2,4,6-Trinitroxylene: Igniting a Closer Look
The Story Embedded in Every Molecule
My fascination with chemicals that changed history always lingers at the intersection of science, society, and policy. 2,4,6-Trinitroxylene, better known as TNX, carves its own path through that intersection. Many chemicals travel from curiosity in a professor’s notebook to heavy industrial use, and TNX followed this arc in the late 1800s. It emerged in the shadow of trinitrotoluene, known to everyone as TNT, during a time when industries and armies raced to harness explosive compounds. Historical records show scientists pushing ahead with substitutions on aromatic rings to squeeze more bang out of each molecule. TNX soon found itself drafted into military research and commercial pyrotechnics. Scientists saw something interesting about the extra methyl group; on paper, it hinted at changes in power, stability, and handling. In my own journey digging through old chemistry journals and war-era documents, I noticed how researchers shifted their focus from basic discovery to seeking operational improvements—things like lower melting points and safer transport—even though safety still felt inadequate by modern standards.
Unpacking the Substance: A Walk Through the Basics
TNX doesn’t show up on a grocery store shelf, and perhaps that’s for the best. Structurally, it's built from a xylene backbone, each ring position capped by a nitro group, echoing the classic shapes familiar to anyone who’s ever doodled a benzene ring. This gives the molecule its punch and a stubborn persistence in the environment. Look and feel matter in real-world handling: TNX often appears as pale yellow crystals, and it’s got a higher density than most people expect from something engineered to burst apart. Its melting point sticks uncomfortably close to what you’d get inside a car on a sunny day—much lower than many other explosives—meaning improper storage can lead to mishaps much too easily. As for solubility, water won’t do much, but organic solvents open up possibilities, which historically created logistical headaches when shipping or cleaning equipment. My own hands-on work in the lab years ago made me appreciate these details, since things like dustiness and static sensitivity forced me and my colleagues to rethink how we moved materials—no shortcuts, no complacency.
Specs, Labels, and the Real-World Complexity
Technical specifications for TNX sit under the scrutiny of both chemists and safety officers. Density, particle size, and purity mark the basics, but operational labeling tells its own story. Labels must warn of explosion and acute toxicity. That’s not ink on a sticker—it’s a reminder of what’s at stake, a discipline forged by past disasters. In one anecdote shared during my safety training, a mislabeled drum led to an uncontrolled reaction and injuries; nobody in those circles forgets how close they came to tragedy. Modern standards now demand prominent hazard symbols, explicit instructions on storage temperature, and emergency deactivation methods. There's no room for outdated labeling or careless handoffs between departments. The regulatory environment focuses heavily on enforcement, with agencies inspecting not just the paperwork but workplace practice, down to the last glove or spill kit. For anyone hoping to work with such compounds, getting comfortable with this ecosystem remains more than a box to check—it's a matter of professional survival and collective responsibility.
Making TNX: Method with Meaning
Preparation methods for TNX resemble a high-wire act, with no safety net. Chemists nitrify xylene using concentrated acids under precise conditions. Slight drift in temperature or acid strength brings the risk of runaway reactions. Recipes evolved as industrial chemists chased efficiency and profit in the early 20th century, often at the expense of worker safety. Later breakthroughs improved yield and consistency but barely contained the inherent hazards. Cleanroom approaches and fume hoods reduced occupational exposures, but even today, preparation chains leak plenty of risk. Looking back at older manufacturing sites, reports of acid burns and environmental contamination serve as reminders. More recent shifts toward automated synthesis tackle some dangers, but financial barriers stall adoption outside larger facilities. It’s clear from reading industrial accident investigations that carelessness or cost-cutting in these steps spells disaster—not just for operators but for neighbors and emergency responders. No shortcut proves worth the headlines.
What Happens Next: Chemical Reactions and Tweaks
Once in hand, TNX offers both potential and peril in further chemical reactions. With all three available positions on the ring substituted by nitro groups, chances for further direct substitution almost disappear, though reduction and partial denitration open the door to synthesizing related compounds. Laboratory experimentation with TNX demands extra steps to shield personnel from exposure, owing to its low threshold for accidental initiation. The molecule resists most common dilution, and reactive impurities—whether from hastily cleaned equipment or contaminated precursors—can result in violent decompositions. Over the years, chemists explored ways to shape TNX into plasticizers, melt-pour explosives, and even as an additive to stabilize other formulations. No matter the goal, the underlying message keeps repeating: deep knowledge of reactivity saves lives. My mentors passed this down through old stories of accidents and near-misses, which echo across every new experiment or scale-up process.
Names, Aliases, and What They Reveal
TNX goes by several names—2,4,6-trinitroxylene, trinitroxylene, and sometimes just its abbreviation. Such aliases aren’t mere trivia. In regulatory lists and forensic analysis, using the right name avoids misidentification, fines, or worse, accidental exposures. Legal and industrial documents rarely forgive slip-ups, as historical cases show that both overzealous renaming and missed synonyms cost companies hard cash and, sometimes, their operating licenses. This tangle of names expands in international commerce, complicating tracking and enforcement by customs or oversight bodies. Those aiming for transparency in supply chains—something regulators preach—must keep this in mind throughout procurement, labeling, and reporting. A slip here may cascade into international disputes or lost shipments, each with its own set of headaches and liabilities.
Safety and What Hard Experience Has Taught
Handling TNX demands a seriousness built on painful history. Its primary threats—explosion, severe toxicity, and combustion—are personal, not abstract. In my days working around energetic compounds, safety drills drilled home the reality: you either respect the chemical, or you risk everything. Modern protocols lean heavily on double containment, anti-static measures, remote handling tools, and comprehensive training. Constant updates to standard operating procedures reflect the lessons learned from each new incident—whether in remote plants or sophisticated defense labs. I remember colleagues recounting stories where a moment’s lapse, one spark, or an overlooked tool left entire projects on hold due to investigations. It’s no exaggeration to say that a culture of constant vigilance saves not just the bottom line, but human lives and family futures.
Application Areas: More Than Just Explosives
Most think only of military or mining applications, but TNX’s utility doesn't end there. Beyond blasting agents, it found a place in specialized fireworks, pyrotechnic signals, and research into controlled detonation devices. Early efforts to repurpose TNX for demolition, shaped charges, or underwater blasting often pushed its technical limits, yielding both innovative results and messy failures. Periodic attempts to tame TNX for industrial cutting or propellant research wavered as safety incidents set back momentum. While newer, less hazardous chemicals have edged TNX out in many sectors, enthusiasts and some defense agencies keep a close eye on its unique strengths, especially in extreme temperature applications. The bottom line: TNX remains a tool that only the well-trained and well-equipped dare to use, but its role in driving practical advances in engineering, weaponry, and emergency response is hard to ignore.
Research Keeps Moving Forward
Academic and industrial research into TNX focuses on managing risk and unlocking untapped utility. Persistent questions: Can we improve stability or reduce sensitivity? How does the compound behave under pressure, shock, or mixed with flame suppressants? One trend in recent papers highlights the hunt for green synthesis routes that cut down hazardous waste—a nod to the increasing focus on sustainability. Laboratories across the globe compare reaction conditions, handler exposure levels, and accident statistics, always hoping for a breakthrough. During a recent visit to an explosives chemistry conference, I heard first-hand how teams leverage computer simulations to model detonation events or predict aging effects, a leap from hands-on trial-and-error of previous generations. These discussions always lead back to practical implementation, because in energetic materials, an elegant theory offers no protection without close attention to detail at every scale of handling and use.
Toxicity: Risks That Stay With Us
Toxicology research confirms TNX packs a punch not just in explosions, but also in harm to human health. Acute exposure leads to nausea, headaches, and, at higher levels, organ damage. Long-term effects stay poorly characterized, in part because most exposures documented occurred during explosions or accidents, leaving toxicologists to work with limited case histories and animal studies. Environmental persistence stays a sore subject—TNX lingers, resists easy breakdown, and travels through soil and water, raising fears in communities near production sites. Some remediation advances help, but nothing so far delivers a total fix. Regulatory pressure for better worker protections and community monitoring grows with every spill or contamination incident, and rightfully so.
Looking Ahead: Future Prospects for TNX
The long view on TNX holds as much trepidation as promise. Demand won’t vanish entirely so long as applications remain in military tech, demolition, or niche industrial uses, but advances in alternative compounds keep shrinking its role. Future progress likely hinges on successful innovation in safety and environmental management: automating dangerous steps, finding more elegant synthesis, or deploying efficient clean-up tech. Strong research networks and global cooperation offer some optimism, but the lessons from TNX—care, respect, continual improvement—should travel alongside every barrel. The story hasn't ended. For science and society, each new development reminds us that progress comes with big responsibilities. Those who forget the past risks may find themselves reliving them in the next news cycle.
What is 2,4,6-Trinitroxylene used for?
Finding Power in Chemistry
2,4,6-Trinitroxylene doesn’t show up in most people's daily conversations, but it plays a role in some heavy-duty work. Its structure, based on xylene with three nitro groups, means it has a lot of stored energy. That fact alone explains why industries that deal with energy release—especially controlled explosions—look at it closely.
Explosives and How They Shape Our World
Let’s talk about its main use: explosives. Whether talking mining, construction blasting, or other industries needing serious breaking power, engineers need materials that have a punch but that they can handle safely. 2,4,6-Trinitroxylene delivers energy in a relatively stable package until detonated with the right conditions. It’s part of the reason why mining operations can reach minerals buried deep in the ground or why road projects make their way through tough rock formations.
Military groups pay attention to these compounds, too. Energetic materials like this explode with enough force to move earth or disable structures, but their handling requires a disciplined respect for safety protocols. Careless storage or transportation can lead to tragic outcomes. In the past, mistakes with similar materials sparked warehouse disasters or transport accidents, sometimes claiming lives. Old headlines about ammonium nitrate explosions remind us of the double-edged sword these materials represent.
Balancing Strength and Responsibility
I spent several years covering stories from heavy industry. Blasting experts let me watch their process. After every charge, a safety officer swept the site. Everybody respected the blast zone as though one wrong step could change their lives. In those moments, I understood the trust they put in science and the rituals of double-checking every wire and barrel.
It’s tempting to think about chemicals like 2,4,6-Trinitroxylene as magic bullets for tough jobs. But the story doesn’t end at the blast. Responsible users have to manage residues, monitor air and water around blast sites, and monitor soil for years if needed. The upside to compounds like this comes with an obligation to prevent unintended harm—an idea that keeps returning in debates about chemical production and use.
Making Safety Part of the Equation
In my experience, the people who work with high-energy chemicals carry an intense level of training. Engineering rules—the ones that seem so strict on paper—actually save lives. Companies improve their systems through lessons learned from near misses, new research on environmental impacts, and regular drills. In regions with strict enforcement, accident rates drop. Emergencies stay contained. Workers return to their families after each shift.
The broader public probably won’t ever see or handle 2,4,6-Trinitroxylene, but its existence highlights the need for oversight and ethical use. Factories and storage sites require constant monitoring. Emergency responders practice their responses for scenarios involving energetic materials. Governments around the world are rewriting regulations as chemists discover new compounds with even more power or different safety profiles.
Looking Ahead
As demand for raw materials and infrastructure grows, safe explosive handling will only become more important. Investment in research, worker training, and transparent safety records keep the risks in check. Everyone benefits when those closest to the work keep learning from science and from each other—turning dangerous potential into progress, with safety at the center.
Is 2,4,6-Trinitroxylene explosive or hazardous?
Understanding 2,4,6-Trinitroxylene in Plain Terms
2,4,6-Trinitroxylene—sometimes cropped up in old chemistry texts as TNX—pops up every now and then in discussions about nitrated aromatic compounds. Many people hear a name like that and imagine something thunderous, probably because it sounds a lot like the infamous TNT (2,4,6-trinitrotoluene). It’s important to check the facts before jumping to conclusions about chemicals with long, intimidating names.
Is 2,4,6-Trinitroxylene as Dangerous as It Sounds?
The structure of 2,4,6-trinitroxylene has three nitro groups stitched to a xylene core. Nitrated aromatic compounds usually get associated with explosives, and for good reason. TNT, for example, has powered everything from blasting tunnels to military operations for more than a century. Similarly, picric acid set off factory explosions decades ago when folks didn’t realize how touchy these materials could be. So, it’s easy to look at trinitroxylene and get nervous.
Here’s where reality breaks away from old habits. 2,4,6-Trinitroxylene does have the building blocks of a typical explosive—the aromatic ring, the nitro groups. But not every combination of these makes something suited for bomb-making or demolition work. TNX doesn’t share the same reactivity as TNT. While there is limited literature, everything we know points to a material that doesn’t ignite or detonate with the ease or speed of familiar military stuff. One reason lies in the position of the nitro groups and the methyl groups on the xylene ring, which tamp down its ability to fall apart violently when shocked or heated.
The best assessment out there is that TNX can be called a “potential explosive.” Under special conditions—high heat, enough friction, or deliberate chemical provocation—it could break apart rapidly. For most workers in labs or factories, TNX doesn’t leap into flames, doesn’t explode from a drop, and doesn’t scatter shrapnel. Mishandling any nitrated aromatic is foolish, but the risks rank below those for dynamite, blasting caps, or TNT.
Hazards Don’t Stop at Explosions
There’s a different angle, though: health hazards. I’ve watched plenty of chemists work with nitrated compounds. Gloves, goggles, and fume hoods aren’t just bureaucracy—they save fingers, eyes, and lungs. Nitrated aromatics can irritate skin, harm the liver, and some are likely to cause long-term harm. Just because a material doesn’t explode doesn’t mean it’s safe. TNX belongs on the shelf of “approach with care” chemicals. It could stain skin, cause allergic reactions, or pose risks if inhaled as dust.
Disposal challenges lurk in the background, too. Pouring leftovers down the drain or tossing them in the dumpster sets up problems for water supplies and landfill workers. In practice, the right way to handle waste calls for specialized disposal through companies certified to deal with hazardous materials.
What Can Be Done to Lower Risks?
Clear labeling and locked storage keep trinitroxylene away from the wrong hands. Training helps everyone on site know the signs of trouble—strange smells, dust, or leaks. Spill kits and emergency plans keep small messes from growing into big news stories. Regulators and safety officers deserve a shout-out for setting the bar higher each year. Proper ventilation keeps fumes low, and personal protective gear shields the people at the frontlines.
Science and experience teach the same lesson again and again: Don’t let technical names fool you, but don’t get too casual. Explosive power isn’t the only risk worth watching. Health hazards matter as much, if not more. With solid habits, good equipment, and a bit of respect, 2,4,6-trinitroxylene stays just another specialty chemical, not front-page news.
What are the safety precautions for handling 2,4,6-Trinitroxylene?
The Risks and Why Precautions Matter
Anyone who’s spent time in or near an explosives lab knows one simple rule: don’t trust a compound just because it looks familiar. 2,4,6-Trinitroxylene may look like its chemical cousins, but this one doesn’t forgive mistakes. Ignoring safety routines can end up in serious injury, destroyed equipment, or even loss of life. This stuff creates powerful blasts. Incidents with similar nitroaromatic compounds have caused workplace disasters before, reminding us that even a moment’s carelessness has consequences that go far beyond the lab.
Personal Protective Gear
No one should even be in a storage or handling area for trinitroxylene without proper gear. Regular cotton or polyester clothing doesn’t cut it. People wear flame-resistant lab coats, goggles that actually block chemical splashes – not those cheap plastic shields – and gloves designed specifically for chemical exposure. Respirators come out if there’s a risk of dust or vapors since inhaling nitroaromatic compounds carries long-term health hazards, not just burns or rashes. Overshoes help keep residue off the hallways and out of your car, so you don’t track danger home.
Safe Storage and Handling
I’ve seen more than a few storage rooms with barrels stacked on top of each other, far too close to heat vents or sunlight. For trinitroxylene, that’s a recipe for disaster. This material sits in cool, dry, well-ventilated spaces – away from anything flammable, oxidizing agents, or sources of friction. People keep it separated from strong acids, bases, and metals that might set off a reaction. You never see open flames, hot tools, or even cell phones used nearby, since one spark turns a regular morning into a panic.
Spill Response and Emergency Planning
It’s tempting to rely on hope, but spills happen. Emergency kits stay fully stocked with absorbent materials—non-static, non-sparking mops and sweepers, not ordinary cotton towels. Quick cleanup requires training, not just guesswork. Folks practicing regular drills handle minor spills safely, avoid creating dust clouds, and never sweep or dry mop. Contaminated materials go into steel containers built for explosives. Fire extinguishers sit in easy reach, but team members know that sometimes evacuation is safer than trying to fight a fire caused by nitro compounds. Local fire departments collaborate on planning; they know the risks and don’t get caught off guard.
Monitoring and Waste Disposal
Monitoring air quality isn’t just paperwork. Portable sensors flag high vapor concentrations before workers feel symptoms. Waste from trinitroxylene processing piles up quickly if routines slip, so staff separate and label it in line with official regulations. Disposal means working with firms certified for hazardous and explosive materials, never dumping something down the drain or in the regular trash. Mismanaging leftover compounds taints local groundwater and puts the wider community in harm’s way.
Training and Ongoing Culture
Training doesn’t stop after a single course. Supervisors keep checking for shortcuts and slipping standards. Posters with chemical safety tips cover the break room walls, not because management wants to nag, but because new faces show up all the time and long-timers get too comfortable. Real safety grows out of habit and a willingness to call out unsafe actions – even if it means risking a tense moment with coworkers.
Looking Forward with Vigilance
Chemistry labs keep finding new ways to reduce risk, from better gloves to smarter sensors. Regulation can feel frustrating, but every step roots in real-life disasters. For anyone working with compounds like 2,4,6-Trinitroxylene, staying alert and respecting the danger isn’t about paranoia – it’s about making sure everybody walks out the door the same way they came in.
What is the chemical structure and formula of 2,4,6-Trinitroxylene?
Understanding 2,4,6-Trinitroxylene
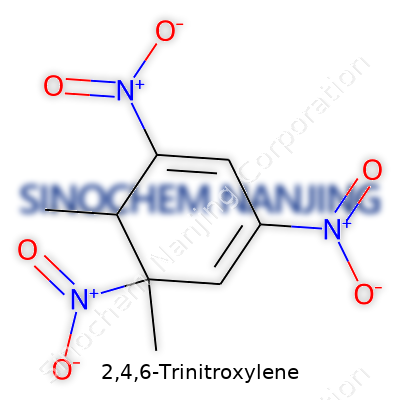
2,4,6-Trinitroxylene sounds like a mouthful. Lucky for those who tackle complicated molecules daily, chemists break it down into something more manageable. Known in short as TNX, this compound is a nitroaromatic derivative of xylene. The nitro groups, three of them, line up at positions 2, 4, and 6 on the benzene ring. The xylene backbone brings in two methyl groups attached to the aromatic ring. The structure matters—a lot—because where you stick those nitro and methyl groups changes everything in terms of how the molecule behaves and reacts with other chemicals.
What the Chemical Formula Tells Us
For TNX, the chemical formula spells out as C8H7N3O6. Chemists will see a ring made of six carbons. Two spots wrap up methyl groups (like off-ramps from a busy highway), and nitro groups attach to three other carbons. Visualizing it helps—it’s a benzene ring at the core with methyl twins at positions 1 and 3, and nitros inserted at the alternate positions. It looks crowded—probably not the best compound to happily dissolve in water or stroll through biochemical reactions.
Why the Nitty-Gritty Structure Matters
I spent a summer scraping by in a dusty chemistry lab, mixing similar nitro compounds. What struck me: just by shifting a methyl group or nudging a nitro to a new position, properties swing from boringly safe to dangerously powerful. Explosives—think trinitrotoluene (TNT)—owe their bite to arrangements like this. TNX isn’t as well-known outside chemistry circles, but its blueprint sits close to TNT, just swapping one methyl for two on that benzene ring.
The triple-nitro punch means this molecule stores a serious amount of chemical energy. Organizations like the National Institute for Occupational Safety and Health track nitroaromatic exposure because their toxicity pushes into concerning territory. Safety guidelines exist for very good reasons. Even a whiff of the dust off compounds like TNX can hurt your health. Regulators in the U.S. and EU take these risks seriously and list strict limits on handling and transport.
Real-World Impact and Responsible Chemical Use
Experts keep digging into how new nitro compounds interact with the environment. The United Nations and others highlight contamination issues from legacy industrial activity. Nitroaromatics, whether spilled from munitions or dye production, linger in soil and water for decades. High stability brings persistence. Some folks have called for updated waste management strategies—shifting from just burying chemical waste to new methods like bioremediation. Some bacteria break down complex nitro compounds, but it's slow, and the field struggles to scale up.
Chemists have started looking beyond classic explosive compounds. There’s renewed focus on green chemistry. This means designing molecules with performance, safety, and environmental impact in mind. Some research teams work on making xylene derivatives less persistent and easier to break down. Others push for better labeling, safety practices, and worker education. Even a textbook example like 2,4,6-Trinitroxylene, mostly discussed around reaction mechanisms and synthetic routes, finds new relevance because of these broader concerns.
Looking Forward
The profile of 2,4,6-Trinitroxylene—dense with nitro groups stuffed into an aromatic backbone—captures the classic pitfalls and possibilities of twentieth-century chemistry. Its formula, C8H7N3O6, sketches not just a structure but a challenge for those who handle, study, or clean up after powerful molecules. Real progress depends on science and policy working together—using lessons from the past to shape a smarter, safer future.
How should 2,4,6-Trinitroxylene be stored and disposed of?
Serious Risks Hiding Behind the Chemistry
2,4,6-Trinitroxylene sounds obscure to most people, but those who have spent years working in laboratory safety know that working with high-energy compounds never comes easy. This chemical features a structure similar to the notorious trinitrotoluene (TNT), which means it belongs to a group known for volatility and a history of nasty accidents. Stored on a shelf beside common solvents or in a warm supply room, trinitroxylene can go from quiet and stable to downright dangerous without much warning. I’ve seen colleagues grow too relaxed around such compounds, but all it takes is a bump, some heat, or an unexpected reaction to trigger hours of emergency response.
Storage: No Room for Shortcutting
Some things in life you can innovate around; explosives and energetic materials don’t fit in that category. 2,4,6-Trinitroxylene keeps its composure only under strict temperature and humidity. Experienced chemists never store it in glass jars mixed on the same shelf with acids or metals. Several best practices come straight from safety data sheets and hard-earned lessons:
- Dedicated explosion-proof magazine: Meant for storing energetic materials, these are built with thick walls, blast relief, and remote siting away from main buildings.
- Consistent temperature and low humidity: A climate-controlled environment keeps risk low. Humidity rusts containers, heat triggers decomposition—both spell trouble.
- Keep amounts small: Large quantities make response plans go from tricky to nearly impossible. Most regulations cap what you can store—stick with that limit.
- Labeling and inventory tracking: Forgetting what sits in that corner cabinet breeds mistakes. Date every batch, check expiration, and review logs regularly.
Years ago, a neighboring lab ignored some of these points. Their storage space faced the afternoon sun. Someone restocked an old bottle and left it on an upper shelf alone; within months it showed signs of decomposition. Local firefighters had to coordinate a full removal, shutting down a block for hours. Situations like that reinforce the need for careful planning and controlled environments.
Disposal: No Shortcuts, No Improvising
Disposing of 2,4,6-trinitroxylene isn’t for the faint of heart or a job to hand off to “someone from maintenance.” This job remains firmly in the wheelhouse of hazardous waste professionals. People sometimes ask about neutralizing these kinds of chemicals at home or down the drain—don’t even think about it. Here’s what experience and the literature recommend:
- Partner with licensed hazardous waste contractors: These folks handle the removal, transport, and eventual destruction—most often by controlled incineration or chemical decomposition under strict protocols.
- Document everything: From transfer paperwork to transportation logs, government agencies want a paper trail. Cutting corners invites fines or investigation.
- Emergency plans in place: Even if you follow the rules, mistakes or container failures happen. Spill kits, evacuation plans, and regular training give teams a fighting chance if things go wrong.
I once watched a team spend an entire day neutralizing less than a kilogram of old trinitroxylene. The process required blast shields, remote handling arms, and full-body protection. It’s a good reminder that when disposal becomes necessary, professionals bring not only the right gear but also years of experience.
Smarter Storage and Responsible Disposal Keep Everyone Safe
No shortcut or improvisation ever justifies the risk associated with energetic materials. Regulations reflect real dangers, and the smallest slip can send waves of consequences through a community. The right approach grows out of a respect for the chemical—not fear, but clear-eyed, responsible management and an understanding that safety is earned through constant attention.
| Names | |
| Preferred IUPAC name | 1,3,5-Trimethyl-2,4,6-trinitrobenzene |
| Other names |
TNX 1,3,5-Trinitro-2,4,6-trimethylbenzene |
| Pronunciation | /ˌtraɪˌnaɪtrəʊˈzaɪliːn/ |
| Identifiers | |
| CAS Number | 82-70-6 |
| Beilstein Reference | 86721 |
| ChEBI | CHEBI:75915 |
| ChEMBL | CHEMBL290512 |
| ChemSpider | 16293 |
| DrugBank | DB13225 |
| ECHA InfoCard | 04000000823955-52-0000 |
| EC Number | 2.4.6 |
| Gmelin Reference | 83988 |
| KEGG | C19645 |
| MeSH | D014286 |
| PubChem CID | 69281 |
| RTECS number | XP2100000 |
| UNII | BZG6A6Q1S2 |
| UN number | UN0395 |
| CompTox Dashboard (EPA) | DTXSID3014769 |
| Properties | |
| Chemical formula | C9H9N3O6 |
| Molar mass | 227.14 g/mol |
| Appearance | yellow crystalline powder |
| Odor | odorless |
| Density | 1.82 g/cm³ |
| Solubility in water | insoluble |
| log P | 1.98 |
| Vapor pressure | 4.44E-4 mmHg at 25 °C |
| Acidity (pKa) | 3.16 |
| Basicity (pKb) | 13.86 |
| Magnetic susceptibility (χ) | -37.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.6260 |
| Viscosity | 3.17 cP (20°C) |
| Dipole moment | 3.78 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 240.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | –65.9 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3255.0 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS01,GHS02,GHS07,GHS09 |
| Signal word | Danger |
| Hazard statements | H201: Explosive; mass explosion hazard. |
| Precautionary statements | P210, P220, P221, P280, P305+P351+P338, P370+P378 |
| NFPA 704 (fire diamond) | 2,4,6-Trinitroxylene: 2-4-2-Explosive |
| Flash point | 79 °C |
| Autoignition temperature | 534 °C |
| Explosive limits | 0.5–6.0% |
| Lethal dose or concentration | Lethal dose (LD50, oral, rat): 340 mg/kg |
| LD50 (median dose) | LD50 (median dose): 308 mg/kg (oral, rat) |
| NIOSH | TT2300000 |
| PEL (Permissible) | PEL: 1.5 mg/m³ |
| REL (Recommended) | 1.5 mg/m³ |
| IDLH (Immediate danger) | 50 mg/m3 |
| Related compounds | |
| Related compounds |
2,4,6-Trinitrotoluene Trinitrobenzene 2,4,6-Trinitro-m-xylene Tetryl Nitroxylene |

