A Commentary on 2,4-Dinitroanisole: Unpacking Its Journey, Uses, and Challenges
Digging Into the History of 2,4-Dinitroanisole
Exploring the roots of 2,4-Dinitroanisole, or DNAN, reminds me that every chemical seen in today’s industries usually traces back to needs that reach far beyond the laboratory. Dating to the early twentieth century, DNAN came about as part of broader efforts to find explosives safer than traditional ones like TNT or picric acid. During tough periods, such as the world wars, the search for less sensitive compounds that could still deliver performance became a race against time. DNAN emerged from these trials, sometimes overshadowed by giants like TNT, but it found its purpose when researchers realized how its properties could solve issues of safety and stability.
Understanding What Sets DNAN Apart
There’s a lot to unpack with DNAN as a chemical. Its most distinct trait as an explosive stems from its lower sensitivity to impact and friction, compared to older compounds. I remember learning how high explosives often balance on a knife-edge when it comes to stability. DNAN, with its molecular weight sitting at 198.13 g/mol and its melting point around 94°C, gave those in the field a sense of relief. Liquid at moderately warm temperatures, solid at room temperature, it offers users the option to process and handle it with less worry about unexpected reactions. Its pale yellow crystals can be dissolved in organic solvents, making it compatible with other materials for various energetic formulations.
Examining Technical Aspects and Labelling Conventions
Lab work involving DNAN isn’t especially forgiving, but it’s not known for wild unpredictability in the lab. Formulators look for high purity thresholds, usually above 99 percent, ensuring the material suits critical end uses in energetic material blends. Industry experts group DNAN under the UN number for explosives, and its Material Safety Data Sheets assign R/S phrases that underscore risks associated with exposure and storage. One glance at labeling, and experienced operators recall that while DNAN scores points for lower volatility and shock, it’s never risk-free, given its chemical family. I’ve seen it classified under chemical abstracts names like 2-Methoxy-1,3-dinitrobenzene and trade names depending on the country and end-use.
Walking Through the Preparation Method
Synthesis boils down to a couple of key steps: methylation of dinitrophenol stands at the core. Industries typically start with 2,4-dinitrophenol, using methylating agents like dimethyl sulfate or methyl iodide. Some producers innovate with greener processes, tweaking reagents or temperature profiles to cut waste or up yields. The reaction tracks a straightforward route in principle, but large-scale work tightens requirements for efficiency and safety. I’ve seen reports where even small jolts to conditions create impurities or, worse, hazardous byproducts requiring careful disposal. Scale matters: Synthesis shifts in complexity going from grams in the lab to tons at a plant, and oversight moves from chemists to whole teams monitoring temperature, pressure, and emissions nonstop.
Exploring the Chemistry: Reactions and Modifications
DNAN’s molecular structure resists many common reactions, which boosts its reputation for stability under normal handling. Nitrogen groups hold tight to the aromatic ring, fending off hydrolysis, oxidation, or reduction under mild conditions. Intense heat or strong reducing agents still crack it open, liberating energy in a way prized in military and civilian energetics. I’ve watched research teams fiddle with related compounds, shifting nitro or methoxy groups around to tune properties for specific needs. Chemists can reduce DNAN or use it as a building block, but the main reputation always circles back to its performance in explosive charges in combination with less sensitive binders and plasticizers.
Names and Synonyms: More Than Just Another Label
In scientific circles, 2,4-Dinitroanisole may masquerade under a handful of aliases, each reflecting a different facet of its chemistry or use. Much of the literature refers to it by its IUPAC-format name. Synonyms, including 2,4-DNA or its registry ID, crop up in both hazard documentation and procurement forms. Real-world applications rarely pause for semantic debate, but names matter when rules around control and transport rely on technical nomenclature. More than once I’ve seen confusion escalate into supply delays because a shipping manifest listed a mismatch in naming.
Safety and Handling: A Hard-Learned Lesson
No story about DNAN avoids the tightrope of safety standards. While it boasts a better safety profile than traditional explosives, operators in the field and at the plant still face real hazards. Dust, spills, and even static can trigger incidents, calling for layers of controls. The difference experienced staff make always stands out to me: Regular training, rigorous procedures, and quick access to neutralizing agents commands respect. Environmental controls matter as well, since runoff or airborne particles introduce risks both to humans and ecosystems. I’ve seen operational standards evolve, especially after incidents that put both people and local communities in danger. Nearby population density only raises the stakes, and regulatory agencies insists on tight inventories, secure storage, and strict adherence to operational guidelines.
Where DNAN Goes To Work: Application Areas
DNAN sits double duty in research and fielded military systems. Its main application links back to insensitive munitions — a need that keeps growing due to safety regulations and performance requirements. Blended with other energetic compounds, DNAN’s job is to deliver explosive force on target, but without the risks of accidental detonation during storage, handling, or transport. Beyond military applications, experimental uses crop up in mining and demolition, though those face commercial hurdles compared to established compounds. Interest in DNAN increased in the wake of incidents involving older explosives, forcing governments and industry to look for alternatives that can handle accidents or external threats with less catastrophic outcomes.
Research and Development: Learning New Tricks With Old Compounds
The research world never gives up on finding improved ways to use DNAN. Frequent studies drill into the smallest details of crystal structure, decomposition temperatures, or compatibility with plasticizers and binders. Innovation doesn’t just sit with bench researchers; process engineering teams seek safer, greener, and more economical production methods. Some teams investigate encapsulation approaches or coating technologies to further suppress sensitivity. I recall a push in recent years to model detonation characteristics with sophisticated computer simulations, shown alongside live testing to validate output. Universities and government labs often collaborate across disciplines, pressing chemistry, materials science, and even computational physics to squeeze the most out of DNAN’s unique traits.
Toxicity and Health Research: Scrutiny That’s Here to Stay
On the toxicity front, DNAN’s record cuts both ways. Some early claims suggested lower acute risks than traditional explosives, but further research revealed slow-to-show effects. Studies point to concerns with chronic exposure: Liver and kidney effects emerge as areas for caution. I’ve seen toxicologists trace breakdown products through waterways and up the food chain, calling for close scrutiny of waste and spill management. Regulatory bodies now push for more data on DNAN’s persistence in the environment. Workers exposed to vapors or dust face mandates for protective gear and health monitoring, especially with mounting evidence for possible mutagenicity and reproductive impacts in some animal trials. The demands for transparency and open reporting in this space feel stronger than ever, reflecting lessons learned from past industrial mishaps.
The Road Ahead: Future Prospects and Open Questions
Looking ahead, DNAN faces both opportunity and challenge. Safety requirements for insensitive munitions only get stricter, so the compound seems poised for continued, possibly expanded use. I see research teams digging deeper into less hazardous synthesis, smarter product stewardship, and more precise risk modeling. The gap between field reports and laboratory data drives calls for real-world testing and transparent sharing of lessons learned. There’s also a push to identify biodegradable or rapidly degrading alternatives that meet similar performance metrics, answering community concerns over long-term pollution. Persistent questions linger: How far can industry push down the risks of handling energetics? Can greener chemistry bring those tradeoffs without new hazards? My own view, shaped by close study and direct conversations with experts, is that every step forward needs more than technical know-how — it takes a resolve to weigh impacts on workers, communities, and the environment alongside every batch produced and every field test logged.
What is 2,4-Dinitroanisole used for?
A Closer Look at 2,4-Dinitroanisole
2,4-Dinitroanisole, often called DNAN in technical circles, slips under the radar for most people. You probably won’t spot it on grocery shelves, and nobody sings its praises on talk shows. Yet, inside defense labs and military stockrooms, it catches plenty of attention. For many years, research pointed out DNAN as one of the alternative ingredients for making safer explosives. The old standbys like TNT brought environmental headaches and safety risks, so scientists turned to options like this one, searching for answers.
Why the Military Picks DNAN
Explosives often get a bad reputation because of their destructive use, but they remain a necessary part of national security. A major point with DNAN is its behavior during accident scenarios. Compared to older ingredients, it doesn’t react as violently under shock or heat. I’ve chatted with a few folks who spent time in explosive ordnance units—they will tell you, any reduction in unpredictability can mean the difference between a safe mission and disaster. Nobody wants to handle an explosive that sets off just because you dropped a box or sat it near a running generator.
DNAN also stands out because it breaks down differently in the environment. Old explosives such as TNT leach into groundwater and linger in soil for years. DNAN poses fewer long-term threats, giving environmental engineers a bit more breathing room. Safety isn’t just about handling; it’s about what happens once these materials leave storage or the battlefield. Countries that want to clean up after military activities look toward ingredients like DNAN to lower the toxic footprint.
Challenges Behind the Chemistry
Some pushback comes from people worried about switching from TNT or RDX to a new molecule. DNAN isn’t perfect. For starters, the production process involves handling other hazardous chemicals, raising questions about risks in the manufacturing stage. And while it’s less sensitive to being set off by accident, chemists still keep an eye on its stability over time. There’s also the question of cost. Changes in military procurement happen slowly, especially if a material carries a higher price tag or its supply chain hasn’t matured yet.
Searching for Balance: Safety, Cost, and Clean-Up
Switching over to safer explosives like DNAN takes more than a government memo. It demands buy-in from chemists, soldiers, policymakers, cleanup crews, and local communities. I once worked alongside a team cleaning up an abandoned munitions plant. The toxic leftovers from the old explosives didn’t just cost money to remove—they disrupted life in the neighborhood for months. Communities remember these events, and many push to avoid a repeat.
If policymakers invest in bridging the costs and logistics for DNAN manufacturing, the benefits stack up. Proper training, strong oversight, and updated environmental guidelines can help get DNAN to the front lines without repeating the mistakes of the past. A few countries already started blending DNAN into their munitions stockpiles. The world watches their outcomes closely, hoping the promises hold up in the field and in the soil where they matter most.
Building on Research—and Responsibility
Real progress won’t come from the chemistry lab alone. It comes from listening to the people closest to these challenges, learning from every setback, and putting safety and environmental health ahead of short-term savings. As the science moves forward, DNAN’s story shows what it looks like to take responsibility in areas most folks rarely see—on the assembly lines, at military bases, and in the ground where old explosives sometimes still lurk.
Is 2,4-Dinitroanisole hazardous or toxic?
Understanding 2,4-Dinitroanisole
2,4-Dinitroanisole, known in chemistry circles as DNAN, started turning up in new explosives over the past two decades. It caught the interest of military scientists who hoped it might be a “safer” replacement for TNT. You see, TNT’s reputation has suffered, whether from groundwater contamination or its effect on workers’ health. DNAN looked better on paper—less sensitive to shock and a bit harder to set off by accident. That sounded promising, but hazard claims always need a closer look.
Toxicity: Digging Into the Facts
Looking at research, the biggest question has always been: How dangerous is DNAN if it gets into living things? Studies so far show DNAN causes harm to fish and aquatic organisms at pretty low levels. For humans, lab studies indicate DNAN can damage red blood cells. Animals tested with DNAN showed signs of anemia, even at exposure levels that could happen accidentally in a work setting. Waste from munitions plants or improper disposal poses serious risks if it leaks into nearby waterways or soil.
People sometimes compare DNAN’s toxicity to TNT and declare it’s “less toxic.” That’s not good enough. Less harmful doesn’t mean safe. Even at lower toxicity, build-up in soil or water could hurt wildlife, disrupt water supplies, or lead to chronic health issues. No new explosive really gets off scot-free—tradeoffs just shift the risk around.
Exposure Risks in the Real World
Factory workers, soldiers, and cleanup crews deal with explosives every day, so the stakes are high for them. As someone who has worked with hazardous chemicals, I know the difference good safety training makes. Even so, accidents happen. Respiratory protection, gloves, and strict handling rules lower risks, but leaks and spills hit communities living downstream. You only need one wastewater discharge or awkward barrel to start contamination. That’s how legacy chemicals like dioxin turned into national headlines decades after people thought everything was fine.
Environmental Impact: Looking Long-Term
DNAN doesn’t break down as quickly as some hoped. Researchers observed breakdown products sticking around in soil and water, which can make cleanup harder. Some of these leftovers also carry health risks, especially for aquatic life. Persistent chemicals in the environment end up in our food, our rivers, and sometimes our own tissues. I’ve seen how difficult—and expensive—it is to clean up after the fact, especially in low-income or rural communities where pollution quietly lingers.
What Can We Do About It?
Chemical safety depends on more than the molecule itself. Real progress comes from smart engineering and policy. Factories using DNAN must invest in treatment systems for wastewater and air emissions. Managers need to check for leaks and train staff with more care than the regulations might require. That means reevaluating exposure limits, updating personal protective equipment, and running biomonitoring for workers.
Communities near production and disposal sites deserve up-to-date environmental monitoring. Clarity in labeling and public reporting helps people learn about what’s happening in their area. Independent audits and stronger enforcement of clean-up requirements make a difference, too. It’s crucial to involve environmental health researchers who aren’t linked to the chemical’s manufacturers in setting safety standards.
Building Toward Safer Alternatives
Safer chemistry doesn’t come from swapping one risky ingredient for another. Pushing for alternatives that leave fewer toxic leftovers, or materials that degrade faster, gives future generations a break from cleanup headaches. Funding fresh research and inviting feedback from frontline workers and nearby towns keeps the focus on real-world safety—not just what looks appealing in lab reports.
What is the chemical structure of 2,4-Dinitroanisole?
What Makes 2,4-Dinitroanisole Stand Out?
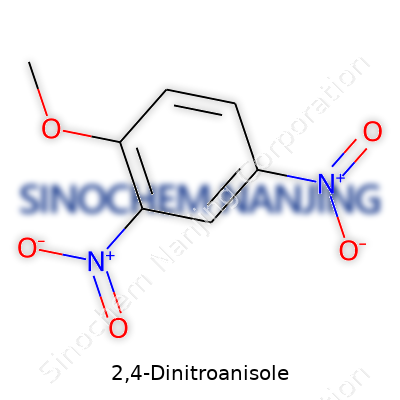
2,4-Dinitroanisole shows up a lot in conversations about safer explosives and smart approaches to energetic materials design. Folks in the field know it as DNAN. What makes it jump out is its molecular design and how that impacts both performance and environmental impact. The backbone of the molecule—a benzene ring—carries two nitro groups and a methoxy group, all organized in a pattern that brings unique chemistry to the table.
The Shape of the Molecule
The name ‘2,4-dinitroanisole’ tells the story. The core is a benzene ring. At carbon 1, you’ve got a methoxy group—an oxygen atom bonded to a methyl, hanging off the ring. Move to carbon 2 and carbon 4, and you hit the nitro groups (NO2). Three substituents pointing off the ring, each changing the molecule’s reactivity and stability.
Here’s why this layout works: the nitro groups increase the explosive potential, but the methoxy side introduces stability that traditional nitroaromatics often lack. That’s not just a lab curiosity—this stability cuts risks during handling and lowers the risk of harmful detonation by accident.
Why Know the Structure?
Getting a handle on the structure of DNAN matters. Anyone who’s worked in a lab with energetic compounds knows the value of predictability. Small changes in a molecule’s design can swing safety profiles and environmental effects. DNAN’s specific arrangement means it doesn’t break down in water as quickly as something like TNT, but it also doesn’t form the same level of toxic byproducts. Researchers at national labs and defense organizations have hammered out countless tests to confirm these differences.
The structure even matters when it comes to mixing and pressing the material into its final form for industrial use. The methoxy group pushes up the melting point, which allows safer melt-casting compared to other nitrated hydrocarbons. Factories and field techs running melt-pour operations often pay close attention to this feature since it means fewer nasty fumes and less risk of fire.
Field Experience: Safety and Sustainability
In conversations with colleagues and EHS specialists, I’ve heard countless stories comparing disasters involving older explosives to the relatively tame behavior of DNAN under stress. You can drop or bump products containing DNAN and not expect instant catastrophe. That reassurance hasn’t always been easy to come by. Production staff especially appreciate the lower vapor pressure and reduced dust hazards tied to DNAN’s makeup.
Environmental chemists see another angle. By swapping in DNAN for more toxic ingredients, contamination at test sites drops, recovery times improve, and wildlife stands a better shot at bouncing back after training exercises. DNAN has a lower potential for bioaccumulation because of its less aggressive breakdown, which makes a big difference for groundwater protection.
Looking for Smarter Solutions in Chemistry
Chemists work toward better solutions every year by tweaking groups on molecules like DNAN. They look for new ways to enhance safety, minimize pollution, and support tough industrial standards all at once. Regulations keep tightening, so the pressure stays on to refine these designs. Colleges and research agencies have grants dedicated specifically to alternative aromatic nitro compounds, aiming to balance performance with responsibility.
Every project in energetic materials teaches something about tradeoffs. DNAN serves as proof that small choices in chemical structure can ripple out into large changes for workers, communities, and the environment.
How should 2,4-Dinitroanisole be stored and handled safely?
Understanding the Risks
Everything I’ve learned about 2,4-Dinitroanisole comes down to respect. This yellow crystalline solid often ends up in the hands of professionals who know sour-smelling chemicals spell danger if people get careless. Nobody likes to think about what goes wrong, but history leaves reminders—touching, inhaling, or mismanaging such substances carries real risks, including harm to human health and the environment.
The Essentials of Secure Storage
Storing 2,4-Dinitroanisole isn’t a job for improvisers. Keep this chemical in a cool, dry, well-ventilated room, always inside sealed, appropriately labeled containers. I’ve seen too many near-misses when folks didn’t double-check their bins or skipped the humidity controls. Humidity drags water into the mix, which can cause the dinitroanisole to degrade or react.
Metal containers seem sturdy but don’t play nicely with every compound. Polyethylene or glass offer better peace of mind—no rust, no sneaky corrosion. Keep the storage room away from direct sunlight. Sunlight tends to raise temperatures, which increases pressure in sealed containers. Sudden temperature jumps sometimes make lids pop and release fumes. Store away from oxidizers, acids, reducing agents, and open flames. A tiny spark and a vapor leak spell trouble.
Proper labeling matters, too. This isn’t about office neatness. If you can’t read the name or the hazard, you and your co-workers might grab the wrong jar, or not take enough precautions. Label every vessel with the full chemical name, hazard class, and date received.
Personal Protective Equipment That Works
Lab coats and gloves sound basic, but there’s science behind their use. Nitrile gloves shield skin far better than latex. A full-face respirator trumps a dust mask, blocking fine particles and fumes that can irritate lungs or worse. Splash-proof goggles keep your eyes safe during transfers or mixing, and they aren’t just for chem students. Wear closed-toed shoes and long sleeves; loose attire or sandals create shortcuts for accidents.
I’ve seen too many workers skip the final check. Gear can fail without warning—a rip in a glove, a loose strap on a mask. That quick glance before handling the chemical can save a trip to the ER.
Everyday Handling Without Shortcuts
Measuring and transferring 2,4-Dinitroanisole always deserve full attention. Use tools—scoops, spatulas, funnels—instead of bare hands. Never pipette by mouth. Isolating small batches keeps reactions controlled and manageable if anything spills. Clean up drips right away, using non-sparking tools on anti-static surfaces. Mishandling powder or letting it pile up around the workspace increases exposure risk.
Spills can happen, so it pays to keep absorbent pads and waste bins nearby. Dispose of all rags, gloves, or broken containers in labeled hazardous waste drums, following all local regulations. Just tossing contaminated gear into ordinary trash only offloads the risk to others down the line.
Training and Emergency Readiness
Regulations, like OSHA’s Hazard Communication Standard, require ongoing training. In practice, hands-on refreshers help more than lectures. Drill for emergencies—chemical exposure or fire—so your team moves fast instead of freezing. Keep eyewash stations and safety showers accessible. Post emergency numbers and keep safety data sheets (SDS) within arm’s reach.
Manufacturing, university, and defense labs are only as safe as the least-prepared handler. Every rule written after an accident started out as a suggestion. Trust experience and facts: 2,4-Dinitroanisole is safest with the right gear, a clear label, a solid storage plan, and a culture where safety always comes first.
Where can I buy or source 2,4-Dinitroanisole?
Searching for 2,4-Dinitroanisole: What Drives the Question?
Type the name “2,4-Dinitroanisole” into a search bar and you may not feel much at first. To most people, it's just another lab compound. For chemists, though, 2,4-Dinitroanisole carries weight. It pops up in research journals as a potential ingredient in energetic materials, especially with modern moves toward “greener” explosives. It’s not the kind of thing you buy on impulse for a home experiment. The average individual rarely considers what it takes to buy a compound with this sort of profile.
The questions about sourcing 2,4-Dinitroanisole usually do not come from casual curiosity. I remember as a grad student, piecing together lists of needed chemicals, then spending hours on vendor websites, always double-checking what could and could not be shipped to a campus address. The hurdles went way beyond a simple digital checkout.
Why So Many Hurdles?
2,4-Dinitroanisole stands out because of how it can be used. Its chemistry attracts attention from defense researchers and regulatory agencies alike. In 2018, the U.S. Army cited it for replacing traditional explosives in some munitions. Government oversight doesn’t relax just because a compound has “potentially greener” applications. Shipping rules, licensing, and background checks kick in long before any actual bottle arrives at a lab door.
Reputable suppliers like Sigma-Aldrich or TCI America stock this chemical. Still, they don’t treat it as a common item. A customer must prove credentials, present legitimate institutional ties, and sometimes even submit end-use declarations. This keeps the path narrow—only for research institutions, certified companies, and government-approved buyers. Private buyers run into brick walls. The chemical supply industry as a whole builds thick layers of screening for anything linked to explosives, toxins, or tight regulatory lists. No easy one-click. No simple personal delivery.
Trust in the Supply Chain Matters
Even for legal buyers, trust in suppliers makes a big difference. I once watched a colleague order a similar “restricted” chemical. Delays stacked up, all because a vendor wanted to check whether our university lab followed proper storage and disposal rules. They walked through approvals step by step, often with local authorities double-checking paperwork. These weeks-long waits weren’t about mistrust of scientists. Vendors wanted to protect themselves from liability and serve as another checkpoint against illegal or unsafe use.
Reducing the Risks, Improving Access for Legitimate Research
The facts behind these roadblocks are clear: safety matters for the public and for research. Roland P. et al. published in "Propellants, Explosives, Pyrotechnics" that new energetic compounds come with unpredictable hazards. The chemical industry adapts by tracking buyers closely. Tighter tracking, clear online information about who qualifies, and harmonized global rules would help scientists. Legitimate researchers should push for streamlined reporting and international cooperation. Industry networks can post more transparent guidance about purchasing rules, so students and researchers waste less time chasing unavailable substances.
Every gate surrounding chemicals like 2,4-Dinitroanisole has its reasons. It protects people, whether they’re in the lab or on the street. At the same time, future breakthroughs in safer energetic materials rely on improved pathways—strong checks paired with clear guidance for those in science and industry.
| Names | |
| Preferred IUPAC name | 1-methoxy-2,4-dinitrobenzene |
| Other names |
DNAN 1-Methoxy-2,4-dinitrobenzene 2,4-Dinitro-1-methoxybenzene |
| Pronunciation | /ˈdaɪ.naɪ.trəʊ.əˈnɪ.səʊl/ |
| Identifiers | |
| CAS Number | 119-27-7 |
| Beilstein Reference | 363956 |
| ChEBI | CHEBI:77918 |
| ChEMBL | CHEMBL331546 |
| ChemSpider | 6707 |
| DrugBank | DB11975 |
| ECHA InfoCard | 100.007.750 |
| EC Number | 208-107-8 |
| Gmelin Reference | 1041 |
| KEGG | C18621 |
| MeSH | D017972 |
| PubChem CID | 8197 |
| RTECS number | UX8575000 |
| UNII | R8C7J8L66O |
| UN number | UN1321 |
| CompTox Dashboard (EPA) | DTXSID4044220 |
| Properties | |
| Chemical formula | C7H6N2O5 |
| Molar mass | 198.13 g/mol |
| Appearance | Yellow crystalline solid |
| Odor | Odorless |
| Density | 1.653 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | 1.98 |
| Vapor pressure | 0.00016 mmHg at 25°C |
| Acidity (pKa) | 13.86 |
| Basicity (pKb) | 12.52 |
| Magnetic susceptibility (χ) | -49.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.635 |
| Viscosity | 80.1 mPa·s (at 80 °C) |
| Dipole moment | 3.01 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 207.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −161.7 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | −4055 kJ·mol⁻¹ |
| Hazards | |
| Main hazards | Harmful if swallowed, causes serious eye irritation, may cause respiratory irritation, may cause damage to organs through prolonged or repeated exposure. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06, GHS09 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335, H351, H373 |
| Precautionary statements | P210, P250, P264, P270, P273, P280, P301+P312, P302+P352, P305+P351+P338, P308+P313, P330, P363, P370+P378, P403+P235, P405, P501 |
| NFPA 704 (fire diamond) | 2,4-Dinitroanisole NFPA 704: "2-2-2 |
| Flash point | 253 °C |
| Autoignition temperature | 410 °C |
| Explosive limits | 1.2–7.0% |
| Lethal dose or concentration | LD50 (oral, rat): 932 mg/kg |
| LD50 (median dose) | LD50 (median dose): 932 mg/kg (oral, rat) |
| NIOSH | WA3850000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.01 mg/m³ |
| Related compounds | |
| Related compounds |
2,4-Dinitrophenol 2,4-Dinitrochlorobenzene 4-Nitroanisole 2-Nitroanisole Anisole |

