2,4-Dinitrobenzyl Chloride: Chemistry with Consequence
Historical Development and Product Overview
Chemicals carry stories whether those tales show up in the lab or change an entire industry. 2,4-Dinitrobenzyl chloride belongs to a group of nitroaromatic compounds that earned a mixed legacy almost a century ago. The journey of this compound kicked off during the early developments in organic synthesis, back when chemists looked for ways to tailor-make intermediates for dyes and pharmaceuticals. The nitrobenzyl family, born from the high-octane drive for better explosives and coloring agents, later bled into biochemistry. As a student in a synthetic chemistry class, holding a bottle bearing that bright yellow tinge and sharp musty bite, I remember realizing just how many chemical lineages cross through one simple reagent. Dinitrobenzyl chloride itself emerged as both a tool and a hurdle: crucial for some transformative reactions but finicky to handle.
Physical and Chemical Properties
Anyone with a lab course under their belt remembers how physical properties dictate daily choices and strategy. 2,4-Dinitrobenzyl chloride, with its pale-yellow crystals, signals caution before the first test tube even gets uncapped. Water doesn’t greet this compound warmly, as it's more at home in organic solvents like ether or chloroform. The nitro groups pumping through the molecule shift its reactivity, making it more electrophilic and, for lack of better words, testy. Heat nudges it from solid to vapor in the neighborhood close to 90 degrees Celsius, but relying on heating for purification asks for trouble, since decomposition risks grow fast. The smell sticks in memory, and the dust can provoke more than a cough—one reminder why simple gloves and a solid fume hood feel less like options and more like basic respect.
Technical Specifications and Labeling
Labeling isn’t about just ticking off boxes for compliance. With 2,4-dinitrobenzyl chloride, a label’s more than a warning—it’s a flashback for anyone who has cleaned up a spill or gotten called out for lax storage. Most containers arrive marked with the classic hazard statements, and with pictograms that hammer home irritation and sensitivity dangers. Details show purity grades, batch information, and shelf-life. One lesson that sticks with professionals: hand-marked or faded labels spell trouble. Restlessness in storage conditions, combined with its tendency to degrade, turns simple chemical management into a real operational priority.
Preparation Method
Synthesizing 2,4-dinitrobenzyl chloride isn’t just a stepwise recipe from a textbook. The route most engineers learn starts with 2,4-dinitrotoluene, which gets doused in chlorine—itself no picnic to handle. The substitution transforms a methyl side chain into the reactive chloride, and, as easy as teachers may make it sound, controlling the temperature, pressure, and byproduct formation calls for patience and discipline. Each batch brings chances for hazardous fumes or exotherms, pressuring everyone around to treat “routine synthesis” with proper attention. I’ve witnessed postgrad students staying late to babysit a reaction flask, worried that one slip in timing or cooling could foul a whole run or, worse, put others at risk.
Chemical Reactions and Modifications
Chemists see 2,4-dinitrobenzyl chloride as a genuine workhorse. That exposed benzyl chloride moiety begs for nucleophilic attack, making it a regular choice for protecting group chemistry—especially for shielding alcohols or amines while more delicate steps unfold elsewhere. Some reactions exploit those nitro groups to modulate electron flow, letting the compound act as a springboard for more radical transformations. Benzylations, displacements with soft nucleophiles, and linkage formation drive its usefulness across making dyes, drugs, and even certain pesticides. In an academic setting, I’ve seen its structure described on whiteboards dozens of times, as professors remind students how leaving groups and electron-withdrawing substituents push chemistry in more productive directions.
Synonyms and Product Names
Monikers stack up quickly in chemical supply catalogs. The same bottle might read 2,4-dinitrobenzyl chloride, DNB-Cl, or even 1-chloro-2,4-dinitrobenzene depending on how the supplier handles IUPAC rules or regional preferences. This web of names can breed confusion, especially for anyone sorting through research papers from different decades or countries. For undergrads slogging through late-night database searches, recognizing synonyms is a rite of passage. Miss that subtle variation, and a critical reference could slip through the cracks.
Safety and Operational Standards
Anyone with scars from a careless lab day knows that safety warnings on a compound like this aren’t overblown. 2,4-dinitrobenzyl chloride wants tight controls and direct respect. The compound dust, vapors, or even stubborn residue linger enough to cause respiratory and skin problems. Beyond gloves and goggles, everyone leans on exhaust hoods and careful waste disposal protocols, because breakdown products can be as ugly as the starting material. Safety data sheets point to both acute effects—coughing, burns, eye irritation—and longer-term concerns tied to regular low-level exposures. In my time overseeing undergraduate labs, it was common to see students roll their eyes before their first spill. One scare has a way of tuning attention back to protocols. Ignoring proper labeling and containment standards flips lab bustling into organized panic in seconds.
Application Areas
Chemistry supplies the backbone for life-saving drugs and less savory products alike. 2,4-Dinitrobenzyl chloride jumps between roles in organic synthesis. It’s gone into protecting group strategies, a staple trick in pharmaceutical manufacturing, where it shields certain functional groups for selective manipulation. Some paths lead it into dye manufacturing and agricultural chemistry. While it’s tempting to focus solely on its technical appeal, ethical shadows show up fast—dinitro compounds have histories both useful and harmful, from medicinal research to explosives. Experience in interdisciplinary teams, like when I tutored in a joint chemistry and pharmacology program, revealed how quickly a single intermediate can slip from laboratory miracle to environmental headache if disposal and tracking fall behind.
Research and Development
The world of chemical research feels like a perpetual series of moves and countermoves. Researchers spend months, sometimes years, exploring shortcuts for cleaner syntheses, higher yields, or safer handling—all around compounds like 2,4-dinitrobenzyl chloride. A major chunk of recent work pivots on greener synthesis, cutting down on harsher reagents, or swapping out chlorination pathways for less hazardous ones. Industrial chemists and regulatory experts keep searching for new angles to reduce human and ecosystem exposure. Even at the graduate level, debates rage about the best alternatives or replacements, as labs and companies try to meet regulatory shakedowns. I’ve seen team meetings where a promising new catalyst or unexpected property shifts research focus overnight.
Toxicity Research
Risk isn’t just academic for anyone who has watched a colleague deal with an accidental splash or inhalation. Toxicology data on 2,4-dinitrobenzyl chloride isn’t kind. The nitro groups themselves, common to many hazardous compounds, raise flags—these structures can cause systemic toxicity, mutagenic risks, and in some cases, ugly surprises in animal models. Chronic low levels pose particular worry for operators who spend years with open bottles and poor ventilation. More studies target metabolic breakdown in soil and water, tracking persistent byproducts and their dangers for both humans and local fauna. Whenever new workers get briefed on this compound, the session shifts quickly from technical vocabulary to straight talk about gloves, masks, and why you never cut corners—even for “just a quick bottling.”
Future Prospects
Shifting from the legacy of crude syntheses to more responsible management shapes the outlook for 2,4-dinitrobenzyl chloride. Chemists face mounting pressure to swap out legacy reagents, either because of safety, legal scrutiny, or green chemistry mandates. As analytical techniques keep improving, detecting trace exposures gets easier, and new limits pop up on what’s considered acceptable risk. The future for compounds like this doesn’t vanish overnight, though. Established manufacturing routines, specialized synthetic pathways, and legacy research habits mean adaptation takes time. I’ve watched as once-ubiquitous chemicals slowly fade as new, safer reagents step into the spotlight. Foresight, ongoing education, and prudent substitution shape the only realistic path forward for compounds with tricky histories and enduring utility. The world doesn’t stand still, and trusted old reagents pick up new responsibilities alongside new restrictions.
What is 2,4-Dinitrobenzyl Chloride used for?
Chemistry Goes Beyond the Textbook
Chemicals like 2,4-Dinitrobenzyl chloride don’t usually spark much conversation outside of laboratory circles, but they play a bigger role in the world than most folks imagine. This compound, with its fairly menacing name and strong punch of bright yellow crystals, often finds itself at the heart of many research-driven processes. In my years spent watching benches covered in glassware and slightly nervous graduate students, I’ve seen the compound pop up in a handful of interesting applications.
A Key Player in Making the Building Blocks
Ask anyone who’s worked a few years in a synthetic chemistry lab and they’ll mention how benzyl chlorides make things happen. Among them, 2,4-Dinitrobenzyl chloride acts like a reactive powerhouse. Its strong electron-withdrawing nitro groups set up the molecule for making connections that others struggle with. I remember working on a project that needed to introduce protecting groups – those little “shields” for parts of a molecule you don’t want to react. This compound makes a good benzyl protecting group, especially for alcohols and amines. It stands out for its reliability and straightforward removal later in the synthesis process.
This usefulness extends into the pharmaceutical world. Some complex drugs and candidate molecules need parts of their structure temporarily blocked or modified. Medicinal chemists, aiming for clean and controlled reactions, often rely on this reagent. It gives them a tool that works, even when other protecting groups can’t take the heat of more rigorous stages of drug design.
Pushing Research and Development Forward
The world of academic research likes 2,4-Dinitrobenzyl chloride, especially in making custom-designed chemical probes and substrates. In biochemical studies, scientists want to track or control certain reactions inside cells. This chemical can help create molecules that light up or turn “on” in response to a specific trigger, thanks to its unique reactivity. I remember coming across a publication that used the compound to design enzyme assays, giving researchers better insight into the complex web of cellular life.
This approach means scientists can test new drugs or understand diseases faster. Well-known companies and government agencies invest millions each year in reagents that streamline and sharpen these experiments. They don’t do that lightly; instead, they follow the path to what’s proven to work.
Risks and Responsibility
Just because something works in the lab doesn't mean it belongs everywhere. 2,4-Dinitrobenzyl chloride brings its own batch of health and safety concerns. Its high reactivity means anyone handling it has to treat it with real respect. Skin contact burns, inhaling fumes turns into a nightmare, and spills become a panic. Those lessons get drilled in early for anyone working with it, and I’ve known more than one person forced to take a break after exposure.
There’s also a shadow that hangs over some uses. The sensitive, react-with-almost-anything nature means bad actors covet this kind of chemical for making illicit substances. Regulations already limit supply, but unscrupulous sourcing still takes place. Open conversations around supply chain transparency and routine checks stand as the best ways to catch lapses before trouble starts.
What Can Be Done?
Smart labs treat storage, use, and disposal as a major priority. Regular audits, rigorous training, and total traceability limit accidents. Leading companies and universities invest in green chemistry alternatives where possible, and stronger international rules keep distribution in check. With knowledge, control, and respect, 2,4-Dinitrobenzyl chloride can keep fueling discovery without adding more problems than solutions.
What are the safety precautions when handling 2,4-Dinitrobenzyl Chloride?
Why 2,4-Dinitrobenzyl Chloride Matters for Lab Safety
2,4-Dinitrobenzyl chloride stands out in a chemical storeroom. The yellow solid signals trouble—skin, eyes, and lungs don’t like it. Touching this stuff can leave you with burns or rashes, while inhaling the dust or vapors can irritate your throat or make it hard to breathe. It’s not a substance that excuses carelessness, especially with its toxic and potentially explosive profile. Anyone who has spent long afternoons coaxing reactions out of stubborn reagents knows some chemicals require more respect than others; this is one of them.
Personal Experience in the Lab
My first job working with nitrated aromatic compounds taught me not to gamble with basic safety. Colleagues joked about “the nitro sniff” when opening the bottle—even brief contact with your skin burns for hours, and a single sneeze can turn a hood into chaos if protective wear isn’t perfect. People get used to shortcuts, but all it takes is one fumbled glove for a week of headaches, or worse, a trip to the hospital for chemical burns. Nobody forgets the time a bottle spilled during a summer internship—cleanup stopped everything for a day, and no one missed the message.
Basic Protective Gear: No Exceptions
Nitrile gloves go on before you open the bottle. Strap on goggles with side shields; a face shield works even better. A cotton lab coat covers everything, snapped right up to your neck—synthetics melt if hit by solvent splash. Respirators don’t always feel necessary, but air extraction systems rarely clear fumes as fast as you think. Respiratory cartridges marked for organic vapors keep you out of trouble if proper fume hoods aren’t strong enough.
Setting Up for Success: Preparing the Workspace
Before measuring out the chemical, check your fume hood. Put a piece of tissue near the sash to test airflow; the paper should blow inward. An open window doesn’t count. Trays and absorbent pads below your beaker catch drips that always seem to escape, even on good days. Only set out what you plan to use; clutter multiplies accidents. Keep wash bottles filled with water or ethanol within arm’s reach to rinse skin or bench surfaces, but never fall back on winded improvisation. Label all containers, and never use food containers or unmarked flasks.
Handling, Storage, and Disposal
Carefully scoop or pour with tools reserved just for hazardous solids. Don’t transfer with bare hands or in a rush. Seal bottles tight right after use and store them away from sunlight or any heat source, inside a vented cabinet meant for toxic solids. Mixing waste doesn’t end well—collect liquids and solids in separate, labeled waste bottles, with date and name. The department’s hazardous waste officer should schedule frequent pick-ups; nobody wants a box full of leaky, ancient chemicals forgotten in a dark closet.
What Strong Safety Really Means
Trying to outsmart 2,4-dinitrobenzyl chloride never ends well. Even experienced chemists talk openly about near-misses or overconfidence. Good practices multiply your confidence and keep accident reports off your desk. Read the safety data sheet before you work, and don’t hesitate to ask for help organizing safe workflow. Properly enforced rules make sure everyone gets home with all their fingers, free from burns or worse.
How should 2,4-Dinitrobenzyl Chloride be stored?
Knowing What You Handle
Working with chemicals like 2,4-Dinitrobenzyl Chloride has taught me to stay alert. This compound doesn’t forgive careless habits: it causes irritation, reacts sharply with moisture, and gives off toxic fumes if it breaks down. A sniffle becomes more than just a cold in the lab, and gloves start feeling more like an investment than a formality. If you’ve ever opened a poorly stored container, you’ll remember that sharp, acrid slap in the nose. Lessons stick when they hurt.
Why Dry and Cool Storage Matters
Most seasoned lab workers will tell you: moisture ruins more than just your lunch. With 2,4-Dinitrobenzyl Chloride, water spells disaster. This compound hydrolyzes, turning into something more hazardous. I once saw a supervisor toss a careless container straight to hazardous waste after finding condensation inside. The fix is simple—keep it dry. Desiccators, well-sealed bottles, and a clean, clearly labeled area do the heavy lifting. Forgetting silica gel or ignoring a cracked cap means losing both product and safety.
Temperature control always comes up in storage discussions. The compound decomposes if left in the heat. Cold storage, around 2-8°C, keeps things stable. Too cold, though, and you deal with glass cracks or leaking stoppers. I keep eye-level thermometers, and every serious chemist I know tapes daily range checklists to the fridge door. A working refrigerator for chemicals isn’t just a nice-to-have—it prevents real headaches.
Ventilation and Segregation
Storing strong chemicals together brings real trouble. Organic chlorides, acids, and bases react in surprising ways. Keeping 2,4-Dinitrobenzyl Chloride away from acids, strong reducers, and open flames sounds obvious. Yet, overfilled shelves and lazy labeling breed confusion. I’ve spent late evenings sorting out “mystery” jars, tracing faded marks, and making sure incompatible bottles don’t sit shoulder-to-shoulder. Separate flammable storage cabinets, complete with automatic locks, do more than tick a box on the safety audit.
Ventilation keeps toxic fumes under control. Even a sealed bottle can seep out vapors if handled roughly or left too long. Chemical storage never belongs in a sealed, windowless closet. Airflow matters. A few years ago, our lab upgraded from drafty cupboards to proper vented cabinets—no more headaches, and no scary surges in air quality readings. That change turned storage from a daily risk into simple routine.
Clear Labeling and Emergency Preparedness
Everyone slips now and then, but mislabeling costs more time and money. Permanent markers, printed stickers, and bold pictograms cut confusion during busy shifts. Emergency gear within arm’s reach—a full eyewash, gloves you trust, and a clear spill kit—keeps everyone ready for the unexpected. Even veterans forget protocol during a real spill, so regular drills and visible instructions become lifelines. A safe storage habit grows from small, consistent practices. Good habits get reinforced the moment something goes wrong and you’re glad you took the extra step.
Common Sense Beats Panic
Safety isn’t about fear; it’s about control. If you find yourself second-guessing where a chemical fits or how it behaves, check the latest safety datasheet. Talk with experienced colleagues who’ve learned lessons the hard way. No substitute for daily vigilance exists, and nobody should leave safety up to hope or luck.
What is the chemical structure and molecular formula of 2,4-Dinitrobenzyl Chloride?
Chemical Structure and Formula
2,4-Dinitrobenzyl chloride draws its name from its benzene core, with two nitro groups and a chloromethyl group dancing around the ring. Picture a benzene ring. Add nitro groups (-NO2) at the second and fourth positions. Then attach a chloromethyl group (-CH2Cl) at the first spot, right on the ring’s edge.
The full molecular formula stands as C7H5ClN2O4. Lay it out on paper and the structure packs a punch: the electron-withdrawing nitro groups pull electron density away from the ring, while the chloromethyl group gives the compound its reactivity. Synthetic labs remember handling this molecule with great care — those same nitro groups that make nitration reactions possible also raise concerns for health and safety. It’s not just about numbers on a sheet, it’s about understanding how simple changes in structure can turn an aromatic ring into something packed with energy, both literal and chemical.
Why Structure Matters
Anyone who’s ever run a reaction with a benzyl chloride derivative knows the impact of electron-withdrawing groups like nitro. The presence of two such groups ramps up the compound’s activity as an alkylating agent. You get a molecule that reacts quickly and sometimes more aggressively, altering how chemists select their solvents, conditions, and even personal protective gear. A little oversight, and instead of a smooth alkylation, you’re troubleshooting side reactions or tossing away precious product. Every high school chemistry textbook talks about “activating and deactivating groups.” You see it play out in real life with compounds like 2,4-dinitrobenzyl chloride. The electron-withdrawing power sits front and center, and you can feel it with every small change in your workflow.
Uses and Hazards Go Hand-in-Hand
Because of this molecule’s structure, it shines as a reagent. It’s a useful player in the protection of amino groups during peptide synthesis, giving researchers a handle to add or take away at the right moment. The same features that make it go-to for scientists also bring caution. Several colleagues remember stories from organic labs: sharp smells, warnings about inhalation, and protective gloves as standard. There’s no glossing over its toxicity or irritancy. Good ventilation and awareness keep the molecule useful, not dangerous.
The nitro groups don’t only bring reactivity, they also raise environmental and health concerns. Waste products containing nitroaromatics need specialized disposal. Regular chemical disposal routines don’t cut it. Miss a step, and you might trigger environmental oversight or even safety shutdowns. It’s a pattern repeated in industrial settings where carelessness can turn a synthetic feat into a regulatory headache.
Solutions Rooted in Experience
Years spent in both academic and industrial labs taught me the value of appreciating not just the structure, but also the real working risks. Stricter safety training, clear labeling, improved extraction procedures — these aren’t bureaucratic boxes to tick, they keep you working safely. A practical tip: closed reaction vessels and local fume extraction systems cut down on vapors and spills. On top of that, investing in robust training for newcomers saves time and resources. It stops trouble before it starts.
For those handling disposal, treating waste streams on-site before release, and working with trusted third-party firms, makes a world of difference. Staying informed about green chemistry alternatives may shrink the role of toxic reagents over time, but for now, handling 2,4-dinitrobenzyl chloride safely lets its strengths shine without letting the drawbacks take over the story.
What are the potential health hazards of 2,4-Dinitrobenzyl Chloride?
What Is 2,4-Dinitrobenzyl Chloride?
2,4-Dinitrobenzyl chloride crops up often in chemical labs. Teams working in organic synthesis know it as a reactive tool. It has a yellowish look and gives off an irritating odor that reminds me of how some paints make your nose sting. While I have spent time in labs handling tough chemicals, I always felt a prickle of worry around this one. There is a good reason. This compound raises several red flags regarding human health.
Health Risks from Direct Exposure
Anyone exposed to 2,4-Dinitrobenzyl chloride can quickly run into trouble. If the vapors get into your eyes or if you touch the material, expect irritation. Pain, redness, burning sensations all follow. Immediate flushing with water usually helps, but sometimes you do not notice until it is too late. Chronic exposure can make these symptoms linger. I have seen colleagues experience rashes or start coughing after a poorly sealed bottle let some vapor escape.
Inhaling this compound brings special risks. It irritates the respiratory tract, causing symptoms like sore throat, chest tightness or short breath. At higher exposure, dizziness or headaches show up. There is always the threat of long-term effects, which stick around even after the exposure ends. Prolonged breathing of dust or fumes could lay the groundwork for chronic respiratory troubles. The nitro group in the structure spells extra danger, known to spark oxidative stress in tissues. Scientists have linked this type of damage to an increased risk of cancer or systemic toxicity.
Damage to Organs and Chronic Toxicity
Absorption of 2,4-Dinitrobenzyl chloride through skin or inhalation can end up affecting more than just the area of contact. Once inside the body, it can accumulate and disrupt internal organs. Animal studies indicate this chemical targets the liver and kidneys. Repeated exposure caused darkened urine or jaundice in lab rats—clear signals that the body is struggling to detoxify the compound. The nitro groups may generate methaemoglobin in the blood, which reduces the ability of blood to carry oxygen. In practical terms, workers could develop fatigue or confusion after regular contact, even if they don’t recall a direct splash or spill.
On-the-Ground Realities: Who Faces the Greatest Risk?
From my time working with research chemicals, graduate students and industrial workers face the sharp end of the stick here. Ventilation systems can only do so much, and small spills often get overlooked in busy labs. I have heard of janitorial staff getting exposed simply by taking out the trash. Even people outside the lab are not off the hook. If this compound escapes during production or transport, it could contaminate community air or water supplies.
Strategies to Reduce Harm
Strong safety training forms the backbone of harm reduction. Everyone handling this compound should suit up with goggles, gloves, and face shields. Chemical fume hoods offer a real shield—not just for volatile fumes but for those accidental splashes that nobody expects. Labeling and storage deserve relentless attention. I remember putting extra reminders on containers to keep everyone alert. Quick access to emergency showers and eyewash stations turns a disaster into a minor scare. Environmental controls matter, too. Regular air monitoring stops problems from becoming chronic hazards. Some labs double wrap their waste and contract out specialized disposal to prevent contamination of regular trash streams.
Final Thoughts
2,4-Dinitrobenzyl chloride offers useful chemistry but punishes carelessness. Awareness, respect for the risks, and clear protocols save lives. Across labs, factories, and neighborhoods, prioritizing education and investment in controls keeps people healthy so the research and industry can keep moving forward—without sacrificing anyone’s safety along the way.
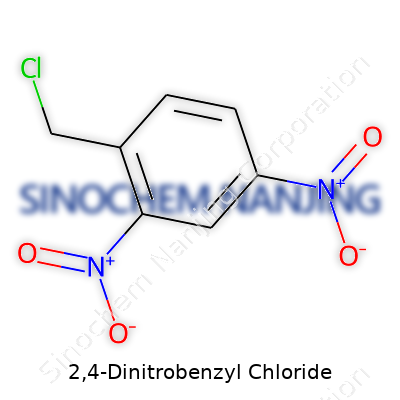
| Names | |
| Preferred IUPAC name | 1-chloro-2,4-dinitrobenzene |
| Other names |
2,4-Dinitrochlorobenzene 1-Chloro-2,4-dinitrobenzene 2,4-DNBCl 2,4-Dinitrobenzylchloride |
| Pronunciation | /ˌtuː fɔːr daɪˈnaɪtroʊˈbɛnziːl ˈklɔːraɪd/ |
| Identifiers | |
| CAS Number | 89-61-2 |
| Beilstein Reference | 87883 |
| ChEBI | CHEBI:51841 |
| ChEMBL | CHEMBL15479 |
| ChemSpider | 17501 |
| DrugBank | DB14106 |
| ECHA InfoCard | 03bce425-36fb-4da3-89d4-2fcfd0a6562b |
| EC Number | 202-863-0 |
| Gmelin Reference | 186158 |
| KEGG | C13999 |
| MeSH | D000070658 |
| PubChem CID | 153325 |
| RTECS number | DH9625000 |
| UNII | 9V5Y53L9DT |
| UN number | UN1576 |
| CompTox Dashboard (EPA) | DJ37T8202K |
| Properties | |
| Chemical formula | C7H5ClN2O4 |
| Molar mass | 202.55 g/mol |
| Appearance | Yellow crystalline powder |
| Odor | Odorless |
| Density | 1.571 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | 1.86 |
| Vapor pressure | 1 mmHg (108°C) |
| Acidity (pKa) | 12.4 |
| Basicity (pKb) | 10.86 |
| Magnetic susceptibility (χ) | -42.5·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.639 |
| Viscosity | Viscous liquid |
| Dipole moment | 3.97 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 333.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -34.1 kJ/mol |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and eye irritation, may cause respiratory irritation, suspected of causing genetic defects. |
| GHS labelling | GHS02, GHS05, GHS06, GHS08 |
| Pictograms | GHS02,GHS06,GHS07 |
| Signal word | Warning |
| Hazard statements | Hazard statements: H302, H312, H315, H319, H332, H335, H400 |
| Precautionary statements | P261, P264, P271, P280, P301+P312, P305+P351+P338, P308+P313, P330, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-1-F |
| Flash point | 93 °C |
| Autoignition temperature | Unknown |
| Lethal dose or concentration | LD₅₀ (oral, rat): 360 mg/kg |
| LD50 (median dose) | LD50 (median dose): 750 mg/kg (rat, oral) |
| NIOSH | DN1225000 |
| PEL (Permissible) | PEL: 1 mg/m3 |
| REL (Recommended) | 0.1 mg/m³ |
| Related compounds | |
| Related compounds |
2,4-Dinitrochlorobenzene 2,4-Dinitrobenzoic acid 2,4-Dinitrobenzyl alcohol 2,4-Dinitrotoluene Benzyl chloride |

