2,4-Dichloronitrobenzene: A Close Look at Where Chemistry Meets Industry
Historical Development
If you dive deep into the archives of chemical industry history, 2,4-dichloronitrobenzene stands out in the lineup of substituted nitrobenzenes. Its story traces back to the years when synthetic dyes and agrochemicals were just beginning to change modern agriculture and manufacturing. Researchers tinkering with nitration and halogenation of aromatic compounds soon realized that adding chlorine atoms to nitrobenzene opened new doors. This compound quietly powered advancements through its use as both a building block and an intermediate, turning traditional industries more innovative. Amid global industrial growth after both world wars, chemists took hold of this molecule, tailoring it for specialty uses in crop protection, pigments, and pharmaceutical synthesis. In labs and factories, practical knowledge about safe handling and process optimization grew alongside production, gradually shaping reliable supply chains. Those early choices continue to influence the chemistry workforce, safety standards, and environmental protocols seen today. 2,4-dichloronitrobenzene, once a curiosity, now connects a chain stretching from research to results in the field and on the production floor.
Product Overview
People who encounter 2,4-dichloronitrobenzene in industry recognize its pungent odor and pale yellow crystals, often arriving in drums destined for chemical plants. Known among chemists and manufacturers as a key intermediate, it rarely stays idle on a shelf. The compound forms the backbone of many syntheses, particularly where specific arrangement of chlorine atoms is essential. Over the years, its consistent physical properties and predictable chemical behavior have made it reliable in large-scale production. At a time when raw material volatility can disrupt entire supply chains, 2,4-dichloronitrobenzene provides some certainty for companies banking on uninterrupted operations. The market depends not only on purity but also on clear labeling and technical transparency, which allows buyers to assess suitability for particular applications ranging from herbicide precursors to intermediates for dyes and pharmaceuticals.
Physical & Chemical Properties
A closer look at the substance reveals a compound that stands out both for its utility and its risk profile. It melts at around 53°C, presenting as yellow crystals that dissolve well in organic solvents but resist water. The nitro group and two chlorines fixed on the benzene ring shape its reactivity and storage needs. Because of these functional groups, it responds well to nucleophilic substitutions and reductions, opening a path to plenty of end-use chemicals. Chemists value this predictability: the compound doesn’t surprise during standard synthetic reactions, which makes process design and scaling less fraught with uncertainty. Practical experience says don’t ignore inhalation risks, so workspace ventilation and containment remain front-line priorities. Over the years, stringent standards emerged to keep inhalation, skin absorption, and contamination under tight control, echoing lessons learned from generations of industrial operators.
Technical Specifications & Labeling
On a shop floor or in a procurement office, clear labeling and accurate specifications are non-negotiable. Buyers look for defined purity levels—often above 98%—because impurities can wreck downstream reactions, waste batches, or build toxic byproducts. Every shipment comes tagged with CAS numbers, batch data, and hazard warnings identified through years of global harmonization. End-users scan for trace metals and moisture levels since even small amounts throw off sensitive catalytic or pharmaceutical processes. This traceability lets outfits in different sectors—be it agrochemical manufacturing or specialty dye production—commit to compliance, especially as national and international regulations grow stricter.
Preparation Method
Chemistry students cutting their teeth on aromatic substitution soon learn about the preparation of 2,4-dichloronitrobenzene. It’s produced by nitrating 1,3-dichlorobenzene, a process that relies on careful control of temperature and addition rates in the presence of concentrated acids. Each stage of refinement takes experienced operators who know the hazards of nitrating agents and waste acid handling. Decades of factory experience point toward recycling acids and minimizing effluent as cost-saving necessities more than just environmental checkboxes. Frequent inspection of reactors and evolving automation techniques have helped reduce the risk of runaway reactions, cutting the number of plant accidents and helping ensure long-term operability.
Chemical Reactions & Modifications
Hardworking chemists spent years mapping how 2,4-dichloronitrobenzene behaves in the lab and commercial reactors. The nitro group offers a handle for reduction to 2,4-dichloroaniline, a precursor to a broad portfolio of products, from dyes to herbicides like 2,4-D. Chlorines on the aromatic ring unlock other substitutions useful in specialty materials. In my own lab work, we learned to respect selective reduction—the difference between a clean product and costly rework comes down to steady control of pressure, temperature, and catalyst. Understanding these subtle mechanics means teams can minimize waste and wring more value from every batch, a lesson that sticks with any chemist aiming to keep production both efficient and responsible.
Synonyms & Product Names
Anyone digging through chemical catalogs or regulatory filings will find a small mountain of alternate names for this compound. Most call it 2,4-DCNB, with synonyms popping up in product orders, research literature, and customs paperwork. CAS numbers help cut through confusion, but field staff and old-school buyers often rely on shorthand—quirks from decades of industrial tradition. Keeping terminology straight isn’t just academic; it’s about safety and logistics. Getting the wrong product because of an ambiguous name can ruin an entire week’s production, or worse, introduce hazards nobody expected. Common sense in communication—double-checking, using unmistakable names—turns out to be a key pillar in smooth operations.
Safety & Operational Standards
Working with chlorinated nitrobenzenes brings memories of training seminars where hard truths get shared: improper handling leads to exposure risks, fires, and regulatory headaches. Factories keep personal protective equipment and air monitoring as daily routines, not suggestions. Plant layout and emergency shutoff systems look boring on paper until they prevent a real crisis. Decades of incident records underscore the necessity of training and maintenance drills. That culture of preparedness—reinforced through audits, third-party checks, and internal reporting—has saved lives and property time and again. On any site dealing with 2,4-dichloronitrobenzene, experience with good housekeeping and adherence to safety data sheets keeps the workforce healthy and reputations intact.
Application Area
2,4-dichloronitrobenzene holds a central role in several industries, especially as an intermediate for synthesizing herbicides and dyes. In agrochemicals, it forms the launchpad for making selective weedkillers. For dyestuff producers, this compound’s specific arrangement of nitro and chlorine atoms is ideal for constructing stable, vibrant pigments. Pharmaceutical labs occasionally turn to it during the pursuit of specialty building blocks, especially where other solutions fall short. In each case, the ability to manufacture or buy high-purity material directly impacts product quality and market competitiveness. Any hiccup in quality or supply can ripple through to farmers, textile producers, and even the pharmaceutical supply system, raising the stakes for everyone involved.
Research & Development
Spending time in research labs, I’ve seen the constant pressure to make 2,4-dichloronitrobenzene safer and greener. Academic teams and corporate R&D units have pushed greener synthesis routes, low-waste separations, and more benign alternatives. Tech transfer offices look for scalable improvements, such as heterogeneous catalysis or solvent recycling, hoping to meet increasing regulatory scrutiny and environmental stewardship goals. Research also keeps open the door to new uses—perhaps fluorinated analogs or advanced materials—driven by fresh thinking and university-industry partnerships. The field remains lively, with the fundamental chemistry still revealing new wrinkles even a century after being first mapped out.
Toxicity Research
No one who’s worked closely with 2,4-dichloronitrobenzene shrugs at toxicity data. Repeated studies, both in-house and independent, have shown acute and chronic risks from overexposure. Inhalation irritates the respiratory tract, and skin absorption can bring systemic effects. Chronic exposure questions linger, making medical surveillance and strict engineering controls non-negotiable. Looking back at modernization trends, safer handling protocols and automation grew from deeper understanding of real-world health risks. Long-term, sound toxicity research informed both facility upgrades and tighter worker protection rules, ultimately reducing incidents and keeping industry viable against evolving legal and social expectations.
Future Prospects
Looking ahead, the story of 2,4-dichloronitrobenzene reads a lot like a playbook for responsible chemical production. Market demand stays tied to growth in agriculture and specialty chemicals. Environmental compliance, waste minimization, and sustainable synthesis have become as important as yield and cost. Upstream, investment into continuous-flow reactors and better waste acid handling keeps plants competitive and neighborhoods safer. Hopefully, ongoing advancements in catalysis and green chemistry will cut down both direct exposure risks and the environmental footprint. As regulations tighten worldwide, producers who invest in safer operations, transparent reporting, and better toxicity management stand to gain market confidence—and maintain their business for the long haul.
What is 2,4-Dichloronitrobenzene used for?
Beyond the Chemistry: Real-World Uses of 2,4-Dichloronitrobenzene
2,4-Dichloronitrobenzene can seem distant, like something tucked away in laboratory glassware. This chemical touches a surprising number of real-world products, even if most people never hear its name. Its main job comes from acting as a building block, especially in the manufacturing of dyes, pigments, pharmaceuticals, and pesticides. The chain reaction starts with a simple molecule, setting off a whole process behind the scenes that affects many industries and, in turn, our lives.
The textile world relies heavily on dyes made with this compound. Synthetic dyes require special molecules to bring out bright, long-lasting colors in fabrics. As someone who often shops for clothes, the variety you see on store racks wouldn’t exist without chemical intermediates like this one supporting dye production. Factories use 2,4-Dichloronitrobenzene to create vivid and colorfast shades in everything from denim jeans to printed t-shirts.
Supporting Pharmaceutical Progress
Its role in the world of medicine might surprise some people. Drug manufacturers depend on it to create active ingredients, particularly for fever and pain relief. Before reaching pharmacy shelves, these medications pass through many complex steps involving this chemical and others. Rigorous quality checks and strict regulations try to ensure that what starts in an industrial setting ends up safe for human use. Much of this process happens out of sight but has a direct impact every flu season.
As I’ve watched generic medicine offerings grow in local drugstores over the years, it’s clear that manufacturing advances help make treatments affordable and accessible. Developing key intermediates such as 2,4-Dichloronitrobenzene has pushed this growth, giving pharmaceutical companies more ways to meet demand while still respecting strict safety guidelines.
The Agriculture Connection
Crops face more threats than ever before, and modern agriculture leans on chemistry to keep up. Many herbicides and pesticides start with 2,4-Dichloronitrobenzene. It provides the backbone for molecules used to target weeds and pests without harming food plants. Farmers depend on these products to protect yields and keep food on tables across the globe. My own backyard garden experiments taught me that effective pest control often decides between a bumper crop and frustration.
Traditional practices like crop rotation still matter, but large-scale farms use science-driven tools to stay efficient. Even then, there’s a growing push toward sustainability, and regulators keep a close watch on the safety of these chemical aids. The process remains challenging, balancing productivity with environmental and health concerns.
Handling Risk and Seeking Improvements
Production and use of 2,4-Dichloronitrobenzene carry significant risks. Handling it exposes workers to hazards—skin contact, inhalation, and contamination can all have serious consequences. Strict safety regulations exist for a reason, guiding proper storage, use, and disposal. As the chemical industry makes progress, there’s increasing attention on minimizing worker exposure and reducing waste.
Innovation continues to drive change. Green chemistry approaches seek processes that cut down on hazardous byproducts and energy use. More investment in safety training and state-of-the-art equipment means that the risks seen in the early days of industrial chemistry can be reduced. This focus not only protects workers and communities but moves industries closer to truly sustainable practices.
2,4-Dichloronitrobenzene’s presence sits in the background, supporting diverse industries yet calling for ongoing responsibility. Understanding its uses reminds everyone that chemistry’s reach goes well beyond laboratory walls—impacting products and lives in often unseen ways.
What are the safety precautions for handling 2,4-Dichloronitrobenzene?
Understanding the Risks
2,4-Dichloronitrobenzene often appears in labs and factories focused on chemicals, dyes, and pharmaceutical work. The deep yellow crystals might not look threatening, but even a small amount can cause serious health effects. Skin contact often leads to rashes or burns, and breathing in the dust isn’t something to take lightly. I’ve seen colleagues develop headaches and breathing trouble from lapses in protocol. The chemical doesn’t play well with most protective gloves, either. Knowing how to handle it changes not just chores in the lab, but also your safety over months or even years.
Personal Protective Equipment: Non-Negotiable
Working with 2,4-Dichloronitrobenzene means wearing chemical-resistant gloves—nitrile or butyl rubber gloves tend to last longest under exposure. Goggles with side shields prove essential, since any dust in your eyes can cause lasting issues. Full-length lab coats might not look cool, but they save you from accidental splashes no paper towel could contain. Respirators become important in confined spaces or during any cleaning jobs. I always double-check the filters are fresh. The smell might seem faint, but it signals particles in the air.
Ventilation and Storage
Nothing beats a good fume hood. You work, and the fans pull away dust and vapors before breathing becomes a risk. Regular rooms build up the chemical over time, even if you don’t smell anything. 2,4-Dichloronitrobenzene doesn’t only bring risk during active use; old rags, wipes, and residues in corners continue to off-gas. That’s why sealed, labeled containers, preferably away from direct sunlight and heat, keep everyone safer. Fires involving this stuff get out of control fast, so keeping it away from flammables turns out to be smart, not just a suggestion.
Spills, Decontamination, and Training
Spills demand fast, calm action. Granular absorbent works well for minor messes. Shoveling up unsafe powder with bare hands or using a random shop broom only spreads it around—and a mop never gets rid of all residues. Moistening down powder before cleanup cuts dust spreading, lowering what you might breathe in. The right answer means following clear instructions and not ignoring training refreshers, even if they seem repetitive. I know experienced techs still run drills, and real emergencies feel much less overwhelming after practice.
The Bigger Picture: Responsibility To Others
Nobody handles chemicals alone. Each lab mate brings their own habits, and one mistake ripples out fast. I’ve seen how labeling shortcuts, ignoring “keep out” signs, or poor waste management make accidents worse. Those who take five seconds to check labels, close containers, or reroute foot traffic during transfer reduce risk for everyone. Sharing accurate safety data sheets and up-to-date emergency procedures saves time and lives. Medical staff in any facility also need quick access to these details, since early response to exposure or inhalation can make all the difference.
Making Progress: From Policy To Practice
Organizational commitment means investing in regular training, providing well-maintained gear, and never pressuring anyone to rush. I’ve worked in places where safety corners were cut to save time; the cost always comes later through illness, injury, or lost productivity. Honest reporting of near-misses shapes better prevention plans. Open communication about incidents leads to smarter, safer routines.
Building A Culture Of Safety
Staying vigilant with 2,4-Dichloronitrobenzene isn’t just about checking off a list. It comes down to staying present in your work, respecting both the risks and your teammates. Every layer of care—personal gear, right storage, proper cleanup—adds a barrier between you and harm. Safety starts as an individual habit and turns into a group standard, and that’s how science and industry keep moving forward without unnecessary setbacks or scars.
What is the chemical structure and formula of 2,4-Dichloronitrobenzene?
Looking at the Molecule Up Close
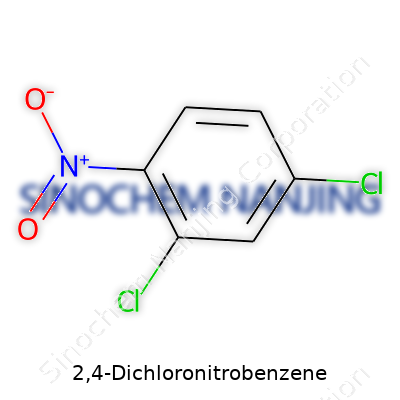
In any lab, the small brown bottle labeled 2,4-Dichloronitrobenzene stands out for a reason—a lot happens in its six carbon ring. Structurally, the chemical carries a benzene ring as its backbone. Attached to this ring, you’ll find two chlorine atoms, one sitting on the second carbon and another on the fourth. Add in a nitro group (NO2) on the first carbon, and you get the whole package. Its formula comes down to C6H3Cl2NO2. These placements might sound simple on paper, but they matter a lot for the reactivity and safety profile of this substance.
Real-World Uses and Everyday Concerns
Plenty of chemists start their journey with 2,4-Dichloronitrobenzene in a synthetic pathway, especially for dyes, rubber chemicals, or pharmaceuticals. All those little modifications—adding a chlorine here, a nitro group there—end up changing how entire supply chains run. The chlorines bring stability but lower the activating potential of the ring, making precise reactions possible without wild side-products. That’s a big reason companies stick to this compound for specialty chemical building blocks.
People sometimes overlook how tightly regulations watch compounds like this. The nitro group transforms the molecule into a potential hazard with respect to both toxicology and environmental persistence. Chlorinated nitrobenzenes don’t break down easily in nature, so they can stick around in soil or water. That makes careful use, disposal, and handling essential—not just a suggestion in a safety sheet. I’ve handled it in a double-gloved setup with a fume hood, thinking not just of the lab but what could end up downstream if I cut corners. The history is full of cases where improper disposal built up over time, leading to real problems in local ecosystems and even affecting nearby drinking water.
Health Perspectives and the Science of Risk
Personal protective equipment isn’t a formality with 2,4-Dichloronitrobenzene. Its molecular setup can allow for slow but steady absorption through the skin or lungs. That puts lab workers on alert for headaches, dizziness, or even more serious neurological symptoms. One sobering experience in a university lab taught me the value of regular air monitoring and quick action when a spill happens. Permanent substitution may not always feel realistic in research, but engineering controls and education bring the risk down to a tolerable level. The bottom line—if the molecule’s structure means it’s reactive or toxic, taking shortcuts isn’t worth it.
Pushing for Safer and Greener Chemistry
Green chemistry tells us to always look for ways to streamline syntheses and reduce waste. Some researchers have switched to less persistent alternatives or figured out better transformations that swap out nitrobenzenes for milder intermediates. Closed-loop processes that recycle spent chemicals or byproducts also help. Working with a clear understanding of each atom in a structure like 2,4-Dichloronitrobenzene, chemists now put more focus on minimizing exposure and building in environmental safety right at the drawing board.
Every line and angle in the chemical structure ties directly into lab decisions that shape safety, industry, and the sustainability of future products. The lesson carries over—even years after that first gloved experiment.
How should 2,4-Dichloronitrobenzene be stored?
Paying Attention to Risks
I’ve watched enough people treat industrial chemicals like office supplies to see how fast a minor mistake grows into a crisis. 2,4-Dichloronitrobenzene ranks alongside other tough organic chemicals—not something you’d leave out next to the coffee machine. Due to its toxic and potentially combustible nature, storage calls for precision, commitment, and a lot of attention to detail.
Setting Up the Right Storage Spot
Long before a drum gets delivered, workers need a clean, cool, ventilated, and dry dedicated area that keeps moisture and heat levels in check. Concrete floors with secondary containment save the day, not just for environmental reasons but for personal safety. Nobody wants their workspace turning into a hazardous slip-and-slide. To really protect people and the product, I always advocate for separate rooms or lockers for flammable substances, away from oxidizers, food, or general supplies. This one step prevents countless headaches, both regulatory and physical.
Choosing the Right Containers
Steel drums or HDPE containers—original, sturdy, sealed—beat makeshift bottles, every time. Cracked gaskets, battered lids, or reused containers all invite trouble in the form of leaks and contamination. Every label on these containers deserves a big, bold font: chemical name, warning information, date of arrival. Labels not only help in day-to-day recognition but reduce risks during emergencies when seconds count.
Protecting Human Health
Walking into a warehouse where 2,4-Dichloronitrobenzene sits for months means facing strong vapors. Good air movement and exhaust systems keep concentrations low. Workers equipped with proper gloves, goggles, and long sleeves stand a much better chance of staying out of the doctor’s office. Strictly no open flames, no smoking, and minimal static—all these habits should be hardwired into every employee who gets anywhere near chemical stockpiles.
What Happens in a Spill?
Working in chemical plants taught me that things don’t always go right. Every facility should lay out a clear spill protocol, starting with containment (using sand or inert absorbents, not water) and followed by proper cleanup practices. All waste—leftover chemicals, used absorbents—heads right into labeled hazardous disposal containers. Local authorities and hazardous waste contractors carry out the haul, not the regular trash service.
A Place for Regular Checks
Containers degrade and seals crack over time. Inspections every few weeks—checking for bulging drums, leakage, or corrosion—build a safer work environment. Records aren’t just paperwork; they give early warnings before anyone gets hurt or a regulator comes knocking on the door. Training goes hand-in-hand with inspections, so workers know what to look for and what to avoid.
Finding the Safer Route
Storing 2,4-Dichloronitrobenzene safely protects workers, property, and the environment. Following proper engineering controls, handling habits, and clear communication keeps workplace stories about triumph—not disaster. Investing in good practice never wastes money; it just keeps everyone breathing easier.
What are the health hazards and first aid measures for 2,4-Dichloronitrobenzene exposure?
Getting to Know the Risks
2,4-Dichloronitrobenzene shows up in some chemical manufacturing, mostly tied to dyes and pesticides. It’s not a household name, but people working in production facilities or handling waste could run across it. Touching or breathing this chemical can cause more problems than a simple skin rash. Inhaling dust or fumes may trigger coughing, sore throat, headaches, or more serious lung irritation. If the skin soaks up a bit, red patches or even blistering sometimes follow, especially after long or repeated contact.
Eyes don’t fare well, either. Getting splashed or being near the powder creates a risk for burning, watering, and even temporary vision problems. Swallowing any amount usually causes nausea, vomiting, dizziness—sometimes the heart races or the body feels confused and weak. It’s not only about short-term trouble. Taking in nitrobenzene compounds over time brings other concerns. These chemicals can damage the liver and kidneys, leading to fatigue or, over years, chronic illness. Some have linked long-term, heavy workplace exposure to blood issues, including methemoglobinemia, where blood struggles to carry oxygen. The signs can be blue-tinted lips, shortness of breath, or, in rare situations, collapse.
What Experience Tells Us about Preventing Trouble
Sitting through safety briefings and reading MSDS fact sheets taught me that personal habits make a difference. Wearing gloves, goggles, and a mask gets old fast, but skipping them sometimes ends with regret. People who ignore ventilation or don’t check equipment for leaks tend to run into chemical headaches sooner. Good hand washing and keeping hands away from the face helps reduce accidental exposure—old advice, but it works.
First Aid That Makes a Real Difference
Accidents will happen, and a quick reaction changes the outcome. If someone gets the chemical on their skin, removing contaminated clothing and rinsing under cool, running water for at least 15 minutes often limits further harm. It sounds simple, but some skip this step, thinking “just a little can’t hurt.” Never use hot water—it opens pores and helps the chemical get in faster. For splashes in the eyes, hold lids apart and flush with water. Most guides say to keep going for at least 15 minutes, and that has saved vision in more than one near miss I’ve seen firsthand.
Inhaling high amounts calls for moving to fresh air, preferably outdoors, right away. Watch for breathing trouble—if it’s hard to talk or someone acts confused, call emergency services. If there’s chest tightness, coughing, or signs of oxygen trouble (like blue lips), don’t wait. Fast medical help counts. Swallowing this chemical does not just irritate the gut. It poses a risk to organs. Experts say skip the old advice of making someone throw up or offering anything except water unless a doctor says so. Medical professionals have tools for poisonings—a trip to the ER wins out over home remedies every time. Bring a sample of the chemical’s label, if possible, so doctors know what they're up against.
Taking Responsibility: Where Solutions Start
Treating chemicals with respect starts with culture, not just warning labels. Creating a workplace where speaking up about leaks or unsafe conditions feels normal protects everyone. Regular updates on training help staff recall what steps matter most—and demonstrate management cares about their health, not just rules on a poster. Whether working in an industrial setting or responding to emergencies, staying a step ahead makes the real difference between an injury and a close call.
| Names | |
| Preferred IUPAC name | 1,3-dichloro-2-nitrobenzene |
| Other names |
1-Nitro-2,4-dichlorobenzene 2,4-Dichloronitrobenzene 2,4-DCNB NSC 8611 |
| Pronunciation | /tuː, fɔːr daɪˌklɔːroʊˌnaɪtroʊˈbɛnziːn/ |
| Identifiers | |
| CAS Number | 611-06-3 |
| Beilstein Reference | 1209230 |
| ChEBI | CHEBI:34760 |
| ChEMBL | CHEMBL16563 |
| ChemSpider | 12767 |
| DrugBank | DB07923 |
| ECHA InfoCard | 03d198d4-8e18-4844-9e38-9be4a78931b1 |
| EC Number | 202-711-9 |
| Gmelin Reference | 89070 |
| KEGG | C14122 |
| MeSH | D004073 |
| PubChem CID | 7915 |
| RTECS number | CZ9625000 |
| UNII | R8YCK5QY1V |
| UN number | UN1598 |
| Properties | |
| Chemical formula | C6H3Cl2NO2 |
| Molar mass | 202.00 g/mol |
| Appearance | Light yellow crystalline solid |
| Odor | Odorless |
| Density | 1.50 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 2.98 |
| Vapor pressure | 0.00022 mmHg (25 °C) |
| Acidity (pKa) | 12.41 |
| Basicity (pKb) | 8.46 |
| Magnetic susceptibility (χ) | -75.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.609 |
| Viscosity | 1.275 mPa·s (25°C) |
| Dipole moment | 2.81 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 229.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -44.9 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1071.0 kJ/mol |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and eye irritation, may cause respiratory irritation, toxic to aquatic life. |
| GHS labelling | GHS02, GHS06, GHS08, GHS09 |
| Pictograms | GHS06,GHS09 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335, H410 |
| Precautionary statements | P261, P264, P270, P273, P301+P312, P305+P351+P338, P308+P311, P312 |
| NFPA 704 (fire diamond) | 2,4,0,▲ |
| Flash point | 113 °C |
| Autoignition temperature | 600°C |
| Lethal dose or concentration | LD50 oral rat 576 mg/kg |
| LD50 (median dose) | LD50 (median dose): 890 mg/kg (oral, rat) |
| NIOSH | CN9625000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of 2,4-Dichloronitrobenzene: 2 mg/m³ |
| REL (Recommended) | 0.1 mg/m3 |
| IDLH (Immediate danger) | IDLH: 25 mg/m³ |
| Related compounds | |
| Related compounds |
1,3-Dichloro-2-nitrobenzene 2,6-Dichloronitrobenzene 2,4-Dichloroaniline 2,4-Dichlorophenol 2,4-Dichlorobenzyl alcohol |

