2,5-Diaminotoluene Sulfate: A Deep Dive into Its Past, Present, and Future
Historical Development
People have tinkered with aromatic amines for well over a century, finding early promise in synthetic dyes and pharmaceuticals. 2,5-Diaminotoluene Sulfate grew out of this era of experimentation, with advances piggybacking on breakthroughs in dye chemistry in the mid-1800s. Early chemists took what coal tar and aniline dyes offered and began to craft more complex molecules, trying to get better colors and more stable colorants. Through this journey, 2,5-diaminotoluene popped up as a reliable intermediate for the production of high-performance dyes and pigments. Over time, stricter regulations and improved synthesis methods led to purer forms and reduced contamination, making this compound safer and more predictable for industrial needs. It's striking how developments in industrial chemistry have shaped not just science but also fashion and consumer health, since hair coloring technology owes much to amines like this one.
Product Overview
2,5-Diaminotoluene Sulfate lands on the lab bench as an off-white or slightly violet crystalline powder. Not flashy, but manufacturers and researchers know this compound for its consistent performance in coloring processes, particularly in permanent hair dyes. In most routines, it comes mixed with a sulfate salt — stability matters, and the sulfate form resists air and moisture better than the free base. Its utility isn't limited to hair dyes, either. Some niche uses in polymer chemistry and organic synthesis highlight its adaptability. Behind these uses, there's a balance between reactivity and control. Scientists often prefer sulfate because the salt form reduces volatility and boosts shelf life, making transportation and storage easier in practice.
Physical & Chemical Properties
This compound carries the formula C7H10N2·H2SO4, with a molecular weight determined by which salt or polymorph is being handled. It's not particularly soluble in non-polar solvents, but dissolves readily in water or dilute acids. The crystalline habit looks ordinary under a microscope, but careful attention to its melting point — usually around 260°C (decomposition) — serves as a vital check for purity. As a diamine, it's reactive toward electrophiles, aromatics, and oxidants. Exposing it to strong sunlight or air leads to discoloration, a property that hair dye formulators must account for. It holds up to moderate heat in applied formulations, though excessive heat or acidification during processing brings out a strong amine odor and can trigger unwanted chemical side-reactions.
Technical Specifications & Labeling
Suppliers typically list 2,5-diaminotoluene sulfate by its purity, with higher grades reaching above 99%. Color, odor, and melting point support quick identification in most labs. Labels call out the CAS number 615-50-9 and include hazard pictograms under GHS labeling, reflecting the compound’s irritant properties and possible sensitizing effects. Handling recommendations hinge on reducing skin exposure and ensuring good ventilation. Researchers pay extra attention to potential contaminants like aniline or heavy metals, since those impurities shift toxicological profiles, especially in regulated industries.
Preparation Method
Chemists have stretched their creativity to tease out this molecule from toluene precursors. Traditional methods start with nitration of toluene to give 2,5-dinitrotoluene, followed by reduction using iron filings and hydrochloric acid or catalytic hydrogenation, and then transformation into the sulfate salt. Current approaches aim to minimize side products and ramp up yield. For folks concerned with green chemistry, less waste and improved selectivity represent major steps forward, since aromatic amines have a reputation for generating messy byproducts. Consistent batch-to-batch quality rests on controlled reaction conditions and efficient purification — easier said than done at scale.
Chemical Reactions & Modifications
The molecule’s two amino groups open up a wide range of functionalization routes. It's a strong nucleophile, handy for making azo dyes and for coupling reactions under mild conditions, particularly in the presence of diazonium salts. Researchers exploit these groups to attach diverse substituents and build complex dye molecules with improved brightness or fastness. Oxidative polymerization and coupling with phenols or naphthols further multiply its uses in pigment design. In my experience, tweaking either amine or methyl group shifts the end dye’s color and stability, highlighting the impact even minor modifications make on end-user performance.
Synonyms & Product Names
Anybody scouring technical literature comes across a stack of alternate names, including 2,5-toluenediamine sulfate, Toluene-2,5-diamine sulfate, and para-toluenediamine sulfate. In hair dye industry documents, this molecule sometimes appears as "PTD sulfate". Such varied naming often leads to confusion, especially across international suppliers and regulatory filings, but a quick CAS number check usually clears things up. Buyers and researchers have learned to cross-reference these aliases for clarity.
Safety & Operational Standards
Aromatic amines draw attention from safety experts for good reason. Skin sensitization and toxicity worries surround compounds like 2,5-diaminotoluene, particularly with repeated or prolonged exposure. EU regulations lay out concentration limits in finished products, pushing manufacturers to monitor residual amounts strictly and implement effective risk management. Workplaces handling this powder need solid ventilation, gloves, protective eyewear, and regular training. Fume hoods and spill kits aren't optional extras. Waste streams rich in aromatic amines face extra scrutiny before disposal or treatment. End users — especially in the cosmetics sector — depend on supply chain transparency, since product recalls or consumer lawsuits can follow lapses in safety.
Application Area
Cosmetics, especially permanent hair dyes, represent the epicenter of demand for 2,5-diaminotoluene sulfate. Formulators value the clean, enduring color this amine brings to the table, especially for dark brown and black shades requiring robust color-fastness. Side industries in coatings, plastics, and specialty pigments also draw on its chemical versatility. Though pharmaceuticals keep their distance due to toxicity considerations, the ability to build stable colorants with customizable hues keeps the compound in circulation among industrial chemists. In my time consulting for beauty brands, risk assessment surrounding this class of amines has shaped not just ingredient choice but also how companies train staff and address consumer concerns.
Research & Development
Academic and in-house R&D teams aren't standing still. Green chemistry researchers investigate ways to limit hazardous byproduct formation, reduce energy inputs, and find renewable feedstocks. Analytical chemists go after ultra-sensitive detection methods in finished goods and wastewater, using advanced chromatography and mass spectrometry for better trace analysis. Formulation scientists chase after combinations that stabilize color, block unwanted degradation, and limit sensitization potential in hair dyes. In recent years, advances in microencapsulation and slow-release technology look promising for lessening direct skin exposure. Competitors to 2,5-diaminotoluene sulfate occasionally emerge from natural colorant lines, but nothing yet has matched the cost and performance balance needed for large-scale dyeing.
Toxicity Research
Toxicologists have raised flags over aromatic diamines for decades, given links to allergic reactions and potential genotoxicity in laboratory settings. Regulatory bodies in Europe, the US, and elsewhere have scrutinized this compound for its role as both skin sensitizer and possible carcinogen under certain use conditions. Chronic exposure studies in animals show some reason for caution, particularly with respect to DNA damage and tumor formation at high doses. Safety assessors in cosmetics keep their work cut out for them, running in vitro and in vivo assays and tracking consumer reactions. Tight regulatory controls cap allowable concentrations, and ongoing research tries to pin down dose-response thresholds, metabolic pathways, and mechanisms for safer use. In the end, everyone from chemists to salon professionals shares a stake in understanding and communicating these risks honestly.
Future Prospects
New colorant technologies and ever-stricter environmental expectations put pressure on traditional amines like 2,5-diaminotoluene sulfate. Breakthroughs in bio-based dyes and non-aromatic alternatives could threaten its standing over the next decade. Still, its track record for performance, physical properties, and relatively low cost make it stubbornly persistent. Forward-looking companies invest in cleaner synthesis, improved exposure controls, and next-generation formulations to hedge against regulatory surprises. Consumers are demanding more ingredient transparency, spurring research into safer analogs and better risk communication. The future leans toward green chemistry and safety, but the transition won’t snap into place overnight. Those of us who have followed changes in dye chemistry appreciate the careful dance between innovation, safety, and performance.
What is 2,5-Diaminotoluene Sulfate used for?
The Color Behind the Bottle
Most people scan the label without thinking too much about the names they see. One of those names, 2,5-Diaminotoluene sulfate, often pops up in hair dye ingredients, especially the permanent or semi-permanent types. It surprises some that such a specific compound plays a part in the shades many choose at the salon or on the supermarket shelf. This chemical helps create the dark, rich colors that stick around through several washes—something most people expect and demand.
Why Hair Dye Formulas Depend on It
If you’ve dyed your hair, especially to a dark brown or black, you’ve probably used a product containing this compound. 2,5-Diaminotoluene sulfate reacts with hydrogen peroxide and other agents in the dye, forming molecules that lock into the hair fiber. Without it, shades would look faded after just a few shampoos. The ingredient helps cover grays and produces deep, intense tones. Many brands include it because high color performance makes or breaks their reputation. Negative online reviews spread fast, so producers look for ingredients that deliver what people want.
Safety Concerns and What We Know
Any chemical in contact with skin or hair long enough raises questions about health. Years ago, worries about hair dye safety led to several studies. The European Union, US Food and Drug Administration, and other watchdogs have looked closely at 2,5-Diaminotoluene sulfate. Research has shown this compound causes skin irritation and other health issues if not used properly. People with sensitive skin sometimes react with redness or itching. Regulations now set maximum allowed concentrations and demand clear warning labels on products with this substance. Salons and home users both need to follow these rules, especially when leaving dye on for longer periods.
Trust in consumer products means relying on decades of oversight and regular reevaluation. Regulatory groups require ongoing safety testing for ingredients like this, and companies must stick to strict guidelines for every batch made. Worst case, users can switch to alternatives or “natural” dyes, though these often show less reliable and shorter-lasting results.
Taking Small Steps for Safer Choices
Some people prefer to use gloves and apply barrier cream to protect their skin when coloring hair. Simple measures help lower risk. Reading the directions and patch-testing a product before coloring the whole head give early warning about allergies. For those with concerns about chemical exposure, looking at the label and picking brands that follow regional safety standards brings peace of mind. Those who seek to avoid 2,5-Diaminotoluene sulfate altogether have more options than ever, thanks to rising demand for plant-based recipes and advances in research.
What matters most is staying aware of what goes near your scalp. Many push for stronger oversight and better transparency on product labels. Trust comes from knowing how ingredients affect the body and having choices that don’t force a tradeoff between style and health.
Is 2,5-Diaminotoluene Sulfate safe to use in hair dyes?
Understanding the Chemical Behind the Color
People want effective and lasting hair color, so the industry uses chemicals like 2,5-Diaminotoluene Sulfate. It helps deliver deep, dark results. You might not notice it unless you carefully read the label on a box, but this ingredient turns up in many permanent and semi-permanent hair dyes. The fact that a substance works in coloring hair is one thing, but many wonder if using it on skin and scalp brings bigger risks.
Why People Worry About 2,5-Diaminotoluene Sulfate
For years, scientists and doctors have examined ingredients in cosmetic products because skin contact can mean those chemicals get absorbed. Reports link some aromatic amines—including 2,5-Diaminotoluene Sulfate—to allergic reactions, irritation, or in rare cases, toxicity at high exposures. Europe's scientific committees and the U.S. Food and Drug Administration both keep this ingredient under close watch.
Studies show most people do not develop allergic responses with average use, but there’s a bigger risk for anyone with sensitive skin, eczema, or a family history of allergies. Doctors sometimes see cases where repeated dyeing leads to redness or rash on the scalp or ears. These reactions force users to change products or stop dyeing entirely.
Regulation and Real-World Limits
In Europe and North America, authorities set tight rules for how much of this chemical companies can add to the dye mix. Manufacturers must respect strict concentration limits and provide clear safety warnings. In my years helping friends navigate the options at beauty stores, clear labeling helps plenty of shoppers decide if they want to take the risk. Still, not everyone checks or understands the fine print.
I’ve spoken with stylists who keep one eye on new regulations and another on client health. They often recommend patch tests—a small spot test behind the ear or on the wrist—before all-over application. Patch testing makes sense, since it is the best way to find out if skin will react badly without taking a major risk.
Looking at Safer Alternatives
More people have started talking about “clean beauty.” This movement pressures brands to find gentler alternatives. Plant-based dyes and products with a shorter ingredient list get popular, especially with consumers who value low-risk beauty routines. But these gentler options may not deliver the same long-lasting results or the same shade range, so people make trade-offs.
Navigating Hair Dye Choices
Some people weigh personal safety based on family allergy history, age, or how often they color. I’ve noticed some friends dye their hair less often, stretch out visits to the salon, or switch to partial treatments like root touch-ups. Others stick with familiar brands and do careful patch tests every time.
Dermatologists and toxicologists keep studying long-term safety, especially for frequent users. Those findings shape future rules and products. In the meantime, reading labels, doing patch tests, and talking with knowledgeable stylists help people make informed decisions.
Moving Forward with Awareness
Hair color boosts confidence and style, but the small print on the box deserves careful attention. 2,5-Diaminotoluene Sulfate continues to show up in many hair dyes, but paying attention and making thoughtful choices can help limit problems. Asking the right questions at the store or salon supports both health and self-expression.
What are the storage conditions for 2,5-Diaminotoluene Sulfate?
Why Care About How We Store This Chemical?
Working in laboratories and handling chemicals, I’ve seen how small details make a big difference. 2,5-Diaminotoluene sulfate doesn’t need a watchful eye because of hype—safe storage actually keeps workers out of harm’s way and prevents waste. This compound often shows up in dye manufacturing and personal care industries. Its impact on health and safety makes storage much more than a technicality.
What Happens If Things Go Wrong?
Improper storage has wrecked samples in our lab before—faded colors, degraded potency, sometimes even reactions we didn’t expect. For 2,5-diaminotoluene sulfate, storing it in humid, warm areas can trigger slow breakdown. Spoiled samples not only leave labs with unreliable results, but stray particles or vapors could affect anyone handling it. The risk of respiratory or skin irritation goes up fast, especially since the compound can release dust on handling.
Key Storage Rules From Experience
Keep it dry. Moisture runs the show when it comes to chemical spoilage. Humidity can set off slow chemical reactions, leading to lumps or hard clumps. Our storage room uses a dehumidifier all year round—not only to protect this compound but because moisture in the air is excellent at ruining chemicals in general.
Temperature matters, too. Room temperature—say, between 20 and 25°C—works for this substance, but keeping it away from sunlight or sources of heat is vital. Sunlight does more than just warm things up. It can actually change the chemical makeup of sensitive dyes, especially ones that companies put into hair coloring products.
Ventilation is everyone’s friend here. I learned the value of a well-ventilated storage space during college, after a sealed closet left chemical odors hanging in the air. With 2,5-diaminotoluene sulfate, even a low-odor sample needs airflow. A working fume hood or vent system means you're not risking slow exposure—people think about the hazards from a big spill but forget about long-term exposure from breathing airborne dust.
Containers play a hidden but important role. Only use sealed, airtight bottles—ideally those made of glass or thick plastic. I’ve watched poorly sealed bags get invaded by moisture and even airborne dust. Repackaging into tight, labeled containers meant fewer samples had to be tossed. Labels matter too: nobody wants to handle a mystery powder, especially in a shared lab.
Responsible Storage Means Less Waste
Many times, I’ve seen labs and small manufacturers buy excess because chemicals keep degrading before use. Tight storage controls meant we bought less, used more of what we had, and spent less money. Good practice saves cash—and for something with health impacts like 2,5-diaminotoluene sulfate, less mess means fewer accidents, which helps morale and keeps everyone safer.
Steps People Can Take
Review handling protocols with the team. Train new workers on hazards and storage requirements, not just with lectures but by walking them through the storage room and labeling procedures. Regular checks on temperature and humidity, plus logging which bottles get opened and closed, help catch issues before there’s real waste or risk.
Most of this comes down to consistency. Storing chemicals with care isn’t just about following rules from the safety data sheet. It’s about keeping staff healthy, reducing costs, and being mindful about waste. Secure storage for 2,5-diaminotoluene sulfate shows respect for the science, the business, and the people handling it.
What is the chemical formula of 2,5-Diaminotoluene Sulfate?
Understanding the Formula
2,5-Diaminotoluene sulfate shows up in the lab as a familiar friend for anyone working with dyes and pigments. Its chemical formula, C7H10N2·H2SO4, reveals a lot about its structure. Combining a core aromatic molecule, C7H10N2, with a single molecule of sulfuric acid, this compound forms a sulfate salt that's stable and workable. I’ve spent time in dye chemistry labs where knowing this exact makeup determined the outcome of many processes. A wrong formula often leads to failed experiments, wasted resources, and poor reproducibility.
Why Details Like This Shape Quality
In manufacturing, no one wants surprises in their raw materials. Even small shifts in composition turn production planning into a guessing game. 2,5-Diaminotoluene sulfate goes into hair dyes, printing inks, and even biological stains. An incorrect chemical formula means trouble: inconsistent color, compromised safety, and regulatory headaches. The chemical formula lays out the foundation for how this molecule interacts with other substances. It directly informs safety protocols, storage decisions, and the quality controls set by regulatory agencies across the world.
Health and Safety Implications
Anyone who has handled aromatic amines can tell you about possible health risks. 2,5-Diaminotoluene itself can cause skin sensitization and has been scrutinized for links to toxicity. The addition of sulfate changes how the molecule dissolves, how it behaves during use, and how it should be disposed of. Research and regulatory agencies base their assessments—such as the European Chemicals Agency—on chemical formulas to set safe exposure limits. Mislabeling or misidentifying those details puts workers and consumers at risk. So, the chemical formula isn’t just catalog information—it helps people make informed decisions about use and protection.
Role in Innovation
Getting formulas right is a gateway to responsible innovation. The cosmetics industry uses 2,5-Diaminotoluene sulfate in permanent hair dyes because its stability and solubility create long-lasting color. But as consumer demand for safer and eco-friendly ingredients grows, chemists turn to molecular structures—like the ones described by formulas—to find safer alternatives. In my own research into green chemistry, I’ve found that a clear understanding of molecular composition fuels the development of substitutes that maintain performance without sacrificing health or the environment.
Solutions for Safer Use
As the demand for personal care and industrial colorants continues, transparency and traceability stay crucial. Manufacturers can do a lot by ensuring rigorous chemical verification, clear labeling, and open disclosure of chemical formulas. Regulatory enforcement then works best when matched with up-to-date databases and ongoing toxicity reviews. For end users—whether hair stylists, artists, or students—education about the substances in use encourages mindful handling and proper protection. Learning to interpret a chemical formula like C7H10N2·H2SO4 doesn’t just promote curiosity, it supports responsible science and safer workplaces.
How should 2,5-Diaminotoluene Sulfate be handled and disposed of?
Daily Risks and Real Safety Measures
In the world of industrial chemicals, 2,5-diaminotoluene sulfate shows up in places like hair dye labs, pigment shops, and research benches more often than people realize. Some see it as a means for vibrant color, but up close, it brings its own set of hazards. My first run-in with it happened during a university internship. A spilled vial left me with a sharp lesson on skin irritation and the smell lingered, reminding everyone that protective habits matter far more than theory. Gloves, face masks, and lab coats aren’t optional extras–they block toxins that can sneak through skin or nose. Good ventilation keeps certainty high and headaches low. This isn’t about being dramatic; skin rash, respiratory irritation, and even long-term health risks draw a clear line: respect the chemical, or pay the price.
Storage Practices Make a Big Difference
It’s tempting to treat chemical storage like an afterthought, to toss containers on whatever shelf has space. I’ve seen that lead to ruptured bags and mystery spills. 2,5-diaminotoluene sulfate stays safest when tucked away in tightly sealed, clearly labeled bottles, kept in a cool, dry spot away from any acids or oxidizers. Never trust makeshift containers or faded labels. It’s worth investing in lockable, dedicated cabinets. I’ve watched teams waste money and time cleaning up accidents that could have been avoided by a simple check before storage. If you can’t guarantee the area’s secure, you end up putting coworkers at risk–their trust isn’t something to gamble with.
Disposal: More Than a Quick Toss in the Bin
After years working in both academic labs and manufacturing, I’ve learned that chemical waste doesn’t magically turn safe in the trash or down the sink. 2,5-diaminotoluene sulfate destroys plumbing, damages ecosystems, and risks public health if handled carelessly. Pouring leftovers down the drain just shifts the problem downstream. On a few occasions, lax rules sent fumes into common areas, putting more than just the users in harm’s way. The safest route involves collecting all waste (including rags and gloves) in robust, leak-proof containers marked for hazardous materials.
Incineration at a licensed facility remains the standard for this chemical. This stops most toxic byproducts from ever reaching the outside world. Local hazardous waste programs offer scheduled pickups, and some suppliers now include take-back options in their sales agreements. Relying on approved methods matches both the ethical and legal obligations companies and researchers sign up for. It sends a message that shortcuts don’t have a place in a safe, responsible lab.
Room for Better Policies and Community Effort
No one solves safety problems alone. At my last job, regular safety drills and open conversations about near misses gradually shifted the culture away from “get it done fast” to “get it done right.” Comprehensive training—done before accidents, not after—lowers rates of exposure. Companies and schools can limit how much is kept on hand, tightening their sources. Community awareness helps prevent the misuse or accidental mixing of incompatible substances.
Turning knowledge into habit makes room for less stress and more trust within work teams. Good safety depends less on equipment lists and more on what people actually do when no one is watching.
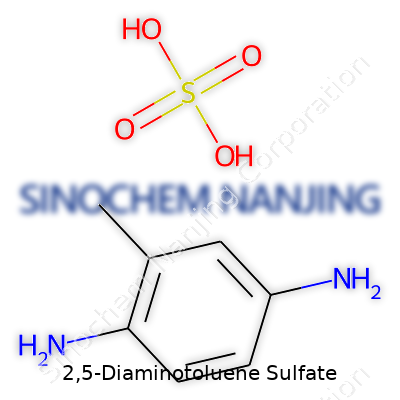
| Names | |
| Preferred IUPAC name | 3-Methylbenzene-1,4-diamine sulfate |
| Other names |
2,5-Diaminotoluene Sulphate 2-Methyl-1,4-benzenediamine sulfate 2-Methyl-p-phenylenediamine sulfate p-Toluenediamine sulfate |
| Pronunciation | /ˈtuː faɪ ˌdaɪ.əˌmɪn.oʊˈtɒl.juːiːn ˈsʌl.feɪt/ |
| Identifiers | |
| CAS Number | 615-50-9 |
| Beilstein Reference | 82263 |
| ChEBI | CHEBI:37743 |
| ChEMBL | CHEMBL185054 |
| ChemSpider | 23713 |
| DrugBank | DB11337 |
| ECHA InfoCard | 100.045.243 |
| EC Number | 212-773-7 |
| Gmelin Reference | 137108 |
| KEGG | C06461 |
| MeSH | D08.811.807.900.900 |
| PubChem CID | 676294 |
| RTECS number | XS9625000 |
| UNII | AD6P21936M |
| UN number | UN2811 |
| Properties | |
| Chemical formula | C7H10N2·H2SO4 |
| Molar mass | 246.32 g/mol |
| Appearance | Dark gray to black-violet crystals |
| Odor | odorless |
| Density | 0.688 g/cm³ |
| Solubility in water | soluble in water |
| log P | -1.2 |
| Vapor pressure | Negligible |
| Basicity (pKb) | 7.88 |
| Magnetic susceptibility (χ) | -54.0e-6 cm³/mol |
| Dipole moment | 1.41 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 192.6 J/mol·K |
| Std enthalpy of formation (ΔfH⦵298) | -91.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3427 kJ/mol |
| Pharmacology | |
| ATC code | D08AX05 |
| Hazards | |
| Main hazards | Harmful if swallowed, in contact with skin or if inhaled. Causes serious eye irritation. Suspected of causing cancer. |
| GHS labelling | GHS05, GHS07 |
| Pictograms | GHS07,GHS08 |
| Signal word | Danger |
| Hazard statements | Hazard statements: H302, H317, H319, H341, H411 |
| Precautionary statements | P261, P264, P270, P271, P273, P280, P301+P312, P302+P352, P304+P340, P308+P313, P312, P321, P330, P363, P405, P501 |
| NFPA 704 (fire diamond) | 3-1-1 |
| Flash point | > 212 °C |
| Lethal dose or concentration | LD50 oral rat 237 mg/kg |
| LD50 (median dose) | 1,470 mg/kg (rat, oral) |
| NIOSH | RN822 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 10 mg/m³ |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
2,5-Diaminotoluene p-Phenylenediamine o-Phenylenediamine m-Phenylenediamine 2,4-Diaminotoluene 2,6-Diaminotoluene |

