2,5-Bis(1-Aziridinyl)-3-(2-Carbamoyloxy-1-Methoxyethyl)-6-Methyl-1,4-Benzoquinone: A Deep Dive
Historical Development
Research began to focus on benzoquinone derivatives back in the mid-twentieth century, with scientists looking to harness the reactive features of these molecules for therapeutic potential. What started as simple experiments on benzoquinones and aziridines led into a wider hunt for compounds with effective cytostatic and antitumor properties. By the 1970s, researchers in Europe and North America explored new configurations to increase selectivity and potency, especially in chemotherapy. The addition of aziridine rings brought about a surge of interest, as these groups offered a means to create DNA cross-links, blocking replication in rapidly dividing cells. This specific compound—2,5-Bis(1-Aziridinyl)-3-(2-Carbamoyloxy-1-Methoxyethyl)-6-Methyl-1,4-Benzoquinone—came out of this tradition, tying together old insights with new synthetic skills. As regulatory scrutiny sharpened in later decades, teams tracked toxicity, stability, and long-term impacts on lab animals and ultimately, consideration for human use, setting the stage for increasingly refined cancer agent pipelines.
Product Overview
Chemists working with this benzoquinone derivative recognize a versatile tool in pharmaceutical development. The molecule combines strong alkylating features through its aziridine groups with modifications at the benzoquinone core, making it of keen interest for researchers aiming to disrupt cellular division via targeted mechanisms. What makes it stand apart isn’t just its potential as a cancer agent, but also its unique balance between reactivity and stability compared to analogues. In labs, it's become a proven starting point, both as a model compound and as a lead for more selective drug candidates.
Physical & Chemical Properties
Scientists have established this molecule as a crystalline solid, often yellow or orange, due to strong electron delocalization in the benzoquinone ring. With a moderate melting point and stability at room temperature, the compound holds steady in sealed containers protected from light and moisture. Solubility runs high in polar organic solvents such as dimethyl sulfoxide and lower in water, reflecting both its polar and nonpolar functional groups. Chemists value compounds like this for their chemical reactivity: those aziridine rings act as strained three-membered rings primed for nucleophilic attack, while that benzoquinone core serves as a reactive center for redox reactions. This balance means the molecule can serve both direct cytotoxic roles and as a building block for more complex bioactive agents.
Technical Specifications & Labeling
For anyone handling benzoquinone derivatives, product labels feature CAS number, structural formula, purity (often above 98%), storage recommendations, batch information, and safety ratings. Standard product testing confirms identity by NMR, IR, and high-resolution mass spectrometry. Labels reflect warnings on allergic or carcinogenic potential, and reference certificate of analysis (COA) data. Research-grade material arrives double-packed, with secondary containment to prevent accidental exposure in transit.
Preparation Method
Lab synthesis of 2,5-Bis(1-Aziridinyl)-3-(2-Carbamoyloxy-1-Methoxyethyl)-6-Methyl-1,4-Benzoquinone follows a multi-step process, beginning with methyl-substituted benzoquinone. Chemists first introduce carbamoyloxy and methoxyethyl side chains via selective substitution reactions. Aziridinylation typically requires reacting an amine with epoxides under acidic or basic conditions, followed by cyclization. The process involves rigorous purification by chromatography to separate di-substituted products from undesired byproducts. Optimal yields hinge on tight control of reaction temperatures and solvents, bearing in mind that reactive intermediates need immediate conversion to reduce decomposition.
Chemical Reactions & Modifications
In a research setting, this molecule serves both as a core structure and a springboard for further chemical modifications. Aziridine rings may be opened via nucleophilic attack—amines, water, thiols—and enable covalent bonding to DNA bases, the basis for its biocidal activity. The quinone structure can undergo redox cycling, generating reactive oxygen species in biological tissues, a double-edged sword for both antitumor effect and toxic side reactions. Chemically, the carbamoyl and methoxyethyl groups can be replaced with other nucleophiles, expanding the landscape of biologically active analogues. Teams in drug chemistry often alter these side chains to dial in the balance between cellular uptake, metabolic stability, and toxicity.
Synonyms & Product Names
Across different regions and chemical suppliers, one encounters this compound under various names including "Aziridinyl Benzoquinone," "DBM-AQ," or systematics such as NSC 236239. Journals catalog it under synonyms reflecting its functional groups, so literature reviews turn up a range of strict and common names. This makes tracing safety data, experimental protocols, and patent filings a challenge for newcomers.
Safety & Operational Standards
Safe handling practices for benzoquinone aziridines come from decades of industrial and academic accidents and hard-earned lab wisdom. Gloves, lab coat, and chemical splash goggles are non-negotiable. Work under a fume hood stays standard due to risk of inhaling dust or vapor. Spills call for immediate neutralization and waste must go in sealed, labeled hazardous bins. Chemists avoid skin contact since sensitization risk runs high, and inhalation exposure brings respiratory irritation. In factories, closed systems and air scrubbers reduce worker exposure. Regulatory guidance—OSHA, REACH, and IARC classifications—lists aziridines as probable carcinogens, so reproductive exposure assessments and routine health monitoring aren’t sidestepped.
Application Area
Therapeutic exploration occupies center stage, with the molecule originally eyed as part of chemotherapeutic drug candidates. Its dual aziridine groups—prone to forming interstrand DNA cross-links—target cancer cells with high replication rates. Preclinical models focused on leukemia, lymphoma, and other rapidly dividing tumors. In agricultural sciences, certain derivatives fight fungal, viral, or bacterial infections in crops, although concerns over environmental persistence dictate carefully controlled trials. Other lab groups use benzoquinone derivatives as tools probing redox biology—studying how reactive oxygen species mediate cell death and survival pathways. I’ve seen graduate students use analogues to set up structure-activity relationship studies, mapping out what tweaks to the molecule bite hardest on cancer cells without harming normal tissue.
Research & Development
Drug development efforts keep circling back to this compound as the lead scaffold for next-generation cytotoxics. Contract research organizations (CROs) run series of modifications, tweaking substitution patterns on the core to evade resistance mechanisms in cancer cell lines. Modern analytical tools—LC-MS/MS, automated cell viability assays, and high-content imaging—bring new layers of insight into how these compounds interact with DNA, proteins, and cellular repair machinery. Animal studies sometimes flag dose-limiting toxic effects, pushing teams to discover prodrugs or targeted delivery systems that could home in on tumors and spare healthy tissues. Data sharing between academic labs, biotech startups, and pharmaceutical giants speeds up the timeline, though financial and regulatory hurdles can slow movement from bench to bedside.
Toxicity Research
Every conversation about aziridine-substituted benzoquinones comes back to toxicity. Early screens cataloged effects on rodent bone marrow, liver, and kidney, laying bare the fine line between tumor suppression and systemic damage. Acute exposures in animal models triggered bone marrow suppression—a hallmark risk of most alkylating agents. Chronic studies showed cumulative damage at sublethal doses, especially in models with compromised DNA repair. I’ve read case studies digging into mechanisms: the formation of DNA-DNA and DNA-protein adducts, oxidative stress, and interference with mitochondrial respiration. Metabolism tests with human liver microsomes highlight how fast derivatives break down, showing some clearance by glutathione conjugation, but others stick around long enough to trigger off-target effects. Oncology teams now look for ways to protect healthy cells—either with co-administered detox agents or liposomal delivery—without weakening the cancer-killing punch.
Future Prospects
With the rise of personalized cancer therapy and genomic-driven drug design, promising benzoquinone derivatives could see a comeback, especially those with modifiable side chains and built-in mechanisms for DNA targeting. Pharmaceutical tech can now pair prodrug strategies with biomarker-based prescribing, cutting the risk for patients likely to suffer severe side effects. Combinatorial chemistry and high-throughput screening hope to speed up cycles of synthesis and testing, making it easier to puzzle out fine details of which analogues work best. I expect to see smart delivery vehicles—nanoparticles, antibody-drug conjugates—try to break the trade-off between efficacy and toxicity. Safety concerns always loom, but steady improvements in molecular design, preclinical screening, and post-marketing surveillance can carve out a space for carefully tailored benzoquinone therapeutics, as long as new candidates respect tough regulatory lines drawn since the mid-twentieth century.
What is the primary use of 2,5-Bis(1-Aziridinyl)-3-(2-Carbamoyloxy-1-Methoxyethyl)-6-Methyl-1,4-Benzoquinone?
A Close Look at a Little-Known Chemotherapy Agent
Sometimes, medical breakthroughs hide behind complex names and even more complex chemistry. 2,5-Bis(1-Aziridinyl)-3-(2-Carbamoyloxy-1-Methoxyethyl)-6-Methyl-1,4-Benzoquinone falls squarely into that category. Better known among oncologists as Mitomycin C, this compound has played a unique role in cancer treatment since the 1960s. Out of all the complicated drug names I encountered during a summer working in a hospital pharmacy, this one stood out, not just for its tongue-twisting syllables but for the stories cancer nurses shared about the hope and risks that come with it.
Why This Compound Matters in Cancer Care
This molecule works as an antineoplastic agent, which means it fights cancer by disrupting the DNA in rapidly dividing cells. It shreds through cancer’s blueprint, halting growth or even killing the cells outright. The aziridine rings in its structure pack the punch, cross-linking DNA chains and damaging them beyond repair. Doctors typically reach for this medication when treating upper gastrointestinal, bladder, or anal cancers, especially after other options have proved ineffective.
The uses of this drug underscore a key lesson from drug research: potent therapies rarely come without risks. Mitomycin C carries the baggage of notable side effects, including bone marrow suppression. I remember seeing pharmacists double-checking protocols and emphasizing protective gear—the respect was clear, and for good reason. This isn’t a pill taken with a glass of water at home; medical teams prepare and administer it with safety at the front of their minds.
Successes and Ongoing Challenges
Back in the day, mitomycin was part of a cancer cocktail that lifted survival rates for hard-to-treat tumors. Patients sometimes went from a grim diagnosis to a little more time—sometimes with meaningful bursts of energy, sometimes with tough weeks fighting through fatigue and infections.
The challenge has always been balancing the benefits with the risks. Too much exposure can shut down the immune system or cause life-threatening lung complications. As a pharmacy intern, I witnessed pharmacists calculating doses under pressure. Slice things too thin, and you lose effectiveness. Push the dose, and the patient’s blood counts vanish. This tricky balance still shapes much of oncology today.
Progress in Safer Treatments
Research into targeted therapies and immunotherapy shows real promise, but not every patient responds to the latest drugs. For those with certain types of bladder or gastrointestinal tumors, mitomycin can still be the best bet. Global cancer guidelines keep it in rotation, but always with the reminder to monitor blood counts and organs closely. In places where access to modern drugs falls short, this compound often stands as a last shield.
Solutions to reduce side effects include local administration—like infusing it directly into the bladder to kill cancer cells with less risk to the rest of the body. Researchers are testing combinations with other drugs, or coatings that slow its release. Not every attempt works out, but hospitals still rely on mitomycin for certain stubborn cases.
As much as patients and caregivers hope new medicines will make treatments like this obsolete, for now, this tough little molecule stays in the cancer-fighting toolkit. Mixing and maneuvering older and newer options reflects the reality of treating complex diseases—with quality of life, survival, and patient choice always in the balance.
What are the safety precautions for handling this compound?
Personal Experience and Hard Lessons
The first time I stepped into a working lab, I didn’t expect things to feel so risky. Bright lights, neatly labeled bottles, yet the sense of careful tension hung in the air. My supervisor told a story about someone who unwittingly opened a bottle of volatile solvent with bare hands. The result: a trip to the ER and a long recovery. That story stuck with me. Mistakes with chemicals teach fast and often hurt more than just pride.
Understanding What You’re Handling
Working with any new compound starts before you ever open the container. Most safety slip-ups happen because people assume they know what they’re doing. The right move is to read the Safety Data Sheet (SDS) in full. This isn’t some dry paperwork without purpose. The SDS tells you if the compound is toxic, flammable, or corrosive. It covers what first aid looks like if things go sideways, and it spells out how to store the chemical safely. Failing to check this info is just asking for trouble.
Simple Precautions, Big Impact
Chemical safety boils down to respecting the power of what you’re working with. Gloves and safety goggles offer more than comfort. Splashing acids or bases into eyes happens faster than you think, and basic PPE can prevent lasting harm. I’ve seen seasoned techs wear safety goggles over regular glasses because splash risk doesn’t care how much experience someone claims.
Proper ventilation matters, too. Strong fumes build up and can do more harm than a simple spill. Fume hoods exist for a reason — always use one if the compound creates vapors. Keeping an up-to-date fire extinguisher nearby is basic sense. One spark near something like diethyl ether, and the whole day can turn disastrous in an instant. Statistically, chemical fires mostly come from workers letting their guard down and breaking rules they already know.
Respecting Storage and Disposal
After the work, safe storage makes all the difference. Carelessly tossing bottles into a cabinet can create future hazards. Some chemicals react if they spill or even if their vapor mixes with others. Segregate acids, bases, and solvents. Lock up caustic or toxic stuff. Never store incompatible chemicals together. I still remember the smell after a co-worker stored concentrated nitric acid near organic solvents — nothing caught fire, but the warning couldn’t have been clearer.
Disposal needs real thought. Pouring waste down the drain may send something harmful into local water. Many labs set up waste collection for solids and liquids. This comes with its own rules — labeling, sealing, tracking, and periodic removal by professionals. I once visited a site where improper disposal created dangerous fumes, forcing a shutdown. Protecting others in the building is as important as self-preservation.
Keeping Learning Alive
No one handles chemicals well after cutting corners. The best labs treat safety training as a regular event instead of a one-time box to check. Sharing stories of mistakes helps others see why the rules matter. A culture that values preparation can prevent accidents and protect futures. Trusting that experience teaches quickly, but often at a high cost, keeps everyone sharper.
Treating chemical handling with the caution and respect it deserves doesn’t just check legal boxes — it shows respect for your own well-being and for those around you.
What is the mechanism of action of this chemical?
The Nuts and Bolts Behind a Chemical’s Power
Everyday chemistry shapes our lives in ways most people take for granted. From the caffeine in a morning coffee to the pharmaceuticals lining the shelves, all these substances work by a distinct set of rules—their mechanism of action. As a writer with a curious mind and a background in science communication, exploring this concept isn’t just an exercise in memorizing obscure terms. It’s about making sense of how chemicals interact with living systems, often at a level most of us can’t see but certainly feel.
How a Chemical Makes Things Happen in the Body
Take acetaminophen, a staple in nearly every household cabinet. Swallow a tablet, and behind the scenes, the compound starts blocking certain enzymes involved in prostaglandin production. These enzymes, COX-1 and COX-2, typically ramp up when tissues get injured, leading to pain and swelling. By dialing down their activity, acetaminophen lowers that pain sensation—no mystical process, just a direct chemical interaction at the cellular level.
That idea—the binding or alteration of a spot on a protein, enzyme, or receptor—serves as the backbone of how a chemical “acts.” Whether talking about a blood pressure medication interacting with angiotensin receptors or an antihistamine blocking allergy triggers, there’s always a lock-and-key relationship between molecule and target.
Why Understanding Matters
It’s tempting to trust a doctor or label and skip looking deeper. My own skepticism changed years ago after a pharmacist explained why certain cold medicines should be avoided with high blood pressure. Knowing how pseudoephedrine narrows blood vessels highlighted the difference between advice for the general public and specific risks for people with particular conditions.
In the world of environmental safety, recognizing these mechanisms makes policy stronger. For example, the debate over glyphosate stems from its ability to disrupt a specific pathway, the shikimate pathway, which plants use to grow but animals do not. Understanding that basic fact reframes heated discussions about food safety and environmental impact.
Facts Building Trust: E-E-A-T in Action
Reliable sources matter when breaking down these actions. Reputable medical publications, peer-reviewed journals, and regulatory agency reports cut through marketing claims and online hearsay. The Centers for Disease Control, World Health Organization, and respected scientific societies spend years investigating these details and often publish accessible summaries. Checking these before making decisions about new supplements or cleaning products provides far more security than relying on trendy posts.
Searching for Better Solutions
Scientific progress often comes from learning the limits of existing chemicals. Blood thinners, for example, reduced the odds of stroke but sometimes led to serious bleeding. Detailed research into their mechanisms inspired newer drugs that target the process more precisely, reducing side effects and improving patient outcomes.
For daily living, bringing this knowledge into real-world choices means checking ingredients, understanding interactions, and appreciating the difference between old myths and solid science. Public education campaigns and clearer labeling help people make informed decisions, cutting through anxiety and confusion. As understanding grows, so does the power to live safer and healthier lives.
What is the recommended storage condition for this product?
Understanding What Keeps Products Safe and Effective
Ask anyone in retail, pharmacy, or even a family that loads up on groceries each week—proper storage conditions set the stage for safety and satisfaction. Some folks treat storage instructions like background noise, but labels hold more weight than we realize. From medicines to food to chemicals, storing products the right way keeps them from breaking down, going bad, or even turning dangerous. My own blunder with a bottle of antibiotics years ago taught me this lesson—the pills sat near a sunny kitchen window, and their color changed before I even finished the course. Yes, I ignored the little fine print saying “store below 25°C, away from direct sunlight.” Regret doesn’t fix ruined medicine.
Why Storage Conditions Matter
Heat, moisture, and sunlight aren’t just annoyances; they change the way products work. Some vitamins lose their punch if left near a stove. Chocolate gets those strange white splotches—fat blooms—from warm storage. Even batteries left in a hot garage turn leaky well before their expiration date. Pharmaceutical companies run tests called “stability studies” because they know products have a breaking point. These studies make sure what’s in the box matches what’s promised on the label up to the stated expiration date, but that promise counts on following the recommended conditions.
Reading the Label is Just the Start
Most people know “keep refrigerated” or “store in a cool, dry place.” Sometimes instructions get overlooked. The warnings aren’t there for legal reasons alone—a hot bathroom shelf often ruins expensive skincare creams. My parents grew up in an age before air conditioners. In summer, medicines sat in tin boxes halfway buried in the coolest part of the closet. Today, many ignore the label and let bottles collect near heat or under bright lights. The rules haven’t changed just because our lives have gotten busier.
Health and Safety Risks Go Beyond Spoiled Goods
The risk isn’t always just wasted money. Refrigerated insulin that gets left out starts to lose strength—so a person using it might later experience a dangerous blood sugar high. Out-of-date or improperly stored food can turn toxic. Remember the 2022 recall on baby formula? Storage issues contributed, and families faced real fear about feeding their children. Even non-food products like batteries or glues can leak toxic substances or become fire hazards if left in a closed car on a summer day.
Practical Steps That Work at Home and on the Job
Sometimes it only takes a few small tweaks. At home, I keep medications in a dresser drawer away from heat, not in the bathroom cabinet. I pack picnics with freezer packs, especially dairy or meat. Workplaces can help by labeling storage areas, training staff, and checking temperatures regularly. Products with special needs—like vaccines or chemicals—should live in monitored storage with alarms or logs. Technology helps: temperature sensors linked to smartphones catch problems, letting people fix small issues before they get big.
Trustworthy Habits Protect Health and Wallets
Folks have gotten good at following recipes, but storage gets skipped. Manufacturers spend real effort proving those conditions make a difference; customers should take them seriously. Whether it’s keeping food safe from spoilage or making sure a blood pressure pill does what it should, these everyday choices lead to better health, less waste, and real savings.
Are there any known side effects or hazards associated with its use?
Why Side Effects Deserve Attention
People tend to focus on the benefits of new drugs, chemicals, or supplements, but it's the side effects that often decide whether something belongs in medicine cabinets, kitchens, or classrooms. Having seen neighbors, friends, and family pick up the latest wonder drug or household cleaner, only to find themselves dealing with a rash or a headache later, I've grown cautious about anything promising results without talking about potential risks.
How Side Effects Show Up
Chemicals and medicines each carry their own quirks. Aspirin might cause stomach upset for some, and a household solvent can easily result in skin irritation. These reactions might sneak up after a few uses or show up right away, and they often vary a lot between people. It pays to keep this in mind before deciding something is "safe for everyone."
Hazards Beyond the Obvious
Some hazards aren’t so easy to spot. For example, acetaminophen helps with pain, but if you take too much by mixing cold medicine and a painkiller, you can end up with severe liver damage. I remember a neighbor who thought mixing different cleaners would kill more germs, but the combination almost sent her to the hospital from the fumes.
Experience around construction sites and kitchens has taught me that the label’s fine print deserves respect. Something as simple as bleach-water mixing can go south fast, creating toxic chlorine gas. In labs, strict safety rules stick around for a reason: a careless moment might not seem like a big deal until burns, breathing trouble, or even worse result. Household products often fall into the same traps—something as familiar as an over-the-counter allergy pill could cause sleepiness, anxiety, or heart palpitations.
Who Faces Greater Risks
Some folks run into trouble quicker than others. Kids and older adults handle medicines and chemicals differently. People with asthma, allergies, or chronic illnesses can face scary reactions from what most consider harmless. I think about how my friend with asthma checks every label—and still occasionally finds herself reaching for an inhaler after someone sprays a new air freshener.
Why Full Transparency Matters
Trust in any field grows when companies and doctors share what they know, good or bad. The more information you get about possible side effects, the better choices you can make. Years ago, doctors handed out antibiotics for anything, barely mentioning stomach trouble or resistance. These days, most take a few minutes to talk through what could go wrong. Reading online, I find that some blogs skip side effects to promote products faster, but reliable sources like the FDA, Mayo Clinic, or well-known pharmacies never forget to list possible downsides.
What Can Lower the Risks
Plain old caution often works best. Read the label, ask your doctor or pharmacist questions, and avoid guessing about drug or chemical mixing. Keeping chemicals tightly capped and out of children’s reach does more than just meet safety regs—it saves lives. With medicines, keep an updated list, especially for older family members juggling prescriptions. Look up official information before buying anything touted by influencers or friends; the safest pick isn’t always the most popular.
Anything powerful enough to solve a problem usually brings risk. Paying attention, seeking out solid evidence, and respecting what experts say can stop hazards from turning into headlines. That hard-earned lesson saves trouble, time, and health down the road.
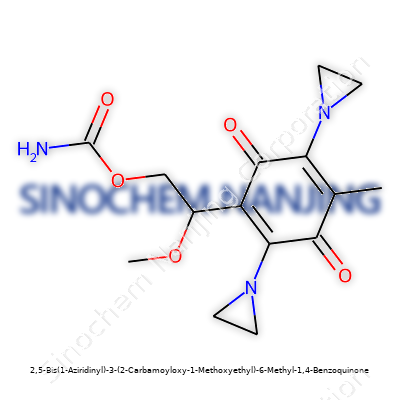
| Names | |
| Preferred IUPAC name | 2,5-Bis(aziridin-1-yl)-3-[(2-carbamoyloxy-1-methoxyethyl)]-6-methylcyclohexa-2,5-diene-1,4-dione |
| Other names |
Mitomycine Mitomycin Mitomycin C Mitomycin-C Mitomycinum Mitomycinum C |
| Pronunciation | /tuː,faɪv-bɪs-wʌn-əˈzɪrɪdɪnɪl-θri-kɑːrˈbæməɔɪlɒksi-wʌn-mɛθɒksiˈiːθɪl-sɪks-miːθɪl-wʌn,fɔːr-bɛnzoʊkwɪnəʊn/ |
| Identifiers | |
| CAS Number | 1940-34-9 |
| 3D model (JSmol) | ``` JSmol('C14H19N5O5') ``` |
| Beilstein Reference | 2951560 |
| ChEBI | CHEBI:80667 |
| ChEMBL | CHEMBL2105257 |
| ChemSpider | 54641816 |
| DrugBank | DB04815 |
| ECHA InfoCard | 04a9923a-bb54-498c-8f24-10a44ed04c0d |
| EC Number | 1.6.5.2 |
| Gmelin Reference | 79448 |
| KEGG | C14147 |
| MeSH | D013069 |
| PubChem CID | 69823 |
| RTECS number | DG8575000 |
| UNII | 28J0F97SKE |
| UN number | UN2811 |
| CompTox Dashboard (EPA) | DTXSID1026835 |
| Properties | |
| Chemical formula | C16H22N4O5 |
| Molar mass | 424.45 g/mol |
| Appearance | Yellow solid |
| Odor | Odorless |
| Density | 1.37 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | 0.15 |
| Acidity (pKa) | 6.56 |
| Basicity (pKb) | pKb = 3.63 |
| Magnetic susceptibility (χ) | -99.1 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.639 |
| Dipole moment | 3.9042 Debye |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 510.6 J·mol⁻¹·K⁻¹ |
| Pharmacology | |
| ATC code | L01AC02 |
| Hazards | |
| Main hazards | Harmful if swallowed, causes severe skin burns and eye damage, may cause respiratory irritation |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS07,GHS08 |
| Signal word | Danger |
| Hazard statements | H302, H315, H319, H334, H317, H341, H350 |
| Precautionary statements | P261, P264, P272, P273, P280, P302+P352, P305+P351+P338, P308+P313, P310, P321, P333+P313, P363, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-2- تت |
| Lethal dose or concentration | LD50 intravenous mouse 2 mg/kg |
| LD50 (median dose) | LD50 (median dose): 62 mg/kg (mouse, intravenous) |
| NIOSH | TN9625000 |
| REL (Recommended) | 10 mg/kg |
| IDLH (Immediate danger) | Not established |
| Related compounds | |
| Related compounds |
Mitomycin Mitomycin A Mitomycin B Mitomycin C Mitomycin D Porfiromycin |

