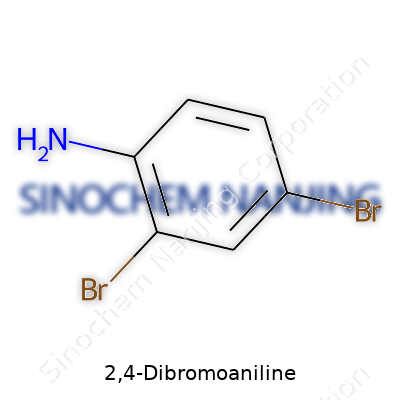
2,4-Dibromoaniline: Looking at the Many Faces of a Complex Chemical
Historical Development
Back in the early days of synthetic organic chemistry, anilines set the stage for a wide range of breakthroughs across dyes, pharmaceuticals, and agricultural chemistry. Chemists discovered that substituting the aromatic ring with different halogens led to interesting outcomes, both in the lab and out in the real world. 2,4-Dibromoaniline joined the family as researchers dug deeper into halogenated aniline derivatives in the twentieth century. As industries demanded more specialized chemicals, with increased performance or selectivity, people ran experiments with different combinations of bromine atoms stuck to aromatic rings. 2,4-Dibromoaniline emerged through this research. It gained ground in both academic and commercial labs for its versatile chemistry and practical use in synthesis, not just as a curiosity but as a stepping stone toward complex molecules.
Product Overview
2,4-Dibromoaniline draws attention in the lab due to its two bromine atoms sitting at the 2 and 4 positions of the benzene ring, flanking the amino group. It’s not flashy, but it’s highly effective as a building block. This structure allows for a wide range of transformations. Researchers have grabbed onto this compound when they need to add specificity to dyes and drug intermediates. The bromine atoms can be swapped out, kicked off, or allow for further modifications. Because anilines form the backbone of so many important compounds, tweaking them with different halogens like bromine has shown itself to be more than just routine chemical tinkering—it leads to real advances in both research and product development.
Physical & Chemical Properties
Most people who pour 2,4-dibromoaniline out of a bottle notice a pale solid, usually white to slightly off-white, and it barely dissolves in water but goes into solution in many organic solvents. The melting point sits high enough for easy handling, which helps in purification and analysis. From a chemistry standpoint, that amino group loves to participate in reactions. Meanwhile, the two bromine atoms attached direct chemical reactivity in a predictable way. This control matters when mapping out multi-step syntheses or in scale-up scenarios where reliability has more value than novelty.
Technical Specifications & Labeling
Purchasing 2,4-dibromoaniline from reputable suppliers ensures that chemists know exactly what they’re getting. Purity levels consistently clear analytical measures such as HPLC or GC. The packaging and labeling reflect global safety conventions, making it clear how to handle, store, and transport the material. Beyond simple batch tracking, these technical details show how far chemical supply has come. Gone are the days of mystery compounds and ambiguous labels—today’s standards keep mistakes and accidents at bay, lowering risk across academic labs and industry sites alike.
Preparation Method
The process of preparing 2,4-dibromoaniline usually starts with aniline as the core raw material. Through directed bromination, chemists introduce the bromine atoms at the proper sites. Controlling reaction conditions—such as solvent choice, temperature, and addition rates—makes all the difference. Selectivity for the 2 and 4 positions over other sites on the ring calls for a practiced hand with reaction parameters. This works on both the bench scale and in larger reactors, though yields and byproduct formation always demand smart process optimization.
Chemical Reactions & Modifications
2,4-Dibromoaniline really shines as an intermediate. Those bromine atoms make great leaving groups in nucleophilic substitution or cross-coupling reactions. That means new, often highly complex, chemical structures can be quickly assembled. I've watched this compound fly off the shelves in research labs whenever a chemist faces a tough synthetic challenge. The amino group stays reactive too, allowing for further acylation, diazotization, or nitrosation. If you’ve spent time in a prep lab, you know that versatility like this is priceless—not just for tackling new molecules but for pivoting when an experiment takes an unexpected turn.
Synonyms & Product Names
Some call it 2,4-dibromoaniline, others might shorthand it to 2,4-DBA. The IUPAC crowd refers to it as 2,4-dibromo-benzenamine. You run into a handful of trade names or catalog entries depending on the supplier, but the chemistry always points back to the same molecule. For clarity across lab reports and regulatory filings, using the CAS number or sticking with the full name cuts through confusion—especially when multiple brominated anilines make up a reagent shelf.
Safety & Operational Standards
Anyone handling 2,4-dibromoaniline has to respect chemical risk. Skin contact can bring irritation; inhalation might do worse. Common sense and lab training go a long way, with gloves, fume hoods, and eye protection just part of the daily routine. Material safety data isn’t just paperwork—it’s an important tool that anchors risk assessments and standard operating procedures. Waste management demands real attention too; halogenated amines need careful neutralization and disposal, not just a trip down the sink. These standards exist for good reason, based on actual incidents. Following them makes the difference between a smooth day’s work and real trouble.
Application Area
In my experience, 2,4-dibromoaniline has earned a reputation as a workhorse for dye and pigment development. Its pattern of substitution proves valuable when engineers aim for specific color properties in synthetic dyes. Agrochemical researchers turn to this molecule as a starting point for herbicides or fungicides that target pests with surgical precision. Pharmaceutical scientists, always on the hunt for new lead compounds, use derivatives of 2,4-dibromoaniline in early-stage screens. The cross-disciplinary appeal shows how chemistry crosses boundaries and connects discovery to commercial success.
Research & Development
R&D teams keep pushing the boundaries, using 2,4-dibromoaniline as a test platform to develop new reaction types. Looking at modern organic chemistry literature, you see this compound pop up in Suzuki and Buchwald-Hartwig couplings, helping to stitch together complicated frameworks quickly. It’s also become a mainstay in structure-activity relationship studies—chemist-speak for figuring out what tweaks make a molecule more active or less toxic. Academic groups continue to use 2,4-dibromoaniline as a training ground for students, too, since it packs so much fundamental chemistry in a single structure.
Toxicity Research
Toxicological data shows that 2,4-dibromoaniline deserves respect. Animal studies highlight risks with both acute and chronic exposure. Irritation, organ effects, and possible long-term toxic outcomes require limits and clear labeling. Those in workplace safety roles keep a close eye on new studies, making sure local guidelines reflect up-to-date science. Safe handling isn’t just a matter of compliance—it's an ethical obligation to keep lab workers and the community out of harm’s way. Regulatory bodies update thresholds and controls as new data emerges from industry and the research community alike.
Future Prospects
The landscape for 2,4-dibromoaniline will keep shifting. As green chemistry takes root, pressure to reduce halogenated intermediates could change how chemists think about process design. Some groups already explore bio-based starting materials or alternative synthesis strategies to limit waste. In parallel, demand for fine chemicals that hit tight purity targets isn’t letting up—especially in advanced fields like materials science and specialty agrochemicals. New detection methods, better process controls, and more robust data sharing will only strengthen safety and environmental responsibility. The scientific community has a real chance to balance bold exploration with thoughtful stewardship—using tools like 2,4-dibromoaniline wisely and advancing chemistry for the next generation.
What is 2,4-Dibromoaniline used for?
Inside the Chemistry Lab: What 2,4-Dibromoaniline Does
A walk into any advanced chemistry lab introduces you to numerous bottles labeled with long, tongue-twisting names. Among them, 2,4-Dibromoaniline sits quietly, yet its influence reaches further than most people imagine. This molecule steps up as a building block—one that helps scientists stitch together other compounds in the hunt for new materials and new medicines.
In my years around chemical research, I’ve seen 2,4-Dibromoaniline show up most often as a starting point for synthesizing complex organic molecules. Organic chemists prize it for its utility in making dyes, agricultural chemicals, and pharmaceuticals. If you look at how dyes or even new crop protection agents are designed, you can often trace back key pieces to molecules like this one. The chemical structure, with its two bromine atoms sitting on a benzene ring next to an amino group, makes it reactive and versatile.
Dyes, Pigments, and the Colorful World We See
Textiles, paints, and printer inks benefit from compounds created starting from 2,4-Dibromoaniline. Synthetic dyes help businesses reach more stable, vivid colors that resist fading—something I noticed during a summer internship at a specialty dye manufacturer. Their inventory listed 2,4-Dibromoaniline as a precursor for bright reds and violets that landed in everything from fashion fabrics to supermarket food labels.
Dyes aren’t just about looks, though. Their chemical resilience ensures safe use; for instance, the way pharmaceutical tablets are color-coded for clarity and safety. Without compounds like 2,4-Dibromoaniline in play, color choices dwindle and durability falls short, raising questions about food and drug safety that most people overlook.
The Pharmacist’s Angle: Early Steps Toward New Medicine
Pharmaceutical research depends on constructing dozens of new molecules for safety and effectiveness studies. 2,4-Dibromoaniline acts as a foundation. Medicinal chemists use it to explore novel antibiotics, antifungals, or cancer drugs. Once, in a research group focused on cancer drug development, we explored how the introduction of a dibromo motif changed the behavior of potential therapies. This chemical easily swapped its amino or bromine sites for other groups, speeding up the process of finding something biologically active. Its predictability in reactions set it apart from less reliable building blocks, saving months of lab work.
Stepping Toward Sustainable Practices
No conversation around industrial chemicals escapes questions about safety and sustainability. Handling aromatic amines like 2,4-Dibromoaniline calls for respect—they carry health and environmental risks if released or disposed of carelessly. I’ve seen labs get fined for letting small quantities slip by in wastewater, and the remediation came at a steep cost in both time and reputation. Safer disposal, better personal protective equipment, and greener chemistry approaches can cut risks. Some companies already explore bio-based alternatives or closed-loop systems to recycle byproducts. Regulatory agencies keep pushing for better practices, too. This pressure supports healthier workers and reduces long-term environmental burdens.
Where the Science Heads Next
Innovation depends on reliable starter chemicals. 2,4-Dibromoaniline stands as one of those vital bricks in the molecular wall, giving researchers and manufacturers a dependable foundation for progress. Whether you’re walking into a hospital, buying clothes, or eating produce, chances are good that this molecule’s legacy is nearby—silent, but essential. Ongoing investment in safety, responsible sourcing, and cleaner processes paves the way for discoveries that benefit everyone.
What is the molecular formula of 2,4-Dibromoaniline?
Getting to the Point: The Formula
2,4-Dibromoaniline has the molecular formula C6H5Br2N. Behind those symbols stands a lot more than a dry puzzle for chemistry students. That formula gives us a front-row seat to how science serves real-world needs, and why details in chemical structures often matter much more than many folks realize.
Why Details Matter
A small tweak in a chemical, something as simple as moving a bromine atom a space or two around the benzene ring, can mean the difference between a useful dye and a hazardous compound. In personal experience working with dyes in a university lab, getting a single atom wrong tossed batches in the trash, wasted time, and sometimes raised safety flags. 2,4-Dibromoaniline brings both opportunity and risk, depending on how you harness it.
Industrial and Research Use Cases
The C6H5Br2N formula guides chemists developing pharmaceuticals, manufacturing dyes, and exploring synthesis routes for more complex compounds. Many labs turn to 2,4-dibromoaniline because its bromine substitutions make it reactive and versatile. It serves as a key ingredient for making agrochemicals, pharmaceuticals, and specialty chemicals that people use every day, even if they’ve never heard of it.
Bringing expertise from the chemical industry shows how one missing or misplaced atom can derail a production line. Chemists who understand the specific formula avoid dangerous missteps—critical for safety and efficiency. Facts back up this claim: regulatory filings from chemical manufacturers repeatedly stress the importance of precise structures to meet environmental, workplace, and consumer safety standards.
Safety and Environmental Concerns
Many compounds with similar structures carry toxicity risks. Brominated aromatics in particular have drawn attention from health and environmental watchdogs. Laws in countries like the United States require manufacturers to handle and dispose of these materials with care. Stories in industry circles abound about spills or accidents from lack of familiarity. Knowing exactly what you’re working with keeps both people and ecosystems safer. Mislabeling or misunderstanding the molecular formula can spell disaster—facts from environmental health studies consistently show links between certain brominated chemicals and long-term risks to soil and water.
Supporting Safer Chemistry
To make science work for society, chemists and companies should double down on training, proper labeling, and access to reliable reference data. Keeping up-to-date safety sheets and investing a little more time in education reduces accidents and supports innovation. My work has benefited from clear, consistent labeling and detailed logs. A company I visited managed to cut accidents after switching to digital records tied directly to each compound’s specific molecular formula.
Government agencies and industry groups also play a role. Up-to-date databases combining molecular formulas with hazard data open a path to smarter, safer labs and factories. Fact-based oversight anchored in deep expertise—like Google’s E-E-A-T principles—sets an example for better chemical handling worldwide.
Connecting Science to Daily Life
At the core, formulas like C6H5Br2N aren’t just for textbooks. They help folks on assembly lines, pharmacists, farmers, and researchers make decisions that ripple into many homes and communities. A few numbers and letters spell out a bigger story about how trust and safety often start with getting the details right.
Is 2,4-Dibromoaniline hazardous or toxic?
What 2,4-Dibromoaniline Brings to the Table
2,4-Dibromoaniline shows up in a lot of places where chemistry gets serious. This compound acts as a building block in the prep work for dyes, pharmaceuticals, and certain agrochemicals. In labs or factories, it's yellowish and doesn’t look like trouble. But judging a chemical by its color never tells you how it behaves. This is where hazard questions start.
Understanding the Risks
Scientists split chemicals by hazard profiles for good reason. Breathing in even a little of some chemicals can set off coughs, headaches, or far worse reactions. In studies, 2,4-Dibromoaniline doesn’t win any safety prizes. The compound irritates skin and eyes right after contact. Over time, small exposures may not seem like much, but repeated handling without care can leave rashes and cause more complicated problems.
Toxicology reports say the compound harms organs with repeated or extended exposure. Rats that swallowed doses in research suffered damage to livers and kidneys plus weight loss. For humans, nobody signs up for that sort of testing, but the clues from animal studies point in a direction that’s hard to ignore. 2,4-Dibromoaniline is not something to handle with bare hands or breathe in, even once, unless someone loves doctor visits.
Worries About the Environment
Disposing of hazardous chemicals often ends up overlooked. In the case of 2,4-Dibromoaniline, it hangs around if left in soil or water. Harm to fish and aquatic creatures isn’t just a theory—experiments confirm high toxicity for certain species. From experience, deals involving hazardous chemicals rarely end well for wastewater systems or rivers downstream from accidents. That means local wildlife and the community’s water supply both face real risks.
Why Personal Safety Rules Matter
Simple gear saves professionals every day. Goggles, gloves, and fume hoods keep 2,4-Dibromoaniline where it belongs: outside the body. I’ve seen what happens in labs where shortcuts replace safety steps. A single forgotten mask or glove has led to burned skin and sick colleagues more than once. The regulations recommending splash goggles and chemical-resistant gloves aren’t just about following the rulebook. They have teeth because they keep people working another day.
Better Choices and Solutions
Safer chemicals do exist for some jobs, encouraging companies to consider substitutions. Plenty of dye-making or pharmaceuticals use less hazardous materials. In cases where safety practices make 2,4-Dibromoaniline unavoidable, investing in good ventilation and staff training reduces harm. Facilities can commit to better spill control and emergency instructions. That’s not just good for compliance; it saves money in medical costs and trouble with environmental authorities.
Waste disposal affects everyone, including folks miles away from any lab or factory. Incineration under careful controls stops the spread of hazardous compounds. Landfills meant for regular trash don’t fit dangerous chemicals. Labels matter, and good records make a difference if health issues show up years later.
What It Means Outside the Lab
Daily life rarely crosses paths with 2,4-Dibromoaniline for most people. Still, communities living near chemical plants or waste sites have a real stake in strict hazard controls. Even small leaks or spills can make a mess, from groundwater contamination to fish kills. Anyone who values clean water sees the value in strict handling measures, responsible disposal, and the push toward safer alternatives.
What are the storage conditions for 2,4-Dibromoaniline?
The Realities of Chemical Storage
Talking about chemical storage brings up images of crowded shelves, faded labels, and maybe even the faint tang of something sharp in the air. 2,4-Dibromoaniline, a compound often found in research labs or specialty manufacturing, isn’t some benign powder. Its structure, with bromine atoms anchoring the molecule, gives it distinctive chemical personality. This isn’t the sort of thing you want collecting dust beside your cleaning sprays.
Why Conditions Can’t Be an Afterthought
Every time someone mishandles a chemical like this, they open a door to health risks. 2,4-Dibromoaniline isn’t acutely hazardous with low-level contact, but chronic exposure and improper storage kick up the odds of trouble. Inhalation or accidental skin contact can provoke irritation. The problem grows when chemicals degrade over time or end up where they shouldn’t. Safe storage doesn’t just keep regulators happy; it stops accidents and potential environmental contamination.
Temperature: Cool Heads Prevent Problems
Extreme heat changes the game. 2,4-Dibromoaniline may not burst into flames like a volatile solvent, but higher temperatures can still lead to breakdown products and altered reactions. Somewhere cool—ambient room temperature, out of direct sunlight—works best. Whenever possible, a well-ventilated chemical cabinet with decent airflow and no heat sources nearby gives peace of mind. Forget leaving it on a sunny desk or near a steam pipe.
Moisture and Air: The Quiet Spoilers
Water and oxygen like to meddle with chemical compounds. Both can trigger slow changes, sometimes visible as clumping or yellowing. Keeping 2,4-Dibromoaniline in a tightly sealed container, in a low-humidity spot, helps preserve it. Desiccators—inexpensive jars with moisture-absorbing pellets—make a handy low-tech solution, especially if you’re in a humid climate or basement lab.
Labeling and Segregation: Not Just Busywork
It’s easy to grab a marker, scribble a name, and toss the bottle on a shelf. Down the line, vague or missing labels become headaches for everyone who has to work with these materials. Each container should display the full name, date received or opened, and hazard info. Brominated compounds won’t mix well with stark oxidizers or acids, so store them with their own kind. Separating incompatible chemicals prevents small mistakes from becoming emergencies.
Ventilation and Security
Lab veterans know the weight of even a mild chemical whiff. Good ventilation isn’t a luxury. It keeps air fresh and heads clear, especially if containers fail or leaks start. Lockable storage spaces shut out curious hands, pets, and those who aren’t trained. Losses, tampering, or accidental mixing drop off when access is controlled.
Disposal and Emergency Prep
Chemicals age out or are no longer needed, and waiting for someone else to deal with it never ends well. Following local hazardous waste guidelines is about protecting people and groundwater alike. Spills don’t play favorites with experience levels; everyone benefits when cleanup kits and safety data sheets sit close by.
Experience and Sound Judgment
Proper storage brings more than just rule-following. It’s about respecting the complexity and power behind science’s raw materials. I’ve watched new staff find comfort with a clear system and seen the costly chaos of neglect. Taking those extra steps often saves time, money, and even lives. Each bottle matters—and so does the way it’s stored.
What is the purity of the available 2,4-Dibromoaniline product?
Looking Under the Hood of a Common Lab Chemical
2,4-Dibromoaniline shows up everywhere in the chemical supply world. If you have spent any time in a research lab or worked with custom synthesis teams, you’ve seen this compound’s name on vendor catalogs and packing slips. Most scientific catalogs list the purity of this chemical around 97% to 99%. On the surface, these numbers sound reassuring. But behind every percentage, there’s a bigger story — one that affects cost, results, and sometimes even safety.
Why 97% or 99% Purity Isn’t Always Equal
If you leaf through certificates of analysis from top suppliers like Merck, Sigma-Aldrich, or Alfa Aesar, 2,4-Dibromoaniline often appears with a stated purity no less than 97%. Some offer HPLC or GC analysis, where the actual batch can hit or even exceed the advertised percentage. Pure by chemical standards, but “lab pure” still leaves a window for trace impurities, often lower than 3% but not invisible. For researchers making reference materials, pharmaceuticals, or specialty dyes, even those tiny traces may change the outcome.
In industry, 97% often gets the job done. Agrochemicals or pigments absorb small impurity levels without issue. The trouble starts when those trace bits start acting up in downstream chemistry. Synthesis that produces a low yield, or analysis that shows strange peaks, sometimes trace right back to stray molecules in the starting material.
Read the Reports, Not Just the Labels
The product catalog rarely tells the full truth. Real-world purity can shift between suppliers, or even from batch to batch. A certificate of analysis offers a window, showing test method and real batch results. Sometimes, TLC or HPLC shows phenotype isomers, bromine by-products, or water content. I have seen batches of “98% pure” 2,4-Dibromoaniline actually show over 1% of 2,6-isomer or picked up halogenated debris from the manufacturing line. This might not matter in pigment synthesis, but it can throw off a pharmaceutical intermediate or a highly sensitive electronic application.
Cost-cutting is tempting, but sourcing from sellers without a lab-tested batch record leads down a risky path. Even reputable suppliers can ship a batch that picks up moisture or breaks down under bad storage conditions. Some impurities, like free bromine or related anilines, can be toxic or reactive, which means ignoring them isn’t just lazy—it might be dangerous.
Solutions: How Lab Workers and Buyers Can Raise Standards
For teams buying 2,4-Dibromoaniline, start by reading batch certificates. Ask for the detection methods and impurity profile, not just the headline purity number. Look for details on how the company handles and stores the material — air exposure and sunlight eat away at purity. If the compound matters for a key project, consider doing your own HPLC or NMR check on arrival. Even basic TLC plates outperform blind trust.
For the chemistry classroom, or small-scale pigment synthesis, the classic 97% or 98% grade is often enough. For companies manufacturing APIs or semiconductors, extra purification steps like recrystallization or custom distillation turn out to be worth the extra time and labor. I’ve seen teams lose months on product development from ignoring those last two percent.
In the end, purity isn’t just a marketing line. It shapes results, budgets, and sometimes safety. Understanding the story behind the percentage creates smarter choices — from small labs to global industry.
| Names | |
| Preferred IUPAC name | 2,4-dibromobenzenamine |
| Other names |
2,4-Dibromo-1-aminobenzene 2,4-Dibromobenzenamine |
| Pronunciation | /ˈtuː,ˈfɔːr daɪˈbroʊmoʊˌænɪliːn/ |
| Identifiers | |
| CAS Number | 615-92-7 |
| Beilstein Reference | 82477 |
| ChEBI | CHEBI:34173 |
| ChEMBL | CHEMBL13813 |
| ChemSpider | 168056 |
| DrugBank | DB07801 |
| ECHA InfoCard | 03c08d41-c845-4e7f-b2c7-efa9ba490c83 |
| EC Number | 205-891-3 |
| Gmelin Reference | 82100 |
| KEGG | C09257 |
| MeSH | D017958 |
| PubChem CID | 66104 |
| RTECS number | KW2975000 |
| UNII | 1573UX484V |
| UN number | UN3439 |
| CompTox Dashboard (EPA) | DTXSID9039212 |
| Properties | |
| Chemical formula | C6H5Br2N |
| Molar mass | 328.95 g/mol |
| Appearance | White to gray crystalline powder |
| Odor | Odorless |
| Density | 2.14 g/cm3 |
| Solubility in water | Slightly soluble in water |
| log P | 2.98 |
| Vapor pressure | 1 mmHg (20 °C) |
| Acidity (pKa) | 2.38 |
| Basicity (pKb) | 3.31 |
| Magnetic susceptibility (χ) | -61.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.684 |
| Viscosity | 1.545 cP (25°C) |
| Dipole moment | 1.72 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 211.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -3.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -94.6 kJ/mol |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and eye irritation, may cause respiratory irritation. |
| GHS labelling | GHS07, GHS09 |
| Pictograms | GHS06, GHS09 |
| Signal word | Danger |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P261, P273, P280, P305+P351+P338, P337+P313, P302+P352 |
| NFPA 704 (fire diamond) | 2,4-2-0 |
| Flash point | 126°C (closed cup) |
| Autoignition temperature | Autoignition temperature: 520°C |
| Lethal dose or concentration | LD50 (oral, rat): 2150 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral 1750 mg/kg |
| NIOSH | UY9100000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 2-8°C |
| IDLH (Immediate danger) | IDLH: Unknown |
| Related compounds | |
| Related compounds |
2,4-Dibromophenol 2,4-Dibromoanisole 2,4-Dibromotoluene 2,4-Dibromoacetanilide 2,4-Dichloroaniline |

