2,4-Dinitronaphthol: A Deep Dive into a Unique Industrial Chemical
Historical Development: Following the Tracks of Synthesis and Curiosity
Few compounds from the early 20th century chemical boom show the layers of utility and challenge found in 2,4-dinitronaphthol. Chemists once scoured naphthalene’s derivatives searching for dyes, explosives, and intermediates with enough complexity to drive both industrial and academic interest. My graduate advisor used to joke that aromatic nitro compounds like this “drew new pathways on every lab notebook page.” That is not far from the truth. Early literature marks the synthesis of dinitronaphthols as a true point of progress in organic nitration, bridging the gap between basic aromatic substitution and more sophisticated, controlled modifications of fused-ring systems. Their journey parallels much of what shaped the organic and dye chemistry industries for nearly a century.
Product Overview: Beyond Simple Descriptions
Stepping past lists and stock photos, 2,4-dinitronaphthol sits among a family of nitroaromatics that earned their keep in fields as varied as pigment production and chemical analysis. Some folks take these molecules for granted, seeing little besides yellow crystalline powder when they crack open a bottle. Yet, looking deeper unlocks a host of possibilities—chromatographic standards, intermediates in pharmaceuticals, and, less commonly today, as precursors in dyes and oxidation processes. Its practical value connects with industries like specialty coatings and analytical chemistry, which often push for greater chemical purity and specificity.
Physical & Chemical Properties: More Than Meets the Eye
Opening a jar, a faint odor escapes—a reminder of its naphthyl roots, laced with the edge of nitro groups. The yellow color stands out, making contamination hard to hide. Like others in its class, this compound dissolves slowly in water, much more readily in organic solvents, and holds up under moderate heating before decomposing. The stability in storage depends strongly on dryness and freedom from reducing agents; moisture and heat push it toward slow degradation, sometimes leading to loss of potency in critical applications. In the hands of a careful chemist, dinitronaphthol functions with the reliability necessary for finely-tuned syntheses.
Technical Specifications & Labeling: Navigating Clarity and Caution
Anyone who’s spent time in a synthetic organic lab has seen the black and yellow hazard stickers, the bold font demanding attention. Storing a compound like 2,4-dinitronaphthol means respecting the details—grade, purity, packing lot consistency. Seeking high assay material for analytical or preparative work can require digging through supplier documentation, cross-comparing melting points and spectral signatures to sidestep surprises that could derail careful experiments. Labels warn about irritancy and combustion risk, which many old-timers in the field consider more than just regulatory overkill. Small details in specifications become the difference between a clean chemical reaction and wasted time, money, and equipment repairs.
Preparation Method: The Roots of Nitric Acid Chemistry
Synthesis follows a script that echoes through generations of organic chemists. Classic nitration, using concentrated nitrite sources and sturdy acid-handling skills, turns naphthol starting material into the dinitro product, often in yields that frustrate but rarely disappoint outright. Batch-to-batch reproducibility depends on acid concentrations, temperature control, and reagent freshness. Those who have run such reactions often mention the deep yellow stains on glassware as badges of both effort and experience. Developing cleaner, greener methods—reducing hazardous acid waste and improving energy efficiency—represents an ongoing challenge tackled in today’s labs.
Chemical Reactions & Modifications: Possibilities and Pitfalls
Dinitronaphthols show a rare versatility for modification, but that flexibility comes at a price. The nitro groups activate the ring toward nucleophilic attack, opening options for substitution and reduction, but introducing instability that has tripped up more than a few chemists with sudden exotherms or incomplete conversions. I remember spending weeks balancing selectivity and stability during one particularly stubborn reduction, only for impurities to reveal themselves late in the analysis. It’s these practical hurdles that make mastering dinitronaphthol chemistry so rewarding: the molecule holds potential as a platform for custom synthesis, analytical standards, and even complex dye structures.
Synonyms & Product Names: Navigating the Chemical Lexicon
Anyone searching for this compound in literature or supplier catalogs will turn up a patchwork of names—2,4-dinitronaphth-1-ol, naphthalenol derivatives, and systematics from different chemical traditions. These names aren’t just labels. They show a legacy of regional and industrial naming practices that sometimes create confusion. Finding the right synonym leads to the right product, saving time in procurement and preventing errors in communication between teams working across continents and time zones.
Safety & Operational Standards: Lessons Learned and Hard Rules
Old-timers in chemistry tell stories about loose safety rules and near-misses with dry, powdered nitroaromatics. Handling 2,4-dinitronaphthol calls for more than casual respect. Eye and skin protection, good ventilation, and careful storage away from both heat and reducing agents remain non-negotiable. Industrial labs often go further, using explosion-proof equipment in scale-up settings. Sloppy handling still leads to burns, inhalation risk, and, in rare cases, fire. Every new chemist learns from the stories, not just from the manuals—treating each jar as both a useful tool and potential hazard shapes a culture of preparedness and trust.
Application Area: The Push and Pull of Modern Use
Changing times have shifted the demand for compounds like this. Its historical footprint stretches across dye manufacture, pigment intermediates, and analytical chemistry. Most modern uses lean toward specialty pigment synthesis and as a reagent for chemical analysis, especially in academic research and quality control work. Some niche areas in organic electronics and advanced polymer science still use it as a building block. The balance between usefulness and the search for safer, greener alternatives challenges both established users and new entrants.
Research & Development: Navigating Innovation
Every new project with 2,4-dinitronaphthol starts with the same question: Can we use this old stalwart in smarter, safer ways? Recent R&D trends focus on developing alternative synthesis routes that cut down on hazardous waste, as well as tweaks to the molecule for greater reactivity in advanced materials or coupling chemistry. Green chemistry has become a rally point, pushing researchers to reimagine processes that once relied on bulk acids and solvents. Collaborations between academia and industry look for tweaks in process conditions or catalysts that reduce energy demands and minimize toxic byproducts.
Toxicity Research: Balancing Risk, Cost, and Regulation
The nitro functional groups bring a real toxicity concern. Chronic exposure—either by inhalation or skin contact—has the potential for both acute and long-term effects, echoing the broader risks found in aromatic nitro compounds. Regulatory pressure increases from both environmental and occupational health agencies, driving stricter exposure limits and raising disposal costs. Toxicity studies, both classic and recent, weigh heavily in cost-benefit analyses for new applications. Parallels exist with past compounds phased out for health and safety reasons, serving as a cautionary tale for those looking to bring 2,4-dinitronaphthol into new consumer-facing products.
Future Prospects: The Road Ahead for 2,4-Dinitronaphthol
A molecule with a storied past and a practical present often prompts tough questions about what comes next. In some ways, the future of 2,4-dinitronaphthol depends on our ability to retool old processes, limit environmental impact, and improve worker safety. If new green synthesis methods reach scale and regulators maintain current exemption levels for specialty chemicals, places in advanced polymer and dye research remain open. On the other side, the ongoing search for less hazardous alternatives could truncate its commercial reach. The dynamic rests on innovation, smart regulation, and the pressure to keep both people and the environment at the forefront of the chemical industry’s priorities.
What is 2,4-Dinitronaphthol used for?
What Makes 2,4-Dinitronaphthol Stand Out?
2,4-Dinitronaphthol has sparked interest across chemical and environmental sectors. Chemists notice this compound for its nitro groups, which do more than just set off a reaction in the lab. Growing up around manufacturing plants, I watched the way chemicals like this entered real-life conversation—not only as curious substances on paper but as agents that shaped products found around homes.
You won’t find 2,4-dinitronaphthol on a pharmacy shelf or in a grocery store cleaning aisle. This compound shows up far deeper upstream, often serving as an intermediate in the production of dyes, pigments, and sometimes explosives. Its chemical backbone, derived from naphthalene, gets modified in the lab for use in industrial colorants. While bright colors catch the public’s eye, what goes into that vivid dye rarely gets a spotlight.
Why Is 2,4-Dinitronaphthol Popular with Manufacturers?
Manufacturers looking for durable, consistent colors often choose naphthol-based intermediates because they bond with fabrics and surfaces where many other compounds wash away. Workers in textile or leather factories probably touch something that’s traveled through a stage involving 2,4-dinitronaphthol. The need for precise, long-lasting dyes pushes companies to choose reliable intermediates.
Some research papers describe its explosive potential. This doesn’t turn every use into a hazard, but it does require strict controls. Anyone who’s handled chemical supplies or managed storage knows that safety protocols matter. The storage cabinets with heavy doors at my old job came from a need to keep compounds like this under close watch, avoiding both accidental mixing and exposure.
Where Risks Come In
2,4-Dinitronaphthol doesn’t just sit quietly after production. Once in the waste stream, it can pollute water, soil, and even move through the food chain. Multiple studies point out its toxicity to aquatic life and potential long-term effects on human health. I remember environmental clean-up teams struggling to extract similar substances from riverbeds near former dye plants. That struggle started due to regulations lagging behind the pace of industrial change.
Workers involved in its production or use need reliable personal protective equipment, strong ventilation systems, and detailed protocols. Exposure, even in small amounts, may cause harm—short-term symptoms like headaches and nausea, along with longer-term risks according to occupational health reports.
How Can Impact Be Limited?
Stronger oversight and regular audits of storage and use cut down on emergency calls and environmental damage. Real waste treatment—using advanced oxidation processes, for example—has made a difference in sites that previously released colored water into nearby streams. I’ve seen local factories improve outcomes from simply investing in the right filtration.
Schools and laboratories moving to ‘greener’ chemistry experiments give hope, guiding future chemists away from reliance on hazardous intermediates. Government policies can help, but industry transparency gives the public a chance to hold companies accountable. I’ve always appreciated conversations with neighbors who demand to see what’s coming out of factory pipes.
The Road Ahead
Careful monitoring, safer alternatives, and engaged communities all factor into responsible management of chemicals like 2,4-dinitronaphthol. It’s not just about achieving deeper color on a dress shirt or a cleaner cut in a specialty explosive—it’s about valuing health and environment long after the reaction stops. Taking lessons from real-life scenarios, future generations can do better by questioning and improving upon the chemicals used today.
What are the safety precautions when handling 2,4-Dinitronaphthol?
Understanding the Risks
2,4-Dinitronaphthol stands out in chemical labs for its vibrant yellow color and serious health risks. This compound shows off its hazardous side with its ability to irritate skin and eyes, spark respiratory issues, and even threaten life if inhaled or ingested in large enough quantities. Just a handful of chemicals have made my palms sweat as much as this one, especially after I saw the damage it caused from a single splash on unprotected skin. That image stays in my mind and shapes how I talk about lab safety.
Prioritizing Personal Protective Equipment
A proper lab coat never feels optional with 2,4-Dinitronaphthol nearby. I’ve watched colleagues wash fragments of this powder off their gloves, thankful for the extra layer that kept their hands safe. Nitrile gloves – not latex, not reusable dish gloves, but sturdy nitrile – act as the basic guard. Chemical splash goggles help too, forming a strong barrier between eyes and those yellow particles. In spots with a higher chance for airborne dust, a full-face shield and a certified respirator turn what could be a dangerous task into a manageable one.
Smart Storage and Respectful Handling
I store 2,4-Dinitronaphthol in a cool, well-ventilated cabinet, far from sunlight and incompatible materials (no acids, bases, or flammable solvents nearby). The best practice comes from never keeping more than you plan to use immediately. Transfer it gently, avoiding spills, and use a dedicated spatula to prevent cross-contamination – small steps, but after one flawed experiment, I won’t risk any shortcuts. The sight of even a hint of yellow powder outside its jar is enough to trigger a cleanup routine.
Cleaning Spills and Emergency Measures
No matter how careful you are, spills happen. Immediate containment means less chance of airborne dust and fewer chances for exposure. I keep absorbent pads and non-sparking tools within arm’s reach. Affected surfaces always get neutralized with mild detergent and cold water. The smallest trace deserves proper disposal in a sealed, labeled bag sent off as hazardous waste, not in a regular trash caddy. Anyone exposed – even slightly – heads straight to the eyewash or safety shower followed by a medical check. These aren't overreactions. Delays turn small accidents into big problems.
Getting Training and Sharing Responsibility
Working with hazardous chemicals takes training that goes way beyond watching a safety video. I’ve helped new lab members practice emergency drills until responses come naturally, not just reflexively. Questions and walk-throughs matter; they build confidence and save time during real incidents. Taking stewardship in a safe lab culture means looking out for each other, correcting risky moves, and sharing those stories – even the embarrassing ones.
Disposal Isn’t an Afterthought
Everything that touches 2,4-Dinitronaphthol, from gloves to pipettes, follows a strict disposal flow. Once tainted, lab wares never return to general use. I always separate solvents and solutions to prevent dangerous interactions, signing each waste container with detailed contents for the disposal team. Local regulations trump personal habits here; that habit keeps legal and environmental trouble far from the lab door.
Building Trust in Safe Practices
Looking back, it’s clear that respect for hazardous compounds grows from real experience. A single oversight leaves lasting consequences. Protecting your health, your lab mates, and your workspace means treating every task with vigilance. I still double-check my gear before handing a beaker of 2,4-Dinitronaphthol to a colleague. In safety, extra minutes mean peace of mind. Foresight always trumps regret when dealing with chemicals of serious risk.
What is the chemical structure of 2,4-Dinitronaphthol?
Understanding 2,4-Dinitronaphthol
Naphthol compounds have carried heavy value in chemistry circles, especially for dyes and the synthesis of specialty chemicals. 2,4-Dinitronaphthol isn’t as widely discussed as its cousin, naphthol yellow, but it brings some interesting lessons about how nitro groups change the behavior of an aromatic structure. The molecule features a naphthalene backbone—a flat, double-ring system that forms the core of countless chemical reactions in both industry and research settings.
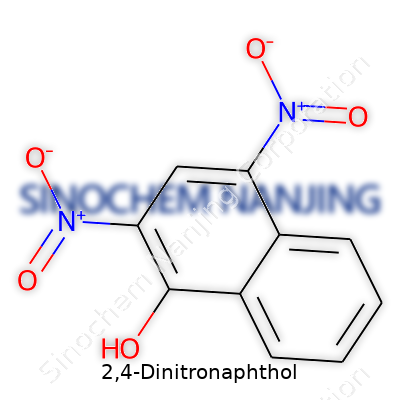
What really marks 2,4-Dinitronaphthol is the presence of two nitro groups and a single hydroxyl group. The nitro groups claim their spots at the second and fourth positions of the naphthalene skeleton, while the hydroxyl group sticks to the first carbon. The positions matter—a seasoned organic chemist immediately recognizes how these electron-withdrawing nitro groups, sitting right next to and across from a hydroxyl, alter the electronic landscape of this aromatic system.
Breaking Down the Structure
In its cleanest form, the molecule carries the formula C10H6N2O5. The naphthalene core looks a bit like two benzene rings fused together. Nitro groups (–NO2) stick out like antennae, each one cranking up the reactivity by pulling electrons from the surrounding ring. These positions—the 2 and 4—mean both nitro groups are ortho and para relative to the hydroxyl group. Nitro groups at those positions create a tug of war on electron density, making the whole molecule more acidic compared to plain naphthol. This extra acidity changes how the molecule behaves in water and how it binds with metal ions, which has opened doors for uses in analytical chemistry and even for niche industrial pigment production.
Why Structure Shapes Function
Electron-withdrawing groups such as nitro play rough with aromatic rings. Painters notice this every time they pick up an old-school yellow pigment: dinitronaphthols and their relatives stubbornly hold their color, often showing strong resistance to breakdown by light and moisture. In the lab, that extra stability comes from resonance stabilization—the nitro groups accept electron density from the ring and the attached hydroxyl, locking the whole system into place. That effect brings durability but also toxicity concerns. Exposure to dinitronaphthols in workplace air and wastewater means we need to pay attention to both process safety and waste management.
Toward Safer Use and Better Alternatives
Nitroaromatic compounds, including 2,4-Dinitronaphthol, can pose serious health risks if handled carelessly. Chronic exposure links to skin irritation and, at higher exposures, possible organ damage. Factories and academic labs set up local exhausts and enforce glove use. Environmental regulations demand scrubbers and chemical neutralization steps before any waste streams leave the site. In my time running a small lab, working with naphthol derivatives never came without gloves or goggles. Mistakes stick—cleanup takes real effort, and nitro compounds never truly mix with water, which slows natural breakdown dramatically.
Some chemists have started shifting toward safer substitutes, especially in textile printing and dye synthesis. Research into less toxic alternatives with similar pigment properties continues, though few simple molecules can rival the stubborn stability of nitro-substituted naphthols. Until true green replacements scale up, the best step is still a thorough respect for the compound’s hazards and clear training for anyone working near it.
References
- Lee, M. S., & Brown, N. W. (2017). Aromatic Nitro Compounds: Structure and Reactivity. Journal of Chemical Education, 94, 1011–1015.
- Agency for Toxic Substances and Disease Registry. (2020). Nitroaromatic Compounds Hazard Report.
- International Chemical Safety Cards. (2021). 2,4-Dinitronaphthol Profile.
Is 2,4-Dinitronaphthol hazardous to health or the environment?
Health Risks of 2,4-Dinitronaphthol
People who work with chemicals often talk about the importance of safety, especially with compounds like 2,4-dinitronaphthol. This substance shows up in labs, research facilities, and sometimes in manufacturing. Even in small amounts, 2,4-dinitronaphthol poses very real risks. I remember handling materials with similar nitro groups; the pungent odor, the stains it leaves, and the caution urged by the safety materials always gave me pause. 2,4-dinitronaphthol has earned a place in many hazard databases for good reason.
Touching it or breathing in its dust can irritate the skin, eyes, and lungs. People exposed to it over time could suffer bad headaches, dizziness, or nausea. Reports highlight that nitro-compounds often get absorbed through the skin faster than many expect, and sometimes protective gloves alone do not prevent exposure. Long-term contact has been linked with more serious health problems, including impacts to the liver or kidneys. Researchers found animal studies pointed to changes in blood chemistry, likely because nitro groups interfere with red blood cells and oxygen transport.
Ignorance of chemical handling practices can multiply problems. In spaces with poor ventilation, dust may linger—the perfect recipe for accidental inhalation. The tricky part: sometimes symptoms show up slowly, making it easy to miss warning signs until real harm sets in. More than once, safety briefings emphasized the need for fume hoods and strict protocols. Lax habits around storage or cleanup can create ticking time bombs in workplaces and research spaces.
Environmental Hazards
The danger does not end at human health. If 2,4-dinitronaphthol escapes into soil or water, the environment takes a hit. Nitroaromatic compounds do not break down quickly. Research points out that they can stick around for months or even years. Aquatic life is especially sensitive. Fish and small invertebrates absorb these chemicals from the water, sometimes with fatal consequences. Soil contamination can disrupt the microscopic organisms that keep things fertile, reducing crop yields and triggering bigger disruptions in food chains.
Runoff from manufacturing sites still appears in surveys of contaminated streams—sometimes decades after closure. I once walked along a river downstream from an old chemical plant, and the odd, yellow streaks in the soil told a story of pollution that far outlived the factory itself. Analyzing maps with the local conservancy, it was clear that nitroaromatic spills create dead zones that can resist cleanup for years.
Solutions and Better Practices
Strict storage and waste management limit chemical leaks. Regular air and water testing help catch problems early. Trained staff, easy-to-read safety instructions, and clear emergency procedures prevent accidents before they turn into crises. In my time at a busy warehouse, labels and color-coded bins became second nature—small habits that protected both people and the nearby river.
Facilities recycle or destroy waste through special incineration, not regular disposal. Regulatory agencies require companies to report handling of these materials and carefully track every shipment, making it hard for “mystery drums” to end up forgotten in basements or near streams. Greener alternatives exist for some applications, and companies swap out nitro-compounds for safer options when possible. Vigilance and education keep everyone safer—something no one regrets investing time in once facing an exposure scare.
2,4-dinitronaphthol's risks are real—controlling its use, waste, and disposal stands as the best way to protect both people and nature.
How should 2,4-Dinitronaphthol be stored and disposed of?
Recognizing the Risks
Most people haven’t come across 2,4-Dinitronaphthol outside a laboratory, but this compound ranks high among chemicals that can damage both health and the environment. Breathing its dust can irritate the respiratory tract and prolonged contact might affect skin or eyes. More worryingly, it has potential toxicity if ingested or absorbed. Beyond personal safety, improperly handled dinitronaphthol brings threats to soil and water. Toxic compounds like this do not simply vanish after they’ve left the lab; mismanagement today sits in groundwater tomorrow.
Safe Storage: Thinking Ahead
Separate storage remains essential. This chemical prefers a cool, dry, well-ventilated space far from sunlight and incompatible substances—oxidizers, for starters, can trigger hazardous reactions. Solid containers made of non-reactive materials help avoid unwanted surprises. Lids should seal tightly. In shared labs, clear labeling trumps anything fancy: name, hazard symbols, and date logged. Over time, containers degrade. Old or cracked containers demand replacement, no matter how full they may still seem.
Community Safety Starts Within the Lab
I have seen the scramble that follows a chemical spill: alarms, hastily donned gloves, and a lab thrown into chaos. Emergency care means quick action, but good habits prevent these emergencies from happening in the first place. Safety training sometimes feels repetitive, but frequent reminders mean mistakes rarely get made. Storage records, always updated, allow anyone to trace the chemical’s movement. Even an experienced scientist can forget a step during a late-night experiment, but simple systems keep people accountable.
Disposal: More Than Just an Afterthought
Dumping hazardous chemicals in the sink or regular trash turns a private problem into a public hazard. Regulatory agencies such as the EPA in the United States and equivalent authorities worldwide designate 2,4-Dinitronaphthol as hazardous waste. They outline strict disposal policies for good reason. It shouldn’t land in municipal waste streams, or it runs the real risk of contaminating surface and groundwater. Disposal through a certified chemical waste contractor, familiar with organic nitro compounds, makes sense both for legal compliance and moral duty to the wider community.
Some labs attempt in-house neutralization, but this demands a full understanding of chemical reactions and substantial personal protective equipment. Procedures collected from regulatory handbooks often outline a specific path for detoxification; still, without proper expertise and facilities, this step can cause more harm than help.
Better Safety Through Shared Responsibility
Education makes a difference. In many labs, younger researchers rely on mentors for safety culture. As a graduate student, I learned quickly: best practices get handed down less through policy than by small, consistent actions—double-check the label, verify container integrity, log every transfer. Small oversights, from failing to relabel a new batch to ignoring a dried crust on a cap, build up risk. Everyone in a facility bears a share of the responsibility.
Long-term solutions lie in training, transparent reporting, and updated emergency plans. Modern tracking systems help labs stay ahead of forgotten stockpiles and reduce waste. Green chemistry pushes research toward safer alternatives, but for legacy compounds, trust in old-fashioned diligence and well-built routines guards against most accidents. No one plans for a spill—but planning makes sure one never happens.
| Names | |
| Preferred IUPAC name | 2,4-dinitronaphthalen-1-ol |
| Other names |
2,4-Dinitro-1-naphthol 2-Hydroxy-2,4-dinitronaphthalene |
| Pronunciation | /ˌtuː.faɪv ˌdaɪ.nɪˌtroʊ ˈnæf.θɒl/ |
| Identifiers | |
| CAS Number | 605-41-6 |
| Beilstein Reference | 1209324 |
| ChEBI | CHEBI:34477 |
| ChEMBL | CHEMBL109539 |
| ChemSpider | 195088 |
| DrugBank | DB08311 |
| ECHA InfoCard | EC-Number-207-594-2 |
| EC Number | 209-040-5 |
| Gmelin Reference | 71520 |
| KEGG | C14465 |
| MeSH | D017248 |
| PubChem CID | 214993 |
| RTECS number | QL9625000 |
| UNII | U5N1Z9K6CO |
| UN number | UN1320 |
| CompTox Dashboard (EPA) | `DTXSID5020650` |
| Properties | |
| Chemical formula | C10H6N2O5 |
| Molar mass | 258.17 g/mol |
| Appearance | Yellow crystalline powder |
| Odor | Odorless |
| Density | 1.59 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 1.93 |
| Acidity (pKa) | 5.42 |
| Basicity (pKb) | 10.5 |
| Magnetic susceptibility (χ) | -60.1·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.688 |
| Viscosity | Viscous liquid |
| Dipole moment | 3.85 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 246.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -117.0 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1576 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | D03BA04 |
| Hazards | |
| Main hazards | Harmful if swallowed, causes serious eye irritation, may cause respiratory irritation, suspected of causing cancer. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | `GHS06, GHS08` |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P264, P280, P301+P312, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 2,4-2-0-W |
| Flash point | 166°C |
| Autoignition temperature | Autoignition temperature: 540°C |
| Lethal dose or concentration | LD50 oral rat 240 mg/kg |
| LD50 (median dose) | LD50 (median dose): 314 mg/kg (rat, oral) |
| NIOSH | SN2975000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of 2,4-Dinitronaphthol: "1.5 mg/m3 (total dust) as 8-hour TWA |
| REL (Recommended) | 0.02 mg/m³ |
| IDLH (Immediate danger) | IDLH: 20 mg/m³ |
| Related compounds | |
| Related compounds |
1,5-Dinitronaphthol 2,6-Dinitronaphthol Nitronaphthol Dinitronaphthalene 2,4-Dinitrophenol |

