Reflecting on 2,4-Dichlorobenzoyl Chloride: History, Science, and the Road Ahead
Tracing the Path: Historical Development
Digging into the story of 2,4-Dichlorobenzoyl Chloride, you find it tied to a larger wave of synthetic chemical development that marked the twentieth century. Interest in chlorinated aromatic compounds picked up speed after the Second World War, when the chemical industry was hunting for more efficient ways to produce herbicides and specialty chemicals. The history of this substance intertwines with the development of agricultural chemistry. Companies learned to tweak benzoyl chloride by introducing chlorine atoms, chasing better performance and more reliable synthesis. Each advance relied on a mix of organic chemistry insights and gritty industrial innovation. The legacy of these old experiments lives on: many widely used chemicals owe their roots to choices made decades ago, both appreciated for progress and scrutinized for safety.
The Basics: What We’re Dealing With
You won’t scroll past 2,4-Dichlorobenzoyl Chloride on a hardware store shelf. It serves as a specialty intermediate, a building block used by professionals in tightly controlled settings. In everyday speech, people lump it into the large family of benzoyl chlorides, but chemists pin it down with its own structure: a benzene ring with two chlorines at the 2 and 4 positions, and a reactive acyl chloride group sticking off the side. Despite being comparatively obscure to the public, its presence underpins polymers, pharmaceuticals, and agrochemical products — all thanks to this molecular backbone. Focusing on such intermediates, rather than final products, shows the deeper plumbing behind modern material science.
Matter of Science: Physical and Chemical Truths
The stuff looks like a crystalline solid, pale to off-white, not the sort of thing one would mistake for a household cleaner. Its sharp scent hints at reactivity. Its melting point sits a little above room temperature, so indoor air conditioning probably won’t affect its form, but a modest rise in temperature can do the trick. Many chlorinated aromatics call for careful storage since they react with water and give off acid gases. 2,4-Dichlorobenzoyl Chloride does not break with tradition. It needs moisture kept away, and contact with bases or nucleophiles ends with release of hydrochloric acid. Chemists recognize its dual nature: stable during careful handling, but ready to leap into reactions with the right partners.
Inside the Lab: Technical Details and Labeling
Anyone working with this compound knows the routine: proper labeling and clear hazard signs. Regulatory standards in the EU, US, and Asia assign specific codes and pictograms, ensuring no confusion about corrosive properties or required precautions. Labels often highlight the chemical’s irritant and corrosive dangers right up front. In technical terms, the substance demands containers made of materials that won’t dissolve, ventilation to keep fumes away, and strict logbooks to track its movement. Real-world chemistry won’t tolerate shortcuts; misunderstanding the rules around such compounds leads directly to health and liability problems.
Building Blocks: Preparation and Synthesis
Producing this material relies on well-worn chemical strategies. One common approach involves chlorinating benzoyl chloride, with catalysts or light nudging the process forward. Industrial chemists select conditions that push for substitution at the right spots on the benzene ring. Sometimes older methods produced messy byproducts, but newer protocols have cut down on waste, partly responding to environmental scrutiny. The trick is maximizing yield without generating a storm of toxic leftovers, balancing efficiency with responsibility. Watching these synthesis choices change over years shows the field adapting—not just inventing, but also cleaning up after itself.
Transformations: Typical Reactions and Modifications
Organic chemistry thrives on group transformations, and 2,4-Dichlorobenzoyl Chloride fits right into this dance. The acyl chloride group snaps onto amines or alcohols, forming amides and esters — new compounds with targeted uses. The chlorine atoms can also influence further reactions, steering outcomes or boosting selectivity. These properties allow chemists to use this intermediate in making dyes, advanced pharmaceutical precursors, and other specialty molecules. While the textbook reactions look neat, real-life chemists deal with side products and operational headaches — and constantly hunt for ways to control every variable.
What Else It’s Called: Synonyms in Use
Chemical naming keeps both clarity and confusion alive. Besides 2,4-Dichlorobenzoyl Chloride, you’ll spot names like Benzoyl Chloride, 2,4-dichloro- and 2,4-DCBC, plus a scattering of registry numbers in databases. Each label points at the same molecular identity, but context matters: a patent application, a supplier’s inventory, or a research paper could each use a different name. For every synonym, there’s a chance of mix-ups or misunderstandings, especially among newcomers or across language barriers. Awareness of this naming diversity keeps mistakes at bay.
Straight Talk on Safety and Handling
Experience shapes respect for chemicals like this. Its chlorinated structure and acyl chloride functionality pack a punch — strong enough to cause burns and respiratory irritation on contact. Labs and factories implement rigorous controls: gloves, splash-proof goggles, fume hoods, and emergency protocols. Training and supervision matter as much as protective gear. People make mistakes or cut corners only once if they’re lucky. The ghost of older accidents haunts chemical safety manuals, pushing managers to insist on double-checking valves and emergency vents. As much as chemistry advances, the basics of safe handling stay constant.
Real-World Roles: Where It Gets Used
This chemical ends up as a junction point more than a destination. Its main use lies in crafting molecules for further industrial and pharmaceutical processing. In agrochemistry, it helps birth specific herbicides and pesticides; in pharmaceutical labs, it becomes parts of compounds seeking activity against bacteria or cancer. The fine chemicals industry values such intermediates precisely for their ability to unlock harder syntheses. What most people touch as drugs or consumer products often began their life in settings where compounds like this enabled some crucial step. The reach extends farther than many realize.
On the Frontier: Research and Innovation
Chemists and engineers still push boundaries with this compound, looking for greener synthesis methods, better yields, and safer reaction pathways. Academic labs chase new organometallic reactions or asymmetric modifications, while industrial researchers attack process bottlenecks from a cost and safety angle. Emerging green chemistry approaches seek to cut chlorinated waste or avoid problematic catalysts. Research also revisits older chemistry, asking if automation and real-time monitoring can trim risk or save time. Every tweak that trims a hazardous byproduct or tightens quality control brings improvements that ripple through entire supply chains.
Toxicity and Human Health Studies
Scrutiny of chlorinated aromatics goes back decades, driven by both workplace exposures and environmental concern. Studies expose laboratory animals and cell cultures to gauge acute and chronic toxicity; they help set workplace exposure limits and personal protective equipment standards. The main message? Direct contact or inhalation poses meaningful risks. Regulatory agencies review new toxicity data as it emerges, sometimes updating safety standards if new dangers come to light. Long-term exposure studies often focus on cancer risk and organ effects, and while this compound is handled mostly by experts, the knock-on effects of spills or improper disposal have kept health agencies watchful.
Looking Ahead: Future Prospects
New regulations and public expectations keep pressure high on chemical producers. Industry isn’t just tasked with steady output; it faces a future where tightening environmental rules and trade restrictions challenge old ways of working. At the same time, demand builds for purer, safer intermediates—especially those that fit with greener processing. Supply chain resilience, digital inventory tracking, and modular reactor designs are shaping the next chapter. Research keeps finding ways to recycle reagents, recover byproducts, and minimize hazardous waste. The path forward isn’t just about making more; it’s about making responsibly, with eyes open to long-term effects. Each turning point in the story of 2,4-Dichlorobenzoyl Chloride reminds us that science, safety, and stewardship walk hand in hand.
What is 2,4-Dichlorobenzoyl Chloride used for?
A Hidden Backbone in Chemical Synthesis
2,4-Dichlorobenzoyl chloride rarely gets the limelight outside chemistry circles. I spent several years working in the chemical industry and remember how many specialty chemicals trace back to simple building blocks like this one. It's no front-facing product, but its fingerprints show up all over advanced manufacturing and pharmaceutical production. In my lab days, a bottle of this compound sat among the core reagents, handled with caution but relied on for serious results.
Applications in Pharmaceuticals
Pharmaceutical development leans on key intermediates for creating complex drugs. 2,4-Dichlorobenzoyl chloride carries a reactive group that attaches with ease to nitrogen or oxygen atoms in target molecules. Chemists use this to build up antibiotics, antifungals, and several anti-inflammatory medications. For example, it's essential for linking specific molecular fragments in drug candidates. Missing it, several medication pipelines could grind to a halt. Drugs on the market or in trials often hide a whole chain of such chemical steps, sometimes designed decades earlier.
Impact on Agrochemical Production
Beyond pharma, the world of crop science uses 2,4-dichlorobenzoyl chloride in the creation of herbicides and fungicides. Farms rely on these compounds to keep weeds and crop diseases under control, boosting yields and slowing food price hikes. I visited an agrochemical plant once and saw firsthand the giant reactors where such reactions take place. Each batch relies on this compound for a specific bond or protective group, setting the stage for the final active molecule. This underlines the real-world importance for food security and the economics of agriculture.
Role in Dye and Polymer Synthesis
Colorants and resins come to life in labs through painstaking chemistry. This compound appears in dye manufacturing, giving textile and printing companies reliable colors with strong durability. On the polymer side, chemists use it to introduce specialized properties, such as resistance to heat or specific solvents. Many high-performance plastics in electronics or automotive parts get some of their key traits from reactions started off with 2,4-dichlorobenzoyl chloride.
Health and Environmental Considerations
Handling comes with its own set of worries. My own experience taught me to respect this chemical—protective gloves and ventilation become essentials, not afterthoughts. It can cause severe skin burns and respiratory issues, and waste management must follow clear, regulated protocols. The EPA and OSHA have strict rules for storage and disposal for good reason. In the wrong hands, a spill or exposure risks causing real harm to workers and the environment.
Pushing for Safer Solutions
Chemical engineers constantly look for safer alternatives that deliver similar results. In some processes, green chemistry offers swaps, but for others, no adequate substitute exists yet. Ongoing research matters here. Encouraging safer production techniques and recycling unused chemicals could shrink potential hazards. Firms that invest in employee training and regular audits see fewer mishaps and protect their reputations. As a chemist, I know progress often comes in small steps, adjusting formulas, updating safety protocols, and listening to the concerns of those actually handling the substances.
Staying Smart About Hidden Ingredients
So many modern conveniences depend on invisible building blocks like 2,4-dichlorobenzoyl chloride. Keeping a focus on safety, supporting innovation for safer alternatives, and holding firms accountable for clean practices create benefits that reach far beyond the lab bench. These chemicals touch lives even when we barely notice them. Attention to detail—on the shop floor, at the company level, and from regulators—makes sure the benefits outweigh the risks.
What is the molecular formula of 2,4-Dichlorobenzoyl Chloride?
Pulling Apart a Name to Find Its Meaning
2,4-Dichlorobenzoyl chloride sounds like something pulled from dense chemistry textbooks, but the name tells a clear story if you take a moment to break it down. It builds off a benzene ring, which is a hexagon made entirely of carbon, with alternating double bonds that let it stay remarkably stable. Add two chlorine atoms in the 2 and 4 spots on that ring—counting one spot as the “1” and moving around. Toss in a carbonyl chloride group in the “1” position, and you’ve got this chemical in full.
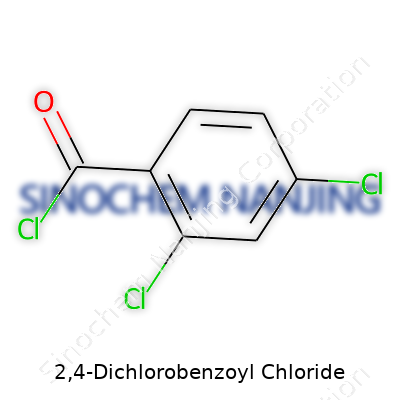
This boils down to a formula: C7H3Cl3O. That’s seven carbons, three hydrogens, three chlorines, and a single oxygen atom. Each piece in the formula serves a purpose: chlorines amp up the reactivity, the carbonyl chloride does the heavy lifting during chemical transformations, and the aromatic ring backbone keeps things sturdy and predictable.
Why Chemists Pay Close Attention to the Formula
I’ve worked in a laboratory, handling compounds not too different from this one. Clarity about the molecular formula isn’t just about ticking a box on a data sheet. Knowing exactly how many atoms make up a molecule guides safe handling, tells us which gloves to put on, and lets us predict how that molecule might behave if splashed on a countertop or mixed with something else. If a molecule has extra chlorine, like 2,4-dichlorobenzoyl chloride, it gets a lot more reactive. So, I always triple-checked labels and double-checked formulas before pulling out any reagents for research projects.
Impact on Industry and Research
This compound often finds its way into the manufacturing of pharmaceuticals, crop protection chemicals, and dyes. Large-scale industries look for consistent molecular formulas because a single missing atom can turn something useful into a potential hazard. Chlorinated compounds like this one fall under strict regulation since they sometimes break down into persistent pollutants. My experience has shown that precision on a molecular level translates directly into environmental fitness and worker safety in real-world settings.
Common Issues and Where Solutions Start
Any chemist can tell you that errors pop up often during manufacturing and analysis. Mislabeling happens, contaminated batches show up, and the wrong formula can mean ruined experiments and wasted money. Routine spectroscopic analysis like NMR and mass spectrometry, which I leaned on heavily in the lab, confirms what’s actually in that bottle. Fact-based protocols, transparent ingredient sourcing, and public reporting all help dodge supply chain mishaps.
Chemical education still needs close attention to detail, not just rote memorization. In my time helping students or junior colleagues, I noticed that teaching them to quickly sketch structural formulas by hand helped prevent mistakes down the road. If someone isn’t familiar with the way each atom fits together on paper, it’s easy to lose track in a sea of numbers and letters.
Smart Steps for Moving Forward
Supporting open, detailed chemical databases online is the most effective way to help researchers double-check themselves and each other. Encouraging companies to share plain-language safety info, alongside the dense technical data, brings everyone up to speed. At the bench and in the plant, knowing the formula for 2,4-dichlorobenzoyl chloride isn't just trivia—it keeps people safe and scientific projects on track.
What are the safety precautions when handling 2,4-Dichlorobenzoyl Chloride?
Recognizing the Risks
In the world of chemical handling, 2,4-Dichlorobenzoyl Chloride stands out as one that can quickly cause trouble in a lab or factory if respect goes out the window. This chemical packs a punch with its reactive nature; it doesn’t hesitate to bite your lungs or skin if you give it a chance. The fumes alone can turn a careless afternoon into a trip to the emergency room. Anyone who’s spent time with reactive acyl chlorides knows the itchy throat, the chest tightness, even from a whiff. It’s not bravado to treat it with caution; it’s common sense.
Dressing for the Job
Protective clothing is not an inconvenience; it’s the first line of defense. Goggles that form a tight seal, a face shield for splashes, sturdy gloves made for chemical resistance—these are your daily armor. Lab coats and long pants do more than just “look professional.” I’ve watched careless folks in short sleeves scramble after unexpected spills. It’s tempting to cut corners to save time, but chemical burns and allergic skin reactions don’t work on your schedule. Nitrile or neoprene gloves hold up far better than cheap latex.
Opening Containers: Not a Blind Task
Popping the top off a bottle of 2,4-Dichlorobenzoyl Chloride outside a fume hood courts disaster. The irritating vapors spread fast, especially if the room’s still air. Every time I run a reaction involving this stuff, that sash stays as low as possible; no exceptions. Fume hoods are not just props—they actively suck away the danger. The right ventilation system helps everyone in the room, not just the person holding the pipette.
Avoiding Water: No Joke
This is one spot where chemistry class turns real. Water or strong humidity near this chemical spells trouble. 2,4-Dichlorobenzoyl Chloride hisses with acid when it reacts with water. Hydrochloric acid vapor does harsh things to lungs and eyes. Keep bottles tightly closed, use only dry glassware, and store far from sinks or open containers of liquid. Desiccators work well for longer-term storage.
Spill Response—No Room for Hesitation
Accidents happen, even if you do everything right. Speed and calm decide the outcome. Absorb spills with suitable materials—vermiculite or sand, not paper towels that might just get shredded by acid. Clean every trace, ventilate, and warn others nearby. I’ve learned the hard way that ignoring a small spill often leads to bigger headaches later, including damaged surfaces or persistent odors that stick around for weeks.
Disposal: Don’t Wing It
Pouring leftovers down the drain is a mistake with real consequences for both people and the environment. Waste streams need proper segregation; this chemical goes with organics and corrosives, not with bland solvents. Label waste containers, keep them shut, and contact licensed disposal services. Treating chemical disposal as an afterthought risks fines, shutdowns, and dangerous reactions in the trash or plumbing.
Training and Vigilance
Shortcuts are easy to spot once you know what disaster feels like. Safety protocols only matter if people actually use them, so regular practice drills and up-to-date training become routine. The best labs and factories run through emergency plans just like fire drills—familiarity stops panic. Trust between coworkers grows strongest when everyone watches each other’s back.
Practical Takeaways
Everything comes down to respect for chemical risk, not fear. The right equipment, habits, and teamwork keep 2,4-Dichlorobenzoyl Chloride from turning a workday into a story nobody wants. Every bottle, every label, every step in cleanup marks a decision: play it safe or pay the price. The choice, every day, is in your gloves.
How should 2,4-Dichlorobenzoyl Chloride be stored?
Knowing What You’re Dealing With
2,4-Dichlorobenzoyl Chloride carries more bite than its technical name lets on. This material, often found in labs and chemical plants, brings both usefulness and risk. Unpacking how it fits into day-to-day practice means getting real about the risks sitting inside every drum or bottle.
Separation and Containment
This chemical does not play well with moisture. Even a tiny amount of water can cause it to break down, throw off irritating gases, and start a reaction you’d rather avoid. On humid days, you can almost hear the worry over every improperly sealed cap. Proper storage means setting up a spot where humidity rarely creeps in—think cool, dry rooms with good air flow. I’ve seen more than one expensive supply written off because someone thought “close enough” meant tight storage. It doesn’t.
You want to stash this material in high-quality glass or plastic containers that won’t react with sharp chemicals. Fact: metal can corrode, and weak-sealing caps turn every shelf into a slow, risky experiment. I’ve learned to favor containers that provide a good, clean seal and show if they’ve been messed with. This is not paranoia. Checking things twice saves hassle and money.
Distance from Heat and Ignition Sources
Heat and reactive compounds are the enemies here. Even a sunbeam across a warehouse floor or a radiator humming nearby can push things in the wrong direction. At one plant, a forgotten HVAC duct cooked more than it cooled. Chemicals stacked too close got warm, and that heat started ruining perfectly good raw materials.
Nobody wants a warehouse fire. Rules on no smoking, no open flames, and keeping flammable chemicals separate from oxidizers seem obvious until you catch someone cutting corners just to save a few steps. You don’t ignore these details: it’s not just regulation, but a matter of everybody getting home in one piece.
Labeling and Access Control
Any container carrying 2,4-Dichlorobenzoyl Chloride should leave no doubt about what’s inside. Even after years in a lab, I’ve seen seasoned staff reach for the wrong bottle when tired or rushed. The right label, written clearly with hazard information, makes hesitation automatic and mistakes less likely.
On top of good labeling, locking cabinets and limited access work better than trust alone. Training makes the difference here. Only folks who know what they’re doing have business pulling these chemicals off a shelf. I still remember a rookie mistake during my early days—one mix-up caused hours of cleanup and a hard lesson for everyone.
Thinking Ahead: Emergency Response
Storage gets real meaning on the day something goes wrong: leaks, spills, or accidental exposure. A clear spill kit nearby, emergency eyewash stations, and posted instructions for what to do pay off every time. Teams who walk through drills and carry up-to-date contact numbers don’t freeze up when things go sideways.
Building a Culture of Safety
Keeping 2,4-Dichlorobenzoyl Chloride secure isn’t a box to check; it asks for discipline and common sense backed by facts. I rely on peer-reviewed guidelines, national safety data sheets, and personal experience—and every layer you add helps. When people see safety in action, they learn what it looks like and pass it forward.
Responsibility means more than following rules; it’s a daily attitude. A workplace with safe storage pays attention, holds each other up, and doesn’t let small errors stack up. That’s where real protection lives.
What are the physical properties of 2,4-Dichlorobenzoyl Chloride?
Getting to Know 2,4-Dichlorobenzoyl Chloride
Anyone who has spent time in a chemistry lab knows the critical role raw materials play in giving us the worlds of pharmaceuticals, polymers, and agrochemicals. 2,4-Dichlorobenzoyl chloride stands in the thick of such interactions. It shows up as a pale to off-white crystalline solid, sometimes coming across as a faint yellow powder depending on purity. People in chemical plants get to know its strong, biting odor—sharp enough to remind them of its reactive nature before they even uncap a bottle.
Melting and Boiling Points Matter Beyond the Lab
I remember melting point determination as one of the first hands-on lessons about the identity and stability of a compound. With 2,4-dichlorobenzoyl chloride, the melting point sits around 46-50 °C. Its boiling point lands near 277 °C but don’t let that broad number fool you; handling gets tricky even at much lower temperatures. Even a mild increase in heat can trigger decomposition, which means you need to keep things cool and steady.
Solubility and Reactivity
Drop this solid into water and you don't get gentle dissolution—you trigger a rapid reaction. Chlorides like these hydrolyze easily, releasing carbon dioxide, hydrochloric acid fumes, and making a mess if left unchecked. Most solvents in the lab, such as chloroform or dichloromethane, dissolve it much more comfortably. Back in my graduate days, anyone who tried to wash glassware with just a splash of tap water soon learned their mistake.
Density and Appearance—No Guesswork Allowed
This compound comes with a density of about 1.43 grams per cubic centimeter, putting it comfortably heavier than water. Easy measurement means you can handle and dose it with precision if you’re running a reaction at small scale. In industry, no one wants surprises from floating or sinking solids where machinery and storage are concerned. That trouble gets avoided thanks to well-documented physical records.
Experience in the Field
Using this substance in the lab impressed on me how quickly mismanagement leads to headaches. Thermal instability combined with reactivity demands airtight containers and cold storage. Open a bottle carelessly and fumes sting your eyes. The technical data what matters most is not just a list in a book; it extends to every step in handling, shipping, and labeling. Chemical burns and inhalation risks drive home the importance of personal protective equipment.
What the Numbers Mean for Safety and Innovation
Physical characteristics like melting point, density, and solubility seem dry on paper until you work up close. These numbers decide how the compound reacts to temperature swings, leaks, or accidental water contact. Adding it to a reaction mixture feels simple until you remember that runaway hydrolysis easily turns routine work hazardous. Factories track every property for a reason: mistakes cost time, money, and safety.
Building Better Practices
Good handling starts with knowing what you’re working with. For those in manufacturing, building tight standard operating procedures—such as inert atmosphere use or controlled refrigeration—protects both workers and products. Regular staff training helps everyone understand why the physical properties of 2,4-dichlorobenzoyl chloride matter in ways that no textbook can fully capture. Integrating real-world experience with technical knowledge makes the difference between risk and innovation in chemical processing.
| Names | |
| Preferred IUPAC name | 2,4-dichlorobenzoyl chloride |
| Other names |
2,4-DCBCl 2,4-Dichlorobenzoylchloride 2,4-Dichlorobenzoic acid chloride Benzoyl chloride, 2,4-dichloro- 2,4-Dichlorobenzoylchlorid |
| Pronunciation | /tuː, fɔːr daɪˈklɔːr.oʊˈbɛn.zɔɪl ˈklɔːraɪd/ |
| Identifiers | |
| CAS Number | [2234-16-4] |
| Beilstein Reference | 1206962 |
| ChEBI | CHEBI:84960 |
| ChEMBL | CHEMBL4260865 |
| ChemSpider | 86501 |
| DrugBank | DB08597 |
| ECHA InfoCard | 100.012.003 |
| EC Number | '211-969-1' |
| Gmelin Reference | 132165 |
| KEGG | C19216 |
| MeSH | D015242 |
| PubChem CID | 66010 |
| RTECS number | DG8925000 |
| UNII | HTM9E1Q2TX |
| UN number | UN2581 |
| Properties | |
| Chemical formula | C7H3Cl3O |
| Molar mass | 197.45 g/mol |
| Appearance | White to light yellow crystalline powder |
| Odor | Pungent |
| Density | 1.43 g/cm³ |
| Solubility in water | Solubility in water: Reacts |
| log P | 2.9 |
| Vapor pressure | 0.03 hPa (20 °C) |
| Acidity (pKa) | 1.05 |
| Magnetic susceptibility (χ) | -84 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.588 |
| Viscosity | 47 mPa·s (20 °C) |
| Dipole moment | 1.92 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 341.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -234.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -5284.7 kJ/mol |
| Hazards | |
| Main hazards | Harmful if swallowed, causes severe skin burns and eye damage, may cause respiratory irritation. |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | H302, H314, H335, H400 |
| Precautionary statements | P261, P264, P271, P280, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P310, P312, P332+P313, P337+P313, P362+P364 |
| NFPA 704 (fire diamond) | 3-2-1-W |
| Flash point | 80 °C (closed cup) |
| Autoignition temperature | 250°C |
| Lethal dose or concentration | LD50 oral rat 1,154 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50: 1650 mg/kg |
| NIOSH | DC2625000 |
| REL (Recommended) | 0.5 mg/m³ |
| IDLH (Immediate danger) | IDLH: 5 ppm |
| Related compounds | |
| Related compounds |
Benzoyl chloride 2,4-Dichlorobenzoic acid 2,4-Dichlorotoluene 2,4-Dichloroaniline 2,4-Dichlorophenol |

