2,4-Dinitrosoresorcinol: A Close Look at a Storied Chemical
Historical Development
Dive into the story of 2,4-dinitrosoresorcinol, and you uncover a trail that traces back to the classic age of organic chemistry. Chemists working in the late 1800s, driven by the curiosity of examining every shade of chemical reactivity in aromatic compounds, first isolated and studied nitroso derivatives of resorcinol. These scientists noticed that introducing nitroso groups to the resorcinol core didn’t just change its color or odor—it dramatically altered its chemical profile, offering insights into the ways electrons behave inside aromatic rings. As scientific tools got sharper and society’s industrial appetite grew in the 20th century, this compound found itself in labs dedicated to everything from dye development to reagent design. The evolution of its synthesis methods over decades tells a story about chemists chasing purer yields, safer reactions, and new applications, each step shaped by technological advances and shifting research priorities.
Product Overview
Look beneath the name 2,4-dinitrosoresorcinol and you find a compound with strong character, marked by two nitroso groups attached to resorcinol’s benzene ring. The structure’s symmetry gives it a particular charm in the eyes of synthetic organic chemists. Its bold coloration and responsiveness to certain reagents make it more than a sterile lab curiosity. Every bottle, whether used in chemical education or advanced research, embodies the practical reality of transforming basic chemicals into specialized tools for further discovery. Because of the narrow window of commercial interest, you don’t see it stacked on shelves outside advanced laboratories, yet its unique chemistry keeps it relevant where targeted modification of aromatic systems is needed.
Physical & Chemical Properties
Meet 2,4-dinitrosoresorcinol in person and its appearance jumps out—a distinctive solid, usually yellow to orange, flashing a warning to anyone who knows vivid coloring often signals strong reactivity. It holds modest solubility in water and dissolves better in organic solvents, mirroring trends seen in other nitroso aromatics. This compound carries a signature aroma, familiar to anyone who’s spent hours around nitroso substances. Its chemical stability rests somewhere between resorcinol and the more volatile nitrosobenzenes: light and heat coax it into degradation, but ordinary handling in a standard lab, with reasonable caution, doesn’t spell trouble.
Technical Specifications & Labeling
Labels on commercial bottles reference its purity, typically exceeding 97 percent—a nod not only to the capabilities of modern chemical suppliers but also to the practical demands of research. You frequently spot the chemical structure printed right on the package, helping to cut down on error and reminding users what’s really inside. Alongside CAS numbers and molecular weights, suppliers often provide melting points and safety notes—details that support both handling and experimental planning. In the real world, a well-marked container goes a long way, especially in busy environments where confusion could cause accidents or unreliable outcomes.
Preparation Method
Making 2,4-dinitrosoresorcinol starts with resorcinol as a foundation. Chemists often introduce nitroso groups using nitrosating agents, with sodium nitrite and hydrochloric acid as classic players in this transformation. The trick involves tightly controlled temperatures and careful addition of reagents to guide substitution at the 2 and 4 positions—details learned through decades of trial and adaptation. After reaction completion, the mixture usually gets filtered before a few washing and recrystallization steps refine the product. It sounds simple, but the real challenge lies in preventing side reactions that rob yield or complicate the purification. Anyone who’s tried this synthesis in a teaching lab remembers stains on their gloves and the tension of managing reactive intermediates.
Chemical Reactions & Modifications
2,4-dinitrosoresorcinol doesn’t just sit passively—it reacts with metal ions to form brightly colored complexes, making it a classic choice for analytical chemistry demonstrations. Its structure welcomes further transformations too, especially reductions and couplings that take advantage of the activated nitroso groups. Chemists have explored its use as a building block for heterocyclic compounds, evidence of its playful reactivity. Its behavior with nucleophiles and electrophiles provides educational insights and continues to pop up in curriculum experiments that highlight the push and pull of electron density in aromatic systems. Advanced researchers keep digging into its transformation pathways, finding new ways to exploit its dual nitroso arrangement for synthesis of specialty molecules and materials.
Synonyms & Product Names
In catalogs and textbooks, you might see it listed as 2,4-dinitrosobenzene-1,3-diol or by its more systematic IUPAC moniker. Trade and lab colloquialism boil it down to variations like DNR or resorcinol dinitroso. These synonyms grow out of different naming conventions, and being aware of them cuts back on confusion, particularly when hunting through literature or ordering from multiple suppliers. This diversity of names is a reminder that chemistry isn’t just formulas; it’s communication, and clarity matters as much in nomenclature as it does in experimental technique.
Safety & Operational Standards
Every chemist who’s handled nitroso compounds has learned to respect their risks. 2,4-dinitrosoresorcinol requires wearing gloves, eye protection, and working in a well-ventilated hood. Its dust and solutions can irritate skin, eyes, and respiratory passages. Incidents of careless handling underscore why lab protocols—the sort students sometimes grumble about—became standards in the first place. Waste management demands particular care, as many nitroso derivatives have long-term environmental effects. Institutions keep safety data sheets on hand and reinforce safe practices every semester, not out of bureaucratic habit but from the hard experience that even small spills or accidents have big consequences.
Application Area
This compound mostly finds work in the trenches of research and teaching. Analytical chemists use 2,4-dinitrosoresorcinol to develop methods for detecting metals, taking advantage of those sharp color changes. In organic synthesis, its reactive nitroso groups open pathways for making specialized heterocycles and dyes. In classrooms, it brings color and drama to lessons about aromatic substitution and ligand chemistry. Its limited industrial footprint has less to do with shortcomings and more with regulatory caution around nitroso compounds and the specific niche it fills—a case where targeted utility outweighs bulk demand.
Research & Development
Over the years, 2,4-dinitrosoresorcinol has worked its way into numerous graduate theses and specialty publications. Researchers track modifications to its core, seeking improved stability or new properties. In analytical chemistry, efforts focus on optimizing its metal binding for selective detection. Synthetic chemists test out new ligands and potential pharmaceuticals by tweaking the dinitrosoresorcinol skeleton. This continual R&D helps keep the compound relevant, illustrating how even century-old molecules evolve with the shifting goals of the chemical sciences. Grant proposals and journal articles still look to classic compounds like this one to solve new problems, linking tradition with innovation in real, hands-on experimentation.
Toxicity Research
Toxicological studies have explored the risks posed by nitroso derivatives like 2,4-dinitrosoresorcinol. Researchers find links between the structure of nitroso compounds and their ability to interact with cellular systems, spurring ongoing caution. Acute toxicity appears moderate, but chronic exposure issues remain less well mapped. Worker safety guidelines often default to assuming potential carcinogenic risk, a legacy of the broader concerns with aromatic nitroso chemicals. Universities and industrial labs err on the side of caution, keeping exposure to a minimum and emphasizing full reporting and transparency in case of spills or accidental contact. Anyone thinking about expanding its use knows that detailed toxicity profiles play a central role in regulatory decisions and responsible practice.
Future Prospects
The next chapter for 2,4-dinitrosoresorcinol will likely come from niche research, specialty synthesis, and advanced analytical methods. Its utility in forming metal complexes and modifying aromatic frameworks remains a steady draw. Regulatory hurdles and toxicity considerations limit direct industrial expansion, but growth in precision chemistry, tailored sensors, and possibly advanced materials could offer new directions. Success rests with creative researchers who find fresh applications that pair the compound’s unique reactivity with genuine needs. Keen eyes on both safety and sustainability will shape every step, ensuring that even as its story continues, the lessons of the past are never out of reach.
What is 2,4-Dinitrosoresorcinol used for?
What is 2,4-Dinitrosoresorcinol?
2,4-Dinitrosoresorcinol brings out curious looks in most chemistry discussions. Thanks to its bright yellow hue and nitroso groups, this compound often crops up when people talk about analytical chemistry and specialty chemical research. Unlike basic household substances, it lives a quieter life in research labs, not in day-to-day cleaning products or over-the-counter medicines.
Analytical Chemistry and Beyond
Scientists use this chemical for detecting and separating metal ions. The nitroso groups make it a good ligand, which means it can bind to certain metal atoms. By adding it to a solution, researchers see color changes if specific metals are present. Think of it as a kind of litmus paper for metals like cobalt, iron, or nickel. This comes in handy for industries that need to keep a close watch on metal contamination or want to purify metals for engineering and manufacturing.
The Research Perspective
Research journals describe 2,4-Dinitrosoresorcinol as part of experiments aimed at developing new ways to test water, soil, and even air for toxic heavy metals. Environmental chemists have a responsibility to protect water sources from pollutants, and tracing metals moves the needle on public health. Using sensitive, targeted chemicals like this one gives specialists tools to spot problems before they get too big.
Safety and Experience in the Lab
Spending years in university labs taught me that not every chemical is equally risky, but fluorescent powders and strong dyes always demanded respect. Handling 2,4-Dinitrosoresorcinol means reaching for gloves and goggles—no question. The nitroso group shows up in compounds that sometimes generate hazardous fumes. Good ventilation and training go with the territory. Students learn fast that skipping safety steps invites trouble, especially with unfamiliar or reactive chemicals.
Where Problems Begin: Metal Contaminants
Factories and labs discharge tiny traces of metals into rivers, and these metals build up in fish and plants. Heavy doses cause illness in humans and wildlife. This actually pushes small compounds like 2,4-Dinitrosoresorcinol into a bigger spotlight. Quick, accurate metal detection gives communities some control, letting them set cleanup priorities and pressure polluters. Governments write standards, but actual enforcement comes down to technology—and the people using it.
The Road to Better Solutions
Not every town or small lab can afford advanced analytical tools. Test kits built on chemicals like 2,4-Dinitrosoresorcinol make regular screening possible. Simplifying sample preparation and creating clear instructions grows access in underfunded regions. Looking down the line, researchers are working on biodegradable, less-toxic detection chemicals. That matters, because environmental chemistry shouldn’t add its own footprint.
Trust in the Science
Care, accuracy, and transparency anchor the work around these specialized chemicals. The best labs publish data and list possible side effects, plain and simple. If a product or test relies on a substance like 2,4-Dinitrosoresorcinol, it serves everyone to ask: Is it reliable? Is it safe? Does it help solve the problem? My own time buried in chemical testing always came back to that last question. In science and policy both, results matter most—especially when clean water and public health hang in the balance.
What is the chemical structure and formula of 2,4-Dinitrosoresorcinol?
Getting to the Core: The Structure That Matters
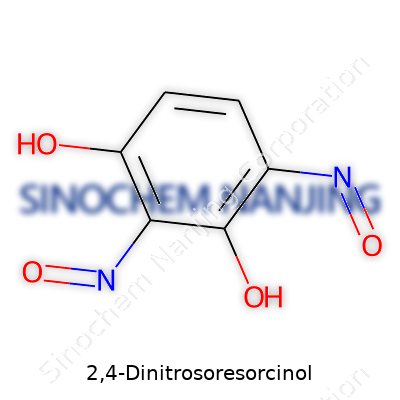
The name 2,4-dinitrosoresorcinol might sound complicated at first, but you boil it down, it's about the arrangement of atoms on a benzene ring. Let’s start visualizing: resorcinol brings two hydroxyl groups sitting at positions 1 and 3 on the ring. For 2,4-dinitrosoresorcinol, these groups are joined by nitroso groups (-NO) at positions 2 and 4. The chemical formula sums up like this: C6H4N2O4.
Within that formula, you see a carbon framework typical of aromatic chemistry. Six carbons from benzene, four hydrogens surviving after substitutions, two nitroso and two hydroxy groups tacked on as functional pieces. Drawing this on paper, you’d lay out the benzene hexagon, assign the –OH groups to carbons one and three, fit –NO to carbons two and four, and let the whole structure balance those electron clouds.
What Those Groups Really Do
Hydroxyl groups help with reactivity. That makes sense based on what we know about phenols, as they tend to form hydrogen bonds, lend solubility in water, and sometimes influence acidity, both in reactions and in the lab when you’re measuring properties. Nitroso groups are a different story. Here, they are electron-withdrawing and bring their own set of chemical behaviors. I remember running a reaction in a teaching lab where nitroso compounds smelled sharp and odd—something you never forget after a long day at the bench.
Nitroso groups change how a compound interacts with other chemicals. They pull electrons in, they can act as intermediates in dye manufacture, and sometimes they find their way into advanced organic syntheses. Their presence in 2,4-dinitrosoresorcinol doesn’t just allow for color changes in test tubes; it marks the molecule as useful for analytical chemistry, like colorimetric determination of certain metals or specific testing protocols.
Why Structure and Formula Go Beyond Just Numbers
It’s easy to overlook chemical structure as just a classroom concept, but I’ve seen how having the right groups in the right places changes everything. The arrangement in 2,4-dinitrosoresorcinol creates an intersection of water solubility and strong electron shifts. That means you can get vivid colors, you get the chance to use the molecule for detecting trace metals, and there’s even talk in research about how these compounds could play roles in new sensing technologies.
I’ve had students ask why we care about substitutions on something as simple as a benzene ring. Looking at this molecule, the pattern of substitution is everything. Put the nitroso groups anywhere else, you won’t get the same action or color results. Details matter. In analytical labs, that precision in chemical building blocks is what lets you measure, discover, and verify substances down to the microgram.
Learning From the Details
Chemistry doesn’t work in isolation. Every functional group means a different property and a different set of potential uses. The hydroxy and nitroso pairing in 2,4-dinitrosoresorcinol supports analytical innovations, sometimes in places you don’t expect. If you’re measuring metals in water, the specific reactivity lets you trust your results. A deeper look at this structure highlights how understanding the “why” behind atomic groups pays off further down the road, both in academic chemistry and in the search for new tools in science and industry.
Is 2,4-Dinitrosoresorcinol hazardous or toxic?
Looking Beyond Lab Benches
2,4-Dinitrosoresorcinol sounds like something out of a dense chemistry textbook, but every chemical carries its baggage when we talk about safety and toxicity. This compound pops up in dye manufacture and a few specialized test kits. Before dismissing it as “just another powder,” it's smart to ask, “What risks am I looking at here?” Having spent my fair share of time handling reagents in college labs, I learned that even obscure chemicals deserve attention.
The Hazards in the Data
Let’s lean on evidence and not just gut instinct: Data from organizations like the European Chemicals Agency labels 2,4-Dinitrosoresorcinol as an irritant to eyes, skin, and respiratory tract. Even at moderate exposure, redness, itching, or sneezing can pop up. Swallowing or breathing in its dust would not be a good idea—it kicks up coughing, throat pain, and could irritate the stomach. Going further, the science around its chronic toxicity sits in a gray zone, because long-term effects haven’t been studied in depth. That means questions stay open until more research rolls in.
Wearing Gloves is Just the Start
Ask anyone who’s worked in a lab, and you’ll hear stories about the one time a splash burned a hole through a favorite shirt. Gloves and eye protection aren’t suggestions—they’re essential. Ventilation in the workspace stops fine powder from hanging in the air. The easiest way to prevent trouble is to store it sealed and far from food, drinks, or busy corridors. Lab managers can rattle off a dozen chemical scares caused by carelessness, not the chemical itself.
Toxicity: How Real is the Risk?
Acute poisoning from this compound doesn’t headline news or medical journals, which suggests it’s not a common threat. That doesn’t mean zero risk. Toxic effects of similar compounds—dinitroso and di-nitro chemicals—have caused problems ranging from skin rashes to headaches when used without proper care. In handling 2,4-Dinitrosoresorcinol, the wise approach borrows from these experiences: don’t gamble on luck to stay safe. Skin contact and dust inhalation always find a way to happen if you don’t prep smart.
Better Solutions, Less Hazard
Safer chemical handling flows from three simple actions: clear labeling, regular safety drills, and honest communication about risks. Schools and companies succeed or fail on these habits. Switching to alternatives makes sense if the chemical is only serving a routine function and a less hazardous option exists—green chemistry cuts both exposure and disposal problems. I’ve seen classes swap out questionable dyes in favor of food-safe substitutes; students still learned what they needed without the extra methylated headaches.
Final Thoughts
2,4-Dinitrosoresorcinol doesn’t carry the fame—or the infamy—of big-name toxins, but that never means a free pass. It fits into a long list of lab chemicals that demand respect, not fear. People who know the risks, stay prepared, and don’t take shortcuts find that even tricky substances lose their scare factor. Science and safety carry equal weight, and paying attention to one strengthens the other.
How should 2,4-Dinitrosoresorcinol be stored and handled safely?
Recognizing Real Risks
I’ve worked in labs where strong-smelling powders and unstable crystals line the shelves. Some, like 2,4-dinitrosoresorcinol, demand respect: they can trigger allergic reactions, and their chemical instability carries real risk. Inhaling or touching even small amounts may cause irritation, so every action in the lab needs intention.
Reliable information points out this compound can decompose if left unattended at room temperature or exposed to sunlight. Such reactions can even escalate to explosions if enough heat or shock enters the picture. Storing it in the wrong spot turns troubleshooting into crisis management.
Smart Storage Choices
Not every shelf fits dangerous chemicals. Keep 2,4-dinitrosoresorcinol in a tightly sealed, amber glass container inside a dedicated explosives cabinet. Temperature—cool, around 2-8°C—makes the difference. Bright light speeds up decomposition, so sidestep windows and open lighting. Fume hoods aren’t just for use—they’re safe storage zones too, especially if ventilation pulls fumes away before breathing them in.
Being careful with quantities matters as well. I never stock more than I expect to use in a month. If there’s a spill, smaller batches shrink the scope of danger. Never return unused portions to the main container; cross-contamination or accidental introduction of moisture or impurities makes the compound more unstable with each cycle.
Personal Protection: Gear That Counts
Losing a minute to struggle with gloves or safety glasses isn’t just an inconvenience; it’s common sense. Simple nitrile gloves, a proper lab coat, and splash-proof goggles keep contact risks low. If you rely on only one pair of gloves, the odds catch up—wear double layers during weighing and transfers. Use a face shield when handling larger batches—no one looks cool with a chemical burn.
Good ventilation goes hand-in-hand with good sense. Open containers only under an active fume hood with steady airflow. In rooms without proper venting, work somewhere else. The chemical can release unpleasant gases, and I’ve seen enough to trust the extractor more than my nose.
Spills, Disposal, and Emergency Prep
Spilling volatile substances teaches quick lessons. Cleanup kits with inert absorbents—vermiculite or sand, not paper towels—contain spills without making things worse. Sweeping up with bare hands or vacuuming brings more headaches, since friction or static can set off unstable dust.
For waste, never pour residues down the sink. Collect them in labeled, shatter-resistant bottles—plastic or glass, but never metal because metal shavings can spark decomposition. Once the waste bottle fills a quarter, schedule yourself for a disposal run; delays increase risk. Local hazardous waste contractors help, and official university or company safety officers can set up pick-ups or emergencies on short notice.
Building Routine Safety Culture
Relying on reminders and regular audits creates a strong safety backbone. Double-checking labels, expiration dates, and chemical inventories every month prevents surprises. I make it a habit to note storage dates and rotate stock to use oldest samples first. Non-chemists don’t always follow the news, but those who cut corners on dangerous chemicals usually end a “cautionary tale.” Proper respect, reliable procedure, and the right gear protect both people and experiments, building a lab environment where skill outruns accident.
Where can I purchase 2,4-Dinitrosoresorcinol and is it available in different grades?
Searching for a Rare Chemical
Looking for 2,4-Dinitrosoresorcinol means stepping into a fairly specialized part of the chemical world. This is not a staple of your local hardware store or mainstream lab supply shop. Instead, this compound often turns up in research environments or niche markets. As someone who has worked in university research labs and handled many requests for rare reagents, I can say this: patience pays off. Reliable sources matter much more than speed when dealing with less common chemicals.
Sourcing through Reputable Chemical Suppliers
The safest route to purchase 2,4-Dinitrosoresorcinol is through established suppliers catering to research, academia, or specialty industry customers. Sigma-Aldrich and Alfa Aesar have distributed hard-to-find analytical standards before, but this particular compound does not always appear on the catalog year-round. When it's not available, I have had success reaching out to their sales representatives for inquiries or special orders. Do not expect immediate access or consumer-kit style transactions like Amazon; these channels screen buyers and ask for affiliation or end-use details, as they follow regulatory controls on handling hazardous materials.
Purity and Grades: What Buyers Should Know
In the rare event a supplier lists 2,4-Dinitrosoresorcinol, you likely see choices based not on a wide range but focused on 'analytical', 'technical', or 'research' grade. Most chemical suppliers focus on purity—the more zeros after the decimal, the more likely it suits sensitive analytical purposes. During my years in analytical labs, colleagues cared deeply about trace impurities when working with trace analysis or calibration standards. For educational demonstrations, less absolute purity may suffice. Communicate your intended use clearly; not every supplier stocks pharmaceutical or food-grade products for niche reagents like this.
Regulation and Safety
This compound flies under the radar of most everyday regulatory lists, but don't let that fool you. Many synthetic intermediates draw scrutiny for potential misuse or hazard, and you may run into requests for documentation or institutional purchase only. My own purchasing experiences confirm that even research chemicals face growing control, especially if you ship across country borders. Always review your local hazardous substance lists and SDS sheets before ordering. My inbox has seen frustrated messages from colleagues whose packages got stopped at customs or who faced repeat requests for paperwork. Avoid shortcuts—request the SDS and submit whatever forms suppliers ask for upfront.
Alternatives and Practical Tips
If commercial sources do not pan out, custom synthesis companies represent another route. These businesses quote on-demand jobs for quantities ranging from milligram to kilogram scales. Speaking with peers in academia, most report better responsiveness when they provide background on their institution or intended research. Be prepared for higher costs and longer delivery times—this is not a shelf-stable commodity, and small order synthesis takes planning. A lack of safety data or handling guides can trip up even seasoned lab staff, so ask for all available information before committing funds.
Navigating an Unusual Marketplace
Tracking down 2,4-Dinitrosoresorcinol demands persistence and well-researched outreach. Suppliers will want to know you're a legitimate researcher, not a hobbyist, and may ask extensive questions to prove it. Documentation shows you know what you’re handling. Along the way, stay alert for mislabeling from no-name web merchants—genuine chemical suppliers share certificates of analysis and openly discuss grade and purity. A bit of networking with colleagues or chemists online often leads to the latest updates about who's carrying which rare compounds.
| Names | |
| Preferred IUPAC name | 2,4-dinitroso-1,3-benzenediol |
| Other names |
1,3-Dihydroxy-2,4-dinitrobenzene Dinitrosoresorcinol |
| Pronunciation | /ˌdaɪ.naɪˌtroʊ.soʊ.rɪˈsɔːr.sɪ.nɒl/ |
| Identifiers | |
| CAS Number | 601-47-6 |
| Beilstein Reference | 354172 |
| ChEBI | CHEBI:51341 |
| ChEMBL | CHEMBL147418 |
| ChemSpider | 26939908 |
| DrugBank | DB14005 |
| ECHA InfoCard | 03dcdfb5-8402-41bb-871b-5a9348feffde |
| EC Number | 205-226-9 |
| Gmelin Reference | 200549 |
| KEGG | C14389 |
| MeSH | D010599 |
| PubChem CID | 16713 |
| RTECS number | DW9625000 |
| UNII | 5W8N59234Z |
| UN number | 2811 |
| CompTox Dashboard (EPA) | DTXSID2046530 |
| Properties | |
| Chemical formula | C6H4N2O4 |
| Molar mass | 182.11 g/mol |
| Appearance | Yellow crystalline powder |
| Odor | Odorless |
| Density | 1.62 g/cm³ |
| Solubility in water | insoluble |
| log P | -0.18 |
| Vapor pressure | 4.01E-7 mmHg at 25°C |
| Acidity (pKa) | 10.57 |
| Basicity (pKb) | 11.26 |
| Magnetic susceptibility (χ) | -44.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.747 |
| Dipole moment | 3.85 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 159.3 J mol⁻¹ K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 98.4 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -373 kJ mol⁻¹ |
| Pharmacology | |
| ATC code | V04CL04 |
| Hazards | |
| Main hazards | Suspected of causing genetic defects. Harmful if swallowed. Causes serious eye irritation. Causes skin irritation. May cause an allergic skin reaction. |
| GHS labelling | Warning; H302; H315; H319; H335 |
| Pictograms | ``` "GHS06,GHS08" ``` |
| Signal word | Warning |
| Hazard statements | H302 + H312 + H332: Harmful if swallowed, in contact with skin or if inhaled. |
| Precautionary statements | P280-P261-P264-P271-P273-P304+P340-P312-P305+P351+P338-P337+P313 |
| NFPA 704 (fire diamond) | 2,4-Dinitrosoresorcinol: 2-3-2 |
| Lethal dose or concentration | Lethal dose or concentration: LD50 (oral, rat): 283 mg/kg |
| LD50 (median dose) | LD50 (median dose): 240 mg/kg (mouse, oral) |
| NIOSH | DH9625000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.1 mg/m³ |
| Related compounds | |
| Related compounds |
2,4-Dinitrophenol Resorcinol 2,4-Diaminophenol 2,4-Dihydroxybenzoic acid |

