2,4-Dichloroaniline: Exploring Its Place in Modern Chemistry and Industry
Historical Development
Taking a closer look at the story of 2,4-Dichloroaniline, anyone familiar with the rise of the chemical industry will spot its roots in the flood of new aromatic compounds that came out of European dye houses during the early twentieth century. Once chemists figured out how to juggle chlorine atoms onto the benzene ring and swap in amino groups, doors opened to an entire class of molecules with tailored properties. 2,4-Dichloroaniline stood out for its role as an intermediate in the manufacture of colors and protective agents for crops. Its influence spread, not because it was glamorous, but because it delivered reliable results in practical settings. Stories passed down from plant operators early in my career frequently highlighted how 2,4-Dichloroaniline’s ease of production and dependable reactivity formed the backbone of entire product lines. The chemical’s history intertwines with advances in public health and agriculture—what would a cotton field or railroad right-of-way have looked like without the herbicides built on this very foundation?
Product Overview
At its core, 2,4-Dichloroaniline belongs to a family of chlorinated aromatic amines. Despite its straightforward appearance, this molecule plays a leading role in the synthesis of vital agrochemicals, colorants, and even pharmaceutical ingredients. Over years spent in chemistry labs and production facilities, I have seen first-hand how 2,4-Dichloroaniline’s profile attracts both curiosity and careful handling: its versatility balances against caution, since its derivatives often walk a fine line between beneficial effect and environmental persistence. Producers rely on this substance for its predictable behavior, using it to stitch together more complex products without igniting the intricate dance of unwanted side-reactions. Manufacturers and formulators recognize that a little goes a long way, as few other compounds convert so efficiently into downstream herbicides and dyes.
Physical & Chemical Properties
A pale yellow to light brown solid at room temperature, 2,4-Dichloroaniline carries a distinct odor that quickly becomes familiar to anyone who has worked with it in a chemical plant. Its melting point tends to fall near 70-72°C, with a boiling point high enough to demand careful thermal control during processing. The solid’s low solubility in water means most waste concerns focus on proper containment rather than rapid dissolution. Having handled this compound, I know it resists casual breakdown by sunlight or oxygen—those halogen atoms clamp down on the ring, giving the molecule a stubborn longevity in soil and water. Chemists appreciate that the compound offers a reliable starting point for modifications, thanks to its pair of electron-withdrawing chlorines and its lone amino group holding ready for reaction.
Technical Specifications & Labeling
In any regulated facility, 2,4-Dichloroaniline must come with precise labeling to avoid mix-ups with similarly named or colored compounds. Whether in bulk sacks or glass jars, labels list purity, hazard information, handling instructions, and emergency measures for exposure. The telltale warning signs—marked flammable, toxic, and environmental hazard, according to local and international standards—leave no doubt that this is an industrial chemical, not a consumer good. Product consistency, monitored by regular analytical tests, matters to those downstream. Missteps in specification can produce ripple effects, from compromised crop yields to malfunctioning dye baths. Batch records verify that every step, from shipment to storage, gets logged and scrutinized.
Preparation Method
Manufacturers of 2,4-Dichloroaniline almost always start with chlorinated precursors, often 2,4-dichloronitrobenzene. Industrial operations use catalytic hydrogenation—passing hydrogen gas over the nitro compound in the presence of a specific catalyst, usually under controlled pressure and temperature—draining the yellow-brown slurry, filtering, then recrystallizing the product for purity. This transformation, which I have witnessed on both pilot and commercial scale, typifies how industry adapts academic reactions to robust systems that minimize waste and maximize yield. Safety measures at every stage—including explosion-proof equipment and air monitoring—keep mishaps at bay, an approach borne not of paranoia, but of lessons learned the hard way. Waste streams demand careful treatment; even small leaks or emissions invite regulatory attention and community concern.
Chemical Reactions & Modifications
Chemists love 2,4-Dichloroaniline because it serves as a launching pad for all sorts of molecular tweaks. The amine group lends itself to coupling reactions, from forming complex azo dyes to producing sulfonamides and pesticides. Herbicide manufacturers typically react it with chloroacetic acids or phosgene derivatives to yield powerful active ingredients that keep weeds in check. In the dye industry, sulfonation and diazotization of 2,4-Dichloroaniline open the way to vivid pigments that color everything from leather to synthetic fibers. Given its reactivity, this compound rarely exists as the end product. Instead, it passes through facilities as an intermediate on its way to bigger and sometimes more controversial roles in agriculture or manufacturing.
Synonyms & Product Names
For those who work in procurement or regulatory roles, knowing the various guises of 2,4-Dichloroaniline can mean the difference between a smooth delivery and a costly mistake. The chemical shows up in documentation under names like 1-Amino-2,4-dichlorobenzene, 2,4-DCA, or even shorthand code numbers in some international catalogs. Each name ties back to the same core structure, so cross-checking product identity across languages and suppliers becomes part of daily due diligence. I’ve seen more than one shipment delayed or cross-docked simply because the label listed an alternate synonym, underscoring the importance of vigilance.
Safety & Operational Standards
Handling 2,4-Dichloroaniline demands respect. Years of industrial experience have drilled home that gloves, goggles, and controlled ventilation are not optional. The compound’s toxicity—especially through inhalation and skin absorption—drives strict training for operators. Facilities integrate local exhausts at filling stations, enforce sealed transfer systems, and run regular air monitoring as part of workplace safety programs. Spill response plans get tested at least once every shift. Regulatory oversight has steadily increased, forcing companies to track not just worker exposures but also offsite impacts—air, soil, and water emissions must stay below thresholds set by agencies like OSHA and the European Chemicals Agency. Improved protocols and better engineering controls have reduced incidents, but history warns that cutting corners brings steep long-term costs.
Application Area
The reach of 2,4-Dichloroaniline stretches further than most suspect at first glance. Its biggest claim to fame surfaces in the agrochemical industry, where it builds the backbone of herbicides that keep broadleaf weeds from choking farmlands. Generations of farmers have counted on downstream products, synthesized with this intermediate, to boost crop yields. The dye industry taps into its potential as well, spinning off colorants used in textiles, plastics, and ink. On occasion, chemists have explored its use in pharmaceutical synthesis, though its strongest market remains firmly rooted in controlling unwanted vegetation. Every time I’ve thumbed through registries of commercial chemicals or environmental monitoring results, the traces of 2,4-Dichloroaniline and its related compounds highlight the balance between modern efficiency and environmental legacy.
Research & Development
Ongoing R&D focuses heavily on safer routes to produce, convert, and degrade 2,4-Dichloroaniline while aiming for greener chemistry and lower waste. Researchers dig into catalytic processes that operate at lower temperatures or avoid hazardous byproducts. Analytical methods improve year after year, with labs adopting more sensitive chromatography and mass spectrometry to detect even trace quantities in environmental samples. My own time working with R&D teams showed the tension between innovation and regulatory compliance: new pathways offer efficiency, but they must deliver ironclad safety before regulators give their blessing. Partnerships across academia, industry, and government fund projects that probe both the risks and the remediation of 2,4-Dichloroaniline derivatives, reflecting a shift toward sustainability.
Toxicity Research
Toxicological studies have flagged real concerns with 2,4-Dichloroaniline and its persistent footprint in ecosystems. Chronic exposure in lab animals points to adverse effects on organs and metabolism, while environmental data draw lines connecting the compound to potential disruptions in aquatic systems. Industrial hygiene programs now incorporate biological monitoring for workers and environmental biomonitoring for communities near production sites. Scientific literature describes the compound as an irritant, with risks that climb the longer and more intense the exposure. In my years overseeing site safety, the stories of those who ignored early warning signs often ended with costly medical surveillance, remediation, and regulatory audits. These pitfalls push companies, researchers, and regulators to demand ongoing updates to risk assessments, venturing beyond quick fixes toward deep, science-based solutions.
Future Prospects
The future of 2,4-Dichloroaniline swings on a few clear pivots—tightening regulations, pressure for lower environmental impact, and demand for efficient crop protection and colorants. Calls for greener herbicides and dyes will force manufacturers to rethink production, adopt cleaner technology, and prioritize traceability through the whole supply chain. Replacement strategies that focus on biodegradable alternatives won’t take root overnight, but the writing is on the wall for outdated processes built with little regard for ecological consequences. Teams working in green chemistry, advanced analytics, and waste treatment point the way forward, promising more sustainable approaches without sacrificing agricultural productivity. Industry veterans and new entrants alike see that the compound’s legacy depends on how well stakeholders balance risk, innovation, and responsibility—decisions made today ripple out for decades in both fields and waterways.
What is 2,4-Dichloroaniline used for?
Behind the Lab Door
Anyone familiar with industrial chemistry will run into odd-sounding names. 2,4-Dichloroaniline belongs to that group. At first glance, it looks like something reserved for textbooks and safety data sheets, but daily life brushes up against products built from it. In the real world, this compound shapes things in agriculture, colorants, and also pharmaceuticals, affecting more people than folks realize.
On the Farm: The Hidden Helper
Agricultural fields rely heavily on herbicides to grow enough food, and behind the labels of “weed killer” and “crop protection” sits the chemical backbone of many products. 2,4-Dichloroaniline forms a building block in the synthesis of several common herbicides, including Diuron and Linuron. These get used across the world, especially on crops like soybeans, wheat, and even coffee. Without weed control, crop yields suffer, and many farmers turn to these chemicals to keep pests under control so families have food on the table.
For me, walking through rural towns growing up, the smell after spraying day meant more than odd odors, it meant neighbors betting their seasons on these synthetic molecules. There’s a sense of real risk. If a year’s wheat harvest fails, the entire community feels it. That puts a face on why these chemicals matter.
The Colors We See
My earliest memory of synthetic dyes lies in childhood—plastic toys, bright shirts, food packaging. 2,4-Dichloroaniline plays an unseen role here too. This compound acts as a starting ingredient for manufacturing certain azo dyes. These dyes show up in textiles, leather, even printing inks and paper products. Factories mix intricate organic chemicals to generate those deep reds, yellows, and oranges we see on store shelves.
Behind those colors, there’s a complex web. Producers working on tight deadlines rely on a steady supply of intermediates like 2,4-Dichloroaniline. Any disruption means missed shipping dates, increased costs, and product recalls. In a world where color sells and branding relies on an exact shade, control over these ingredients matters.
Balancing Safety and Progress
Every industrial story has two sides. 2,4-Dichloroaniline helps drive innovation, but it also comes with risks. Research from peer-reviewed journals highlights toxicity concerns for workers handling it, and environmental studies point to persistence in soil and water. Several regulatory bodies have classified it as hazardous. There are calls for tighter controls, better protective gear, and safer disposal methods.
Smart solutions don’t start with bans; they grow from honest conversations. Factories can invest in closed-loop processes to cut down on waste. Farmers can learn about safer application techniques, and governments have the tools to track and control harmful runoff. On top of that, the future might bring greener alternatives that offer the same benefits with fewer side effects—already, universities and companies are putting money into bio-based chemistry research.
Staying Informed, Staying Safe
Folks often glaze over at discussions about obscure chemical names, but understanding their reach makes a difference. Transparency, traceability, and investment in science-backed risk management provide real-world safeguards. 2,4-Dichloroaniline sits in the background of countless useful things; learning how to use it wisely ensures those benefits don’t come at too high a price.
What are the safety precautions when handling 2,4-Dichloroaniline?
Understanding What You’re Working With
2,4-Dichloroaniline serves as a building block in creating dyes, pesticides, and pharmaceuticals, but it poses real risks. This stuff isn’t your everyday household chemical; even a little mishap can mean trouble. Persistent contact with skin or inhalation often leaves workers with rashes, headaches, or worse. More serious or repeated exposure sometimes affects the liver and kidneys. There’s even evidence pointing to carcinogenic potential. The risks demand respect—so does the science, which recognizes its toxicity for people handling it day in and day out.
Personal Stories From the Floor
In my years in industrial plants, I’ve seen firsthand what happens when coworkers put safety to chance. Gloves with pinholes, masks pulled down for a quick conversation, bare arms on a hot summer day—these might seem like small slip-ups until someone lands in the emergency room. I remember a crew member turning bright red from a splash he brushed off as “no big deal.” It set off a long chain of incident reports and sleepless nights. Memories like those shape how seriously I take every bottle and barrel with the 2,4-Dichloroaniline label.
What Works: Safety Steps That Make the Difference
The facts leave no wiggle room. Anyone working with 2,4-Dichloroaniline should suit up. Gloves matter, but not just any pair—nitrile or neoprene lasts far better than bare latex. Coveralls give an extra barrier. Eye protection comes standard, and a full-face shield isn’t overkill when pouring or mixing. Always double-check that gear for breaks before starting.
Some folks toss on a disposable dust mask and call it good, but that doesn’t cut it. The EPA and NIOSH recommend a proper respirator, fitted to your face, with cartridges designed to trap organic vapors. Good ventilation in the workspace helps limit fumes collecting near your nose or mouth, and I swear by fume hoods for any job above a few grams at a time.
Common Mistakes: What Leads To Trouble
Shortcuts pile up over time. Washing gloves and reusing them? Bad call. Not labeling containers or leaving open vessels on the bench? That leads to cross-contamination—and accidents that send people home sick. Eating or drinking anywhere chemicals are used always seems convenient until someone gets a mouthful of residue. Storing 2,4-Dichloroaniline near acids or strong oxidizers increases the chance of a violent reaction. Double-check storage against safety data sheets and don’t improvise shelf space.
Emergency Response: Quick Thinking Saves More Than Time
Every site dealing with chemicals deserves a shower and eyewash station within arm’s reach. A couple lost seconds scrambling for help spell the difference between a mild irritation and a burn that scars. Spill kits should sit ready, with extra absorbent pads, and workers trained to lock down the area before cleanup. Donning the right protective suits matters here, too, since skin absorbs a surprising amount fast.
Stepping Up: Training and Oversight
Regular refresher sessions prove their worth, not only in keeping mandatory paperwork up to date, but also in building a culture that values every team member’s long-term health. Supervisors who model good habits, from double-gloving to checking fume extraction systems, set the bar. Welcoming questions or reporting concerns without fear of being blamed goes a long way toward safer shifts. At the end of the day, the goal is always the same—everyone goes home in the same shape they arrived.
What is the chemical formula and structure of 2,4-Dichloroaniline?
Breaking Down the Chemical Formula
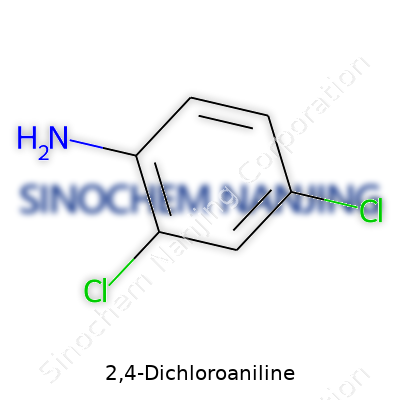
2,4-Dichloroaniline belongs to the family of anilines. Its chemical formula is C6H5Cl2N. In this molecule, two chlorine atoms replace two hydrogen atoms on an aniline ring, specifically in positions 2 and 4 of the benzene ring. That’s not just trivia for chemists—knowing the positions of those chlorine atoms can mean the difference between a compound that’s safe for use and one with unexpected reactivity or toxicity.
Molecular Structure: Looking at the Details
Imagine a benzene ring, which chemists often describe as a hexagon with alternating double bonds. In 2,4-dichloroaniline, an -NH2 (amino) group attaches to position one on that ring. Chlorine atoms sit at positions two and four. I remember the old ball-and-stick models in chemistry class—placing the chlorines near the amino group changes the electron distribution in the ring. That can alter how the molecule interacts with everything from other chemicals to living tissue.
The structure looks like this:
- A six-carbon benzene ring
- Chlorine atoms at positions two and four
- An amino group (-NH2) at position one
Chemists use this molecular arrangement to predict how the compound might behave. The chlorine atoms pull electron density towards themselves, which can make the aniline group less basic. That probably sounds like textbook trivia, but tiny changes in electron density can mean huge changes in reactivity, color, or toxicity.
Why Structure and Formula Matter in Everyday Life
I’ve seen people glaze over when the subject of chemical formulas comes up. These aren't just pointless numbers and letters. Companies use 2,4-dichloroaniline to create dyes, herbicides, and pesticides. The way those chlorine atoms sit on the ring can mean a cleaner, faster reaction during manufacturing, or it can trigger unexpected byproducts that pollute waterways or soil.
Health effects from direct exposure shine a light on why molecular details matter. The arrangement of the two chlorine atoms adds to the compound’s toxicity—studies have linked some dichloroanilines to skin irritation and more serious internal effects in larger doses. Limiting contact and following proper safety measures isn’t paranoia—it’s a direct response to what chemists see on paper, which gets validated by both lab and real-world experience.
Solutions and Responsibility
Growing up in an industrial town, I saw firsthand what happens when chemical formulas cross over from the lab to the environment without much oversight. Factories producing herbicides would sometimes let small spills or leaks go unreported. Over time, people in the area logged more illnesses linked to groundwater contamination. Real change started only when companies took environmental chemistry and public health seriously.
Today, tighter safety protocols, smarter chemical management, and investing in safer alternatives help address the problems associated with compounds like 2,4-dichloroaniline. Synthetic chemists search for ways to tweak these molecules so they break down faster or act less aggressively in the environment. At the basic level, it all circles back to understanding the formula and structure. Only with that knowledge can we strike a realistic balance between industrial progress and staying healthy.
How should 2,4-Dichloroaniline be stored?
Treating Chemicals with Respect
2,4-Dichloroaniline falls into a family of chemicals that demand attention, even before opening the drum or bag. This compound helps produce dyes and agricultural chemicals—valuable on the job, but risky once mishandled. Getting storage wrong risks health, property, and could even mean legal trouble. Past experience with chemical storage rooms sticks with me: a single oversight in handling or temperature control once led to an evacuation and weeks spent cleaning up. That memory sharpened my sense of caution around compounds like this.
Keeping It Cool, Safe, and Shielded
I learned the hard way that storing 2,4-Dichloroaniline means picking a spot protected from sunlight, moisture, and wild temperature swings. This compound breaks down faster with heat or light. In labs and stockrooms, metal containers with secure lids always did better at keeping it dry and uncontaminated. Glass works too for smaller batches, but nothing beats locking all openings tight. This keeps vapors in and keeps the workspace safer for everyone. The right shelving can make a difference as well. Putting this chemical below shoulder height and far from edges or walkways stops accidental drops and spills. Simple changes in storage layout—such as segregating by compatibility—really matter. Don’t leave it beside strong oxidizers or acids. The wrong mix can set off toxic fumes or even fire.
Ventilation Matters
Good airflow gives peace of mind. Even in sealed containers, 2,4-Dichloroaniline has a way of getting into the air if storage gets too warm or a lid slips loose. I saw poor ventilation in small closets turn a minor leak into a major problem, just from fumes building up. Chemical cabinet makers often advertise their vent systems, but even a basic exhaust fan helps. This simple addition can help keep exposure levels safe and meet local safety rules. It’s not just about rules—it’s about health. Chronic exposure can hit the liver or kidneys, and those long effects can take years to show. A quick look at OSHA and EPA guidance shows how real the risks stay, even for users who believe they know best.
Labeling, Training, and Checking In
Handwritten notes and faded ink don’t cut it. I always made sure to use clear printed labels, listing the chemical name and hazard information. Marking the date of delivery or opening helps track shelf-life. Some chemicals break down and lose strength or build up unsafe byproducts if kept too long. Rotating stock keeps accidents low and saves money. Storing emergency supplies—like spill kits and respirators—close to storage areas makes sense. Drills sound silly on paper, but they prepare everyone for real accidents. Any employee who works near 2,4-Dichloroaniline should know how to use protective equipment, respond to a spill, and contact emergency services. Regular walkthroughs catch leaks, corrosion, or slack procedures before they cause trouble. In my experience, the places that checked on staff habits the most ended up with fewest incidents. Culture counts as much as good containers.
Reassessing and Improving
Every storage spot, from big factory to backroom shelf, benefits from review. Safety Data Sheets deserve a careful read—rules change, new studies reveal fresh risks, and old practices get replaced. Following the latest science and tightening up training keeps workplaces and communities safer. For 2,4-Dichloroaniline, taking a hands-on, proactive approach lowers risk for everyone involved.
What are the potential health hazards of exposure to 2,4-Dichloroaniline?
The Everyday Risks Behind the Chemistry
2,4-Dichloroaniline doesn’t show up in everyday conversations, but for plenty of folks working around herbicides, dyes, or specialty chemicals, it feels all too familiar. My own time spent hunting down safety data for clients led me down odd rabbit holes. Few chemicals perk up a safety officer’s nerves quite like this one. Its sharp-smelling, yellowish flakes are a base ingredient in plenty of products but often cause more worry in the warehouse than excitement in the lab.
What Skin and Lungs Go Through
Those who handle 2,4-Dichloroaniline often end up worried about what will happen if it touches their skin or gets in their nose. Several reliable peer-reviewed studies show that skin contact can cause rashes, burning sensations, or even allergic reactions over time. Eyes sting and get red after even the lightest exposure. Inhaling dust or vapors can do more than make you cough—it can trigger sore throats, wheezing, and lung discomfort. The National Institute for Occupational Safety and Health warns that breathing in these fumes day after day can harm airways, especially in workplaces with poor ventilation.
Long-Term Consequences
Researchers have found it lingering in blood samples and the food chain near chemical plants. Chronic exposure becomes a real concern. Repeated contact may start to impact liver and kidney function over time. There are documented cases in areas with chemical spills where residents have reported fatigue, headaches, or trouble with concentration. Animal models exposed for extended periods suggest a possible cancer risk, although evidence remains patchy in humans.
Water and Environmental Issues Roll Back to Health
Communities living near factories can face low doses of 2,4-Dichloroaniline in drinking water or soil. The trouble doesn’t stay at the plant gate—the chemical seeps into rivers, then accumulates in fish or crops. Environmental Protection Agency reports show traces turning up downstream of industrial sites. With every spot it turns up, a new health worry follows, driving a demand for faster cleanup and better oversight.
How to Stay Safe
Protection comes down to more than gloves and goggles. Workplaces must invest in local exhausts and regular air monitoring. I always tell clients not to skip annual health checks for anyone exposed even for short periods—some symptoms don’t show up right away. Small tweaks—like using enclosed mixing systems or scheduling breaks in clean areas—reduce risk. Emergency training should cover skin washing stations and quick first-aid, giving workers better odds to avoid lasting harm.
Finding Healthier Alternatives
Regulators like OSHA and the EPA press manufacturers to lower accidental releases and explore safer alternatives. In my experience, companies that swap this for less reactive ingredients notice far fewer worker absences and complaints. Chemical engineers have started searching for greener processes that produce fewer byproducts, which benefits both factory crews and the neighborhoods next door.
Looking Toward Solutions
People don't always get the choice to walk away from risky materials—more often, the challenge is to make the risks manageable and the workplaces safer. Public pressure ramps up real improvements. Lawsuits, community activists, and honest reporting combine to encourage companies to get serious about protecting the folks exposed. It's a long haul, but every control measure and cleaner production choice pays back in healthier workers and neighborhoods.
| Names | |
| Preferred IUPAC name | 4-chloro-2-chloroaniline |
| Other names |
2,4-Dichlorobenzenamine 2,4-Dichloranilin 2,4-DCA Benzeneamine, 2,4-dichloro- 2,4-Dichloroaminobenzene |
| Pronunciation | /ˌtuː.fɔːr.daɪˌklɔːroʊ.əˈnɪliːn/ |
| Identifiers | |
| CAS Number | 554-00-7 |
| Beilstein Reference | 1209224 |
| ChEBI | CHEBI:34656 |
| ChEMBL | CHEMBL1369 |
| ChemSpider | 8514 |
| DrugBank | DB14055 |
| ECHA InfoCard | 03b7e5e3-7560-41f8-95f9-7cca9b4b1877 |
| EC Number | EC 219-468-7 |
| Gmelin Reference | 80904 |
| KEGG | C06563 |
| MeSH | D003563 |
| PubChem CID | 6769 |
| RTECS number | SS8925000 |
| UNII | 0VH015P3JZ |
| UN number | UN1590 |
| Properties | |
| Chemical formula | C6H5Cl2N |
| Molar mass | 162.04 g/mol |
| Appearance | White to slightly beige crystalline solid |
| Odor | Aromatic odor |
| Density | 1.42 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | 2.49 |
| Vapor pressure | 0.015 mmHg (25°C) |
| Acidity (pKa) | 3.55 |
| Basicity (pKb) | pKb = 9.25 |
| Magnetic susceptibility (χ) | -69.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.589 |
| Viscosity | 1.34 mPa·s at 25 °C |
| Dipole moment | 2.61 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 160.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -43.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3236.6 kJ/mol |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and eye irritation, may cause respiratory irritation, suspected of causing genetic defects, toxic to aquatic life with long lasting effects |
| GHS labelling | GHS05, GHS07, GHS08 |
| Pictograms | GHS06,GHS09 |
| Signal word | Danger |
| Hazard statements | Harmful if swallowed. Causes skin irritation. Causes serious eye irritation. Suspected of causing cancer. Toxic to aquatic life with long lasting effects. |
| Precautionary statements | P261, P264, P271, P273, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P330, P337+P313, P362+P364, P405, P501 |
| NFPA 704 (fire diamond) | 2,4-Dichloroaniline: "3-2-0 |
| Flash point | 150°C |
| Autoignition temperature | 425 °C |
| Lethal dose or concentration | LD50 oral rat 650 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat LD50: 650 mg/kg |
| NIOSH | 'PS7430000' |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for 2,4-Dichloroaniline: 0.1 mg/m³ (OSHA Ceiling) |
| REL (Recommended) | 0.5 mg/m3 |
| IDLH (Immediate danger) | 50 mg/m3 |
| Related compounds | |
| Related compounds |
Aniline 4-Chloroaniline 2-Chloroaniline 2,6-Dichloroaniline 3,4-Dichloroaniline 2,4,6-Trichloroaniline 4-Bromoaniline |

