2,4-Pentanedione: A Close Look Through Science, Safety, and Industry
Historical Development
Long before 2,4-pentanedione found a foothold in laboratories and manufacturing plants, chemists explored the chemistry of simple diketones in the hope of better understanding carbonyl reactivity. The journey kicked into gear during the late nineteenth century, sparked by European research into beta-diketones. As the century turned, synthetic methods improved and the chemical’s role expanded beyond laboratory curiosity. By the time I first set foot in an organic lab, 2,4-pentanedione featured on every stockroom shelf, always ready to help probe metal ions or make more complex molecules. Throughout the twentieth century, industries in Europe, North America, and Asia sharpened production methods, ensuring a steady, cost-effective supply. This substance hasn’t faded—from staple in classic analysis to an essential building block in modern materials science.
Product Overview
Characterized as a yellow, somewhat oily liquid, 2,4-pentanedione stands out for its strong chelating ability. It isn’t just chemistry textbooks that talk about it; anyone working with acetylacetone, as it’s also called, knows its versatility. From its use in analytical chemistry to an ingredient in paints, adhesives, and specialty chemicals, this diketone wears many hats. Rather than a niche product, it appears in daily life more than most would guess, quietly supporting industries like coatings, fuel additives, and pharmaceutical intermediates.
Physical & Chemical Properties
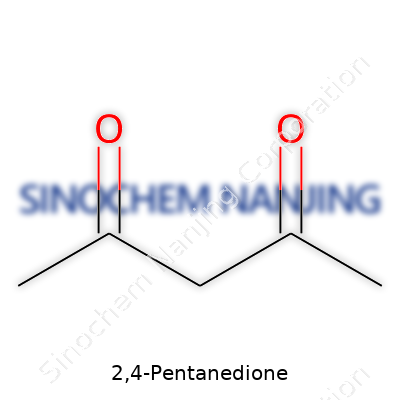
Experience with 2,4-pentanedione means learning its quirks. Its boiling point hovers around 140°C–142°C, and it melts at about -23°C. Its distinctive odor can fill a workroom quickly, unmistakable to anyone who has worked with it long enough. What also matters: 2,4-pentanedione is soluble in water, alcohol, ether, and other organic solvents. Its chemical structure, CH3COCH2COCH3, gives it tautomeric flexibility—enol and keto forms flip-flop depending on the solution or temperature, making it a bit of a shape-shifter in chemical reactions. This property helps it act as a chelating agent, forming stable complexes with many metals, which drives its diverse usage.
Technical Specifications & Labeling
Industry standards expect high purity grades for industrial or laboratory use, ranging from 98% up to 99.5%. Product delivered often meets specifications for acidity, water content, and color measured by APHA scale. Labels warn of flammability, corrosive behavior, and inhalation risks, along with proper UN numbers for shipping. My time handling this material taught me not to ignore safety data sheets; even a small splash requires respect for its irritant properties. Technical data lists flash points, vapor density, and storage conditions—dry, cool, and tightly sealed, always away from oxidizers.
Preparation Method
The main commercial method relies on the acid-catalyzed condensation of acetone. Usually, manufacturers let acetone react in the presence of a catalyst like acidic sulfate, drawing off the product once formed. Efficient distillation finishes the process. Years ago I tried the laboratory version using similar principles—cost-effective, reliable, scalable from gram to ton, provided temperature and acidity stay tightly controlled. Yields can run high, but impurities can slip in, so careful distillation becomes key for pure product recovery.
Chemical Reactions & Modifications
2,4-pentanedione willingly slides into a range of reactions—a chemical Swiss army knife. It forms complexes with transition metals, proving indispensable for spectroscopic analysis and separation technology. Chemists exploit its enol form in organic synthesis, building larger molecules or creating heterocycles. It also takes part in Michael additions, transesterifications, and can act as a ligand in catalysis. For materials science, modifying the diketone structure opens new avenues for creating functional polymers or stabilizers. In one memorable undergraduate experiment, I witnessed its ability to turn copper ions a deep blue — a visual reminder of its coordination chemistry talent.
Synonyms & Product Names
In science and industrial ordering, 2,4-pentanedione often appears as "acetylacetone." Other names include diacetylmethane and 2,4-dioxopentane. International suppliers distribute it under brand names or generic designations, but the CAS number 123-54-6 stays consistent. Regardless of the name, chemical handlers recognize its characteristic label and hazard warnings.
Safety & Operational Standards
Every worker dealing with this liquid learns to keep respect at the forefront. Exposure can irritate eyes, skin, and the respiratory system. The volatility means good ventilation, protective gloves, face shields, and chemical-resistant aprons all play a part in staying safe. Storage away from oxidizers or flames isn’t optional. Best practices follow global safety codes—GHS labeling, OSHA standards, REACH registration. In the workplace, regular training and up-to-date safety documentation back up safe handling. Spills require quick action: absorbent material, contained disposal, and if inhaled, immediate fresh air or medical consultation.
Application Area
2,4-pentanedione bridges lab chemistry and industry. Analytical chemists use it for extracting and quantifying metal ions, and it shows up in protocols for water analysis and soil testing. It serves as a stabilizer for hydrogen peroxide, helping keep solutions effective. Manufacturers of coatings, adhesives, and specialty resins value its reactivity and chelating power. The pharmaceutical world taps it for active pharmaceutical ingredient (API) intermediates. Fuel additive producers rely on it for anti-knock and combustion-improving blends. Its role in metal extraction and catalysis keeps it firmly rooted in both niche and mainstream production lines.
Research & Development
Academic and industrial R&D keeps finding new angles for 2,4-pentanedione. Researchers push beyond classic applications, exploring new catalysts for green chemistry and designing functional materials for electronics or energy storage. The ongoing search for more selective metal capture or ways to tune its reactivity for targeted synthesis keeps the compound central to innovation in analytical science and organic transformations. Green synthesis methods and bio-based production appeal to sustainability-minded teams, aiming to cut emissions and hazardous byproducts.
Toxicity Research
Toxicology data on 2,4-pentanedione shows acute exposure affects central nervous systems, liver, and kidneys in animal studies. Short-term inhalation can irritate mucous membranes, so I never skip the fume hood when handling this diketone. Long-term effects remain under review, and studies showing neurotoxic effects in some animal models have led to stricter workplace exposure limits in recent years. Modern monitoring equipment tracks air concentrations, protecting workers with real-time alerts. Industry and regulatory bodies monitor new research closely, and any emerging health concern prompts immediate updates to safety protocols.
Future Prospects
Current demand in catalysis and analytical chemistry looks steady, but new sustainability targets mean industry wants greener production routes with lower emissions and waste. The trend points toward bio-based acetone feedstocks, solvent recovery, and safer downstream processing. Advanced material science and pharmaceuticals continue to hunt for diketone derivatives with selective properties. With increasing attention on workplace safety and environmental stewardship, the way 2,4-pentanedione is made and handled keeps evolving. As a staple reagent that adapts to new challenges, this compound likely will remain integral to multiple industries for decades to come.
What is 2,4-Pentanedione used for?
Understanding This Forgotten Workhorse
If you’ve spent any time in a university chemistry lab or looked at the label on an industrial solvent drum, you may have come across 2,4-pentanedione. Most folks just call it acetylacetone. Even though its name usually lurks in small print, this compound does heavy lifting across several industries.
What Makes It Worth Knowing?
Chemistry students bump into this liquid early on. Acetylacetone acts as a chelating agent. That means it grabs onto metal ions in a way that lets chemists separate and purify all sorts of metal-based compounds. Without this function, you probably wouldn’t be able to standardize quality when making catalysts or metallic coatings. If a batch of titanium dioxide comes out wrong, sunscreen and paint become a mess. Reliable chelation makes a difference.
I remember a project in grad school where small differences in chelating agent quality turned an entire batch of catalysts into sludge. Reliable sources always used 2,4-pentanedione, and nothing else did as well. It just works every time — and that matters if you care about getting the job done right.
More Than a Lab Tool
Beyond college chem labs, factories rely on acetylacetone for product development. It finds its way into inks and coatings, keeping colors stable and resisting yellowing over time. Some wood stains wouldn’t penetrate as evenly without it. These aren’t glamorous uses, but take them away and problems start to show up: faded billboards, uneven furniture finishes, chemical residues that refuse to scrub away.
Health and safety get a boost too. In medicine, researchers use it to develop radiopharmaceuticals, helping create drugs that diagnose heart disease or cancer. Its ability to grab metal ions lets technicians make these sensitive compounds cleanly, cutting down on the risk of contamination. In some cases, people have found early problems in clinical trials by isolating impurities thanks to reagents like acetylacetone.
Environmental Stakes
Every time a chemical like this enters the water supply or air, it raises questions about long-term exposure. Acetylacetone evaporates easily and doesn’t stick around in the ecosystem, but too much in one place at the wrong time can still become toxic. Factories have to monitor disposal, and researchers keep looking for ways to recycle or break down leftover chemicals. Years ago, an accident at a small plant reminded the town how quickly air quality can shift if someone ignores safety data sheets.
Looking Ahead: Smarter Chemistry
Industry faces a challenge: Make chemicals that perform as well as 2,4-pentanedione without risking health or the environment. This isn’t just about regulations; it’s personal for a lot of chemists. Green chemistry teams explore alternatives—sometimes blending natural compounds or reinventing process steps entirely. I’ve worked alongside researchers who replaced large portions of solvents with water, inspired by the same frustration over “forever chemicals.” Progress comes slow, but it’s possible.
The Takeaway
Behind every reliable batch of catalyst, every clear finish on a table, and every accurate pharmaceutical test, you often find small but crucial compounds like 2,4-pentanedione. Looking closer reminds us that progress depends on punching above your weight and never cutting corners on responsibility—especially when ordinary chemicals carry such unseen promise and risk.
Is 2,4-Pentanedione hazardous or toxic?
Taking a Closer Look at 2,4-Pentanedione’s Effects
I've spent part of my career working alongside chemists who know 2,4-pentanedione as a regular guest in the lab. Sometimes called acetylacetone, this yellowish liquid shows up in the synthesis of medicines, metal chelation, and industrial processes. Its utility has put it into many workspaces, but too many folks overlook how it can bite back if not handled with respect. People wonder if it’s toxic or hazardous, and there's no dodging the evidence: 2,4-pentanedione deserves real caution.
Toxicity Isn’t Just a Technicality
I’ve met technicians who’ve shrugged off pungent solvents, saying, “No big deal, we’ll just turn on the fan.” Let’s be honest: some chemicals demand more than cracked windows. 2,4-pentanedione’s sharp odor does more than wrinkle noses—it signals a substance that irritates eyes, skin, and lungs. Scientific studies make it clear: breathing the vapor may bring on coughing, difficulty breathing, and headaches. My own quick whiff during an unventilated afternoon told me what the textbooks said—keep air moving, or pay the price.
On skin contact, redness or blisters may follow. If 2,4-pentanedione splashes in the eyes, it stings badly, and temporary vision blur isn’t rare. I’ve seen newer lab techs dismiss minor exposures until persistent irritation set in. According to material safety data sheets reviewed by NIOSH and published research, significant skin absorption over time could even have systemic effects—meaning this chemical doesn’t just attack where it lands. Once inside, animal studies suggest it may target organs, especially the liver and kidneys.
Chronic Hazards—Long-Term Isn’t a Free Pass
I’ve come across some seasoned operators who say, “A little exposure won’t kill you.” Chronic low-level exposure brings its own set of risks. Animal trials outline nervous system effects and organ damage after regular contact. Regulatory agencies, including OSHA and the European Chemicals Agency, categorize 2,4-pentanedione as hazardous. It isn’t a known strong carcinogen, but the International Agency for Research on Cancer points to insufficient evidence. Still, the possibility of liver, kidney, or nervous system effects means anyone working with acetylacetone should keep long-term safety procedures tight.
Risk in Everyday Settings
While factories and research labs handle larger quantities, workers aren’t the only ones to worry. 2,4-pentanedione’s volatility means small leaks or spills can drift beyond the bench. Storage mistakes or broken containers can send vapors further than expected; I’ve heard the familiar sweet scent waft down hallways more than once when someone didn’t tighten a cap. Poorly maintained gloves or worn aprons raise the odds of accidental contact—personal stories of irritation speak louder than paperwork warnings.
Mitigating Exposure Isn’t Optional
In my own experience, the best protection comes from simple diligence: nitrile gloves for skin, goggles for the eyes, good fume hoods for air quality. Teaching co-workers about the risks they’re facing keeps everyone alert. Facilities should keep spill kits and wash stations ready, rather than assuming nothing will go wrong. Storing 2,4-pentanedione in clearly labeled, tightly sealed containers, away from heat sources or incompatible chemicals, blocks a range of predictable disasters.
Employers have a duty to educate and protect staff, not just hand out safety data sheets that collect dust. Regular training, proper air monitoring equipment, and open reporting of exposure incidents show respect for the people handling dangerous substances and prevent chronic health stories from piling up.
Safer Practices Give Peace of Mind
Respect for chemicals like 2,4-pentanedione doesn’t mean fear. Smart approaches, up-to-date risk assessments, and clean lab habits make all the difference. Seeing co-workers healthy and going home as they arrived—that’s the benchmark to measure any safety plan against.
What are the storage conditions for 2,4-Pentanedione?
Why 2,4-Pentanedione Demands Respect
2,4-Pentanedione often crops up in chemical labs—a building block for all sorts of reactions. Anyone who's capped a bottle of it knows its strong smell and how it can seem to seep from imperfect flasks. That smell comes with a warning: this stuff evaporates fast and reacts with air and moisture. The bottle it comes in does a decent job, but mistakes with storage can quickly turn into hassle or real safety risks.
Actual Storage Needs
Every lab veteran learns by heart: cool, dry, and dark beats out fancy refrigeration units for a lot of reagents, including 2,4-Pentanedione. Heat speeds up evaporation, so keeping it away from any heat source is non-negotiable. Sunlight can break down this compound over time, so a dark shelf or amber glass container earns its price.
Humidity brings trouble. If the bottle sits in a damp spot, the contents turn acidic. Even tiny cracks in caps can spell “ruined stock.” Desiccators, or dry cabinets, give peace of mind. If those aren’t an option, at least keep the bottle tightly closed and far away from any source of water. Never underestimate how a careless rinse or a spill from a nearby water bath can spoil a good batch.
Ventilation and Isolation
Strong odors always hint at volatility, and 2,4-Pentanedione is no exception. A whiff means vapors in the air, and nobody wants that. Work with it in the hood, and keep its container in a well-ventilated storage area. Don’t put it anywhere near acids, bases, or oxidizers. Mixing storage groups invites disaster if bottles break, leak, or labels fade.
Practical Safety Moves
Lab fires always feel distant until one happens close to home. This solvent goes up in flames easily. Store it far from sparks, outlets, and open flames—never above head height or on cluttered benches. Too many stories start with “I thought it was fine just there for a minute.”
Good labeling helps everyone. Many folks pick up mystery bottles in shared fridges or on shelves. Clear hazard labels, storage dates, and owner initials pull their weight during inventory and emergencies alike.
Handling Leftovers
Open containers just long enough to measure out what’s needed. Wipe threads before recapping to keep seals tight. Spills need quick response—use chemical spill kits, not just a paper towel and a promise to mop up later. Once a bottle nears its shelf life or turns yellowish, treat it as hazardous waste. Don’t let expired stock pile up “just in case.”
Solutions for Safer Storage
Simple steps make big differences. Invest in proper chemical storage cabinets—I’ve seen even tiny departments pool money for one decent flammable cabinet that shifts the whole attitude around lab safety. Create a culture where checking labels, caps, and shelf conditions feels as routine as starting a reaction. Training every lab member, even the interns, is cheaper than calling the fire department.
Smart planning and attention to detail win trust, both among coworkers and with safety inspectors. Small investments in good storage conditions pay off with fewer surprises, less waste, and safer workdays. That peace of mind matters to everyone who spends time near the bench.
What is the chemical structure and molecular formula of 2,4-Pentanedione?
What 2,4-Pentanedione Looks Like on the Molecular Level
2,4-Pentanedione goes by another name many people in laboratories recognize: acetylacetone. Spend a little time in organic chemistry, and you get used to how simple structures can pack surprising properties. At its core, this molecule carries a string of five carbon atoms. Two carbonyl groups, or C=O functional groups, sit at the second and fourth position on the chain. Its structure, written out, looks like CH3COCH2COCH3. Strip away the text, and you have a zigzag carbon chain. Each carbon at either end grabs three hydrogens and connects to an oxygen by a double bond for those two key carbons. The direct chemical formula comes out as C5H8O2.
Why This Compound Matters
Some might wonder why people spend so much time talking about a simple five-carbon chain with a couple of double-bonded oxygens. It turns out acetylacetone plays a vital role in how we build and understand many other things in chemistry. It serves as a chelating agent, binding strongly with metal ions. This helps researchers who want to separate metals, analyze samples, or create special catalysts. During my time working with metal complexes, we used 2,4-pentanedione to form stable, colorful compounds. The color changes tell tales about metal identities. Beyond the lab, industrial production taps into acetylacetone for paints, coatings, and as a stabilizer for fuels and plastics.
Enol and Keto: The Molecular Balancing Act
Acetylacetone never sits still in one form. In solution, it shifts back and forth between two arrangements: the keto form (with two straightforward carbonyls) and the enol form (where one of those turns into a carbon-carbon double bond, and a neighboring hydrogen jumps to the oxygen to form an alcohol group). This kind of balance, called tautomerism, pops up in many important molecules. Experience shows that in water, the keto form dominates. In nonpolar solvents, the enol form can account for more than three-quarters of the molecules present. This balance makes 2,4-pentanedione a valuable teaching model for students learning structure-property relationships.
The Challenge: Safe Handling and Environmental Impact
Many people take chemistry safety for granted until something goes wrong. 2,4-Pentanedione does not smell pleasant, and even a brief encounter with the vapors produces irritation. Chronic exposure brings real risks, with headaches and nervous system effects. In workspaces, following solid ventilation practices and using gloves and goggles keeps accidents away. Disposal often gets ignored, but pouring it down the drain contributes to water pollution. Local hazardous waste programs offer better options, and manufacturers can look for ways to recycle or recover unused solvent when possible.
Looking Forward
Acetylacetone may not spark the collective imagination like a new drug or battery material, but its presence helps countless researchers and industries get down to business. The molecule remains a workhorse—one whose chemical quirks and uses highlight the fascination that can be found in even the simplest organic structures.
What precautions should be taken when handling 2,4-Pentanedione?
Respecting the Potential Risks
Before walking into a chemistry lab, most of us expect the air to carry some tang of chemicals. But a strong, sweet, and pungent odor tends to raise alarms, and that’s the signature of 2,4-pentanedione. It’s more than just an unpleasant smell—it brings headaches, nausea, and irritation to the nose and throat, and it doesn’t stop there. My first day handling this stuff taught me the value of simple habits over expensive equipment.
This chemical manages to get into your body quickly if you breathe it, touch it, or by accident, swallow it. Studies prove that people exposed to high amounts get eye and lung irritation, and some even show clear signs of organ damage with repeated, sloppy handling.
Basic Rules Most Forget
Wearing gloves and goggles seems obvious, but people cut corners. Nitrile gloves usually work, but a spill or a rip means you get burned. Disposable gloves go on before opening a bottle, and they come off before touching your phone or face. A tight lab coat keeps the skin safe, and I keep a separate pair of safety glasses for shifts handling organic solvents.
Face masks or fume hoods matter just as much. Even a few whiffs in a closed space brings on nausea or a cough, and the vapor pressure is no joke at room temperature. If doing any real work—pipetting, pouring, weighing—only a vented fume hood does the job.
Speaking from (Painful) Experience
Once, someone stored 2,4-pentanedione in a fridge where snacks were kept. That batch got tossed. Inhaled vapors from the lingering smell hung around long after we cleaned the shelves, and the entire team learned to separate chemical and food storage. That wasn’t just policy—it became a line we never cross again.
Labeling every container, even a beaker that’s in use for only an hour, helps catch confusion before accidents pile up. With chemicals like this, a routine—label, cover, store in a flammable locker—stops panic before it starts.
Accidents Happen Fast
If 2,4-pentanedione touches skin, wash with cool water for at least 15 minutes; remove clothing if it gets splashed. For eyes, use an eyewash station right away—not after sending a text or grabbing paperwork. If you feel dizzy, leave the room. Don’t try to tough it out.
People sometimes forget spills on the floor make slippery patches and create invisible vapors at foot level. I’ve cleaned a few, using spill pads—not just paper towels—to soak up and contain everything. Waste sits in sealed containers marked clearly for hazardous pickup.
A Path Forward for Safer Labs
Lab managers invest in regular training, not just paperwork. I learned the rules by watching others, repeating habits, and getting corrections. Having a real person explain dangers, demonstrate what burns look like, or walk through an emergency drill—none of that can be replaced by a sign on the wall.
If you’re new to working with 2,4-pentanedione or other solvents, ask questions. Learn about the chemical before you ever touch a vial. And never store, use, or dispose of it without knowing your lab’s rules and where to find the safety data sheet. Everyone in a lab benefits from the shared discipline of prevention, and it rarely costs more than a little attention and care.
| Names | |
| Preferred IUPAC name | pentane-2,4-dione |
| Other names |
Acetylacetone Acac 2,4-Dioxopentane Acetoacetone Pentane-2,4-dione |
| Pronunciation | /ˌtuː fɔːr ˌpɛn.teɪnˈdaɪ.oʊn/ |
| Identifiers | |
| CAS Number | 123-54-6 |
| Beilstein Reference | 1203542 |
| ChEBI | CHEBI:39753 |
| ChEMBL | CHEMBL14150 |
| ChemSpider | 6432 |
| DrugBank | DB01971 |
| ECHA InfoCard | 03a2a1b8-786d-4ff4-b7f4-78436e3006f5 |
| EC Number | 2.4.1.117 |
| Gmelin Reference | 1271 |
| KEGG | C00783 |
| MeSH | D003863 |
| PubChem CID | 311 |
| RTECS number | SA9100000 |
| UNII | 9G2MP84A8X |
| UN number | UN1230 |
| Properties | |
| Chemical formula | C5H8O2 |
| Molar mass | 100.12 g/mol |
| Appearance | Colorless to yellow liquid with a strong odor |
| Odor | Sharp, mint-like |
| Density | 0.97 g/mL at 25 °C (lit.) |
| Solubility in water | Miscible |
| log P | -0.35 |
| Vapor pressure | 3.5 mmHg (20 °C) |
| Acidity (pKa) | 8.99 |
| Basicity (pKb) | 8.88 |
| Magnetic susceptibility (χ) | -47.5×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.445 |
| Viscosity | 0.83 mPa·s (20°C) |
| Dipole moment | 3.11 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 179.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -327.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1680 kJ/mol |
| Pharmacology | |
| ATC code | D06AX12 |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS02, GHS07 |
| Signal word | Danger |
| Hazard statements | H226, H302, H312, H315, H319, H332, H335, H360 |
| Precautionary statements | Precautionary statements for 2,4-Pentanedione: "P210, P261, P280, P302+P352, P304+P340, P305+P351+P338, P312, P337+P313, P403+P233, P501 |
| NFPA 704 (fire diamond) | 2-3-0 |
| Flash point | 54 °C |
| Autoignition temperature | 183 °C |
| Explosive limits | 1.2–8.4% |
| Lethal dose or concentration | LD50 Oral Rat 55 mg/kg |
| LD50 (median dose) | LD50 (median dose): 55 mg/kg (rat, oral) |
| NIOSH | TTQ6425000 |
| PEL (Permissible) | 200 ppm (790 mg/m³) |
| REL (Recommended) | 100 mg/m³ |
| IDLH (Immediate danger) | 120 ppm |
| Related compounds | |
| Related compounds |
1,3-Cyclopentanedione Acetylacetone peroxide Acetylacetonate Hexafluoroacetylacetone Trifluoroacetylacetone |

