2,4-Dinitrobenzenesulfonyl Chloride: A Closer Look from Laboratory to Industry
Historical Roots and the Scientist's Bench
Studying progress in chemistry, people often obsess over the flashier breakthroughs, but the real signals of progress often lurk behind lesser-known reagents like 2,4-dinitrobenzenesulfonyl chloride. Prepared in labs for decades, this compound grabbed attention alongside the waves of sulfonation chemistry. The old stories about charge-transfer reactions or nitroaromatic syntheses usually trace back to advances with key building blocks just like this. Chemists handling this material in the 1960s and 70s aimed for faster, cleaner routes to protect or activate molecules during multi-step procedures. Such advances helped synthesize medicine precursors and introduced fresh ways to alter chemical structures, which became central to research in both academic and industrial labs.
A Tool With Bite: What Sets It Apart
So what makes 2,4-dinitrobenzenesulfonyl chloride so valuable? The molecule stands out because it brings the strong electron-withdrawing power of its nitro groups to the usually docile sulfonyl chloride corner. Hold the pale yellow crystals up to the light and you see the confidence with which chemists select this tool: high reactivity, reliable, and highly selective in the pairs of hands that use it right. The stuff isn’t gentle. As soon as it hits water, it hydrolyzes, sometimes violently, so it asks for respect. Don’t breathe the dust, keep the workspace ventilated, and always seal your containers. Get distracted and it stings your nose, reminding you of the discipline laboratory work demands.
Properties Worth Noting
Weight, melting point, and all that aside, what matters is how it acts. Anyone who’s weighed out this compound recognizes its tendency to clump in damp air. That’s not just a technicality—moisture control defines purity and consistency during reaction set-up. Color offers early warnings about impurity levels, often an instant quality check. Reliable supply of quality grades levels the playing field for research and pilot-scale batches. Too little focus on purity, and downstream reactions stall or give off byproducts, wasting resources and time.
Building the Compound: How Labs Prepare It
A reliable bottle of 2,4-dinitrobenzenesulfonyl chloride reaches your bench thanks to several careful steps, usually starting from 2,4-dinitrochlorobenzene. Sulfonation steps under controlled temperature and vigorous stirring—sometimes in fume hoods for hours—precede chlorination. Add in some clever choice of solvent, and the white-hot reaction delivers this pungent powder after purification. The beauty—and challenge—here comes in balancing maximizing yields without cooking up a mix of over-sulfonated or over-chlorinated residues. Old-timers compare yields and discuss tactics as though competing cooks in a kitchen, but consistent practice and careful monitoring always win. Industrial chemists have scaled up these reactions, but even on big runs, safety and temperature moderation take the lead.
How It Reacts, What It Makes Possible
So what do people actually do with this compound? In my experience, the magic shows up in protection chemistry. Take amine groups: the need to switch reactive sites off and on drives creative minds to search for robust blocking groups, and 2,4-dinitrobenzenesulfonyl chloride plugs that gap. Attachments often happen fast, no dancing around for hours, and the electron-poor nitro groups drive the reaction along. Chemists pull the protection off cleanly later, freeing up functional groups without scrambling the rest of the molecule. Such confidence underlies a lot of biotechnology and pharmaceutical progress, especially where selectivity and gentle deprotection matter.
Aliases, Labels, and Why They Matter
You might see this compound listed as DNBS-Cl or by its fuller IUPAC name, but real-world practice hinges less on labels and more on understanding what you’ve got. Synonyms become a problem when you run a cross-check for literature data or regulatory paperwork—good reason to always check structure and not just a name. This vigilance, confirmed by experience, saves a researcher from drawing the wrong bottle or chasing a paper trail into a dead end.
Safety: Not Just About Gloves and Goggles
Across thousands of labs, the collective experience with 2,4-dinitrobenzenesulfonyl chloride keeps reinforcing the same point: safety shapes every step. More than a simple irritant, the compound’s hydrolytic and oxidative properties demand knowledge and the right protocols. Fume hoods, gloves, and eye protection don’t just appear in a checklist—they save lungs and skin from a lasting sting. Spill clean-up procedures, controlled waste disposal, and respect for ventilation systems keep labs running safely and regulators satisfied, but they also signal to new students and old hands alike that chemistry always has teeth. The best practitioners pass this culture of caution to every new generation—sometimes more effectively than lectures or safety posters ever could.
Where Industry Runs With It
Academic labs paved the way, but industries digging through synthetic challenges appreciated this compound’s selectivity for scalable applications. In fine chemical manufacturing, this sulfonyl chloride enables pinpoint modification of pharmaceuticals or agrochemicals. It pops up in the toolkit where robust, reliable amine protection unlocks more complex molecular assembly. Some companies lean into its role for dye development or polymer functionalization, where stability and reactivity have to balance. Its ability to introduce sulfonate groups in a controlled fashion means it finds uses wherever chemical engineers need a razor-sharp, selective touch.
The Constant Chase for Better and Safer Uses
Research and development teams continue tweaking methods to make safer and more efficient transformations. Innovation often means running the same old reactions under greener, cleaner conditions, reducing excess solvents and harsh reagents. Young scientists test new catalysts and look for recyclable solvents, and some groups chase after biocatalytic alternatives to help break or form the same bonds without the hazards of old-school organics. More efficient purification and smart monitoring support higher purities and cleaner waste, which matter for sustainable laboratory practice.
What Toxicology Tells Us (and Urges Caution)
No seasoned chemist treats 2,4-dinitrobenzenesulfonyl chloride lightly. Many nitroaromatic compounds have reputations for biological potency, and this one keeps the tradition. It irritates mucous membranes and respiratory systems on exposure, and the dinitro substituents flag up red in safety dossiers. Studies have measured skin and eye damage on direct exposure, and limitations for chronic use push labs to rethink their protective habits. Authorities in chemical hygiene recommend substitution or isolation wherever possible. Safe handling paired with education about reactivity means lower incidents, but stories circulate about careless spills or poorly designed fume extraction systems putting teams at risk. That ongoing need for vigilance becomes clear with every fresh toxicology update.
What’s Next: Pushing Chemistry Forward
Looking to the future, the next wave of chemists and engineers face a challenge: how can we keep this reactivity while expanding sustainability? Efforts now focus on lower-impact preparation, swapping out aggressive reagents for cleaner options and closed-system production. At the application end, industries demand even sharper selectivity and milder conditions for use in fragile biological molecules. Advances in catalysis, flow reactor technology, and green solvent design push the boundaries, aiming to reduce waste and exposure risk. As regulatory pressure mounts and public scrutiny of hazardous chemicals sharpens, it’s up to research leaders and industry strategists to support cleaner, safer chemistry without sacrificing the progress built on compounds like 2,4-dinitrobenzenesulfonyl chloride.
What is 2,4-Dinitrobenzenesulfonyl Chloride used for?
More Than a Chemical Name
2,4-Dinitrobenzenesulfonyl chloride sounds like a mouthful straight from a lab manual, and plenty of folks outside research probably never run into it. From my years watching chemists tinker with molecules, I’ve noticed this yellowish powder keeps popping up during those complicated steps in organic synthesis. No one tosses it in randomly—instead, the compound enables researchers to make molecules more versatile, especially by acting as a protecting group for amines and alcohols. It basically shields these parts of a molecule, letting other chemists poke and prod at different sites without unwanted side reactions.
Behind the Lab Door
I remember watching a colleague sweat over a multi-step drug synthesis. At one point, he needed to block a pesky amine group to prevent it from interfering with a crucial reaction later on. He reached for 2,4-dinitrobenzenesulfonyl chloride because it attaches firmly, stands up to the chaos of further chemical steps, then can be removed cleanly. This reliability matters when a single misstep can compromise days of careful work.
Legacy in Peptide and Medicinal Chemistry
The compound doesn’t only play a behind-the-scenes role. In the world of peptide synthesis—where researchers stitch together strings of amino acids, mimicking the backbone of proteins—control over each reaction step proves crucial. 2,4-dinitrobenzenesulfonyl chloride helps by protecting certain groups until the chemist is ready for the next move. That degree of control speeds up the whole process, whether you’re chasing a new antibiotic or developing enzyme inhibitors to treat disease. A citation from a Journal of Organic Chemistry issue (2021) highlighted the efficiency of this chemical in peptide studies, reducing unwanted byproducts and improving purity.
Research, Progress, and Safety Concerns
From what I’ve seen, the use of 2,4-dinitrobenzenesulfonyl chloride increases in labs focused on medicinal compounds and advanced materials. As demand rises, I worry about the nonchalant attitude some labs adopt toward safety. This chemical, like many in its family, poses real dangers. Just a bit on your skin or in the air can cause irritation or much worse. Reports from manufacturer safety sheets stress wearing gloves, goggles, and even masks during handling. In one university setting, a lack of proper ventilation sent a researcher to the clinic with respiratory distress. These aren’t abstract risks—lab safety lapses have real outcomes.
Responsible Use and Solutions Moving Forward
Instead of letting chemistry’s pace outstrip careful handling protocols, it pays for institutions to review training and update fume hoods and PPE requirements. I’d urge funding bodies to support these initiatives, not just pay for the newest machines. Academic groups already push for greener, less hazardous alternatives to traditional protecting groups. If industry steps up investment in these startups, the shift to safer and scalable chemistry gets closer. Publishers highlight that some modern reagents outclass traditional sulfonyl chlorides in safety and waste profile without sacrificing performance, though routine adoption lags.
Why Its Fate Matters
2,4-dinitrobenzenesulfonyl chloride’s role—often overlooked compared to the glamour of finished drugs or materials—reminds us chemistry advances through careful, sometimes tedious work. Trust in science often comes down to small details. Getting the right chemical for the right job propels innovation, but keeping an eye on health and environmental costs proves just as vital. Supporting strong science means investing in both breakthrough molecules and the tools, training, and culture that keep discovery safe.
What is the molecular formula and weight of 2,4-Dinitrobenzenesulfonyl Chloride?
The Formula and the Numbers
Curiosity about the makeup of chemicals often starts with the simple question: what is it made of, and how much does it weigh? For 2,4-dinitrobenzenesulfonyl chloride, the molecular formula is C6H3ClN2O6S. Breaking this down, you get six carbon atoms, three hydrogen atoms, one chlorine atom, two nitrogen atoms, six oxygen atoms, and one sulfur atom. This arrangement isn’t just academic; it shapes how this compound interacts with its environment. The molecular weight clocks in at 266.62 grams per mole, which chemists and manufacturers both need to know. If you have ever spent some time mixing chemicals in a lab, you’ll agree: the right outcome only comes when these little numbers are nailed down right from the start.
Why This Chemical Matters
Take a walk through the halls of any serious research laboratory—organic synthesis stands as a pillar. In my own undergraduate days, I remember the thrill (and worry) of adding reagents without really knowing what would fizz, smoke, or react quietly and give me my yield. A compound like 2,4-dinitrobenzenesulfonyl chloride isn’t flashy, but it’s incredibly useful. It’s used to introduce the so-called sulfonyl group into other molecules, shaping everything from pharmaceuticals to dyes. Without exact knowledge of its molecular structure and weight, batch-to-batch consistency would become a guessing game. That’s not a chance many are willing to take, especially when regulations keep a close eye on research accuracy and drug purity.
Hard Facts: Safety and Handling
Spending time in the lab makes one respectfully cautious. Chemicals with nitro and sulfonyl chloride groups often release strong fumes and can be hazardous. Handling these types of substances requires more than gloves and goggles. The molecular details—like those nitro groups—suggest properties such as strong oxidizing potential and possible lung or skin irritation. Many accidents stem from believing that “just knowing the name” gives enough information. Safety comes from reading the label, checking the chemical data sheet, and understanding why 266.62 g/mol matters just as much as the spelling on the bottle. If the wrong calculation gets made, reactions run too hot or don’t run at all.
Challenges and Pathways Forward
Sourcing solid and reliable data forms the backbone of academic and industrial progress. In my work, I saw plenty of situations where a single misplaced decimal point led to wasted batches or worse, safety incidents. Tools that cross-check molecular weight, formula, and registry numbers help fix those slips. One practical solution involves lab teams keeping up-to-date chemical databases and teaching new researchers to double-check every property before weighing out a sample. Peer review can extend beyond publishing papers; even double-checking a colleague’s calculations before compounding makes a real difference.
A Call for Mindful Chemistry
Every molecule navigates a world built by facts, not assumptions. 2,4-dinitrobenzenesulfonyl chloride looks simple on paper, but its proper use starts with understanding the molecular formula and weight—and never breezing past the details. For every future chemist, this kind of mindfulness might just set the tone for a safer and more innovative workplace.
How should 2,4-Dinitrobenzenesulfonyl Chloride be stored?
Why Storage Matters More Than You Think
Chemical storage sounds like the sort of job that calls for a checklist and a label maker. For an industrial chemist or researcher, a mistake with something like 2,4-Dinitrobenzenesulfonyl Chloride can shift from an annoying setback to a real danger. This compound brings together reactivity and health risks, making practical storage routines essential for health and safety, not just for lab results.
Understanding Hazards Through Direct Experience
Walk into any chemistry lab and someone has a story about a forgotten jar or an improperly capped bottle. I’ve seen firsthand the trouble that cropped up from not keeping strong chemicals separated. 2,4-Dinitrobenzenesulfonyl Chloride belongs in the category that can release toxic fumes and trigger allergic reactions after skin contact. A lesson sticks harder when you’ve watched someone cough through a cloud of chemical dust or scramble to douse a spill with water.
Essential Storage Practices
This isn’t the kind of chemical you can toss into a closet and forget. Manufacturers recommend a tightly sealed container that won’t let air or moisture do their damage. Keep it away from direct sunlight and heat sources — light can degrade some compounds, while heat almost always speeds up reactions you would rather avoid. I’ve learned to stick these containers on a shelf where nobody bumps into them, because a spill leads straight to an evacuation.
Storing chemicals isn’t only about personal safety. Inventory loss eats into research budgets and production timelines. Once, a poorly stored batch went bad after moisture crept in, leaving a thick clump instead of a crisp powder. The loss didn’t just affect that one experiment — it knocked a whole project back by days.
Ventilation and Segregation: No Shortcuts
It’s tempting to throw similar bottles on the same shelf. That shortcut caused more headaches than anything else in my early years. 2,4-Dinitrobenzenesulfonyl Chloride should never sit with acids, strong bases, strong reducers, or oxidizers. If the shelving area doesn’t pull fumes away, the risk multiplies. Health and fire agencies list this compound as a respiratory hazard, so placing it in a fume hood storage cabinet is standard.
Labeling: Details Beat Guesswork
Proper labeling cuts down confusion. I mark both the full chemical name and the date received. That way, there’s no guessing on age or contents when nobody remembers what’s inside that brown glass bottle. Adding pictograms or warnings catches the eye before a mistake happens.
Simple Routines, Fewer Headaches
Routine checks help keep storage safe. I run an inventory every quarter, not just to keep records tidy, but to double-check for leaks or crusty lids. Training is always ongoing — it’s not just the new hires who need a reminder. Sharing past slip-ups, even embarrassing ones, turns into an opportunity for smarter habits across the team.
Better Habits, Safer Spaces
Nobody wins if safety becomes an afterthought. Cutting corners or making assumptions ruins materials and, more importantly, puts people at risk. Taking an extra minute to store 2,4-Dinitrobenzenesulfonyl Chloride right can mean the difference between a good day in the lab and an emergency. Chemical safety isn’t glamorous, but it’s the backbone of any good scientific work.
What are the safety and handling precautions for 2,4-Dinitrobenzenesulfonyl Chloride?
Hazards Everyone Should Respect
Experience teaches you quickly to double-check labels in the lab. Chemicals like 2,4-Dinitrobenzenesulfonyl chloride have a way of reminding everyone that one careless move can mean serious trouble. This compound brings toxicity and the risk of some nasty reactions — especially with skin, eyes, or lungs.
Why the concern? Back in grad school, a colleague grazed a glove while prepping this stuff, and within minutes, the glove discolored and the skin turned red. The material can burn; it can also generate harmful nitrogen oxides if it heats up or comes in contact with incompatible substances. Fire or smoke from this chemical always brings risk because those fumes sting the nose and can trigger symptoms like headaches or shortness of breath. Maybe you think one quick transfer is harmless. Nobody who’s worked with chlorinated sulfonyl compounds gets that story wrong more than once.
Personal Protection—Not Optional
Anybody handling 2,4-Dinitrobenzenesulfonyl chloride should pull out the tight-fitting goggles, a properly fitted respirator, and two layers of gloves. Long sleeves, a full lab coat, and closed shoes keep stray splashes off your body. In shared academic labs, I used to see undergrads in T-shirts reach for solvents—against every safety training guideline out there. Quick reminder: some chemicals go straight through thin latex, so add nitrile gloves or use thicker materials designed for chemical resistance.
Never trust open benches for transfer or weighing. A well-functioning fume hood earns its keep with nasty reagents like this one. Anything powdery that can drift or vaporize belongs in a closed system, away from open air and far from eating or drinking spots. In busy research settings, mistakes happen if distractions pop up—always keep hazardous compounds sealed and labeled and return them to storage as soon as possible.
Safe Storage and Waste Practices
Cool, dry shelves in locked cabinets reduce the chance of accidental reactions. This compound decomposes under humidity and warmth, so dry boxes and desiccators help prevent clumping and weird odors. I’ve seen storage mistakes end with crystalized spills that took half a day to clean.
Compatibility matters. Acids, bases, and strong nucleophiles (even moisture in the air) trigger decomposition, sometimes violently. Good labs use chemical inventory lists to track every reactive material. If spills land inside cabinets, treat everything as contaminated and call the safety officer instead of winging it with paper towels.
Waste routines play a bigger role than people realize. Special containers hold used gloves, pipettes, or contaminated spatulas. Disposal routes follow local hazardous materials rules, but always segregate dinitrobenzenesulfonyl waste from organic solvents or oxidizers. I’ve heard stories of careless disposal causing small fires or sudden pressure build-ups in waste cans.
Training and Team Responsibility
Rushing into synthesis without safety checks creates headaches for everyone. Safety culture grows from open talk and firsthand stories. A good mentor once told me, “If you’re not a little worried, you’re not respecting your tools.” That thought works just as well outside the classroom. Supervisors must push for annual refreshers, spill drills, and honest reviews after every incident—no matter how embarrassed someone feels. Signs, material safety sheets, and checklists matter, but clear communication and steady routines carry the load in tough situations.
Practical Solutions Work
A small investment in chemical-resistant gear and real safety training prevents bigger losses. Fume hoods and sealed containers slow down work a little but help everyone sleep better at night. Working in pairs and reviewing every unusual odor or color change saves more than just time. Everyone in the lab shares responsibility for health—so don’t cut corners just to save a few minutes.
Is 2,4-Dinitrobenzenesulfonyl Chloride soluble in water or organic solvents?
What Kind of Solvent Does 2,4-Dinitrobenzenesulfonyl Chloride Prefer?
If you’ve worked in a university lab, you’ve probably met chemicals that just don’t cooperate with water. 2,4-Dinitrobenzenesulfonyl chloride belongs to that stubborn category. Drop it in water, and you’ll quickly see it isn’t going to dissolve easily. In fact, the bulk of it will hang back, even when you start swirling your flask. The reason lies in the molecule’s personality—loaded with non-polar nitro groups and only one sulfonyl chloride group for polar interaction. There’s also another problem: this compound likes to react with water, breaking itself down instead of staying whole.
Ask any synthetic chemist, and most will point out that the real action happens in organic solvents. Try acetonitrile, dichloromethane, or even tetrahydrofuran (THF), and you’ll see 2,4-dinitrobenzenesulfonyl chloride making itself at home. That matters because most protection and deprotection steps in organic synthesis rely on having everything dissolved and reacting smoothly. I’ve ruined enough reactions to know that forcing a compound into the wrong solvent only wastes your precious starting materials.
Why the Solubility Question Matters Beyond the Flask
In my time running student practicals, nothing throws off a workflow faster than watching a key reagent clump up. This compound’s behavior isn’t just academic trivia—it shapes safety, storage, and waste. If you try to handle it using water, you risk both safety hazards and lower yields. The breakdown products from its reaction with water can be tough to deal with. If you’re in a small teaching lab, that sometimes means more work for your team on clean-up and extra cost for safe disposal.
The flipside, sticking with only organic solvents, introduces its own headaches. Solvents like dichloromethane can evaporate, sending fumes into the air, and some—like acetonitrile—aren’t cheap to buy or easy to ship. Chemists know all these trade-offs, but students rarely see how choosing the right solvent connects to waste generation, cost, and even compliance with chemical safety regulations.
Looking for Practical Solutions
Many research groups have been moving towards more sustainable lab practices. That means asking tough questions: can you switch to less hazardous solvents? Can you scale down reactions so you make less waste? ACS and other chemistry societies now encourage greener choices. But for tricky reagents like 2,4-dinitrobenzenesulfonyl chloride, you usually fall back on a few tried-and-true organics. Companies now label containers with clear hazard warnings, which helps teams in the lab consider risks during setup.
It doesn’t hurt to talk about smarter storage, too. Keeping the solid dry, tightly sealed, and at low temperatures keeps it safer and lasts longer. In one lab I used to work in, just keeping stock bottles wrapped in parafilm and pushed deep in the flammables cabinet kept surprises at bay for years.
Key Takeaways for Students and Working Chemists
Learning which solvents pair well with which reagents is a rite of passage in chemistry. For 2,4-dinitrobenzenesulfonyl chloride, remember: stay away from water unless degradation is what you’re after. Look to organic solvents, but weigh out the risks and the long-term costs. For greener labs, smaller batch sizes, less hazardous solvents, and smart storage all play a role. Each of these choices shapes the outcome of your reactions and the safety of everyone working beside you.
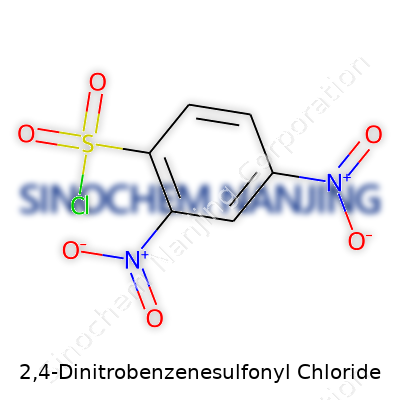
| Names | |
| Preferred IUPAC name | 2,4-dinitrobenzenesulfonyl chloride |
| Other names |
DNBS-Cl DNBS chloride 2,4-Dinitrobenzenesulfonic acid chloride 2,4-Dinitrobenzenesulfonylchloride 2,4-Dinitrobenzenesulphonic acid chloride |
| Pronunciation | /ˈtuː fɔːr daɪˌnaɪtrəʊˌbɛnˌziːnˌsʌlˈfəʊnɪl ˈklɔːraɪd/ |
| Identifiers | |
| CAS Number | 97-02-9 |
| Beilstein Reference | 87864 |
| ChEBI | CHEBI:52043 |
| ChEMBL | CHEMBL3319839 |
| ChemSpider | 14621 |
| DrugBank | DB08503 |
| ECHA InfoCard | 03b08e38-bee1-4c47-b58e-84a0986b15b1 |
| EC Number | 221-009-7 |
| Gmelin Reference | 85924 |
| KEGG | C06250 |
| MeSH | D004238 |
| PubChem CID | 72518 |
| RTECS number | DH9625000 |
| UNII | 8C2O392YH6 |
| UN number | UN1597 |
| CompTox Dashboard (EPA) | DTXSID8020267 |
| Properties | |
| Chemical formula | C6H3ClN2O5S |
| Molar mass | 317.62 g/mol |
| Appearance | Yellow crystalline powder |
| Odor | Odorless |
| Density | 1.75 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | 0.91 |
| Vapor pressure | 3.1 x 10^-4 mmHg (25 °C) |
| Acidity (pKa) | -1.0 |
| Basicity (pKb) | 12.96 |
| Magnetic susceptibility (χ) | Magnetic susceptibility (χ): -60 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.644 |
| Viscosity | 1.324 Pa·s (25 °C) |
| Dipole moment | 3.10 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 389.2 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -324.7 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -1085.0 kJ/mol |
| Hazards | |
| Main hazards | Harmful if swallowed, causes severe skin burns and eye damage, may cause an allergic skin reaction, toxic to aquatic life. |
| GHS labelling | GHS02, GHS05, GHS06, GHS08 |
| Pictograms | GHS05,GHS07 |
| Signal word | Warning |
| Hazard statements | Hazard statements: H302, H315, H319, H335, H351, H373, H411 |
| Precautionary statements | P261, P264, P271, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P312, P321, P332+P313, P337+P313, P362+P364, P405, P501 |
| NFPA 704 (fire diamond) | `2,4-Dinitrobenzenesulfonyl Chloride: NFPA 704 = Health 3, Flammability 1, Instability 1, Special -` |
| Flash point | 81°C |
| Autoignition temperature | 150 °C |
| Lethal dose or concentration | LD50 oral rat 698 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat LD50: 566 mg/kg |
| NIOSH | DH6825000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 10 mg/m³ |
| Related compounds | |
| Related compounds |
2,4-Dinitrobenzenesulfonic acid 2,4-Dinitrochlorobenzene Benzenesulfonyl chloride 4-Nitrobenzenesulfonyl chloride 2-Nitrobenzenesulfonyl chloride |

