2,4-Dinitroaniline: Digging Deeper into a Chemical Story
Historical Development
Chemistry has seen many stories echo the evolution of science, society, and industry, and 2,4-Dinitroaniline remains one of the less glamorous but still consequential stops along that route. Its journey started in the late 19th century, back when the world’s hunger for new dyes and explosives spurred relentless experimentation with nitroaromatic compounds. Researchers hunted for derivatives of aniline, and adding nitro groups became a key move. 2,4-Dinitroaniline was once viewed mainly as a stepping stone toward more potent products, yet it gradually found its own place as technology and demand shifted its uses. By the mid-20th century, labs in the United States and Europe documented its properties and potential, while the chemical industry began to recognize it as more than just a byproduct. Thinking back to my own college lectures, professors described it almost like a sibling to trinitrotoluene, reminding students that every nitro group added more complexity for both synthesis and safety.
Product Overview
In its essence, 2,4-Dinitroaniline stands as an aromatic amine with both nitro groups attached to the benzene ring – at positions two and four, just as its name shouts out. Commercial chemists encounter it most often as an intermediate, slipping it into processes that end up yielding dyes, pigments, or even agricultural products. Its role isn’t always heralded, but industry insiders understand its quiet importance for building up more elaborate molecules. Whenever factories and labs look for a reliable nitroaniline derivative, this compound often lands on the shortlist.
Physical & Chemical Properties
Any person handling 2,4-Dinitroaniline soon recognizes a yellow crystalline powder with a faint odor. It doesn't dissolve well in cold water and prefers organic solvents, reflecting the stubborn nature typical for such aromatic molecules. Melting point hovers in the modest range, usually below 200°C, so it survives most low-temperature reactions easily, but high enough to call for caution near any open flame. The electron-withdrawing nitro groups anchor it as a stable, fairly non-volatile compound under ordinary storage conditions. From a practical standpoint, the overall stability means processors and researchers can manage it with predictability, while still respecting its sensitivity to shock or severe heat, especially if blended carelessly with reducing agents.
Technical Specifications & Labeling
Standards for 2,4-Dinitroaniline reflect its dual life in academia and industry. Chemists seek out high-purity material—often above 99%—to avoid unwanted reactions in downstream processing. Labels must warn about toxicity and potential environmental harm, but those who’ve worked with it recognize the importance of batch consistency and clear identification. Many recall the days before global chemical inventory schemes, but modern protocols demand standardized safety icons and clear identification, reducing risks for warehouse workers and researchers alike.
Preparation Method
Getting to 2,4-Dinitroaniline means following the long tradition of aromatic nitration, usually from aniline as a starting point. Nitration under controlled acidic conditions introduces the nitro groups, with plenty of cooling required to avoid runaway reactions. The procedure involves metering nitric and sulfuric acids carefully to limit byproducts like 2,6-dinitroaniline or even trinitroaniline. My own experience in undergraduate chemistry taught the value of gradual acid addition—a rapid pour could easily spark disaster. Afterwards, filtering, washing, and drying give the familiar yellow crystals. Process improvements over decades reduced waste and improved yields, but the bones of the process barely changed, a testament to both simplicity and challenge in such classic chemistry.
Chemical Reactions & Modifications
For those in synthesis, 2,4-Dinitroaniline opens up reaction possibilities. The amino group allows coupling reactions critical for creating azo dyes, a backbone for vibrant colors in textiles and inks. Nucleophilic substitution on the nitro groups lets chemists target more complex molecules, albeit with careful control over temperature and catalysts. Hydrogenation can remove nitro groups, revealing a way to explore further derivatives. To the trained eye, this molecule becomes a building block for branching into pesticides, medicinal chemistry, and material science. Each new substitution pattern adjusts properties, either increasing solubility, shifting color, or altering biological activity. Real-world labs must consider selectivity, yield, and safety at every step.
Synonyms & Product Names
Common synonyms for the compound reflect its structure—names like Dinitroaniline, 2,4-DNA, and even its IUPAC label show up in research and commerce. Depending on supplier or region, one might run into the more tongue-twisting 1-amino-2,4-dinitrobenzene. The variety in names sometimes trips up newcomers, though seasoned chemists quickly learn never to assume chemical equivalence without double-checking catalogs and CAS numbers.
Safety & Operational Standards
Safety with 2,4-Dinitroaniline means attention to personal protection and strict control over dust and spills. Nitrated aromatics have a reputation for toxicity, and 2,4-Dinitroaniline has earned its place as a chemical of concern, flagged for both human and environmental health. Direct skin contact and inhalation bring dangers, from acute irritation to concerns over long-term organ damage. Most labs and factories require full goggles, gloves rated for chemical resistance, and local fume extraction. The days of cavalier handling thankfully faded as more was learned about chronic exposure in manufacturing environments. Safe disposal matters: uncontrolled release carries real risks, so regulatory agencies set limits on runoff and workplace emissions.
Application Area
People outside the chemical industry rarely realize that 2,4-Dinitroaniline hides inside some of the world's common products. Dye manufacturers value it for creating deep, lasting colors in fabrics. Researchers harness its structure to build pharmaceutical intermediates. Some synthetic routes to herbicides pass through this compound en route to active molecules that control weed growth. Over the decades, its use as an intermediate in industrial pigment and pesticide production drew debates about the balance between benefit and risk. Looking at the wider world of chemistry, 2,4-Dinitroaniline may not gain headlines, but its presence ripples through textiles, agriculture, and pharmaceutical research.
Research & Development
Recent years witnessed a surge in looking at both greener syntheses and reduced environmental footprints. R&D teams work to cut down harsh acid use during nitration, searching for milder, more selective catalysts. Interest in nano-scale applications also picked up, with its rigid structure and modifiable sites appealing for materials science. Toxicological screening and fate analysis remain front-burner priorities, especially as regulatory scrutiny tightens around industrial chemicals. In my own visits to industrial labs, I saw firsthand how vapor monitoring and effluent controls became standard, underscoring the push for safer, less wasteful workflows.
Toxicity Research
Toxicology teams found that 2,4-Dinitroaniline, like its chemical relatives, affects both animal and human health upon sufficient exposure. Studies link it to blood changes and possible organ stress, and animal models showed mutagenic effects at high concentrations. Occupational guidelines recommend strict exposure limits, usually measured in milligrams per cubic meter, to avoid cumulative damage. Regulatory agencies also flagged its breakdown products as potential marine and soil contaminants, leading to monitoring programs near production facilities. From the standpoint of consumer safety, regulations set down maximum allowable residues for its pesticide derivatives in food and water. The upshot is that everyone in the handling chain faces responsibility, from storage to site cleanup.
Future Prospects
Looking ahead, the role of 2,4-Dinitroaniline depends on industry’s ability to reconcile usefulness with responsibility. Tighter regulations on hazardous intermediates could prompt shifts to alternative chemistries, but as long as demand continues for specific dyes and selective herbicides, this molecule likely won’t vanish overnight. Labs exploring greener routes and lower-toxicity modifications could push the field forward, marrying old-school robustness with new safety practices. If sustainable chemistry is going to succeed, compounds like 2,4-Dinitroaniline offer a test case for responsible innovation. Young chemists and environmental engineers alike watch its story, knowing each improvement ripples into broader chemical safety and stewardship.
What is 2,4-Dinitroaniline used for?
Where You Find 2,4-Dinitroaniline
Walk into any large chemical manufacturing facility and you'll probably run across barrels or containers labeled with hard-to-pronounce names. One of those is 2,4-Dinitroaniline. Even though most people haven’t heard of it, this compound touches more lives than most would realize. Factories use 2,4-Dinitroaniline mainly to build something else—serving as a bridge in chemical reactions that drive large sectors of industry. Anyone working in dyes, pigments, or agricultural chemical production has likely seen its name on paperwork or ingredient lists.
The Building Block for Dyes and Pigments
Textiles, plastics, printing inks—these industries all rely on colors that need to last. Synthetic dyes keep shirts from fading and add bright colors to everything from toys to packaging. 2,4-Dinitroaniline plays an important part in making those dyes. Chemists use it to make more complex chemicals, including yellow and orange colors popular in both industrial and consumer goods. The textile world depends on reliable, strong colors, so companies lean on this compound to deliver consistency.
Agricultural Chemistry Connections
Farmers rarely see the chemical itself, but 2,4-Dinitroaniline underpins some weed control solutions. Herbicides need stable, predictable ingredients that allow crops to grow without interference. Scientists start with this compound in the lab and then shape it into more specific molecules that help kill weeds in soybean, corn, and other fields. The agricultural sector sees huge gains in productivity because reliable chemical building blocks mean fewer failed batches and better crop protection.
Safety Concerns and Environmental Impact
People who work around chemicals know health and safety must come first. Even though 2,4-Dinitroaniline sits upstream in manufacturing, it can have a downside. Exposure can cause skin or respiratory irritation, and mishandling in factories sometimes leads to bigger environmental issues. Disposing of leftover chemicals the wrong way can poison water or soil. This fact isn’t lost on regulators or factory supervisors. Regular training, updated protective equipment, and closed systems help lower those risks in responsible workplaces. Governments push for stronger controls, but loopholes and poor enforcement sometimes let pollution slip through.
Toward Safer and Cleaner Use
No easy answer wipes away the hazards, but steady efforts keep making things safer. Well-run factories invest in better air filtration, and research teams experiment with less hazardous alternatives. Big buyers of dyes, pigments, and crop chemicals also start to push for greener supply chains, setting the bar higher for everyone. Coordination across industry, regulators, and researchers leads to fewer accidental releases and better health for factory crews and their neighbors.
Why This Matters Now
Many consumers benefit from reliable colors in clothing and safe food on shelves, but may not realize the chain of chemistry that gets us there. 2,4-Dinitroaniline offers proof that everyday products often start with hidden compounds and big industrial systems. Yet, with every year that passes, pressure grows for cleaner, safer manufacturing. Paying attention to who uses these base chemicals, how they’re handled, and what waste flows out of factories could shape not just products, but also public health and the quality of the environment. Protecting both workers and communities means getting the basics right with chemicals like this and never letting convenience justify carelessness.
Is 2,4-Dinitroaniline hazardous to health?
What Is 2,4-Dinitroaniline?
Most folks outside chemistry circles have never stumbled across 2,4-Dinitroaniline. Don’t let the technical name fool you—it’s used for things like dyes, pigments, and as an intermediate for other chemicals. The bright yellow appearance might attract curiosity, but once you start digging, the concerns become clear.
Health Risks and Exposure
Breathing dust or handling this chemical without proper protection doesn’t strike most people as a big problem, until the symptoms sneak in. Headaches, dizziness, and even nausea can pop up with moderate exposure. The main routes are skin contact or inhaling dust, which can happen easily in factories or labs that don’t prioritize strict safety habits.
Over time, there’s troubling news about longer exposures. The body doesn’t appreciate repeated contact. Some studies in animals show effects on bone marrow and blood. Nitrogen-based compounds like 2,4-Dinitroaniline have a pattern of causing problems in red blood cells, which the experience of workers in dye factories has borne out over decades. Nobody wants to roll the dice with conditions like anemia or irregular heartbeats.
What Science Tells Us
Researchers put effort into finding out whether chemicals like this pose a cancer threat. So far, the verdict isn’t carved in stone, but flags have gone up. Some dinitro compounds have caused cancer in animal testing. 2,4-Dinitroaniline hasn’t been singled out as the most dangerous, but scientists take it seriously and keep it under observation. Real-life data on workers is limited, which muddies the picture.
Other hazards don’t need more evidence. If the stuff gets into your eyes or on your skin, irritation almost always follows. People with allergies or asthma often have worse reactions. The chemical’s tendency to linger in soil and water after factory use doesn’t help matters, either. Bioaccumulation can bring unexpected consequences years down the road—not just for workers, but for nearby communities.
Real-World Experience
Having spent time in chemical plants early in my career, I’ve watched plenty of skilled people develop rashes and respiratory symptoms after a few months handling dyes and pigments. The frustrating thing: some shops had great safety routines, but corners still got cut during busy seasons. Even minor exposure can snowball into something worse if ignored.
Cleaning up spilled powders, I ended up with stained fingers and the familiar itchy skin. Lab friends talked about daily headaches until new ventilation fans arrived. What seemed minor at the time always turned out to have stronger consequences later. It taught me to take even “medium-risk” chemicals seriously—especially when official long-term data stays fuzzy.
What Should Happen Next?
Worksites must demand better protective gear and enforce regular training. Plain cotton masks don’t cut it; respirators and gloves make a bigger difference, especially for dusty jobs. Frequent air checks and proper waste handling play a key role.
Regulators and managers can’t shrug off spotty science either. If a chemical has cousin compounds that trigger cancer or blood disorders, they should crack down on exposure limits, even while further studies unfold. Better reporting about workplace illnesses helps track subtle effects and could save lives. I’ve seen the benefits firsthand—companies that took small risks seriously saw sharp drops in sick days and accidents.
Looking Forward
Communities near factories deserve transparency, too. Anyone living by a facility using chemicals like 2,4-Dinitroaniline should get honest updates. Health departments can encourage regular soil and water checks, plus open up hotlines for workers to report symptoms early.
Staying informed and insisting on stronger controls means fewer surprises. My experience tells me that in health and safety, playing it cautious beats scrambling to fix problems after the fact.
What is the chemical formula of 2,4-Dinitroaniline?
Behind the Formula
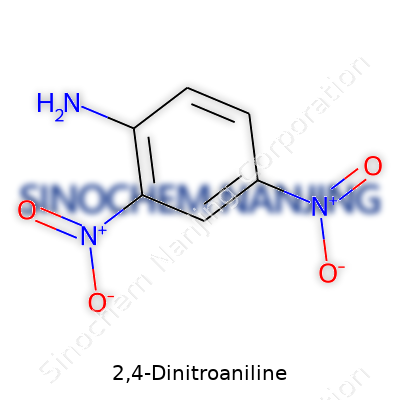
2,4-Dinitroaniline, a name that pops up often in discussions about dyes, pesticides, and even forensic science, has a chemical formula of C6H5N3O4. At its core, the structure features a benzene ring attached to both a nitro group at the second and fourth positions and an amino group. These functional groups shape its identity, affecting everything from color to chemical reactivity and its interaction with living organisms and the environment.
Why This Compound Matters
From personal experience in lab settings, it’s striking how much a small change to an aromatic molecule can ramp up biological activity. Add two nitro groups, and suddenly an otherwise quiet molecule turns potent. In the agricultural world, this makes 2,4-dinitroaniline a backbone for some widely-used herbicides. Farmers rely on these compounds to keep crops healthy and productive, controlling weeds before they take over a field. As a result, millions of people enjoy more stable food prices and greater food security.
Questions on Health and Environment
Any time a chemical lands in soil, air, or water, questions crop up about safety. Data from the US Environmental Protection Agency shows that dinitroaniline-based herbicides persist in soil for months. That spells trouble if misuse or overuse occurs. Research in environmental chemistry often points to groundwater contamination risks, especially in regions with heavy agricultural activity. Simple acts—washing vegetables, handling treated crops—can result in trace exposure. Scientists have documented that high concentrations can stunt plant development and disrupt soil organisms that play a critical part in healthy crops.
Navigating Safety and Sustainability
For workers in chemical plants or on farms, 2,4-dinitroaniline presents clear safety issues. Touch or inhale too much, and it’s not just a mild irritant; some studies show links to chronic health problems with long-term exposure. Training helps, along with gloves and masks, but safer manufacturing and handling practices can make a big difference. In my own work, shifts to less hazardous alternatives helped protect both workers and nearby communities. Newer formulations and precise application techniques, like targeted spraying, cut down waste and runoff.
Paths Toward Smart Use
Several solutions start at the research and regulatory level. Innovators keep searching for biodegradable alternatives and smarter application tools. Lawmakers and agencies can learn from cases in Europe and Japan, where tighter controls led to measurable improvements in groundwater quality and reduced health complaints.
Transparency always helps. Clear labeling, safety briefs, and farmer education programs let consumers and workers make informed choices. Trust builds when science agencies publish exposure data and outcomes, openly showing steps taken to protect public health and ecosystems.
Balancing Progress With Caution
2,4-Dinitroaniline, carrying that simple formula C6H5N3O4, underscores an old lesson: chemicals that serve us well can need careful respect. Responsible use, openness about risks, and investment in safer chemistry hold keys to ensuring benefits without sacrificing tomorrow’s health and environment.
How should 2,4-Dinitroaniline be stored?
Working With a Classic Lab Chemical
For anyone who’s handled organic chemicals, 2,4-Dinitroaniline isn’t just another bottle tucked in the flammables cabinet. The bright yellow crystals pack a punch: toxic by inhalation, skin contact, or swallowing, with a solid record of combustibility. I remember days in the university stockroom, where this compound shared a locked shelf away from sunlight, warmth, and any hope of casual access. Its reputation earned every restriction placed on it.
Dangers Lurking in a Yellow Jar
Let’s not dance around it—2,4-Dinitroaniline stings eyes and burns skin on the lightest touch. Breathing in dust will leave lungs irritated, coughing up harsh reminders for days. Even though it’s not the most unstable nitroaromatic, dry storage and steady temperatures keep it from turning unpredictable. I always see colleagues treat it with the same wariness as other energetic nitro compounds. No burning candles or open flames nearby, because the risk won’t cut you any slack.
Choosing the Right Spot
I’ve walked into labs where 2,4-Dinitroaniline containers shared a shelf with solvents, acids, and even a few sadly neglected glass jars. That’s just asking for trouble. Moisture, heat, jostling, or accidental mixing can turn a simple storage slip into an emergency. I always suggest a separate cabinet, locked up, marked with clear hazard labels—avoid confusion, keep mistakes out.
The storage space should stay cool and dry. In my experience, ventilation matters more than most people think. Weak fans or plugged ducts turn dust or fumes into a growth problem. Don’t stash it by a window or on top of radiators. Sun and heat make things unpredictable, and a hot shelf tempts accidents.
Proper Containers Matter
Glass jars with airtight lids offer the best peace of mind. The original packaging, if intact, adds a layer of confidence—manufacturers design these to keep out moisture and air, and to resist spills. Never swap into plastic unless the chemical compatibility charts check out. Some folks get clever and use improvised containers; I’ve seen too many horror stories to endorse that route.
Thinking Past the Storage Cabinet
It’s not enough to just stash it and forget about it. Every team member should know what sits on the shelf and why it’s a hazard. Training sessions, clear signage, and regular audits remove the ambiguity. In one place I worked, surprise checks uncovered leaky lids and faded hazard labels before trouble found us. Inventory lists kept on hand helped us track usage and spot expired or decomposing stock early.
Disposal and Emergency Readiness
Disposal often gets pushed down the priority list until the last minute. That can mean hazardous waste bins loaded with careless mixes, upping the risk for anyone who moves them. Follow local hazardous-waste regulations every time. I always double-check with environmental safety staff before clearing out any old jars, and recommend not taking shortcuts here.
Regular Checks Save Lives
Routine inspections—real, hands-on checks—keep risk under control. Crusty jars, piles of dust around lids, sticky shelves: those are alarms, not minor messes. Fixing issues early beats making excuses after an incident. If everyone pitches in, stores chemicals correctly, and knows the odds, the lab stays a safer place for everyone.
What safety precautions are needed when handling 2,4-Dinitroaniline?
What Folks Handling 2,4-Dinitroaniline Should Know
Anyone who’s spent time in a chemistry lab learns to respect certain chemicals pretty quickly. 2,4-Dinitroaniline comes with its own set of risks. This yellow, crystalline powder shows up in dye manufacturing and chemical research, but it doesn’t play nice with people or the environment. I remember my first week in an organic research lab—we had a strict talk about nitro compounds. It wasn’t just about avoiding spills, but understanding why you keep your gloves on, why the goggles never come off, and why you never fumble with a mask just to scratch your nose.
Personal Protection: More Than a Checklist
It’s easy to underestimate a powder, but the dangers with 2,4-Dinitroaniline go beyond regular dust. Gentle skin exposure can cause irritation or even burns. Breathing in the particles may trigger headaches, dizziness, or worse. Standard issue is a pair of nitrile gloves—not just latex, since some organics slip right through. Lab coats and splash-proof goggles aren't just for show, even in university settings. Most labs require work in a fume hood by default. The exhaust system isn't there for aesthetics; it reduces the chances of anyone inhaling dust or vapors, which could cause lung problems over time.
Understanding the Fire Risk
The nitro groups make this compound a fire risk. Heating or dropping it next to an open flame could throw sparks. Heat-resistant gloves make sense during any procedure involving temperature changes. Fire extinguishers should sit close by, with sand buckets as backup. If you’ve ever watched a nitro compound ignite, you don’t forget that lesson.
Spills and Disposal: What Happens Next Matters Most
Spills can ruin more than your day—they can put your health on the line. I still remember cleaning up a minor spill, following every step our safety officer drilled into us. Cover the powder with a damp towel to avoid dust, scoop gently into a sealable bag, and label the waste. Anything contaminated—gloves, towels, paper—should go into hazardous waste containers right away. Rinsing down the sink doesn’t cut it. Waste disposal teams handle the rest, using methods that lower risk to groundwater and local wildlife. Regulations for chemical waste may seem like red tape, but they protect more than just the folks in the lab.
Training and Good Habits: The Best Defense
No matter how advanced the equipment looks, accidents start with lapses in memory. Regular training drills helped everyone in our department keep sharp. I’ve seen people get hurt by “just handling something for a minute” or “cutting corners to save time.” The best habit: treat every step with respect, from labeling bottles to cleaning glassware. Supervisors should push hands-on training, not just safety PowerPoints. Peer reviews and safety audits caught mistakes before they became big problems in our lab.
Keeping Risks Low for Everyone
Anyone working with 2,4-Dinitroaniline must focus on people, not just procedures. Good safety culture starts with honest conversations. If someone feels unsure, there must be a way to speak up before an accident happens. Investing in better gear or regular training pays for itself. Science moves fast, but staying healthy must come before every discovery.
| Names | |
| Preferred IUPAC name | 4,6-dinitroaniline |
| Other names |
1-Amino-2,4-dinitrobenzene 2,4-Dinitro-1-aminobenzene Dinitroaniline |
| Pronunciation | /ˌdaɪˌnaɪtroʊˈænəˌliːn/ |
| Identifiers | |
| CAS Number | 97-02-9 |
| Beilstein Reference | 87880 |
| ChEBI | CHEBI:51052 |
| ChEMBL | CHEMBL15600 |
| ChemSpider | 15337 |
| DrugBank | DB12983 |
| ECHA InfoCard | 100.007.032 |
| EC Number | 208-741-0 |
| Gmelin Reference | 69312 |
| KEGG | C14382 |
| MeSH | D003877 |
| PubChem CID | 6941 |
| RTECS number | BO8925000 |
| UNII | IJ26FPT5QN |
| UN number | UN1321 |
| CompTox Dashboard (EPA) | DTXSID0029067 |
| Properties | |
| Chemical formula | C6H5N3O4 |
| Molar mass | 198.13 g/mol |
| Appearance | Yellow to orange crystalline powder |
| Odor | Odorless |
| Density | 1.57 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | 1.98 |
| Vapor pressure | 0.00029 mmHg (25°C) |
| Acidity (pKa) | 18.10 |
| Basicity (pKb) | 13.34 |
| Magnetic susceptibility (χ) | -29.9·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.718 |
| Viscosity | 4.36 cP (25°C) |
| Dipole moment | 3.12 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 144.3 J⋅mol⁻¹⋅K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -9.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -902 kJ/mol |
| Pharmacology | |
| ATC code | D06AA10 |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and eye irritation, may cause respiratory irritation, suspected of causing cancer |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06,GHS03 |
| Signal word | Danger |
| Hazard statements | H301, H319, H335, H351, H400 |
| Precautionary statements | P260, P280, P305+P351+P338, P310 |
| NFPA 704 (fire diamond) | 2,4 Dinitroaniline: "3-2-1 |
| Flash point | 138 °C |
| Autoignition temperature | 570 °C |
| Explosive limits | 0.2–2.0% |
| Lethal dose or concentration | LD50 oral rat 590 mg/kg |
| LD50 (median dose) | 1750 mg/kg |
| NIOSH | DD0875000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.05 mg/m³ |
| IDLH (Immediate danger) | IDLH: 10 mg/m3 |
| Related compounds | |
| Related compounds |
1,3-Dinitrobenzene 2,4-Dinitrochlorobenzene 2,4-Dinitrophenol Aniline |

