2,4-Lutidine: Peering Into a Resilient Building Block of Modern Chemistry
Historical Development: A Walk From Past Curiosities to Modern Utility
People didn’t take much notice of 2,4-lutidine back when pyridine chemistry was still in its early days. In the nineteenth century, chemists discovered many of these nitrogen-containing rings while working out what gave coal tar its bite and usefulness. The early records of 2,4-lutidine surfaced during these initial explorations of fractional distillation. Isolation methods required patience and plenty of material. Researchers in Germany and the UK found several methylpyridines clustered among heavy oils, where they showed up as minor but persistent troublemakers—present, but neither easy to purify nor to ignore. As chemical techniques grew sharper, 2,4-lutidine emerged more distinctly from the pool. By the twentieth century, as people pushed deeper into pharmaceutical research, alkaloid studies, and synthetic dye production, these small heterocycles drew more focus. 2,4-Lutidine’s own chemistry, though never as famous as pyridine or 2,6-dimethylpyridine, gained respect for its reliable quirks. Its stability and predictable behavior kept it in labs, shaping research directions and, sometimes, industrial walks of life.
Product Overview: Using What We Know
2,4-Lutidine, known chemically as 2,4-dimethylpyridine, strikes a balance between reactivity and stubborn structure. It’s an aromatic compound outfitted with a pyridine ring and methyl groups on its second and fourth carbons. In this arrangement, the methyls block much of the ring’s usual attack points, adding some bulk while leaving the nitrogen’s lone pair exposed. People in chemical processing, analytical labs, and pharmaceutical setups keep bottles of 2,4-lutidine handy as a base or a solvent, precisely because it has just the right combination of modest basicity, volatility, and manageable odor —a breathy, pungent scent that lingers even after the bottle is closed.
Physical & Chemical Properties: Knowing What You’re Handling
2,4-Lutidine lands on the shelf as a colorless to pale yellow liquid under ambient conditions, with a boiling point that stays shy of 172°C. It has a density a little lighter than water and dissolves fairly well in most polar organic solvents. The structure doesn’t lend itself easily to hydrogen bonding, letting the molecule move freely in various reaction mixes. Its basicity, though milder than aliphatic amines, stakes out a middle ground within pyridines due to methyl substitution. That shift in electron density draws interest when optimizing reactions that need a base neither too soft nor too aggressive. The methyl groups also add some shielding. That subtle bulk, especially at the 2-position, keeps stray electrophiles from wandering to the nitrogen, making the molecule more enduring in tough conditions.
Technical Specifications & Labeling: Field Realities
Pick up a bottle of 2,4-lutidine in the laboratory, and you’ll see it wears several warning labels. Regulatory authorities flag it for potential hazards: the liquid can irritate the skin, eyes, and lungs, and carries flammability risks. Chemical suppliers grade it by purity, usually above 98 percent for synthetic work, with the balance consisting of related lutidines, water, or trace pyridine. The color and odor offer helpful clues—any darkening hints at age or air exposure. Labs require tight caps, well-ventilated spaces, and gloves. Labels inform users about flashpoints, transport regulations, and incompatibilities, especially around oxidizers and acids, since even a stubborn solvent like lutidine can flip characters under the wrong conditions.
Preparation Method: A Lesson in Synthetic Patience
Years ago, most 2,4-lutidine came from the mammoth piles of coal tar distillation. Extracting milligram yields from barrels of stinking pitch kept chemists humble. Today, synthetic routes hold much more appeal. The Chichibabin pyridine synthesis, using acetaldehyde, formaldehyde, and ammonia, provides a foothold for methylated pyridines, though it generates a mix of regioisomers. Targeted methods have since emerged: the use of acetoacetic esters, enamine reactions, or cross-coupling strategies let chemists steer more cleanly toward the 2,4-disubstituted ring. These reactions, managed carefully, deliver workable yields but call for thorough purification through distillation or chromatography. Even though sources and pathways have shifted from tar pits to sleek reactors, each batch of 2,4-lutidine still carries a legacy of trial and refinement.
Chemical Reactions & Modifications: Versatility in Context
2,4-Lutidine’s mild basicity and hindered structure make it a specialist’s tool in organic synthesis. Researchers use it as a proton scavenger in acylations, alkylations, and some condensation reactions. The molecule’s reluctance to form strong complexes with transition metals—thanks to those methyl groups —means it's less likely to gum up metal-catalyzed reactions as an unwanted side product. That opens doors in advanced synthesis, especially for pharmaceuticals or fine chemicals. Modifications tend to focus on further substituting the ring, oxidizing the methyl groups, or using it as a stepping-stone for complex ligands in coordination chemistry. Each transformation rides on a careful balance: pushing the ring’s reactivity just far enough without compromising its stubborn backbone.
Synonyms & Product Names: The Lingua Franca of the Lab
Chemists like clarity, so 2,4-lutidine also goes by 2,4-dimethylpyridine. You’ll see both names—sometimes just “lutidine” in lab notes, though other isomers lurk under that wider umbrella. While official catalogs may list the CAS registry number for precision, most scientists decide which lutidine they want by the numbers on the ring. Each name carries its own regional and historical nuances, but the molecular formula C7H9N usually clinches the identification. In product listings or among bench chemists, the default always comes back to the numbers, short and direct.
Safety & Operational Standards: Practical Cautions and Everyday Responsibility
Nobody enjoys an accident, so safety with 2,4-lutidine stays front and center. Its sharp odor gives early warning for leaks or spills, but the real risks come from skin contact or inhalation. Gloves, goggles, and a working fume hood take care of most workplace exposures. Fire safety matters: lutidines ignite more readily than water-based chemicals, so keeping them away from flames and heat sources is standard practice. Regulators set occupational exposure limits based on irritation studies and animal models; while the quantities in a synthetic lab are typically small, repeated exposure without ventilation can add up. Waste streams need attention too, as pyridine derivatives resist easy breakdown and can pollute water sources if dumped carelessly. Every bottle thrown away or solution flushed down the drain should go through dedicated organic waste collection, to respect both safety laws and downstream impacts.
Application Area: Beyond the Reagent Shelf
2,4-Lutidine finds work in several corners. In chemical synthesis, it’s a favorite for blocking unwanted side reactions. The pharmaceutical world leans on it to adjust reaction mixtures, especially in processes where standard bases like triethylamine prove too reactive or introduce unwanted byproducts. Analysts sometimes tie its detection to environmental monitoring, measuring levels in industrial effluent to watch for regulatory compliance. In rare cases, lutidines show up in plant alkaloids, drawing biological researchers into the story. Its value rests mostly in the reliability and predictability it brings to tricky organic reactions rather than as a building block for living things. Even as demands change, lutidine’s combination of chemical stability and manageable volatility helps it hang on to a place in the toolkit.
Research & Development: Open Questions and Hands-on Progress
Every laboratory has its stories about 2,4-lutidine — from finding it as a stubborn contaminant in chromatograms, to relying on it as a failsafe in delicate coupling reactions. The search for greener, more efficient bases keeps R&D chemists circling back to pyridines. Recent projects focus on designing cleaner routes for lutidine synthesis, hoping to sidestep the environmental pain points of classic processes. Students learn important lessons about electronic effects in aromatic rings by tracing how different methyl placements tweak basicity and reactivity across the lutidine isomers. New efforts study its role as a solvent for emerging catalytic procedures, sometimes relying on 2,4-lutidine’s unique blend of steric and electronic properties to coax difficult transformations along —work that reflects a broader push to control not just yield but the chemistry’s entire environmental and operational footprint.
Toxicity Research: Looking Out for Trouble Spots
Toxicologists look at 2,4-lutidine with wary eyes, matching its behavioral profile to related pyridine derivatives. Animal tests point to moderate toxicity, particularly after inhalation or ingestion. Studies report liver and kidney impacts at high doses, matching concerns scientists have seen with other nitrogen heterocycles. Human data remains spotty, which means workers stick to gloves, hoods, and routine precautions. There’s ongoing curiosity about long-term effects from chronic low-level exposures, especially for people in poorly ventilated environments. Established limits offer a buffer, but every new finding prompts reviewers to revisit where the safety bar should sit —a reminder that even legacy chemicals keep revealing new wrinkles as research methods sharpen.
Future Prospects: Where Curiosity and Necessity Collide
Looking ahead, 2,4-lutidine stands at the crossroads of tradition and reinvention. The molecule’s solid niche in synthetic chemistry won’t vanish, but tighter environmental rules and new green chemistry priorities cast its future in a shifting light. Scientists aim to reduce reliance on hazardous solvents by engineering more selective, recyclable reagents. At the same time, the search for alternative, biologically-derived bases that can mimic lutidine’s performance without its legacy environmental baggage jumps up the research agenda. Digital chemistry platforms, powered by machine learning, now scan families of methylpyridines for better candidates or unexpected reactivities. Every leap in analytical detection or catalytic application opens doors for new uses beyond the routine. As more industries rethink risk and sustainability, even a familiar face like 2,4-lutidine finds itself adapting, tested equally by tradition and by the inventions just over the horizon.
What is 2,4-Lutidine used for?
Understanding What 2,4-Lutidine Does
2,4-Lutidine isn’t a household name, but it pops up in a surprising number of chemistry labs. This liquid belongs to the pyridine family, with two methyl groups attached. That simple tweak makes it stand out from plain pyridine. In my experience, a small adjustment like this can turn a boring compound into something much more useful, especially for folks working in research or chemical manufacturing.
Why Chemists Reach for 2,4-Lutidine
Most people probably don’t realize how many reactions depend on a good base, one that won’t muck up the rest of the process. 2,4-Lutidine shines here. It neutralizes acids without getting itself tangled in unwanted side reactions. That makes it a go-to choice for chemists doing sensitive work, especially in pharmaceutical labs where precision matters. If you’ve ever waited on a prescription at the pharmacy, you’ve witnessed the ripple effect: small details in a lab can shape the medicines you rely on.
Helping Build New Molecules
2,4-Lutidine often steps up during synthesis, mostly as a non-nucleophilic base. Think of it as a trusty wrench that loosens only the targeted bolt, leaving the rest of the engine untouched. This selectivity means chemists can shape complex molecules without causing chaos in the reaction flask. In organic chemistry classrooms, teachers use examples like this to show students how to build structures with care and intention. Without tools like Lutidine, creating modern drugs and materials would feel like working blindfolded.
Industrial Contributions
Industrial workers don’t usually deal with 2,4-Lutidine by name, but the products they help make often owe a debt to it. Paints, coatings, and some flavors or fragrances trace part of their production back to reactions involving Lutidine. Plants manufacturing these goods depend on reliable intermediates, and this chemical fills that spot well. That’s not just trivia. Jobs and product quality ride on the shoulders of parts like these in the supply chain.
Safety: A Matter of Respect
Nobody should handle 2,4-Lutidine without some knowledge and respect. The fumes irritate, and skin contact can cause trouble. Labs enforce careful handling, good ventilation, and solid training. I’ve seen the difference that attention makes: accidents drop, and people keep their health. Safety protocols aren’t just red tape—they protect real people from real harm.
Reducing Environmental Impact
Chemicals like 2,4-Lutidine don’t just disappear down the drain. Waste from labs and factories adds up, and local waterways don’t need any more challenges. Some companies explore green chemistry methods to use less or reclaim and recycle spent materials. This trend matters. Cleaner chemistry supports community health and builds trust between industry and neighbors.
Looking Ahead
Science never stands still. Students and researchers work on safer, greener alternative bases and drive demand for efficiency at every step. I’ve met young chemists tackling these questions head-on, looking for solutions that don’t sacrifice performance or safety. As these new approaches reach the factory floor, everyone—from the worker to the end customer—wins.
What are the safety precautions when handling 2,4-Lutidine?
What 2,4-Lutidine Is, and Why Serious Precautions Matter
2,4-Lutidine comes from the pyridine family, a class of chemicals found across labs and industries, whether in synthesis or research. Over the years, I’ve worked hands-on with nitrogen compounds that pack quite a punch—plenty of risks, whether you see them or not. It never takes much to trigger a hazard, especially with compounds like this. Even a short lapse can cause harm to your health or those nearby.
Hazards Linked to 2,4-Lutidine
2,4-Lutidine lands on watch lists for a reason. It gives off a strong odor, and vapors don’t just fade into the background—those fumes irritate eyes, skin, and lungs. Lab reports and experience both say acute exposure causes burning sensations and persistent cough. On skin, I’ve seen red, itchy rashes, and if your hands aren’t clean, you risk carrying contamination all over the workspace.
Fire and chemical burns aren’t rare cases. 2,4-Lutidine can ignite from a simple spark, and even the vapor will catch fire in the right mix of air. It’s easy to think gloves and goggles cover it all, but an open bottle sitting too long in an active lab can change everything in an instant.
Protect Yourself with the Right PPE
Chemical splash goggles don’t just keep vapor from reaching your eyes—they save your eyesight. Regular glasses just don't cut it. Nitrile gloves protect against skin absorption and accidental splashes. I stick to lab coats that button all the way up and closed-toe shoes; one spill, and you learn to never slack on coverage.
If you plan to transfer or pour, don’t work with 2,4-Lutidine in a wide-open room. Fume hoods create a physical barrier. Even a little exposure over time builds up, and the headaches or throat pain that come later can be warning signs ignored at your own risk.
Storage: Don’t Give Vapors a Chance
A well-labeled, tightly sealed bottle keeps vapors in check. I always keep the container far from heat sources—heating vents, sunny windows, or spark-prone areas. In my experience, locking up chemicals in a ventilated, flame-proof cabinet keeps unexpected accidents at bay. Folks sometimes forget to check expiration dates, but old stock can become unstable, so that regular inventory sweep pays off.
Spill Response: Speed and Precision
Accidents in a chemical workspace follow no schedule. If a bottle tips, speed and calm matter more than anything. I’ve learned that a simple paper towel won’t cut it. Get a chemical absorbent pad, and always wear gloves and a mask while cleaning. Once you finish, dispose of pads in a chemical waste bin to prevent secondary exposure. Notify a trained supervisor every time, no exceptions—hidden residues can harm anyone who comes after you.
Training, Monitoring, and Culture
Working around chemicals like 2,4-Lutidine only stays safe if you know the risks. Training sharpens awareness, helping you recognize odd smells or spills faster. Safety showers and eye-wash stations never sit as afterthoughts on my checklist. I make a habit to double-check exits and know exactly where to run if something flares up. In busy labs, regular drills create muscle memory and cut panic in half.
Calls for detailed logs and exposure reviews come from real needs—not just bureaucracy. In my time, I’ve seen minor incidents that, with better preparation, could have been avoided. Team meetings, checklists, and clear labels build a safer routine. Each person looking out for the next turns a high-risk environment into a reliable workspace.
What is the chemical structure and formula of 2,4-Lutidine?
Diving Deep into Its Structure
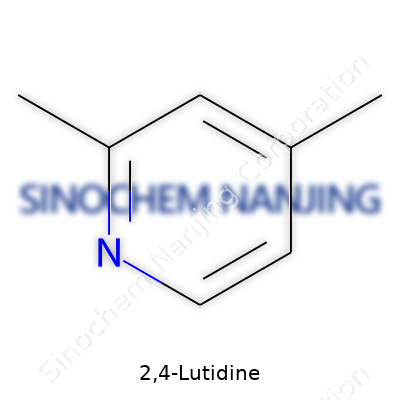
2,4-Lutidine carries one of those names that sounds obscure until you meet it in a laboratory. This compound belongs to the pyridine family, which means its basic structure mimics that of pyridine: a six-membered ring containing five carbon atoms and a single nitrogen atom. What sets 2,4-Lutidine apart is the presence of two methyl groups attached to this ring. As its name suggests, these methyl groups sit at positions two and four on the ring, counting from the nitrogen atom.
Sketching out the molecule, we get its chemical formula: C7H9N. In structural terms, it looks like this: a benzene-like ring, each corner taken by a carbon except for one nitrogen. Methyl groups hook onto the second and fourth positions. This setup makes 2,4-Lutidine less symmetrical and a bit bulkier than its monomethyl cousin, 2-methylpyridine.
Why It Matters in Everyday Chemistry
Walk into a chemical storeroom—a small brown bottle labeled 2,4-Lutidine might stand among other organic solvents and reagents. As someone familiar with basic lab work, I bump into lutidines in synthetic chemistry routines. The two methyl groups make 2,4-Lutidine less reactive than plain pyridine, which changes how it interacts with other reagents. That means it often acts as a base in reactions where a strong base would trigger unwanted side-products. These methyl groups also push electron density toward the nitrogen, tweaking the molecule’s behavior without making it overwhelming.
People working in pharmaceuticals and agrochemicals often lean on compounds in the pyridine family. 2,4-Lutidine shows up as a catalyst or intermediate. Its slightly non-polar nature lets it dissolve in organic solvents with ease. One practical aspect that’s hard to ignore is its smell—pungent, with a sharpness that anyone in a lab will recognize instantly. This isn’t just trivia; safe handling means good ventilation and protective equipment, especially since exposure can irritate skin or eyes.
Implications for Researchers and Industry
Anyone tasked with creating new molecules—research chemists, industrial scientists, even grad students—needs to understand how substitutions like those methyl groups alter the whole personality of a basic ring. 2,4-Lutidine doesn’t just act as a base; it can buffer pH and stabilize transition metals in catalysis. That methyl positioning also slows down the molecule’s tendency to undergo oxidation, which keeps it useful for holding its shape during tough reactions.
From a safety perspective, seeing the 2,4-Lutidine label means looking up exposure limits, keeping it capped when not in use, and dating chemical containers. Studies of pyridine derivatives have raised questions about long-term exposure and environmental build-up, so labs often invest in systems that track usage and disposal. Switching from more hazardous bases to less reactive ones like 2,4-Lutidine can help cut down on waste and lower risks.
Potential for Greener Chemistry
Modern chemistry pushes for greener processes. Choosing 2,4-Lutidine offers a step away from harsher amine bases. Labs that recycle solvents and minimize toxic byproducts pay close attention to compounds like this. Its ability to deliver results in controlled conditions—without igniting spikes in toxicity—shows the payoff in tuning a molecule’s properties with just a couple of methyl groups.
2,4-Lutidine reminds everyone that a small shift in structure brings big changes in function. Its role as a building block offers lessons for chemistry students and professional chemists alike: understand your reagents, read the label, and never underestimate how even a minor substitution shapes the outcome.
How should 2,4-Lutidine be stored?
Understanding What’s in the Bottle
Anyone who’s worked around chemicals for a while knows that safety habits come from equal parts training and hard-knocks lessons. 2,4-Lutidine, a compound you'll often find in research labs, smells strong and acts flammable, so it calls for more respect than some average reagents. It’s a basic heterocyclic amine—a clear, colorless liquid under normal conditions—and its vapors are nothing you want floating around your workspace.
Why Reliable Storage Matters
I’ve seen folks get lax about storage because they figure, "If it’s behind a door, it’s safe." That’s a good way to wind up with a headache—or worse—since 2,4-Lutidine evaporates easily and catches fire when conditions allow. Every chemical safety course drills home the risk of storing volatile organics in open shelves or near ignition sources. I’ve smelled leaking amines from forgotten vials, and I don't want to repeat those headaches.
Reports from both the National Institute for Occupational Safety and Health (NIOSH) and the Safety Data Sheet (SDS) underline the point: store 2,4-Lutidine in a cool, well-ventilated spot, locked away from ordinary office environments and separated from oxidizers and acids. Even if a lab feels large enough, vapors build up where people least suspect—especially in old buildings with poor airflow.
Mixing Common Sense with Protocols
It isn’t enough to label a flask and shove it on the lab’s general-purpose shelf. I’ve seen what happens when flammable liquids are lined up with acids and someone knocks a bottle over. Emergency showers and eyewash stations might be nearby, but nobody wants to test them out. I always recommend using dedicated flammable storage cabinets. Metal, self-closing doors, spark-proof interiors—these details prevent bigger problems. Lutidine’s flash point is around 66°C. Even an ordinary hot plate nearby could light off vapors, so isolation from heat sources is key.
Ventilated storage reduces vapor buildup. Good labs install exhaust fans or vented safety cabinets, but even in smaller workspaces, cracking a ground-level window helps some. The key is reducing inhalation risks. Lab workers exposed to amines report nausea and coughing—symptoms you can avoid by paying attention to airflow and sealing containers tightly. Chemical fume hoods enable safer handling and short-term storage, especially during weighing or dispensing.
Mind the Neighbors: Chemical Incompatibility
Segregation of incompatible chemicals keeps accidents from spiraling out of control. 2,4-Lutidine reacts with strong oxidizers and acids. If containers start leaking or mixing, toxic fumes might result. Store it far from strong acids, oxidizers, and bleach-based cleaners. Label the shelf and cabinets clearly. Routine inventory checks help spot any damaged bottles or evaporated contents before they cause trouble. Even small leaks can corrode shelving or damage equipment nearby.
Working Toward Safer Labs
It helps to set up checklists for staff. The American Chemical Society and OSHA recommend monthly storage inspections: look for loose caps, check expiration dates, and replace old containers regularly. People on the frontlines should have gloves and protective eyewear handy, since Lutidine’s contact hazards go beyond simple flammability. In large research operations, digital inventory logs and barcoded containers make it easier to keep track of who’s using what and where.
Safer labs grow from a culture of careful storage and honest communication. New staff quickly learn from experienced workers who treat every volatile bottle with respect. With enough practice, keeping 2,4-Lutidine secure becomes second nature—just another step in working smarter and protecting everyone nearby.
Is 2,4-Lutidine hazardous to health or the environment?
What Is 2,4-Lutidine?
2,4-Lutidine comes up in labs and industry settings. This colorless liquid falls into the pyridine family—basically chemicals with a strong smell. Folks in research, pharmaceuticals, and chemical plants often cross paths with it. As someone who’s handled plenty of solvents and reagents in research over the years, the moment I smell that sharp, fishy scent, I know to take it seriously. Strong odors from chemicals usually point toward something risky if you’re not careful.
Health Concerns: Not Something to Shrug Off
Just breathing in 2,4-lutidine vapor can irritate the nose, throat, and lungs. Some people feel dizzy, get headaches, or even nauseous after exposure. If it gets on your skin, it can burn or cause rashes. Let this stuff touch your eyes and you’ll know real pain—serious eye damage sits on the table. As common sense and a lot of real-world incidents show, using gloves, goggles, and proper ventilation aren’t just suggestions in labs and chemical plants—those basics save people from a world of hurt.
There’s more to it. Studies in animals show possible nervous system problems and effects on the liver or kidneys after repeated or high-dose exposure. Scientists haven’t linked 2,4-lutidine directly to cancer in people—at least, nothing’s shown up in long-term studies so far. Still, the risk of chronic health effects climbs if caution gets tossed aside during use. Anyone saying, “It hasn’t hurt me yet,” might not realize how slow health issues from chemicals can creep up. A strong safety culture should win out over shortcuts every time.
Environmental Risks: Spills and Long-term Consequences
Every time chemicals see heavy use, there’s a real chance they’ll slip into rivers, air, or soil. 2,4-lutidine floats up into the air faster than some heavier compounds, but it sticks around in water or dirt if nobody cleans it up. Reports show it doesn’t break down easily in groundwater or surface water. What does this mean? If a spill goes unchecked, local wildlife could take a hit. Fish, insects, even the plants along the banks—they all depend on clean water.
I remember a river clean-up after a tannery spill years ago. Nobody noticed anything right away, but one season later, fish started dying in big numbers. The culprit turned out to be leftover chemicals that didn’t break down. Every chemical we use can come back in ways we don’t expect. The trouble with 2,4-lutidine is that it can travel beyond the factory fence and stick around, tagging along with ground or surface water. This stuff doesn’t just vanish on its own.
Better Choices and Safer Practices
Some companies look into alternative chemicals that don’t linger or attack living creatures as harshly. Substituting less toxic compounds works where possible, though not every job fits that swap. Precise guidelines on storage, handling, and disposal come in handy because mistakes—spills, vapor leaks, improper dumping—don’t give second chances. For those of us who’ve managed chemical labs or taught proper waste management to students, setting rules isn’t about red tape—it’s about protecting coworkers and the environment every day.
Real transparency from manufacturers and workplaces makes the whole situation safer for everyone, inside and beyond the lab. Communities deserve to know if a facility handles large volumes, and workers should push for straightforward safety training that doesn’t cut corners. 2,4-Lutidine serves a practical role in research and industry, but treating it with respect keeps people and the environment out of trouble.
| Names | |
| Preferred IUPAC name | 3,5-Dimethylpyridine |
| Other names |
2,4-Dimethylpyridine 2,4-Lutidin 2,4-Lutidinum |
| Pronunciation | /ˈtuː.fɔːr.ljuː.tɪˌdiːn/ |
| Identifiers | |
| CAS Number | 108-47-4 |
| Beilstein Reference | 63605 |
| ChEBI | CHEBI:28925 |
| ChEMBL | CHEMBL14242 |
| ChemSpider | 58116 |
| DrugBank | DB01999 |
| ECHA InfoCard | echa.infocard.100.003.733 |
| EC Number | 202-111-9 |
| Gmelin Reference | 8534 |
| KEGG | C01745 |
| MeSH | D008157 |
| PubChem CID | 7000 |
| RTECS number | UJ4375000 |
| UNII | EDQ288H7Y7 |
| UN number | UN2643 |
| CompTox Dashboard (EPA) | DTXSID7020708 |
| Properties | |
| Chemical formula | C7H9N |
| Molar mass | Molar mass: 107.16 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | pyridine-like |
| Density | 0.925 g/mL at 25 °C |
| Solubility in water | miscible |
| log P | 0.77 |
| Vapor pressure | 2.7 mmHg (20 °C) |
| Acidity (pKa) | 6.70 |
| Basicity (pKb) | 6.77 |
| Magnetic susceptibility (χ) | -58.1·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.475 |
| Viscosity | 1.53 mPa·s (20 °C) |
| Dipole moment | 1.27 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 165.2 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -14.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3407.8 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS06,GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H302, H312, H315, H319, H332 |
| Precautionary statements | P210, P261, P280, P301+P312, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 2,4-Lutidine: "2-3-0 |
| Flash point | 61 °C |
| Autoignition temperature | 525 °C |
| Explosive limits | 2.1% (LEL), 10.9% (UEL) |
| Lethal dose or concentration | LD50 oral rat 2000 mg/kg |
| LD50 (median dose) | LD50 (median dose): 200 mg/kg (oral, rat) |
| NIOSH | SK0450000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of 2,4-Lutidine: 2 ppm (parts per million) |
| IDLH (Immediate danger) | 300 ppm |
| Related compounds | |
| Related compounds |
Pyridine 2,6-Lutidine 2,4,6-Collidine 3,5-Lutidine 2-Methylpyridine 4-Methylpyridine |

