for a convenient time, so knowing chemical spill and fire procedures comes from real-life drills instead of reading about them after the fact. My experience with workplace emergencies taught me that taking shortcuts is never worth the risk.
Improving the Culture of Safety
Manufacturers also play a role. Providing clear and complete safety data, easy-to-read warning labels, and straightforward disposal guidance helps everyone down the line. Substitution sometimes offers the best answer—if a safer chemical can do the job, that’s always the better choice. Chemists and workers benefit from cutting exposure thanks to automation and process changes.
So, while 2,5-dimethyl-1,5-hexadiene isn’t the most notorious toxin on a lab shelf, it demands respect. Keeping a workplace safe depends on treating these hazards seriously and reacting long before a problem turns into a story on the evening news.
How should 2,5-Dimethyl-1,5-Hexadiene be stored and handled?
Understanding 2,5-Dimethyl-1,5-Hexadiene’s Hazards
2,5-Dimethyl-1,5-hexadiene doesn’t look much different from other hydrocarbons, but it brings its own set of risks to the table. It comes as a colorless liquid that can catch fire quickly, sometimes with just a stray spark or an open flame in the room. Anyone who’s spent time around chemical stockrooms knows how important it is to respect substances that seem innocuous—they have a habit of teaching you hard lessons if you let your guard down.
Storing the Chemical
Set aside a cool, well-ventilated space. Choose a storage spot far from sources of heat or ignition. This compound doesn’t like sunlight or high temperatures, since these can make it unstable. Keep containers tightly sealed, using ones that won’t react with organic solvents. Glass or certain specialty plastics hold up better than metal if the integrity of the seal is reliable.
Think about what sits nearby. Do not keep acids, oxidizers, or strong bases on the same shelf or cabinet. If one leaks, the reaction could get messy in a hurry—sometimes with toxic gases or even an explosion. Segregation saves months of trouble, both for health and regulatory paperwork.
Labeling and Awareness
Never assume someone else knows what’s in the bottle. Clear, chemical-specific labels including hazard symbols go a long way to avoid confusion and accidents. In my experience, labels wear out or peel off, so check them regularly. Keep an up-to-date inventory. This isn’t just good practice—health and safety inspectors expect it, and it helps head off small problems before they get bigger.
Safe Handling Routine
Personal protective equipment matters just as much as storage. Splashing this kind of hydrocarbon on skin or eyes leads to irritation or worse. Nitrile gloves do better than latex for organics. Goggles make sense, even if you think you’ll be careful—eyes are precious, and accidents happen fast. A lab coat or apron adds another layer of protection.
Work only in well-ventilated areas—chemical fume hoods come in handy. Breathing in the vapors may lead to headaches or dizziness. Don’t trust a regular desk fan to clear out the fumes; local exhaust systems pull potentially harmful air away from you much more efficiently.
Open containers only after triple-checking the space for ignition sources. No cell phone charging, no hot plates, no loose extension cords lying around. Static electricity sparks can and do ignite vapors, especially in dry winters.
Mitigating Spills and Emergencies
Plan ahead for spills. An absorbent material like activated carbon or vermiculite helps contain leaks. Always clean up as soon as evidence of a leak appears. Properly ventilate the area during cleanup, and dispose of contaminated materials in a fire-safe chemicals waste bin, not down the drain or normal trash.
Keep a fire extinguisher rated for chemical fires nearby. Water sometimes spreads hydrocarbons rather than putting out the flames, so dry chemical or CO2 types usually do the trick.
Fact-Based Recommendations
I’ve learned over the years that routine checks, clear training, and accessible emergency supplies keep both people and property safe—whether you handle a liter or just a few milliliters for an experiment. Constant vigilance trumps shortcuts every time. Proper storage, labeling, and handling of 2,5-dimethyl-1,5-hexadiene make both daily work and unexpected events a lot less stressful for everyone involved.
What are the physical properties of 2,5-Dimethyl-1,5-Hexadiene?
Getting to Know This Compound
Step into any laboratory where new polymers or specialty chemicals get designed, and you might spot a bottle labeled 2,5-Dimethyl-1,5-Hexadiene. On the surface, it looks like a clear liquid—almost like many other hydrocarbons. Pop open the container, though, and you’ll catch a distinctive scent common among diene compounds. It’s not just any everyday chemical. The structure packs a punch: two methyl groups branching off a long chain, plus double bonds sitting at the right places to invite plenty of chemical reactions.
Key Physical Properties Everyone Should Notice
2,5-Dimethyl-1,5-Hexadiene typically flows as a colorless liquid at room temperatures. If you’ve ever handled other open-chain hydrocarbons, you might expect the same—low viscosity, quick to spread, and not sticky at all. Its boiling point sits around 142–143°C, which means it doesn’t disappear into thin air during moderate heating but will start turning into a vapor if left near an open flame or heated beaker. Working with this material, I appreciate how it doesn’t linger in the air (unlike some heavier organics) but evaporates faster than you’d expect during transfers.
The density hovers near 0.75 g/cm³—which makes it lighter than water and most oils. It floats, which comes in handy during separations or when you want to avoid mixing layers in an reactor setup. Pouring it, you notice it runs quickly along glassware. This reminds me why extra care matters—knocking over a flask can lead to a quick spreading of this low-density liquid, making cleanup a challenge in a busy lab.
This compound’s refractive index sits at about 1.423, so if you’ve ever looked down a sample cell, it offers a bit more visual distortion than water. That matters during purity checks or analytical work. It won’t freeze easily in a regular freezer, since the melting point drops down around −81°C. That trait gives researchers room to experiment in colder conditions, or store samples in typical lab freezers without watching out for solidification.
Odor, Flammability, and Handling
No one should ignore the sharp smell that signals you’re working with an unsaturated hydrocarbon. Diene structures often bring a distinct aroma—recognizable and hard to forget once you’ve worked with a few. That’s more than a nuisance; it tips you off to volatility and potential hazards. Like other hydrocarbons of its class, 2,5-dimethyl-1,5-hexadiene lights up in the presence of an open flame. Its flash point drops below typical lab heating conditions, which means safety goggles and well-ventilated hoods become mandatory every time you pop a cap or start a reaction.
I remember trying to run a Grignard addition with this diene and learning quickly that splash shields and quick transfers prevent headaches. Spills threaten both personal safety and experiment integrity, since a volatile diene runs off surfaces fast. Chemical supply catalogs point out proper storage: tight seals, cool dark shelves, and—always—labels warning against open flames or static sparks.
Why Physical Properties Matter for Real Work
Every property mentioned above affects how scientists put this molecule to use. Process chemistry benefits from that moderate boiling point: most distillation columns separate out the diene easily without pushing equipment to extremes. The low freezing point means less downtime during winter shipping. Density helps during organic extractions, setting up clean phase separations.
Using real experience drives this home. If you ever try to scale up a reaction from grams to kilograms and miss a boiling point or density detail, you end up wasting time and resources. Safety, purity, reactor design—even something as simple as picking the right bottle—trace back to physical properties. This diene’s characteristics open creative possibilities for materials research and challenge anyone who overlooks them.
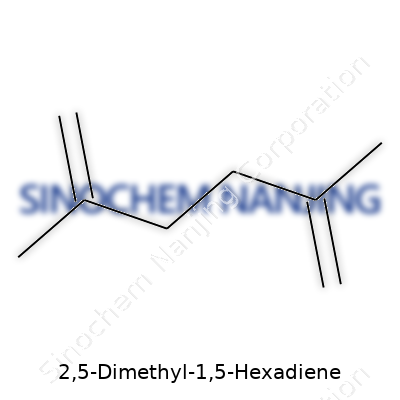
| Names | |
| Preferred IUPAC name | 4,7-Dimethylocta-1,6-diene |
| Other names |
2,5-Dimethylhexadiene 2,5-Dimethyl-1,5-hexadiene 1,5-Hexadiene, 2,5-dimethyl- 2,5-Dimethyl-1,5-hexadiene 2,5-dimethylhexadiene NSC 403640 |
| Pronunciation | /tuː,faɪ dʌɪˈmɛθəl wʌn,faɪ hɛkˈsaːˌdiːn/ |
| Identifiers | |
| CAS Number | 1420-68-8 |
| Beilstein Reference | 1207553 |
| ChEBI | CHEBI:140553 |
| ChEMBL | CHEMBL15360 |
| ChemSpider | 21106361 |
| DrugBank | DB14096 |
| ECHA InfoCard | 07a5b103-5a86-4dac-9955-cf6e9e834207 |
| EC Number | 203-851-2 |
| Gmelin Reference | 101206 |
| KEGG | C08295 |
| MeSH | D003602 |
| PubChem CID | 11566 |
| RTECS number | OI5950000 |
| UNII | A149MY85JS |
| UN number | UN2352 |
| Properties | |
| Chemical formula | C8H14 |
| Molar mass | 110.20 g/mol |
| Appearance | Colorless liquid |
| Odor | mild |
| Density | 0.737 g/mL |
| Solubility in water | Insoluble |
| log P | 2.97 |
| Vapor pressure | 1.6 mmHg (25°C) |
| Acidity (pKa) | 15.5 |
| Magnetic susceptibility (χ) | -0.000066 |
| Refractive index (nD) | 1.4480 |
| Viscosity | 1.68 mPa·s (20 °C) |
| Dipole moment | 0.13 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 390.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 77.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3687.9 kJ/mol |
| Pharmacology | |
| ATC code | |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02, GHS07 |
| Signal word | Warning |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P264, P271, P280, P302+P352, P304+P340, P312, P303+P361+P353, P305+P351+P338, P370+P378, P403+P235, P501 |
| NFPA 704 (fire diamond) | 2-3-0 |
| Flash point | 31 °C (closed cup) |
| Autoignition temperature | 220 °C |
| Explosive limits | 0.9–6.5% |
| Lethal dose or concentration | LD50 (oral, rat): 1670 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 > 10 g/kg |
| NIOSH | NA0480000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | No REL established |
| IDLH (Immediate danger) | IDLH: 500 ppm |
| Related compounds | |
| Related compounds |
1,5-Hexadiene 2,5-Dimethylhexane 2,5-Dimethyl-2,4-hexadiene 3,4-Dimethyl-1,5-hexadiene 2-Methyl-1,5-hexadiene |

