2,4-Dinitrotoluene: Past, Present, and Paths Forward
The Long Road Behind and Where It Started
Every chemical has a trail of stories, and 2,4-dinitrotoluene probably tells a deeper one than most folks guess. Through the years, researchers and industry have relied on its unwavering reactivity, with records of its use going back to the days when the world paid less attention to safety and more to the scramble for progress. Chemists discovered 2,4-dinitrotoluene amid the push for better explosives, and like many things from that era, its value rode on the demand for industrial might. By the middle of the 20th century, large plants were feeding the world’s hunger for synthetic fibers like polyurethane, where 2,4-dinitrotoluene turned from a lab curiosity to a workhorse ingredient. History doesn't give much credit to intermediates like this one, yet without it, the things we take for granted—insulation, adhesives, even spandex—would look pretty different, or might not exist at all.
What We’re Really Talking About
This compound lands on the table as a pale yellow, sometimes flaky solid that packs a lot of chemical reactivity. Its formal name, 2,4-dinitrotoluene, only hints at a broader list of aliases: DNT, DNT 2,4, and others from the trade and science benches. It fits squarely among aromatic nitro compounds, with two nitro groups locked onto a toluene ring. Unlike gasoline’s familiar fumes, the scent here is sharp in a way that spells industrial lab more than backyard garage. Some may not realize how many familiar products owe a debt to DNT. Towed into the limelight by its role in making toluene diisocyanate, DNT often doesn’t get top billing, but its footprint stretches through adhesives, foams, and dyes with a scope few single chemicals can match.
Getting Down to the Nitty-Gritty: Properties and Specifications
Anybody working around 2,4-dinitrotoluene gets used to its solid, somewhat grainy form under room conditions. It melts between 70 and 75 degrees Celsius. Toss it in a lab flask, and it dissolves smartly in most organic solvents, dodging water almost completely. Technicians respect its ability to hold onto a nitro group, which helps both in stored stability and in later reactivity. Its molecular weight and density may look like numbers on a page, but in practice, these traits steer everything from shipping requirements to how mixtures behave down an industrial pipeline. You’d be hard-pressed to overlook the color. There’s something about that yellow—both warning and promise. People keep close tabs on its purity, usually topping out above 98 percent for industrial runs, with tighter margins needed in specialty work to manage unwanted surprises during more complex syntheses. Labels remind workers about its reactivity and the fact that its dust isn’t something anyone wants floating in their airspace.
How People Make It and What Happens Next
Big production usually starts with toluene, a mainstay of the chemical world, and a nitric acid bath. Mix in sulfuric acid, control the heat, and the molecule picks up two nitro groups at set positions, landing you right at 2,4-dinitrotoluene. There’s a knack to balancing reaction temperature and time—push it too far, and your yield falls off; too little, and incomplete products gum up the works. This is chemistry with consequences—not just making the right stuff, but also handling the tangle of leftover acids and byproducts. Technicians have honed processes since the mid-20th century, drawing on lessons written in workplace injuries and environmental scares. Even after the reaction’s done, there’s a checklist of washes, filtrations, and chemical neutralizations before the DNT is ready for its next industrial chapter. Tweak those wash protocols, and product safety leaps forward. Miss a step, and downstream processes get unpredictable fast.
From Simple to Complex: How the Molecule Changes
In a laboratory or plant, DNT’s twin nitro groups invite all kinds of chemistry. Reductions can trim those nitros down to amines, leading straight to toluene diamines, which then unlock toluene diisocyanate—fundamental for polyurethane foams. Add a little tweaking, and you alter the aromatic ring, setting the stage for specialty dyes or modifying the thermal properties for unique plastic applications. Chemists developed selective pathways to harness this potential, turning DNT from a simple intermediary into a robust chemical Swiss Army knife. Modern research centers often look for new catalytic methods to slash waste or cut down energy use. Some labs probe substitutions that replace the core altogether, but DNT’s chemistry keeps it hard to beat for both reactivity and cost. Every change spins off a web of downstream benefits or hiccups, making clear why experience matters as much as innovation here.
Names and Numbers: Keeping Things Clear
Nobody trusts just one name in the chemical business. Call it 2,4-DNT, or use synonyms like para-dinitrotoluene or Dinitro-p-toluene. Catalogs and databases list a stack of alternative names and CAS numbers, backing up decades of regulation and research. This isn’t splitting hairs—emergency responders, warehouse operators, and regulatory agencies need sharp clarity to keep DNT from being mistaken for its more dangerous cousins like 2,6-dinitrotoluene or one of the even heavier nitroaromatics. Mislabel a drum, and even small mix-ups can snowball into big problems, both for workers and for anyone trying to track industrial emissions or workplace exposure.
Safety and the Hard Lessons Learned
Few chemicals teach hard lessons like 2,4-dinitrotoluene. Long before regulators set exposure limits, folks learned to keep their hands protected and their lungs clear of dust. Those yellow crystals don’t mix well with open skin or careless handling. Chronic exposure links back to severe health concerns, from liver and nervous system impacts to suspected carcinogenicity. Agencies like NIOSH and OSHA draw thick lines on permissible exposure, and those numbers go lower as new studies emerge. In the field, this means everything from specialized ventilation to double-checking respirators, especially in older facilities where dust still lingers on rafters and equipment. Emergencies aren’t rare—spills, accidental heating, or just the daily grind of moving hundreds of kilos. Workers rely on routine monitoring and solid emergency plans, and they don’t take shortcuts when it comes to storage or disposal.
Where It Lands: Real-World Uses
Most folks outside the plant floor don’t realize just how much 2,4-dinitrotoluene shapes the stuff around us. Its main gig involves acting as the precursor for toluene diisocyanate, letting polyurethane foam happen in construction, automotive seating, insulation, and footwear. The trickle-down effect lands in modern furniture, coatings for electronics, and even the flexible sponges beneath manufactured wood floors. Some specialty sectors tap DNT for making dyes and explosives, though modern safety rules shrink those footprints compared to a hundred years ago. Paints, varnishes, and some plastic colorants still dip into this chemical’s wide reach, giving products both durability and visual punch. Nobody sees or smells DNT in the finished items, but its fingerprint lingers in every finished edge and strong joint. Efforts to recycle polyurethane or shift toward greener chemicals keep DNT under scrutiny, pushing industry leaders to weigh its irreplaceable chemistry against growing environmental concerns.
What Research Tells Us—and Worries Us
Labs have spent decades probing DNT from every angle: its pathways in soil, its breakdown in drinking water, and its fate in living cells. Toxicologists map out how DNT and its metabolites travel through the body, watching for health effects carried across generations. More recent work zooms in on how DNT behaves in natural ecosystems, especially given its tendency to linger in soils and leach into groundwater. Research flagging reproductive toxicity and possible carcinogenic effects set the tone for tighter controls in both manufacturing and cleanup sites. Scientists now dig into DNA-level disruptions caused by low-dose exposures, pushing regulators to keep closing loopholes where DNT could slip through unnoticed. Industry-backed studies sometimes play defense for current use, but the impartial science almost always calls for stricter safety, better monitoring, and a serious look at legacy pollution sites.
What Lies Ahead
Looking ahead, DNT stands at a crossroads. Researchers grind through new ways to replace it or at least cut its risks, knowing full well how entrenched it stays in the supply chain. The push for green chemistry sends venture-backed labs searching for safer surrogates, though few rivals match DNT’s mix of price, reactivity, and proven track record in foam manufacture. Environmental watchdogs keep pressure on companies to rethink waste streams, close old dump sites, and scrub manufacturing emissions. Advances in catalysis aim to strip out fossil-derived feedstocks, but the learning curve remains steep. The next few years likely hold both challenges and inventions, as the chemical sector wrestles with decarbonization while holding onto practical performance. The real test lies in making safer, more sustainable choices—without tossing aside the hard-won reliability that DNT delivers. Experience, science, and a commitment to health will need to line up, giving this age-old molecule a fair shake at a safer, more responsible future.
What is 2,4-Dinitrotoluene used for?
What Is 2,4-Dinitrotoluene, and Where Does It Show Up?
2,4-Dinitrotoluene, known to some folks in the industry just as DNT, gets pulled off the shelf mostly to crank out polyurethane. Polyurethane makes its way into plenty of things: seat cushions, insulation foam, and even shoes. Most people, myself included, have relaxed on a couch padded with foam that started off its life thanks to chemicals like this one. DNT sneaks behind the scenes into so many comfort products at home, work, and everywhere in between.
Plenty of factories rely on DNT as a chemical stepping stone — they convert it into toluene diisocyanate (TDI), and that’s where the real action in foam-making starts. That means DNT helps make the springy furniture for living rooms, dashboards in cars, and mattresses for tired bodies. Nobody gives much thought to chemicals buried in cushions, but every real-world comfort traces back to a raw material somewhere down the line.
Risks Nobody Should Ignore
Even though DNT plays a big part in the comfort industry, it carries baggage. Every time I’ve read about chemical safety, names like 2,4-Dinitrotoluene come with red flags. You’re looking at something toxic to breathe or touch. Studies link exposure to problems with blood, nervous system, and possibly even cancer. And DNT isn’t picky about where it spreads, drifting into air, water, and even the ground around plants where it’s handled carelessly. CNN’s coverage on contaminated groundwater in factory towns tells the story: pollution from these substances can linger for years, changing the health of entire communities.
Workplaces handling DNT operate under strict rules — minimum protective gear, constant monitoring, lots of gloves and masks. People who punch the time clock in places like these depend on established safety habits, clear ventilation, and up-to-date equipment. But accidents and spills slip through cracks sometimes, often making news when plant workers get sick or neighborhoods see a spike in unexplained illnesses.
Solutions: Protecting People and the Planet
Switching gears from risk to responsibility, it feels clear that industries using this chemical must focus hard on safety and oversight. My experience in manufacturing taught me real safety comes from top-down commitment and clear communication. No “maybe tomorrow” attitude keeps anyone safe in a plant with chemicals that threaten health if handled wrong. Emergency response teams have gotten sharper over the years, stepping up rapid cleanup and restricting leaks, but it always seems smarter to invest heavily in prevention than scramble to undo harm after the fact.
On a bigger scale, researchers and companies keep searching for substitutes to DNT in polyurethane manufacturing. Chemists talk excitedly about greener chemistry, and there’s solid progress. Traditional materials like soy-based polyols or newer bio-based feedstocks show promise. They don’t always slot right in to every current process, but continued research and practical trials could help phase out the worst chemicals, one product at a time.
Public transparency matters too. People who live near chemical plants have a right to know what’s in their water and air, and updates on plant safety practices or contamination levels shouldn’t get buried in paperwork. Where towns have demanded more frequent testing and open reporting, community health trends have started to change in the right direction.
Balancing Progress and Precaution
Modern life builds comfort on some risky foundations, and 2,4-Dinitrotoluene proves it. The challenge comes in never losing sight of the costs that sometimes hide in plain sight. Prioritizing research, pushing safety, keeping communities informed — these steps matter when it comes to building a safer future for everyone.
Is 2,4-Dinitrotoluene hazardous to health?
What is 2,4-Dinitrotoluene?
2,4-Dinitrotoluene is not a word that slips easily into everyday talk. The substance shows up mainly in factories, especially those that make polyurethane foams and explosives. Workers and communities near such plants might not think about it much, but this yellowish chemical deserves close attention because of its health track record.
Health Risks: More Than Just a Chemical Name
People usually connect scary chemicals to heavy industry or long chemical names on ingredient lists. But a closer look at 2,4-Dinitrotoluene pulls it straight into real-life worries. Facts from health research tell a clear story. The International Agency for Research on Cancer (IARC) lists this substance as “possibly carcinogenic to humans.” In plain terms, scientists see enough warning signs to suspect it can cause cancer. Animals exposed in tests often develop tumors in the liver, kidneys, or bladder. Nobody wants to gamble with those odds, even if the threat seems distant.
This chemical has a way of entering the body unnoticed. It can get in through the skin, with the air someone breathes, or in rare cases by swallowing contaminated food or water. Workers in manufacturing settings rarely smell or see it; they may just notice a bitter flavor in the air. Longouts of low-level exposure seem especially risky, and that matters when jobs last decades. In some cases, symptoms don’t even start until years after exposure. It’s a slow-moving hazard, and those can be the trickiest to control.
More Than The Workplace
Health risks go beyond factory floors. 2,4-Dinitrotoluene may leak into groundwater or soil near production sites. Folks living nearby might never work inside the plant, but the effects can reach neighborhoods through drinking water, dust, or homegrown food. Studies in American industrial towns have found elevated traces in local water supplies. People don’t appreciate their health getting caught in a crossfire they never signed up for.
My Experience With Safety Attitudes
Years on construction sites taught me a lot about how people view risk. Anything you can't see—like airborne chemicals—often gets ignored. Most folks want to trust experts to keep them safe. But in places without strong oversight, rules might be bent to save time or cut costs. Good intentions can’t substitute for smart controls. I’ve seen battles over proper ventilation or protective gear, and not everyone takes regulations seriously until sickness strikes. Over time, the costs—medical bills, lost work, worry—pile up for families and whole regions.
Smart Steps Toward Prevention
Clear information helps people make smart decisions. The most effective approach involves regular air and water monitoring around plants, proper labels on containers, and enforced wearing of protective gloves and masks. Many countries use strict rules to restrict how much 2,4-Dinitrotoluene makes it into the environment. They run regular health screenings for workers. Sharing test results with local communities also builds trust and gives individuals the ability to speak up when risks appear. Pressure from community groups and honest reporting—never swept under a rug—make a real difference in protecting everyone’s health.
How should 2,4-Dinitrotoluene be stored and handled?
Chemicals with Baggage: 2,4-Dinitrotoluene Isn’t a Regular Tote-and-Go Compound
Bottling up anything with the nitro group can set off alarms in a chemist’s mind. 2,4-Dinitrotoluene lands right in that red-flag zone. If you’ve worked around industrial settings, the orange-yellow crystals pop up in stories about explosives, dyes, and polyurethane production. They don’t get their reputation just from where they’re used—these molecules pack reactivity and toxicity you can’t shrug off. Some workers only realize the seriousness after headaches and rashes remind them they got sloppy.
Straight Facts: Physical Threats Lurking in the Drums
Ignore the chemical’s dry look—under the wrong push, 2,4-Dinitrotoluene reacts with strong oxidizers and light a fire. Stat sheets say don’t store it with nitric acid, hydrogen peroxide, or open flame. That’s not red tape; it means containers go in a spot with no stray sparks or exposed wiring—think solid concrete room, free from sunlight, where humidity and temperature don’t play games. It can self-heat and run the risk of release if forgotten in a warm, damp warehouse corner. Supervisors must care about those background details, since the compound holds its toxicity in fumes and dust. Break the seal carelessly, and it’s in the air, in your skin, deep in your lungs.
Storing 2,4-Dinitrotoluene: Forget Shortcuts
Most mistakes happen from shortcuts or half-training. Someone once propped open the lid, just planning to check on things “for a minute.” Next week, the smell gave it away. Those fumes can hurt liver and nervous system, and there’s a low but real cancer risk—no scare tactics, just hard evidence from years of occupational medicine studies. Suppliers ship in tight steel drums with gasket-sealed lids for a reason. At every step, keep those containers closed unless you’re ready to measure. Use containment trays. Post clear hazard signs—words mean less than clear images when someone’s in a hurry or new on shift. Never leave repackaged leftovers unmarked. No matter how short the time, always close it up tight, seal it, and lock the storage room. A little heat, a little static—the margin of safety fades too fast.
Handling: Why Gear and Clean-Up Matter More Than You Think
Any worker who ever skipped gloves or went without goggles knows regret stings more than the paperwork. Exposure means blue-tinged skin, odd fatigue, dizzy spells, and not in a good way. Masks, gloves rated for chemical resistance, full sleeves, and boots aren’t optional. Any spill dries out, turns to dust, and travels far. Ventilate work areas, run HEPA filters, and always wet down dust before wiping—never sweep a 2,4-Dinitrotoluene spill dry. Everyone on the crew should know the location of the eyewash and shower, with drills that make it second nature. Don’t trust memory—keep the Safety Data Sheet pinned and up to date.
Smart Moves for Safety
Industrial safety comes from drill and habit, but also from managers who invest in regular training and never ease up on inspections. A mix of warning systems and real-life drills pushes information into daily routines. Nobody stays safe through luck. Industry leaders audit their storage systems and swap out old seals or labels long before they give out. In companies where safety lapses lead to legal trouble or worse, it’s the details—tight storage, PPE compliance, sharp cleanup routines—that draw the line between a clean record and a major incident.
What is the chemical formula of 2,4-Dinitrotoluene?
Treating Chemicals as More Than Numbers
2,4-Dinitrotoluene stands out in any basic chemistry set. You find it not just in dry textbooks or on worn lab chalkboards. It pops up across industries, from explosives manufacturers to pigment research teams. I still remember the first time a college lab instructor set a bottle of 2,4-Dinitrotoluene on the bench: a yellowish, crystalline solid, as if trying to look harmless. Those hands-on encounters stick with you, especially after learning what those innocent-looking yellow crystals actually contain.
The Formula: C7H6N2O4
C7H6N2O4—just nine atoms. At first glance, not so complicated. Seven carbon atoms form the backbone, with six hydrogens darting off like bristles, two nitrogens from the tough nitrate groups, and four oxygens settled where they play a major role. Simple as that? Not quite. Placement matters, and here those two nitro groups find their home at the 2 and 4 positions on a benzene ring, next to a methyl group. Try drawing it properly: it takes some finesse, and I’ve clutched my fair share of broken colored pencils doing just that.
Real-World Importance—Why It Matters
This formula isn’t trivia for chemists or a code to memorize for tests. What you have here is a crucial intermediate in making explosives, particularly TNT. The same molecule works its way into polyurethane production and certain dyes. Communities near production sites keep a wary eye on emissions for good reason. The story of 2,4-Dinitrotoluene reminds us how small compounds can alter huge systems. The U.S. Environmental Protection Agency points out risks tied to this chemical, highlighting long-term exposure concerns, including potential effects on the nervous system and links to cancer.
Ask anyone who has spent time in an analytical lab, and they’ll tell you: 2,4-Dinitrotoluene isn’t something to handle lightly. While convenient for industrial processes, its toxicity calls for respect. Protective gloves, goggles, fume hoods—after seeing firsthand how even a few stray granules can bring lab teams to a halt, the respect becomes second nature. Factories invest in filtration, detection, and waste treatment for good reason. Ignoring safety protocols, even on a small scale, can turn a productive day upside down.
Balancing Use and Safety
Workers and surrounding communities deserve clear safeguards. While companies design systems to keep exposure low, gaps can appear in older facilities or places where regulations lag. The best operators never stop monitoring air, soil, and wastewater. Regular training—always hands-on and never just a checklist—makes a difference.
I once visited a plant that actively recycled and neutralized DNT waste. Seeing those workers take environmental health as seriously as production numbers proved how strong safety culture works in real life. Technologies for remediation, including special bacteria and advanced oxidation, keep improving. Policy makers, scientists, and manufacturers have the tools now to limit harm and improve safety records. Staying rooted in facts, testing, and transparency builds trust in these tough industries.
Fact-Driven Decisions Count
C7H6N2O4 works as both a powerful industrial tool and a potential hazard. The formula tells a story of responsibility and risk. By combining practical knowledge, experience, and consistently strong safety habits, people can protect workers and communities while still meeting economic goals. Chemistry isn’t just about numbers or bonds—real lives depend on getting these choices right.
What safety precautions are needed when working with 2,4-Dinitrotoluene?
Why 2,4-Dinitrotoluene Matters—and Why It’s Risky
I’ve never forgotten the sense of unease that comes from handling dangerous chemicals. Few names bring that uneasy knot quite like 2,4-Dinitrotoluene, or 2,4-DNT. Known in industrial circles for its role in making polyurethane foams, explosives, and dyes, the real story lives in its reputation for serious health risks. This chemical isn’t something you can take lightly: inhaling its powder or mist, skin contact, or accidental ingestion can lead to everything from headaches and dizziness to damaged kidneys, anemia, or even cancer. OSHA and NIOSH don’t just urge caution—they demand concrete steps for anyone with their sleeves rolled up around this compound.
What’s on the Line?
I’ve watched a coworker brush off instructions, only to end up in the emergency room with chemical burns. Two minutes of distraction created hours of chaos and months of regret. 2,4-DNT isn’t just an irritant. Repeated or prolonged exposure means taking risks with your liver, nervous system, and blood. Recent research links long-term contact to cancer in lab animals. It’s tough to shake off the feeling that you owe it to yourself, and your family, to take every precaution offered.
The Steps You Don’t Skip
Start with the basics: nobody belongs in a room with 2,4-DNT without wearing solid, chemical-resistant gloves, goggles with side shields, and full-body coveralls. I know some folks play fast and loose with glove changes or eye protection. After seeing what this powder does to unprotected skin, skipping even one piece of safety gear just doesn’t seem worth it.
Ventilation stands between safe handling and disaster: keep the space under negative pressure and work near fume hoods where practical. Dust and vapor can hang in still air, turning a simple job into a medical emergency if inhaled. Respirators rated for organic vapors and particulates become your lifeline, especially if there’s any doubt about airflow.
The habit of careful hygiene becomes crucial: don’t eat, drink, or smoke anywhere near work areas. Keep extra clothing on hand and wash up before heading home. No trace of yellow powder belongs on your skin, in your hair, or under your fingernails. The urge to get lunch without changing out of your gear just isn’t worth the risk.
Emergency Plans Aren’t Optional
Spills or exposure can’t always be avoided, no matter how disciplined you feel. Emergency showers, eye wash stations, and spill kits should never be locked away or covered with storage boxes. I remember drills where people rolled their eyes; then one afternoon, a spill forced everyone out in under two minutes. Those who knew where the safety gear was cut their health risk down to almost nothing.
Medical attention moves up the priority list instantly. Symptoms don’t always show up right away. The boss who insisted “shake it off” ended up with bigger medical bills and more time off than the cautious one who called a doctor.
Building Safer Habits
Knowledge makes all the difference—smaller, regular group trainings keep instructions fresh. Reading up on the latest handling procedures pays off, since standards get updated as research uncovers new risks.
With 2,4-DNT, shortcuts belong in stories about what went wrong, not in everyday work. The facts are clear: respect the chemical and follow those safety steps without skipping a beat, or expect some tough lessons. Real safety comes from persistence and a healthy dose of humility around chemicals that don’t offer second chances.
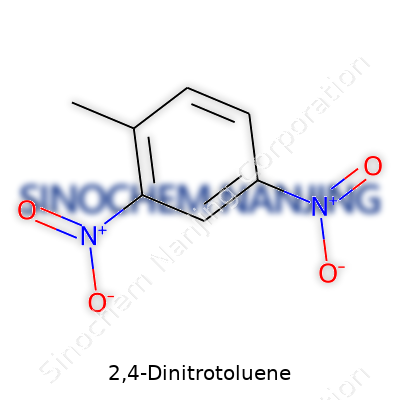
| Names | |
| Preferred IUPAC name | 2-methyl-1,3-dinitrobenzene |
| Other names |
Dinitrotoluene DNT 2,4-DNT 1-Methyl-2,4-dinitrobenzene |
| Pronunciation | /ˌdaɪˌnaɪtroʊtəˈluːiːn/ |
| Identifiers | |
| CAS Number | 121-14-2 |
| Beilstein Reference | 1209235 |
| ChEBI | CHEBI:34671 |
| ChEMBL | CHEMBL14170 |
| ChemSpider | 15413 |
| DrugBank | DB11098 |
| ECHA InfoCard | 100.005.609 |
| EC Number | 208-760-7 |
| Gmelin Reference | 1528 |
| KEGG | C14305 |
| MeSH | D004089 |
| PubChem CID | 8021 |
| RTECS number | XP2075000 |
| UNII | 81W53A8F6D |
| UN number | UN1320 |
| Properties | |
| Chemical formula | C7H6N2O4 |
| Molar mass | 182.14 g/mol |
| Appearance | Yellow to yellow-orange crystalline solid |
| Odor | Odorless |
| Density | 1.55 g/cm³ |
| Solubility in water | 0.04 g/L (20 °C) |
| log P | 1.98 |
| Vapor pressure | 0.01 mmHg (20°C) |
| Acidity (pKa) | 13.46 |
| Basicity (pKb) | 12.82 |
| Magnetic susceptibility (χ) | -71.2·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.546 |
| Viscosity | 2.13 mPa·s at 25 °C |
| Dipole moment | 3.27 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 203.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -56.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3245 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS02, GHS06, GHS08 |
| Signal word | Danger |
| Hazard statements | H302, H315, H319, H332, H351, H373, H411 |
| Precautionary statements | P210, P260, P261, P264, P270, P271, P280, P301+P312, P304+P340, P308+P313, P314, P330, P405, P501 |
| NFPA 704 (fire diamond) | 2,4,0,2 |
| Flash point | 129 °C |
| Autoignition temperature | 636 °C |
| Explosive limits | 0.8–7% |
| Lethal dose or concentration | LD50 oral rat 180 mg/kg |
| LD50 (median dose) | LD50 (median dose): 268 mg/kg (oral, rat) |
| NIOSH | WN1225000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of 2,4-Dinitrotoluene: 1.5 mg/m³ |
| REL (Recommended) | 0.2 mg/m³ |
| IDLH (Immediate danger) | 50 ppm |
| Related compounds | |
| Related compounds |
2,6-Dinitrotoluene Toluene Nitrotoluene Dinitrobenzene Trinitrotoluene (TNT) |

