2,4-Dichlorophenol: Reflecting on a Chemical’s Journey and Its Place in Modern Industry
Historical Development
2,4-Dichlorophenol didn’t arrive in labs by accident. Decades ago, scientists looking for dependable building blocks found that swapping a couple of chlorine atoms onto phenol led to a chemical with new potential. Its story has roots in an age when chemistry meant crawling through stacks of glass vials, each holding a promise of something new or vital for agriculture or manufacturing. Chemists recognized that adding chlorines onto a benzene ring changed the way molecules reacted, smelled, and performed. By the middle of the twentieth century, companies realized this compound could serve as more than a curiosity. It became a stepping stone: essential for making other chemicals, especially herbicides like 2,4-D, and for foray into dyes, wood treatment, and industrial disinfectants. I’ve seen how historic choices about which chemicals get invested in have long tails. 2,4-Dichlorophenol’s path is a classic case—something once on the margins now ends up all over the landscape, literally.
Product Overview
Think of 2,4-Dichlorophenol as a foundation chemical. Straightforward in its structure and function, it packs quite a punch for its size. Manufacturers don’t build household products out of it, but it helps make those products work—especially things designed to fight weeds or control mold. It’s easy for folks to overlook chemicals like this, but those making decisions in agriculture, disinfectant production, and chemical synthesis know that cutting out 2,4-Dichlorophenol means hunting for a new reliable alternative, which isn’t as simple as swapping out a widget. I’ve talked to seasoned plant managers who remember when switching supply chains caused headaches precisely because foundational chemicals like this don’t grow on trees.
Physical & Chemical Properties
This compound enters the scene as a white to off-white solid, bringing a distinctive, sharp odor—anyone who’s worked with it remembers that strong, phenolic punch. The molecule has two chlorine atoms sitting on the aromatic ring, making it less volatile than plain phenol. It doesn’t dissolve well in water, but it dissolves much better in organic solvents like ether or chloroform, which make it useful for synthesis. Its melting point usually hovers around 41 to 43°C. Chemists appreciate its stability, but anyone handling it will notice it reacts aggressively in alkaline conditions. All these details don’t just sit in a reference manual—they guide decisions in labs, and the practical aspects keep operations honest. Years spent working alongside industrial chemists taught me that things like melting points and solubility mean a lot more once you have to run a distillation column or plan routine maintenance in an active facility.
Technical Specifications & Labeling
With a compound like 2,4-Dichlorophenol, accuracy in labeling and handling information matters just as much as the material itself. Industry professionals expect suppliers to mark containers clearly, not just with a name but also with essential details like purity, batch number, hazard symbols, and storage guidance. Inconsistent or inaccurate labels spell real trouble—especially for downstream users blending chemicals or conducting quality control. Hazard communication standards, such as those laid out by OSHA and GHS, set the baseline, but anyone who’s seen a facility audit knows that real safety comes from more than meeting minimum requirements. Over the years, as I’ve reviewed chemical storage rooms, I’ve learned that simple mistakes—a mislabeled drum or missing safety sheet—can snowball fast. Proper labeling isn’t glamourous, but it keeps teams informed, equipment safe, and companies protected from liability.
Preparation Method
Production routines for 2,4-Dichlorophenol rely on chlorinating phenol under controlled conditions. The factory floor often smells sharply for a reason—chlorination emits gases that need strong ventilation. Most processes introduce chlorine gas to phenol dissolved in a solvent or suspended in water. Running the reaction at the right temperature, and keeping time tight, makes the difference between high yield and a soup of unwanted byproducts. It’s chemistry that draws a line between reliable industrial-scale output and unpredictable batch outcomes. Chasing better yields kept R&D teams busy for years; process chemists hunt for catalysts or safer conditions that don’t sacrifice efficiency. This pursuit isn’t theoretical—cost savings on feedstock, fewer hazardous byproducts, and streamlined purification steps help facilities stay competitive and avoid regulatory pushback.
Chemical Reactions & Modifications
At its core, 2,4-Dichlorophenol offers a jumping-off point for making all sorts of specialty chemicals. The two chlorines in place make it a powerful intermediate. Add a bit of chemistry know-how and it’s possible to turn this into herbicides, antiseptics, dyes, and complex laboratory reagents. Reactions range from simple substitutions—swapping out a hydrogen or further chlorinating—to full-on coupling reactions with other aromatic systems. Its phenolic nature lends itself to creation of esters and ethers, which broadens its role in synthesizing pharmaceutical intermediates and advanced materials. Listening to veteran synthetic chemists, stories come up about challenging routes to specialty products that started with this very compound. Chemical versatility often makes or breaks the economics of running a plant.
Synonyms & Product Names
Chemicals wear a lot of hats depending on the context. 2,4-Dichlorophenol goes by names like Dichlorophenol or 1-Hydroxy-2,4-dichlorobenzene. CAS numbers—120-83-2 in this case—help clear up confusion, especially with imports or regulatory filings. During my early years in the industry, sorting through imports for a specialty manufacturer often meant double-checking every label, as the same compound could enter a facility under several trade or common names. Recognizing that a delivery labeled only “DCP” referred to 2,4-Dichlorophenol avoided several arguments and costly delays.
Safety & Operational Standards
Handling 2,4-Dichlorophenol safely presents real-world challenges that go beyond reading a safety manual. The compound is toxic if swallowed, inhaled, or absorbed through the skin. Anyone who’s attended a chemical exposure drill appreciates the importance of personal protective equipment and solid hygiene routines. Facilities with any volume of throughput need engineering controls—good ventilation, clear containment protocols, and fast eyewash/shower access. Beyond physical safety, correct record-keeping and waste management protect both workers and local communities. Getting these protocols right has a direct impact: fewer accidents, faster emergency response, and better regulatory compliance. During a site visit, seeing a facility ignore the basics—unlabeled secondary containers, open vents, lax glove use—left a deep impression that safety standards can’t remain on paper.
Application Area
The reach of 2,4-Dichlorophenol spreads far wider than most people realize. Its most notable job sits within the synthesis of major herbicides like 2,4-D, shaping food production and landscape management across the globe. Alongside that, it finds use in forming antiseptics, fungicides, and wood treatments. Occasionally, it sneaks into water treatment plants as a precursor for certain disinfectants. Specialty chemical manufacturers also use it as a launchpad for making dyes, solvents, and specific advanced materials. My experience reviewing environmental data taught me that even light industrial users manage to purchase the compound, leveraging its versatility in research and nuanced technical projects.
Research & Development
R&D teams keep circling back to 2,4-Dichlorophenol not just because it gets the job done, but because its properties offer a toolkit for chemists designing new molecules. A pattern appears in patent filings and journal articles—modifying the aromatic ring or swapping out functional groups can lead to new products with real-world use. Agricultural companies look for ways to reduce off-target toxicity or environmental persistence. Materials scientists use it for making polymers with customized characteristics. Environmental chemists, meanwhile, study the breakdown pathways and strive to design remediation strategies for contaminated sites. Every generation of researchers hurdles the same obstacles: maximizing efficiency while cutting costs and reducing hazards. Years working alongside chemical engineers highlighted the role collaboration and relentless testing play in finally signing off on a new synthetic route.
Toxicity Research
The legacy of 2,4-Dichlorophenol can’t be separated from its toxicological profile. Animal studies and environmental reports build a clear picture: The compound disrupts biological systems at low concentrations. Skin contact and inhalation present acute risks—burns, respiratory irritation, and in severe cases, systemic poisoning. Chronic low-level exposure lingers as a concern given its persistence in soil and water. Regulatory agencies, including the EPA and WHO, stress exposure limits and require rigorous monitoring wherever the compound appears in significant volumes. Historical cases of environmental contamination motivate stricter standards, especially where local communities depend on ground or surface water. My time consulting in environmental health revealed how persistent compounds like this reshape entire monitoring programs, not just for direct users but all the way down to affected ecosystems.
Future Prospects
2,4-Dichlorophenol stands at a crossroads. Its effectiveness as a chemical building block secures its spot in industry for the foreseeable future, especially for agriculture and specialty synthesis. At the same time, growing pressure to reduce environmental footprints and health hazards drives research into alternatives. Biobased phenols, greener chlorination processes, and tighter emission controls all compete for attention as regulatory standards rise. I’ve watched trade shows and technical conferences fill up with startups and university spinouts pushing non-chlorinated herbicides and faster-degrading substitutes. Nevertheless, dropping a workhorse compound rarely happens overnight—legacy infrastructure, regulatory inertia, and supply chain realities shape the market. The real test for 2,4-Dichlorophenol’s future will be how effectively industry shifts toward next-generation safety standards without sacrificing reliability or cost. The chemical’s story is a mirror for larger shifts: where science, industry, and society all negotiate the value and risks of innovation on a human scale.
What is 2,4-Dichlorophenol used for?
Where 2,4-Dichlorophenol Shows Up
2,4-Dichlorophenol might sound like something straight from a high school chemistry class, but it’s a fairly common name for anyone digging into the world of chemical manufacturing and environmental safety. This compound doesn’t stand alone for long in the real world. Chemical factories use it as a starting ingredient—kind of like flour for bread, but here, we’re talking about herbicides and antiseptics, not sandwiches.
Key Roles in Industry
Big manufacturers lean on 2,4-Dichlorophenol to make 2,4-dichlorophenoxyacetic acid (2,4-D), a pretty famous weed killer. Farmers and groundskeepers spray 2,4-D on their fields to clear out unwanted plants. Middle-aged gardeners recognize the stuff—the smell clings to the back of your throat after working around recent applications. The weed killer’s popularity stretches back to the 1940s, so it’s everywhere: by roadsides, in ditches, behind barns.
Factories also turn to 2,4-Dichlorophenol for making antiseptics and disinfectants. These products help scrub out germs in hospitals, restrooms, and sometimes in homes. Before reaching store shelves, the chemical plays a part in producing other chemicals too—so it’s easy to overlook how far its reach goes. That cleaning product under your sink? It might trace its roots to a plant using this compound.
The Environmental Angle
Tracing where 2,4-Dichlorophenol travels from factories can turn up some harsh truths. Wastewater from industrial processes sometimes carries this chemical along into rivers or lakes. That isn’t just a paperwork problem for regulators. The compound doesn’t break down quickly, so it can linger in soil or water for weeks, pushing worries about long-term effects on fish, plants, and wildlife. Studies point to toxic effects, especially for aquatic life. In one survey, researchers saw inhibited growth in algae and invertebrates when exposed to water containing even low levels of this chemical.
Homeowners flush trace amounts down drains each time those cleaning products do their job. It doesn’t take much to add up, especially in huge cities where thousands of folks clean up every day. The government points to 2,4-Dichlorophenol as a key “priority pollutant”—a flag meaning its presence gets attention from water safety experts and legislators.
Balancing Benefits and Risks
Nobody enjoys living with weeds, pests, or bacteria—so the value of an effective herbicide or disinfectant feels easy to understand. At the same time, folks living near chemical plants or downstream of big farms sometimes report headaches, strange smells, or concerns about their water quality. In my own community, people organize water testing events after heavy rain, since runoff can carry chemicals into streams. Many demand clear answers about what’s in their environment, and whether the water is truly safe.
The EPA requires careful handling and sets strict discharge limits for chemicals like this. But leaks and accidents still happen. I’ve visited sites where the promise of jobs and growth from new factories clashed with concerns from neighbors about chemical smells and odd tastes in the tap water.
How Communities and Industry Can Respond
More folks expect transparency about how chemicals travel from factory floors to neighborhoods. Local governments push for better monitoring. Wastewater treatment upgrades play a role, especially with new filtration technologies that capture more contaminants before they hit rivers. Stronger labeling for household cleaners helps too—letting people make informed choices and encouraging safer disposal habits.
Researchers keep developing alternative chemicals that break down faster or carry less toxicity, and industry groups promote these options for at least some uses. It’s not about stopping chemicals outright but about better balance between what we need for crops, hygiene, and public health, and the responsibility to keep our environment safe.
Is 2,4-Dichlorophenol hazardous to health?
Real Concerns from Real Exposure
I once lived near a lumber yard. The air sometimes carried a sharp odor, a bit musty, noticeably chemical—something you couldn’t quite pin down. Over the years, locals grew worried about health issues popping up. That yard used chemicals for preservation and pest control, including ones related to 2,4-Dichlorophenol (2,4-DCP). This compound, used in making herbicides, disinfectants, and fungicides, doesn’t get the same spotlight as notorious chemicals, yet its story carries weight.
How 2,4-Dichlorophenol Enters Daily Life
Factories sometimes release 2,4-DCP into the air or water. Sometimes, it even shows up in the soil. Households using certain disinfectants or lawn treatments might unknowingly handle trace amounts. I’ve worked on community garden projects where neighbors worried if the soil was safe. Health dangers always felt closer to home than what reports or technical documents suggested.
Health Risks Backed by Science
Researchers have linked 2,4-DCP to a range of health troubles. The U.S. Environmental Protection Agency places it among hazardous substances, and the Centers for Disease Control and Prevention highlights risks if you swallow, touch, or breathe high doses. Reports cite symptoms like headaches, burning skin, throat irritation, and coughing. In animal studies, liver and kidney problems showed up after repeated exposure.
The International Agency for Research on Cancer classifies 2,4-DCP as “possibly carcinogenic to humans.” While that’s not an ironclad verdict, the evidence gives plenty of reason to be cautious. European bans on similar chlorophenols reflect the trend of tightening safety rules globally.
Environmental Ripple Effects
Chemicals like 2,4-DCP don’t just threaten humans. They spread through water and linger in sediment. Fish and other aquatic life often pay the price first. I’ve walked along streams near manufacturing sites and seen stretches where nothing seemed to grow. Community groups have documented mutations and low survival rates in exposed wildlife. These signs point to a bigger circle of harm beyond factory gates or yard fences.
Guarding Health at Home and Beyond
It’s tempting to think big chemical problems belong to someone else, somewhere else. Experience proves they reach well beyond their original sources. People should push for strong air and water monitoring near manufacturing plants and ask for regular, transparent reporting. At home, reading product labels matters. Choosing alternatives without chlorophenols can lower exposure.
Workers face the greatest risks, so protective gear and safety training make a genuine difference. I’ve talked with plant employees who felt safer after their management invested in solid protections. Advocacy focuses public attention and pushes lawmakers to keep up with the science. If safer substitutes exist for a process or product, making the switch helps everyone.
Moving Toward Safer Communities
Communities who’ve dealt with legacy contamination often raise their voices about proper cleanup and responsible waste disposal. Safety doesn’t belong on an industry wish list—it should anchor daily practice. With new research and early detection, health professionals can catch problems sooner. Teaching young people about environmental health builds awareness for the next generation.
Chemical safety isn’t just for experts; it’s for people breathing the air, drinking the water, or picking from backyard gardens. Knowing the facts and acting locally can make a real difference.
What are the storage and handling precautions for 2,4-Dichlorophenol?
Why Care About Storage: Health, Safety, and the Environment
2,4-Dichlorophenol turns up in a lot of chemical processes, especially in herbicide and dye manufacturing. It smells sharp and it doesn’t play nice with skin or lungs. I remember working in a small lab where we only needed small jars of this chemical. Some co-workers started skipping steps, thinking plastic jugs would do for short-term storage—until rough handling cracked a container open. The fumes hit hard, irritating the air and making it clear: shortcuts invite trouble.
What Makes 2,4-Dichlorophenol Tricky to Store
This compound reacts with air, moisture, and strong bases. It can corrode some materials and leach through simple plastic. Over time, vapors leech out and spread a pungent odor. Spills soak into porous surfaces, and scrubbing does little to remove the smell or hazard. In warm or wet conditions, decomposition speeds up and the risk of toxic byproducts rises.
Proper Storage: Practical Steps That Matter
Glass and high-quality polyethylene containers work best for long-term storage. In a cool, dry, well-ventilated storeroom, keep bottles tightly sealed and labeled. Thick gloves and goggles make a difference, since just a quick splash burns skin. Store far from food, water, or any snacks people might bring into a break room. Anyone who’s ever had a lunch ruined by the sharp scent of 2,4-Dichlorophenol wafting through the door knows how far those vapors drift.
Good ventilation does more than keep away bad smells—it prevents fumes from building up to dangerous levels. At one site, a neglected bottle cracked and let out enough vapor to trigger a fire alarm. Extra ventilation cleared the room fast, saving both time and a major cleanup headache.
Don’t Ignore the Waste
Disposal often gets brushed aside, but this chemical requires more than just pouring down the drain. Local laws often require hazardous waste pickup. Pouring in the sink, aside from being illegal in many places, usually means sending the problem downstream, straight toward public water. In my experience, a simple consultation with a waste handler made the disposal question simple and safe. Keep all waste in solid, labeled containers, sealed and stored away from the main work area until pickup.
Training and Daily Practice
Training keeps people safe. New lab members need real examples of what a poorly stored bottle can do—photos, stories, even a whiff of the strong, biting scent that signals trouble. Emergency showers, eyewash stations, and proper spill kits stand ready in every safe workspace. One slip or distraction, and someone can end up rushed to urgent care for chemical exposure. People who know what to expect react faster and think more clearly when accidents happen.
Finding Better Solutions
Some larger companies swap 2,4-Dichlorophenol for less hazardous alternatives. Where that’s not realistic, tight controls, routine checks, and strong safety culture take priority. Digital inventory logs, frequent container inspections, and practical safety briefings every shift all help prevent problems before they start. Every careful step taken now saves much bigger headaches down the line—for people, the building, and the environment everyone relies on.
How should 2,4-Dichlorophenol spills be cleaned up safely?
Nobody Wants 2,4-Dichlorophenol in the Wrong Place
Few people outside chemical plants or labs spend much time thinking about 2,4-Dichlorophenol, but its risks are real. This is a compound used in herbicides and disinfectants, and it’s not what you want anywhere near water or soil outside a tightly controlled setting. I once worked in facilities that handled all kinds of chemicals, and I remember the tension whenever a spill occurred—even a small one. Spills don’t stay contained on their own, and the clock starts ticking for human health and the environment once any toxic substance escapes storage.
PPE Isn’t an Afterthought—It’s Step One
Right out of the gate, personal protective equipment turns into a lifeline: chemical-resistant gloves, splash-proof goggles, long sleeves, and a reliable respirator. I’ve known folks who thought they could just grab some towels and mop things up—big mistake. 2,4-Dichlorophenol can absorb through skin and irritate mucous membranes, so it’s never a case for bare hands or a T-shirt. No one should send staff into a room with unknown vapors or residues, not even for a “quick check.”
Fast Moves Stop Big Trouble
Chemical spill kits don’t gather dust for long in well-run facilities. Absorbent granules or specialized pads should go around the liquid immediately, and that means scooping the right material, not grabbing the old box of kitty litter. In my experience, speed and the right gear win every time. Fumbling around raises risks for the person cleaning up, and also for people nearby. If a puddle runs into a drain, whole systems have to be checked and treated, making everything harder and more expensive. A single minute counts for a lot in spill management.
Ventilation and Containment
Some chemicals release strong fumes at room temperature, and 2,4-Dichlorophenol is no exception. Getting fresh air flowing and quarantining the area reduces exposure and keeps more people from breathing in toxic vapors. Open windows and exhaust fans matter, so does closing off the spill site. Doors stay shut and signs go up. No outside traffic means no spreading the problem onto shoes or tools that head home after a shift.
Proper Waste Disposal Makes a Difference
Collected waste doesn’t belong in the usual trash. Used pads, gloves, and rags have to find their way into sealed containers clearly labeled as chemical waste. Hauling off that material requires experienced hazardous waste handlers. Some places cut corners, sending bags to regular landfills—those mistakes poison water and soil for years. Regulations exist for a reason, and it pays off to follow them by the book. The EPA calls for specifics: incineration or other methods that totally break down these stubborn molecules, leaving nothing harmful behind.
Training and Emergency Plans
Experience tells me accidents catch even the most careful teams off guard. Refresher drills matter more than posters on the wall. Real response plans mean everyone knows who to call, what to seal off, and how to handle equipment. I’ve seen workplaces where people run in all directions; confusion leads to panic and real injuries. Regular practice, not paperwork, builds confidence for the moment something actually goes wrong.
Long-Term: Invest in Prevention
Every cleanup teaches the same lesson: it’s far better to prevent a spill than scramble to fix one. Investing in double-walled tanks, leak-proof containers, and up-to-date detection alarms saves time, money, and health. Taking every small leak seriously—no exceptions—keeps trouble from snowballing. Spills aren’t just an inconvenience: they risk the safety of workers, the community, and our shared environment. Careful cleanup matters, but stopping spills at the source will always be the smarter move.
What is the shelf life of 2,4-Dichlorophenol?
Why Shelf Life Matters in Chemical Storage
Ask anyone who's worked in a lab or an industrial plant, and they’ll tell you—shelf life isn’t just a detail you check off when supplies roll in. For a chemical like 2,4-Dichlorophenol, commonly used as an intermediate in the manufacture of herbicides and antiseptics, understanding how long it stays stable shapes everything from safety decisions to bottom-line costs.
Chemical Stability: What Changes Over Time?
2,4-Dichlorophenol has a reputation for hanging in there. Its molecular structure, with those two chlorine atoms attached to the phenol ring, resists breaking down under normal conditions. In a clean, sealed container, kept away from sunlight, high heat, and moisture, it can sit on a shelf for three to five years before you start seeing degradation. The chemical itself has a melting point around 43°C, and it doesn’t volatilize quickly at room temperature—both facts help it last longer.
I remember a cabinet at the back of our college’s storeroom labeled “aromatic compounds.” Some dusty bottles in there had been unopened for years. We tested a decade-old 2,4-Dichlorophenol batch and, using GC-MS, got results nearly identical to a brand new supply. The difference showed up only after chemical exposure to open air—color darkened a bit, and there was a faint odor shift. That tells you purity can last, but environmental exposure chips away at it.
Risks of Ignoring Shelf Life
Over time, exposure to air and moisture kicks off slow oxidation. That leads to breakdown byproducts. Even trace degradation matters. In industrial production, contaminated intermediates can sabotage reaction yields, throw off quality control, or even form hazardous byproducts. Storage in warm or damp conditions increases the chances of polymerization or hydrolysis, which can gum up lines or lead to dangerous pressure build-up. I’ve seen production lines grind to a halt over something as simple as a cloudy sample in a feed tank.
Outdated stocks also creep into research and development. Use a degraded reagent, and your experiment can go sideways—not because your hypothesis failed, but your input wasn’t pure. Students and junior researchers get frustrated and waste time troubleshooting failed syntheses caused by expired chemicals.
How Manufacturers and Users Can Do Better
For 2,4-Dichlorophenol to stay usable, storage conditions make all the difference. Glass or HDPE containers, with airtight seals, prevent air and moisture exposure. Keeping bottles in a cool, dark facility, well away from direct sunlight and dampness, protects against accidental breakdown. Facilities should label every incoming shipment with a “date received” and practice regular rotation—use the oldest stock first. Nobody wants a backup bottle lurking in the shadows for ten years, forgotten until an emergency.
Manufacturers run regular quality control on stored stocks. A simple TLC or HPLC assay every six months lets you catch degradation early. Large-scale users benefit from keeping digital inventory logs that track expiration periods. Proper hazardous waste protocols for expired material keep people and the environment safe.
Looking Ahead
Ultimately, no magic formula extends the shelf life beyond good storage and regular checks. Responsible stewardship over chemicals like 2,4-Dichlorophenol isn’t just about compliance—it’s about trust, safety, and getting reliable results, every single time. Laboratory culture shapes these habits. Understand shelf life, stay vigilant, and buy only what you’ll use in the next few years, rather than betting on shelf stability to save costs down the road.
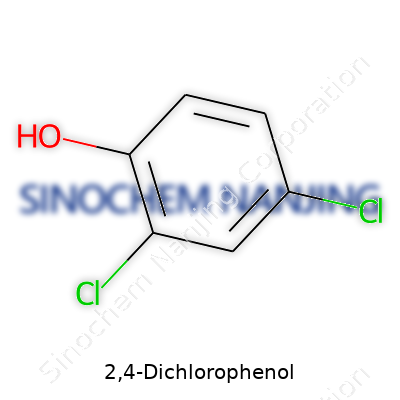
| Names | |
| Preferred IUPAC name | 2,4-dichlorophenol |
| Other names |
1-Hydroxy-2,4-dichlorobenzene 2,4-DCP Dichlorophenol Dichlorophenol-2,4 p-Dichlorophenol |
| Pronunciation | /ˈdaɪˌklɔːrəˌfiːnɒl/ |
| Identifiers | |
| CAS Number | 120-83-2 |
| Beilstein Reference | 1209224 |
| ChEBI | CHEBI:15838 |
| ChEMBL | CHEMBL18872 |
| ChemSpider | 5179 |
| DrugBank | DB02693 |
| ECHA InfoCard | 04e32749-0adf-4066-8a05-bcfa5b63a68a |
| EC Number | 3.1.1.65 |
| Gmelin Reference | 5886 |
| KEGG | C01530 |
| MeSH | D004073 |
| PubChem CID | 1385 |
| RTECS number | SN1575000 |
| UNII | 9C7YQ9NM6L |
| UN number | 2641 |
| Properties | |
| Chemical formula | C6H4Cl2O |
| Molar mass | 163.00 g/mol |
| Appearance | White to yellowish crystals or powder |
| Odor | Phenolic odor |
| Density | 1.38 g/cm³ |
| Solubility in water | 3.0 g/L (20 °C) |
| log P | 2.76 |
| Vapor pressure | 0.04 mmHg (25 °C) |
| Acidity (pKa) | 7.9 |
| Basicity (pKb) | 7.94 |
| Magnetic susceptibility (χ) | -73.0e-6 cm³/mol |
| Refractive index (nD) | 1.553 |
| Viscosity | 2.66 mPa·s (25 °C) |
| Dipole moment | 2.67 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 146.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -234.0 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3757 kJ/mol |
| Pharmacology | |
| ATC code | D08AE01 |
| Hazards | |
| Main hazards | Causes severe skin burns and eye damage. Harmful if swallowed. Toxic to aquatic life with long lasting effects. |
| GHS labelling | GHS02, GHS05, GHS06 |
| Pictograms | GHS06, GHS05 |
| Signal word | Warning |
| Hazard statements | H302 + H315 + H319 + H400 |
| Precautionary statements | P261, P273, P280, P305+P351+P338, P310 |
| NFPA 704 (fire diamond) | 2,4-Dichlorophenol: "3-2-0 |
| Flash point | Flash point: 108 °C |
| Autoignition temperature | 605°C |
| Explosive limits | Explosive limits: 2.4–18% |
| Lethal dose or concentration | LD50 oral rat 580 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral 580 mg/kg |
| NIOSH | SN2075000 |
| PEL (Permissible) | 50 mg/m3 |
| REL (Recommended) | 0.5 |
| IDLH (Immediate danger) | 100 ppm |
| Related compounds | |
| Related compounds |
Phenol 2-Chlorophenol 4-Chlorophenol 2,4,6-Trichlorophenol 2,5-Dichlorophenol |

