2,4-Dimethyl-3-Pentanone: Peering Behind a Quiet Cornerstone of Chemistry
Tracing the Historical Roots
Long before synthetic pathways evolved into crisp computational models and green chemistry hit the limelight, chemists relied on basis molecules like 2,4-Dimethyl-3-pentanone to unlock new frontiers. I remember thumbing through older organic textbooks where its simple structure appeared in practical organic syntheses. Its development stretches back to classic carbonyl chemistry, often overshadowed by sexier reagents, yet it remained a quiet workhorse in research labs. Chemists exploring branched ketone synthesis leaned on this molecule for its modest reactivity and reliable formation through alkylation and acylation, tracing its relevance to advances from the mid-20th century onward. Whenever people laud progress in organic electronics or specialty polymers, they tend to overlook how foundational molecules like this paved the way.
Understanding the Core: Product Overview
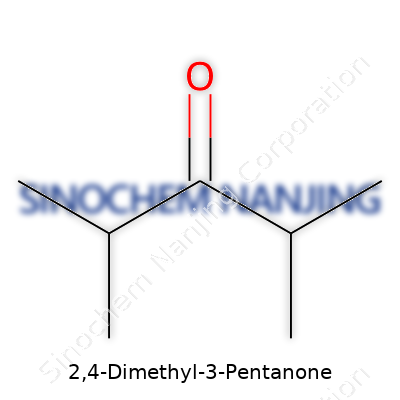
2,4-Dimethyl-3-pentanone falls under the camp of aliphatic ketones. Its formula, C7H14O, hints at a small twist on the simpler pentanone backbone. The methyl groups add a touch of steric flavor, transforming an otherwise unassuming chain into something more: a molecule with unique boiling, solvating, and reactive properties. Commercial supplies usually focus on its use as an intermediate rather than as an end product. This ketone rarely appears on store shelves by name, but its DNA threads through fragrances, fuel additives, and pharmaceutical precursors. Researchers who encounter it early in their studies soon recognize its signature structure in many synthetic routes.
Physical and Chemical Significance
It’s easy to gloss over catalog numbers in lab drawers, but physical and chemical properties matter. 2,4-Dimethyl-3-pentanone presents itself as a colorless liquid at room temperature, exuding a faint odor reminiscent of other small ketones. Boiling in the range of 140–145°C, it offers a higher thermal stability than shorter-chain family members. Its polarity allows some solubility in organic solvents, and its carbonyl group makes it reactive in forming derivatives like oximes or hydrazones. Unlike aromatic ketones, there’s no delocalization to stabilize the carbonyl, so nucleophilic additions react more predictably. I’ve watched newcomers underestimate its reactivity until a runaway addition taught them to respect its carbonyl strength.
Getting Technical with Specifications and Labeling
Industry and academia rely on clear technical specs for chemicals. Every bottle of 2,4-Dimethyl-3-pentanone demands careful labeling, reflecting not just identity but purity grade, allowable water content, and storage recommendations. Chemists track refractive index and density because real-world outcomes change with minor impurities. Without robust standards, experiments stall or fail, and no one wants to explain that to funding agencies. Over time, regulatory agencies like OSHA and REACH have pressed for tighter labeling, and everyone in a working lab learns to appreciate knowing precisely what comes out of each bottle.
How Synthesis Shapes the Molecule
Preparation techniques mark the difference between lab-scale convenience and industrial scalability. Most routes start with starting materials like acetone and isobutyl halides, leveraging alkylation or acylation under suitable catalysts. Modern labs reached for Grignard reagents or enolate alkylations, balancing selectivity, yield, and waste products. Scale-up magnifies every small inefficiency, so process chemists prioritize atom economy and mild conditions. Publications over the last decade describe greener processes using less hazardous solvents or recyclable catalysts, responding to sustainability demands without sacrificing throughput or purity.
Exploring Reactions and Derivatives
The chemistry of 2,4-Dimethyl-3-pentanone comes alive in the hands of creative researchers. Its methyl groups shelter the carbonyl somewhat, but not enough to restrict classic ketone reactivity. I’ve watched how it engages in aldol condensations, forming larger β-hydroxy ketones that build molecular complexity. In pharmaceutical explorations, chemists tweak this backbone through reductive aminations, producing tertiary amines for bioactive compounds. The molecule also enters hydrazone formation and enolate chemistry, standing as a flexible platform for synthesis. Exploring derivatives isn’t glamorous or headline-grabbing, but it drives incremental innovation beneath the surface of major discoveries.
Names and Aliases
Clarity in names matters because confusion kills time and cash. In journals, 2,4-Dimethyl-3-pentanone masquerades under synonyms like diisopropyl ketone or 3-Pentanone, 2,4-dimethyl-. Cross-referencing IUPAC and common names avoids cross-contamination and accidental mix-ups. Regulatory agencies insist on unified nomenclature, but everyday lab chatter always falls back on the shorthand, as anyone who’s spent time in mixed-discipline meetings can confirm.
Learning from Safety Rules and Operational Wisdom
No discussion of real-world chemistry gets far without touching on safety. Like many midweight ketones, 2,4-Dimethyl-3-pentanone poses both flammability and inhalation risks. Training isn’t just bureaucratic; it saves lives and avoids setbacks. Labs enforce proper ventilation and grounding when handling even small volumes, and anyone who’s watched a careless peer spark a solvent fire takes these things to heart. Modern standards demand more than an MSDS printout tucked behind a fume hood—demonstrated competence counts, and so does a community that takes shared responsibility seriously. Chemical hygiene plans often bake in regular refreshers and incident drills, which might seem excessive until a real mishap comes around.
Application Areas: Where It Shows Up
2,4-Dimethyl-3-pentanone has a reach that’s easy to overlook. It serves mainly as an intermediate, feeding into the production of more complex molecules. Pharmaceutical developers use it as a backbone for synthetic pathways, especially where branching proves crucial to biological activity. Some fragrance designers borrow its structure to design volatile components with desired evaporation rates. I’ve run across reports where energy researchers test its blends as possible fuel additives, banking on its branching to tweak combustion properties. In the world of fine chemicals, few outside the field know how many steps trace back to small intermediates like this.
Driving Research and Development Forward
Current R&D around 2,4-Dimethyl-3-pentanone mostly unfolds in quiet academic corners and underfunded industrial pilot lines. Chemists chase greener synthesis: reducing toxic byproducts and maximizing reuse of key reagents. Catalyst redesign experiments tread the line between cost and environmental burden. Scale-up efforts occasionally catch broader attention when a new process slashes waste or energy use. Translational research brings hope, especially as new pharmaceutical candidates emerge from hidden corners of combinatorial chemistry, where intermediates like this get built in bulk.
Toxicity Research and the Responsibility of Knowledge
Toxicology often lags behind innovation, and this holds true for many once-obscure intermediates. 2,4-Dimethyl-3-pentanone doesn't hold the notoriety of benzene or dioxins, but early animal studies have mapped out acute exposure hazards and flagged chronic risks. Inhalation brings classic solvent symptoms: dizziness, headache, and, in higher doses, pulmonary irritation. Labs enforce lower volume handling and diligent waste capture to stay ahead of regulatory shifts. Long-term data still needs to catch up, but the pattern remains clear: precaution beats remediation every time. The field benefits from any push toward alternatives with lower human and environmental impact.
Imagining the Road Ahead
Looking forward, 2,4-Dimethyl-3-pentanone will keep playing a part in layered industrial and academic syntheses. Its potential in more sustainable manufacturing lines holds appeal for teams chasing green chemistry targets. More transparent reporting requirements and improved analytical tracking can ensure that byproducts, exposures, and downstream derivatives remain safer for workers and the environment. Researchers stand to gain from new catalytic scaffolds that cut down on energy use, reduce hazardous waste, and boost overall atom efficiency. In a world increasingly focused on supply chain resilience and environmental stewardship, molecules like this will attract closer scrutiny and smarter engineering, ensuring that progress in chemistry brings as few regrets as possible in the years to come.
What is 2,4-Dimethyl-3-Pentanone used for?
How This Chemical Shows Up in the Real World
2,4-Dimethyl-3-pentanone doesn't grab headlines like some big-ticket chemicals, but it quietly plays a real part in labs and industry. Its structure—two methyl groups attached to a central pentanone backbone—gives it a set of useful physical and chemical properties. The compound flows as a colorless liquid with a slightly sweet scent, making it easy to handle compared to more volatile cousins. That’s not the sort of detail most folks dwell on, but being less likely to go up in vapor helps during storage and lab work.
Key Uses for Science and Industry
This molecule gets real traction as a building block for bigger, more complex chemicals. It acts as a starting point for synthesizing specialty solvents and intermediates—think ingredients in pharmaceuticals, flavors, or fragrances. In the pharmaceutical world, researchers use it to tweak or test new reactions because it delivers reliable results and doesn’t react in unexpected ways under standard lab conditions.
Labs often reach for 2,4-dimethyl-3-pentanone when developing or testing analytical techniques. For example, in chromatography, it helps sort out how well a system can separate chemicals with similar shapes and weights. By turning up in calibration tests, it ensures measurements stay accurate. Quality control teams in chemical plants rely on this kind of chemical consistency to spot trouble before it hits consumers or the environment.
Why Should This Matter Outside the Lab?
Chemical building blocks may seem removed from daily experience, but hidden work like this supports safer, more effective medicines, foods, and consumer products. The right intermediate keeps unwanted byproducts in check, making it less likely for impurities to slip through to the shelf. From an environmental point of view, using stable and predictable ingredients in manufacturing keeps production lines running cleanly without a string of accidents that could leak toxic vapor or waste water.
Balancing Safety and Progress
Like a lot of mid-stage chemicals, 2,4-dimethyl-3-pentanone comes with safety notes. Anyone handling it needs to suit up: gloves, goggles, and good ventilation are standard. Strict labeling and transport rules put checks in place, reducing spills or mix-ups along the supply chain. I’ve seen first-hand how small mistakes in chemical labeling can spiral, sending shipments to the wrong spot or causing confusion in busy warehouses. The safer route means clear rules and people who stick to them, right down to the last container.
Training for chemical handlers isn’t just a box-check. It gives people a fighting chance to spot problems early—maybe a leaky seal, or a spilled drop no one noticed at first. Companies investing in ongoing training, not just annual refreshers, see fewer workplace incidents and reduce legal or financial headaches, too. Those are lessons the field can’t afford to ignore, since the reputation of the entire supply chain depends on trust and transparency about what gets used, where it ends up, and what happens if it’s handled wrong.
Solutions and Smarter Practices
Safer alternatives grab more attention these days, but switching out well-understood chemicals takes real investment in research and infrastructure. For now, plugging safety gaps, investigating greener production techniques, and tightening reporting between suppliers all offer practical steps. A culture of responsibility, not just compliance, keeps everyone from the research team to the end customer in the loop. Chemicals like 2,4-dimethyl-3-pentanone may stay in the background, but the choices around their use ripple much further than most folks expect.
What are the safety precautions when handling 2,4-Dimethyl-3-Pentanone?
Recognizing Hazards in the Lab
Anyone working with chemicals like 2,4-Dimethyl-3-Pentanone knows there’s no room for shortcuts. This compound belongs to the family of ketones, which means it evaporates easily, has a strong smell, and can irritate skin, eyes, and lungs. Chemical burns, headaches, even longer-term nervous system problems—these aren’t just scare tactics. Over the years, I’ve seen what happens when even experienced chemists get a little too comfortable and skip safety steps. Most regretted it right away.
Personal Protective Equipment Really Matters
Back in grad school, I watched a classmate splash a ketone on his hand while pipetting. No gloves, because he “hated the feel.” Ended up with peeling skin and a nasty rash for weeks. Wearing nitrile gloves, safety goggles, and a lab coat makes more sense than a trip to the ER. With volatile vapors, a tight-fitting pair of goggles—never just glasses—keeps your eyes covered if something splashes or vapors drift up. Long sleeves and closed shoes finish the job, making sure you won’t have regrets after a spill.
Good Ventilation Isn’t Optional
A lab bench next to a door or tiny fan isn’t enough. Proper exhaust hoods suck away vapors before you can breathe them in. If you ever stepped out of a room filled with ketone fumes, you know how quickly headaches and eye irritation can hit. Over years, low-level exposure adds up. Smart labs save money in the long run by investing in real ventilation. I’ve run plenty of reactions with the sash pulled down low, and it makes a difference. Portable respirators only come out for spills and accidents, since hoods prevent most issues.
Safe Storage and Handling Techniques
I once opened a corroded metal container because someone stored a reactive ketone in the wrong bottle. Always stick with amber glass containers and label every bottle or flask—no exceptions. 2,4-Dimethyl-3-Pentanone catches fire easily, so it stays far from heat sources and out of sunlight. Fire blankets and extinguishers should be easy to reach close to where the chemical gets used. Any stock containers go into a ventilated flammables cabinet, away from oxidizers and acids.
Planning for Emergencies
It’s easy to laugh off safety drills, but I’ve watched minor leaks turn into emergencies. Spills get contained fast with absorbent pads made for organics. Cleanup crews should have training and a written plan nearby. Emergency showers and eyewash stations need to stay open and accessible, not blocked by extra boxes, just in case something splashes where it shouldn’t. Clean hands after every session and toss gloves in designated waste bins—no shortcuts there, either.
Ongoing Training and Support
People forget things, even those who handle chemicals every day. Regular refresher sessions keep everyone sharp. I’ve sat through my share of safety talks, and the ones that stick are always full of real-life stories rather than lectures. Supervisors and lab managers should set an example—if they’re not using PPE or following the rules, no one else will, either. Sharing close calls helps everyone remember why rules exist.
Staying Responsible
Working with 2,4-Dimethyl-3-Pentanone doesn’t have to be risky. Respect for chemicals, clear training, and the right gear turn danger into safe, productive work. Experience teaches the importance of prep work far more than any textbook ever could.
What is the chemical formula of 2,4-Dimethyl-3-Pentanone?
Getting Acquainted With the Structure
Not everyone enjoys deciphering long chemical names, but every so often, one pops up that gets the mind working: 2,4-Dimethyl-3-Pentanone. The name offers all the clues. The “pentanone” hints at a five-carbon backbone with a ketone smack in the middle. That ketone shows up at the third carbon: hence “3-pentanone.” Two methyl groups attach at carbons two and four. If you count it all up, you’ll find seven carbons, fourteen hydrogens, and one oxygen, giving us C7H14O.
Names and Numbers: Translating to Real Chemistry
Organic chemistry names make you imagine the molecule like a puzzle. Picture this: five carbons in a row. The third carbon carries the ketone group (a double-bonded oxygen), and methyl arms branch off carbons two and four. Chemists use this logical system to avoid confusion in the lab or plant, where little mistakes don’t just lead to wrong math, but real hazards or wasted money. That’s why the art of chemical nomenclature still holds value in a world of software auto-completion.
Why Understanding the Formula Matters
While textbooks dish out formulas, knowing how to visualize them connects people to the heart of chemistry. Take 2,4-dimethyl-3-pentanone—it’s a mouthful, but in classrooms and labs, it’s a chance to flex those mental muscles. More than a test question, this understanding spills into real life: design a chemical, and shape how it reacts, purifies, or breaks down.
Years spent in the lab, glassware in hand, show how one wrong substitution can flip a process upside down. If you grab something mislabeled and expect a simple five-carbon chain, but end up with a branched structure, your reaction path changes. You might lose days—and trust in your process. It’s about moving from ink on a page to products that help make plastics, pharmaceuticals, even flavorings.
Drawing Experience to the Lab Bench
I remember early days studying for organic chemistry exams—sleep blurry, hands stained with ink as I drew structure after structure. Building models out of toothpicks and marshmallows, sometimes just peas from a lunch tray, made sense of these long names. Years later, managing a work team in a plastics plant, those old lessons paid off again. Feedstock purity and exact molecular arrangements felt far from student exercises—if something changed, a production run could fail, costing thousands. That’s why a name like 2,4-dimethyl-3-pentanone carries more than a formula. It’s a code for building, troubleshooting, and improving.
Real-world Impacts and Solutions
Industry still relies on sound chemical information. Good labels and solid training aren’t just regulatory red tape—they make sure people handle the right chemicals the right way. Mislabeling or a bit of overconfidence introduces real risk. Teams that encourage everyone to double-check assumptions often avoid trouble before it starts. Working from a place of respect for chemical detail shapes safer practices as much as better products.
A deep understanding of compounds, from formula to full synthesis route, encourages smarter experimentation. In large-scale manufacturing, for example, verifying both the structure and supply chain for a specialty ketone like 2,4-dimethyl-3-pentanone keeps everything on track. Factoring in what can go wrong helps labs and plants head off problems, save costs, and protect people.
How should 2,4-Dimethyl-3-Pentanone be stored?
The Real Life Stakes of Chemical Storage
Storing any solvent gets personal fast once you’ve handled spills or had containers leak in a makeshift lab. 2,4-Dimethyl-3-Pentanone isn’t rare in the world of organic synthesis, and for anyone who handles chemicals with a flammable or volatile streak, small details in storage habits can draw the line between smooth workflow and expensive mistakes. Anyone working in a lab — whether academic, industrial, or even those small start-up shops — quickly learns the value of taking these things seriously, not cutting corners.
Flammability: The Fire Risk No One Can Ignore
Ask fire marshals, insurance agents, and seasoned lab techs about improper solvent storage. They'll point to the number one risk: fire. 2,4-Dimethyl-3-Pentanone carries a low flash point, making it easy to ignite if it ends up near open flames, sparks, or hot equipment. Never store it near sources of ignition — this means no smoking, no electrical panels, and absolutely no open flames in the vicinity. Store the chemical in sturdy containers approved for flammable liquids, away from sunlight to fend off both heat and UV exposure.
Ventilation and Containment
Fumes build up quickly in poorly ventilated spaces, so a properly ventilated flammable storage cabinet isn't just a suggestion — it's non-negotiable. Long-term exposure to those fumes in confined areas doesn’t just make the job unpleasant; it can affect health over time. A fellow researcher once shared how headaches and strong smells tipped them off to leaks — which later traced back to a half-closed cap within a cramped cupboard. Even small oversights add up.
Humidity, Temperature, and Physical Conditions
Extreme temperatures stress containers and increase evaporation rates, creating pressure build-up and raising leak risks. Always pick a cool, dry area for chemical storage. Humidity in storerooms can cause rust on metal containers, corrode caps, and eventually let vapor escape.
Any container showing signs of swelling, rust, or corrosion deserves replacement right away. Rigid plastic or glass containers tend to be best, with clearly legible hazard labels, so anyone can identify the contents in a hurry.
Separation from Incompatibles
Too many labs treat solvent storage as an afterthought, tossing bottles on a shelf without enough attention to what sits nearby. Keep oxidizers, acids, and other reactive chemicals far away from 2,4-Dimethyl-3-Pentanone. The right move always involves checking the chemical’s SDS — information directly from safety data sheets prevents mixing hazards.
Training and Emergency Preparedness
Even if a storage protocol seems clear, make sure everyone—old hands and new recruits—understands what to do in case of spills or fires. Regular drills matter, as do clear signage and eyewash stations nearby. Spill kits filled with absorbent materials, chemical-resistant gloves, and sturdy goggles need to stay within easy reach.
Practical Improvements and Solutions
Investing in high-quality storage cabinets rated for flammable chemicals often costs less than clean-up or liability after an incident. Keeping inventory tight—never buying more than needed—reduces risk and waste. Digital inventory systems prevent forgotten containers from lurking in back corners.
Finally, ask experienced colleagues for advice. Learning from others’ near-misses saves you from repeating the same mistakes, and it builds a stronger safety culture for everyone in the lab.
What are the physical properties of 2,4-Dimethyl-3-Pentanone?
How Its Structure Shapes Everyday Use
2,4-Dimethyl-3-pentanone doesn’t come up much in household chatter, but chemists know it by its clean, simple build. With a five-carbon backbone carrying two extra methyl groups, this molecule slips between water’s fingers but cozies up to organic solvents. Its clear, colorless liquid form lines up with other small ketones. A hint of sweetness in its scent puts it in the same ballpark as acetone, though it runs a bit heavier in the nostrils. It boils above water—around 115°C (239°F)—so it won’t vanish like alcohol or ether. Liquid at room temperature, it lets you work in open air without rushing against evaporation.
Volatility and Boiling Point: Handle with Care
I remember working with this ketone in a modest academic lab. Left open, it didn’t sting the nose the way acetone did, but it hung around long enough to be a mild distraction. The flash point sits around 25°C (77°F)—well below a warm summer day. That shows it doesn’t take much heat to put it into vapor form, and that vapor can find a spark. I kept my glassware away from any open flames, learned the hard way that spilled drops spread their smell. Anyone using it in bulk indoors should trust their ventilation system. Though not as risky as some solvents, the right focus on safety matters.
Solubility and Miscibility: Boundaries Show Up Fast
Drop this compound into water, and you see where chemistry draws the line. Its nonpolarity, thanks to all those methyl groups, blocks it from mixing well with water. Instead, it heads right for other organics—hexane, diethyl ether, benzene. This isn’t just academic; it shapes how folks in process industries separate, clean, or extract materials. Water won’t pull it away, which steers you toward different disposal options. Awareness of these tendencies means fewer surprises later, especially if you’re trying to clean up afterwards.
Density and State: Why It Still Relates to Practical Jobs
At room temperature, it flows with a density lower than water—close to 0.79 g/cm³. Tip a beaker of it onto a spill tray and it won’t sit still; it creeps fast, following gravity wherever it finds. It resists freezing unless you drop temperatures drastically—freezing point sits around -40°C. For anyone storing it in regular freezers or fridges, there’s no worry of solidification, and that kind of reliability helps in labs focused on consistency.
Potential Hazards and Responsible Use
Anyone who’s ever worked hands-on with organic ketones knows the irritation they can cause. It dries the skin, tingles on a cut, and vapor makes eyes water. Inhaling too much never does a body good, and repeated exposure deserves respect. Regulatory bodies flag it accordingly: you’ll see recommended limits to keep air quality reasonable anywhere it’s used. Gloves, goggles, open windows—the basics carry everyone safely through an experiment or a day on the production line. Common sense, learned early, holds up year after year.
Thinking About Alternatives and Solutions
The world doesn’t stand still on solvents. Some companies, teachers, and researchers look for greener picks, sometimes swapping in less volatile molecules. Rethinking processes that use 2,4-dimethyl-3-pentanone can lower risk and cut emissions. Sometimes that means collaborating with suppliers to secure closed-loop systems or adjusting recipes so less of it escapes into the air. The work isn’t flashy, and it doesn’t make headlines, but steady efforts win the safety and sustainability race in chemistry labs and industrial floors, every day.
| Names | |
| Preferred IUPAC name | 3,5-Dimethylpentan-2-one |
| Other names |
Diisopropyl ketone 2,4-Dimethylpentan-3-one Isopropyl isopropyl ketone |
| Pronunciation | /tuː, fɔːr daɪˈmɛθɪl θriː pɛnˈteɪnoʊn/ |
| Identifiers | |
| CAS Number | `565-69-5` |
| Beilstein Reference | 1209246 |
| ChEBI | CHEBI:89341 |
| ChEMBL | CHEMBL1537087 |
| ChemSpider | 12081 |
| DrugBank | DB01852 |
| ECHA InfoCard | 400011654953 |
| EC Number | 207-419-1 |
| Gmelin Reference | 107932 |
| KEGG | C06642 |
| MeSH | D017824 |
| PubChem CID | 8695 |
| RTECS number | SA8575000 |
| UNII | WAR8456E3P |
| UN number | UN1157 |
| Properties | |
| Chemical formula | C7H14O |
| Molar mass | 100.16 g/mol |
| Appearance | Colorless liquid |
| Odor | sweet |
| Density | 0.801 g/mL at 25 °C (lit.) |
| Solubility in water | Insoluble |
| log P | 1.5 |
| Vapor pressure | 3.7 mmHg (20°C) |
| Acidity (pKa) | 20.6 |
| Magnetic susceptibility (χ) | -55.0x10^-6 cm^3/mol |
| Refractive index (nD) | nD 1.406 |
| Viscosity | 0.76 mPa·s (20°C) |
| Dipole moment | 2.75 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 340.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -246.1 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3253 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | '' |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02, GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H315, H319, H335 |
| Precautionary statements | P210, P280, P301+P312, P303+P361+P353, P305+P351+P338, P370+P378 |
| NFPA 704 (fire diamond) | 1-3-1 |
| Flash point | 41 °C (closed cup) |
| Autoignition temperature | 514 °C (957 °F; 787 K) |
| Explosive limits | 1 to 6.2% (in air) |
| Lethal dose or concentration | LD50 (oral, rat): 1700 mg/kg |
| LD50 (median dose) | LD50 (median dose): 2500 mg/kg (rat, oral) |
| NIOSH | UJ8575000 |
| REL (Recommended) | REL: 50 ppm (230 mg/m³) |
| IDLH (Immediate danger) | 500 ppm |

