2,4,6-Trinitrotoluene: An Explosive Legacy, Real-World Challenges, and Next Steps
Tracing the Past to Understand the Present
Every time I hear about 2,4,6-Trinitrotoluene, or TNT as most people know it, I remember how a single molecule can change history. Factories in the late 19th century churned out materials like dynamite and smokeless powders by trial, error, and more than a few disasters. TNT, found by Joseph Wilbrand in 1863, seemed almost trivial at first because it wouldn't explode easily in the ways black powder would. That slow acceptance gave it a reputation for stability—a blessing in war and a curse for what lingered afterward. Its true value became clear during the World Wars, where governments valued materials that could be transported and handled with less chance of an accidental blast. Now, we see headlines about unexploded munitions popping up in farm fields all over Europe, the UK, Asia, and anywhere conflict pressed forward. TNT’s history isn’t a distant storybook—it’s the reason some land is still off-limits or carries risk, decades later.
The Stuff Itself: A Look at TNT’s Makeup and Properties
TNT stands out in my mind because of the yellow crystals, bitter almonds smell, and stories about mutations in factory workers’ skin color. Chemically, it’s just a mix of nitro groups added to a toluene backbone—pretty routine in organic labs on paper, but the trouble comes from how stable and insensitive it is. Melting just under 81°C, it can fill any container, poured like syrup, but it won’t ignite unless pushed hard. This lets armies fill artillery shells en masse, store them for years, and fire them hundreds at a time. TNT’s chemical behavior—the fact that it resists moisture, sunlight, and shock—made it a staple in explosives, but it also means the material lingers once it gets out. As solvents dissolve it, wastewater flows out orange or pink, showing up in surface and groundwater wherever TNT has been handled or discarded. Looking back at physical and chemical properties, you quickly see how science’s strengths—safety and versatility—also create enormous waste and cleanup headaches.
Technical Tags: Labels Behind the Boom
Most people never see the technical tags that stick onto drums of TNT. These labels mean strict limits on purity, moisture content, and color—nobody wants a mixture where impurities cut the explosive power or generate unpredictable reactions. International transport relies on well-known hazard and UN numbers, but in practice, only trained specialists decode the difference between “castable” and “pressed” forms. Trying to even get near TNT in a working lab reminds you how tightly regulations have wrapped around the compound. Proper storage requires cool, dry spaces with locked doors, sometimes even special ventilation to keep dust from building up. There’s a reason most chemists never see freshly made TNT outside documentaries or defense labs: the standards for handling verge on the obsessive, and even a single gram lost or spilled triggers reports and cleanups worthy of a disaster movie.
Making TNT—Neither Trivial Nor Clean
I’ve listened to old-timers talk about production lines in the earlier half of the 20th century—places that reeked of nitric and sulfuric acid, with steaming vats and workers who sometimes got sick from the fumes. The method for making TNT starts simple: take toluene, treat it with mixed acid, and add more nitration stages under careful conditions. Triple nitration gives you 2,4,6-Trinitrotoluene. Industrial chemists discovered that temperature, ratios, and timing all matter—get any part wrong, and you get runaways or, worse, toxic byproducts like dinitrotoluene. Even with safety controls, legacy pollution follows. TNT’s resistance to water means it washes out in traces, but never really leaves a contaminated site without determined intervention. Factories all over the world left their fingerprints on rivers, soils, and aquifers, and today, this shapes how chemical synthesis gets handled. Limiting exposure and finding less hazardous routes for synthesis has become a pressing job for any chemical manufacturer with a sense of responsibility for both workers and the environment.
Chemical Pathways and How TNT Changes
On paper, TNT looks almost peaceful—an aromatic ring no different from the dyes or drugs I’ve seen in other fields. Its big transformation comes from the nitro groups, which drive everything from its color to its volatility. Expose it to sunlight, and you see rapid fading and chemical change. Slow hydrolysis breaks it down in water, but this is measured in years, not weeks. In soils, microbes nibble at the molecule, but usually leave toxic metabolites. Scientists keep searching for bacterial strains or environmental technologies that speed the decomposition to safer products, but it’s an uphill climb. In the meantime, slight chemical modification—reduction, oxidation, or coupling—produces dyes and intermediates. For now, the major reaction folks care about is the explosive one: mix it with a detonator, and you get a release of hot gases and fast expansion, tearing apart just about anything in its path.
A World of Names and Uses
I’ve heard it called trotyl in Russian, trilite in some mining records, and of course the familiar TNT across most of North America and Europe. These names might look different on paper, but anyone who has worked in demolition, defense, or even mining knows their meaning points to the same thing: a reliable, often dangerous tool for breaking rock, shaping battlefields, or clearing land. Sure, chemical texts list synonyms and CAS numbers, but outside the lab, TNT is still just TNT—a boom for those who use it right, and a lingering health hazard for those stuck cleaning up old stockpiles.
Staying Safe: Lessons Learned the Hard Way
Every industry working with TNT has accident stories. It took decades to learn that nitroaromatic compounds wreck bone marrow, cause anemia, and sometimes trigger nervous system effects with chronic exposure. I’ve read about the “yellow faces” of World War I munitions workers, where jaundice from chemical poisoning was so commonplace that it marked entire cities. Today, factory standards set air and skin exposure limits using everything from sealed rooms to protective suits, but even with new gear, accidents still happen. Most modern countries list TNT as a highly controlled substance, and store anything related to it in bunkers far from roads or water sources. Security and environmental audits happen regularly, and a single suspicious reading gets investigated by both regulatory bodies and company safety teams. This rigidity comes from a long history of explosions, chronic disease, and slow contamination that past generations lived through and modern ones still manage.
Putting TNT to Work—Across War, Peace, and Science
TNT is more than just an explosive. Sure, it shaped the face of major wars, but it also made large civil engineering projects possible—from blasting tunnels through rock to clearing land for roads and dams. I’ve met people who spent years working on pipeline projects, always following strict routines when handling charges, measuring chemical residues, and maintaining blast control logs. Lately, TNT’s role has shrunk. Mining engineers and demolition teams now prefer less toxic, more powerful alternatives like ANFO or clean-emulsion explosives. Yet, there’s no denying how deeply TNT is woven into the legacy of chemical engineering, geology, and military logistics. Chemical analysts still use traces of it as standards when studying soils for past contamination or even when searching for illegal munitions or terrorist stockpiles. Occasionally, artists and activists repurpose deactivated shells for installations or memorials—but even then, the shadow of what the explosive once did is hard to ignore.
Chasing Cleaner Processes and New Possibilities
Researchers face a tough job: break down a stubborn molecule that resists most efforts at quick cleanup, or build safer ways to get the same work done with less harm. Enzymes, advanced oxidation reactions, and clever bioreactors now spearhead projects around the globe, trying to cut environmental impact at old ordnance sites. Some labs focus on finding bacteria that turn TNT into carbon dioxide and harmless minerals—no easy task since nature evolved around breaking down simpler plant chemicals, not tailored explosives. Environmental engineering firms deploy permeable barriers, phytoremediation beds, and even constructed wetlands, hoping to pull the last traces of explosive residue from groundwater and soil. I’ve visited former munitions depots transformed into green spaces, but there’s always the caveat: technicians test the ground at regular intervals, and warning signs stand where contamination persists. On the synthetic side, researchers continue to hunt for alternatives that match—or exceed—TNT’s reliability without its persistent footprint. These efforts range from green explosives based on nitrogen-rich salts to hybrid molecules that break down more readily under sunlight or in wet conditions. Some show promise at lab scale, but scaling up to replace century-old production chains remains a steep climb.
Weighing the Risks: Toxicity, Health, and Clean-Up
The real sticking point for TNT comes down to what it leaves behind long after the blast. I remember reading about wildlife studies near old shooting ranges and army training centers—rabbits, deer, even fish show tumors and reproductive trouble tied to TNT residues. Lab studies back this up, repeatedly flagging cancer risk, immune suppression, and even nervous system changes from chronic low-level exposure. People living near contaminated sites share similar worries, often fighting for health screenings or soil remediation. Cleanup is slow, expensive, and heavily monitored. Direct contact still threatens the lives of workers, while legacy exposure in drinking water remains a challenge for rural communities. Chemical engineers and toxicologists work hand-in-hand with environmental regulators, pushing for stricter monitoring, innovative treatment systems, and tighter site controls. As these collaborations continue, both costs and standards keep rising, but the stakes for public health demand nothing less.
Looking Forward—What Comes After TNT?
TNT’s era ran from the industrial revolution through modern times, but its future looks increasingly challenged by environmental science and regulatory pressure. I watch with interest as universities and defense agencies collaborate on new explosive materials that detonate just as reliably, but break down without the same toxic residue. Some groups focus on energy-rich compounds that pack more power per gram, cutting logistics waste, while others embed “self-degrading” switches that activate after use. Biodegradable explosives seem far-fetched, but history suggests that, with enough trial and error, innovation will catch up. Meanwhile, remediation scientists work to turn back the clock, restoring land and water to safe use. These next chapters demand a mix of stubborn optimism, honest science, and public accountability. Every batch of TNT that gets replaced, every contaminated field that recovers, and every advance in alternative chemistry serves as a quiet marker in the ongoing effort to balance human progress with safety and environmental well-being.
What is 2,4,6-Trinitrotoluene (TNT) used for?
From Battlefield to Quarry: TNT’s Many Roles
Few compounds have shaped the world’s industrial and military landscape like TNT. Chemists first made TNT back in the 19th century, but the world took real notice during the major wars that followed. The reason is simple: it works. TNT explodes only under the right conditions, so people could store, transport, and use it without fearing an accident from a stray jolt or spark. This stability offers a sense of control in work that often feels unpredictable.
In war, TNT changed the game. Factories churned out shells and bombs by the millions, lining artillery and aerial payloads with this yellow powder. Soldiers counted on its reliable performance. There was always risk on the battlefield, but few wanted to imagine packing their grenades and mortars with something less dependable. Over a hundred years later, defense industries worldwide still use TNT as the backbone in munitions. Not because it’s new, but because it gets the job done with less drama than other options.
Blasting Away in the Civilian World
Military use tends to grab headlines, but TNT’s reach stretches into peacetime too. Construction and mining jobs depend on controlled blasts to shift rock and earth. Civil engineers employ TNT to carve out tunnels, clear rockfalls, and even demolish old buildings. Good blasting matters for worker safety and project cost, and TNT allows for precise measurements. Workers can calculate just how much explosive force to use, and TNT responds in a predictable way. I’ve heard from mining professionals who track every ounce of explosive because costs add up, and they can’t afford mistakes—or surprises.
Safety Concerns and Environmental Impact
No discussion of TNT skips the issue of health and safety. Production and handling bring real risks. Yellow-stained hands once marked workers in wartime factories, sometimes signaling health problems like anemia. Factories these days use strict protective controls: gloves, respirators, and better venting systems, to limit toxic exposure. Worker safety has vastly improved, but environmental fallout lingers at old sites. TNT seeps into soil and groundwater, leaving toxic residues that persist for decades. In some communities, old military ranges have sidelined development projects or forced big cleanup efforts.
Alternatives and What Comes Next
People debate the future of TNT a lot. The call to cut pollution and improve worker safety drives innovation. Some military labs have mixed TNT with less-sensitive compounds to reduce blast risks. Others look to “greener” options—explosives that break down faster in the environment or carry fewer toxic byproducts. These newer choices face hurdles: cost, supply, and plain old habit. It’s tough to push aside something that’s worked for generations. Improvements keep coming, but TNT’s reliability keeps it in regular use.
Looking forward, safe storage, responsible cleanup, and ongoing research remain crucial. The world doesn’t turn on a single chemical, but the story of TNT carries lessons worth remembering: even powerful tools deserve respect, and every solution brings new questions. People in the field—miners, soldiers, engineers—count on that kind of balance every day.
What are the health risks associated with handling TNT?
Exposure: Skin, Lungs, and a Yellow Glow
Handling TNT isn’t just a matter of keeping away from fireworks or military-grade explosives. Production workers, demolition crews, and even some miners face risks just from touching or inhaling the stuff. I still remember hearing stories from factory workers in older towns whose skin would turn an unmistakable yellow after a shift. Turns out, prolonged TNT contact does that, staining hands and even hair. The yellow isn’t just a warning sign—it signals toxins absorbing into skin, moving through the bloodstream, messing with your organs.
Research in the International Journal of Occupational Medicine backs up what those workers always suspected. Skin absorbs TNT fast. Many workers describe headaches, dizziness, skin rashes, and even a strange sweet taste in the mouth. All these signals hint at a toxic process deeper in the body.
How TNT Impacts Blood and Organs
The most pressing risk comes from changes to red blood cells. TNT breaks them down, leaving the body low on oxygen. Doctors call this condition “hemolytic anemia.” Long-term cases show up with weakness, fatigue, and the kind of breathlessness that feels like you’ve run a mile. It doesn’t stop there. Some exposed workers end up with enlarged livers, because TNT puts the liver under constant stress processing the chemicals out.
Professional bodies like NIOSH flag the kidneys, too. TNT increases protein and blood in workers’ urine, often a signal of kidney stress or damage. If TNT builds up, organs just can’t keep up with the cleanup. That damage sometimes lasts years after leaving the job.
TNT and Cancer: A Brewing Concern
There’s a bigger worry lurking beneath the surface. Stories and data from munitions factories show a link between working with TNT and higher cancer rates, especially bladder cancer. Medical studies on wartime workers pointed out increased tumors decades later. Bladder cancer in TNT workers appears more often than in the average population. Oncologists explain this happens because breakdown products of TNT pass through the urine. The constant exposure leaves tissues more vulnerable.
Sneaky Routes: Dust, Vapors, and Water
It's not just the direct stuff on the hands. Dust and vapors escape into the air during handling and production. Breathing in these chemicals adds another layer of risk—damaging not just lungs, but also the central nervous system. Sometimes, even the water supply pays the price near poorly managed storage or waste sites, putting whole communities at risk of long-term health effects.
How to Protect Workers and Communities
I learned from safety officers in explosive plants that simple steps go a long way. Wearing gear that covers skin, such as gloves and uniforms changed daily, cuts down skin absorption. Modern plants set up showers and require workers to rinse off before heading home, stopping toxins at the door. Ventilation systems pull dust and vapor out of the air, making a noticeable difference to those inside.
Strict medical checkups catch problems early. Regular blood and urine tests let doctors spot signs of organ stress before things spiral. That early warning can give workers a chance to change roles or get extra care. At the policy level, stopping TNT pollution from reaching groundwater means better waste management and real oversight, not just paperwork. Lessons from the past show why these changes matter to the people actually doing the hard work, and for the neighborhoods nearby.
How should 2,4,6-Trinitrotoluene be safely stored and disposed of?
Why TNT Demands Respect
2,4,6-Trinitrotoluene, or TNT, rarely brings to mind images of dusty old chemistry textbooks. People hear the name and picture explosions in action movies. But in real life, this chemical means business. TNT's stubborn stability lets it sit unlit for decades, but the moment conditions tip, things can go bad fast. At temperatures above 240°C, TNT becomes unpredictable, and friction or static charge poses its own hidden dangers. I once toured a military chemical disposal facility, and the workers spoke about cleaning every tool and surface as if one speck could ruin their day—or several lives.
Storage: Playing by the Rules
I’ve watched teams in explosives warehouses and it’s clear: no corner-cutting happens there. TNT stays in solid form, sealed tight within non-reactive containers, usually lined with polythene or glass. Wood might look old-fashioned, but it helps prevent static buildup. The buildings carrying these containers have thick walls, steel doors, and excellent ventilation. Temperature ripples get tracked obsessively—climate control never gets left to chance. Ignoring this part turns warehouses into time bombs, especially in hotter climates where summers send mercury soaring.
No one wants stacked explosives tumbling onto the floor, so boxes and drums remain low to the ground, spaced out, and never jostled. Those storage rooms keep out sunlight and open flames. Smoking anywhere close draws a sharp rebuke. I remember my guide pointing out every fire extinguisher and sand bucket in the room—no one expects them to stop a big blast, but everyone needs those safeguards anyway.
Safe Disposal: More Than Digging a Hole
TNT doesn’t just disappear once it reaches the end of its useful life. Burying it in the backyard or dumping it in a landfill shoves the problem downstream. Nitrotoluenes linger in soil and leach into water, harming living things for generations. My years of environmental reporting showed me the mess blown-open ordnance can cause: poisoned rivers, sick wildlife, and cancer hotspots where kids should play.
The best way to dispose of surplus or waste TNT is controlled incineration at high temperatures, which breaks the molecules into harmless gases. This only happens in state-of-the-art facilities, where exhaust scrubbing removes lingering toxic gases like nitrogen oxides. Open burning or simple detonation sometimes gets used in emergencies or remote areas, but risk piles on with every shortcut. Explosion pits must be far from homes or crops, and wind direction gets checked daily. Every operator stays ready for unexpected wind shifts or stray embers.
Reducing Hazards: Training and Vigilance
No rulebook replaces steady hands and sharp minds. Everyone in contact with TNT needs real training. That means learning how to spot signs of aging or sweating (when crystals form on the surface)—a clear warning that something’s off. Workers suit up in anti-static clothing and avoid using metal tools. Regular inspections happen at strict intervals, and any spill sparks a lockdown until every grain is gathered and neutralized.
Moving Toward Better Solutions
Safer substitutes are coming, but for now, complete respect for TNT's risks saves lives. More sensors, stricter logging of movement and use, and robust emergency plans reduce accidents. I’ve seen firsthand what care looks like—a fire marshal with a checklist, a chemist teaching the next shift, two workers chatting about where one crate should go. Respect for this substance isn't just a workplace rule; it’s a lifeline everyone relies on, from the storage bunker to the ecosystems just outside the fence.
What are the chemical properties of 2,4,6-Trinitrotoluene?
What Gives TNT Its Edge?
2,4,6-Trinitrotoluene, known to most as TNT, owes its reputation to its remarkable chemical structure. It packs three nitro groups onto a single toluene ring, which cranks up both its potential energy and its stability against random shocks. The nitro groups, each with an eager nitrogen and oxygen team-up, pull electrons from the ring, making the compound less likely to react with stray sparks or static charges. This keeps the substance stable while sitting on a shelf or being shipped across the world, yet ready to release a punch when triggered the right way.
Why Stability Matters in Explosives
A big part of TNT’s fame comes from the way it blends power and safety. Many explosives either blow up too easily or need special handling to set off. With TNT, the molecule stays put until a real detonation source arrives—a strong shock or a reliable fuse. Decent stability lets engineers load it into shells or blocks without constant worry. On top of that, TNT melts at just over 80 degrees Celsius but does not burn unless someone seriously tries. This means people can melt it down and pour it into molds for making military ordnance, which keeps manufacturing steady and consistent.
TNT’s Reactivity Isn’t Like Household Chemicals
Handling TNT gets complicated because of its unique chemistry. Something as simple as a sparkler can't set it off. Unlike substances like nitroglycerin, TNT enjoys great shelf life. Its structure shrugs off most accidental bumps, but if enough energy is delivered—through a detonator, for example—it crumbles fast, smashing its molecular bonds and releasing a cloud of hot expanding gas. That rapid expansion, multiplying in a flash, causes the type of blast used in controlled demolitions or military operations.
Environmental and Health Concerns
TNT’s chemical properties lead to problems far outside the laboratory. It doesn’t dissolve easily in water but doesn’t stay locked in place, so it seeps through soil and groundwater in areas around old munitions factories or test sites. Workers who breathe in TNT dust sometimes develop skin or liver issues. Breaking down TNT in contaminated soil takes patience and the right bacteria—these bugs munch on its breakdown products, but some byproducts stick around and can be much harder to clean. Once I visited a decommissioned military site and spoke to scientists monitoring the cleanup; they walked us through rows of soil-treating bioreactors and kept layers of records about lingering chemical residue.
What Can Be Done About Clean-Up?
There’s no easy fix for TNT pollution, but creative solutions keep coming. Chemists research new ways to trigger safer breakdown, exploring microbes that munch faster or using sunlight to push the molecules apart with less secondary pollution. Tight regulation on waste and storage in modern plants has helped. I met folks volunteering with local water projects; they talked about how capping old waste sites and filtering runoff lets communities slowly heal the scars of past use. These actions don’t wipe out every trace fast, but step by step, science and steady work can chip away at what years of munitions have left behind.
Final Thoughts on Chemical Properties and Impact
TNT’s chemical properties made it the powder of choice for generations. Stable in the right hands, hazardous if mismanaged, it leaves a legacy that stretches from wartime factories to environmental cleanup teams still at work today. Efforts by scientists and engineers remind us that people can innovate better paths forward when they study both the chemistry and the consequences.
Is TNT explosive, and how is it typically detonated?
Everyone Knows the Name, Not the Whole Story
Growing up, I thought TNT was the stuff of cartoons and blockbuster movies. Characters tossed it around or pressed cartoon plungers, and explosions followed like clockwork. Once I actually learned about it, TNT seemed less about cartoon flash and more about science—and why people trust it when things really get serious.
So, Yes, TNT Explodes—but Not Like You Think
TNT (trinitrotoluene) is indeed explosive. Curiously, you can drop, bump, or burn it without blowing yourself up. I remember watching a video of someone hitting a TNT block with a hammer—nothing happened. That isn’t magic; it’s chemistry. TNT doesn’t explode in a snap from heat or shock alone. For all its reputation, it actually keeps a pretty cool head under everyday pressure.
The real danger starts once you give TNT a hard enough shock—something strong enough to jumpstart the molecules into a rapid breakdown. This creates a chain reaction: gases form super-fast, expand in a blink, and you have a blast. The fact that TNT needs this strong jolt makes it safer for transport than some other explosives out there. Soldiers have carried blocks of it on their backs without issue—a fact that probably saves lives in war zones.
How Detonation Actually Works
People often imagine big red plungers or long lines of cord with sparks dancing along. In real life, TNT rarely blows up from a simple flame or match. Demolition experts and miners use a smaller, more sensitive explosive—called a detonator or blasting cap—to set off TNT. Think of the blasting cap as a lighter for a stubborn campfire; it provides just the right punch. Once set off by an electric current or even a fuse, the cap makes a shockwave that ignites the TNT block, unleashing its stored energy in one massive release.
This two-stage process isn’t just for the movies. It saves lives across battlefields, mine sites, and in old construction jobs. A single loose spark doesn’t wipe out a crew. Handling still matters—no one gets casual with any explosive—but TNT’s “coolness” under stress has made it popular since World War I.
Risks Beyond the Blast
Many forget that the aftermath of TNT use lingers beyond the explosion. Soldiers who handled it too long sometimes ended up with skin problems from repeated exposure. Manufacturing plants have left waste behind, posing health risks for nearby people years after production stops. Cleanup teams wrestle with old TNT-laden shells or forgotten storage sites all over the world.
What Makes Good Practice?
Miners, military engineers, and project managers need strict rules. Secure storage, careful tracking of even the tiniest bits, and proper training all add a layer of safety. I've seen how modern crews run through checklist after checklist—sometimes it feels excessive, until you remember the stakes. Regular training drills, even for seasoned teams, drive home just how crucial it is to respect the material. Leaving nothing to chance means fewer tragic headlines and safer communities, now and long after the ground stops shaking.
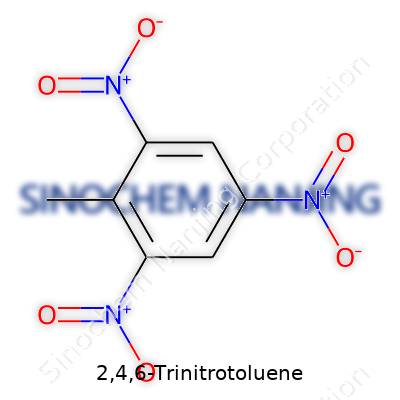
| Names | |
| Preferred IUPAC name | 2-methyl-1,3,5-trinitrobenzene |
| Other names |
TNT Trotyl Trinitrotoluol 2-Methyl-1,3,5-trinitrobenzene Trinitrotoluen Tritol |
| Pronunciation | /ˌtraɪˌnaɪtrəʊˈtɒljuiːn/ |
| Identifiers | |
| CAS Number | TNT" (2,4,6-Trinitrotoluene) has the CAS Number: "118-96-7 |
| Beilstein Reference | 1209234 |
| ChEBI | CHEBI:8615 |
| ChEMBL | CHEMBL1209077 |
| ChemSpider | 8619 |
| DrugBank | DB11625 |
| ECHA InfoCard | 100.003.223 |
| EC Number | 206-147-9 |
| Gmelin Reference | Gmelin 836 |
| KEGG | C02932 |
| MeSH | D014273 |
| PubChem CID | 5936 |
| RTECS number | XN7875000 |
| UNII | AG7JFD7F76 |
| UN number | UN0209 |
| CompTox Dashboard (EPA) | DTXSID2020150 |
| Properties | |
| Chemical formula | C7H5N3O6 |
| Molar mass | 227.13 g/mol |
| Appearance | Pale yellow solid |
| Odor | Odorless |
| Density | 1.65 g/cm³ |
| Solubility in water | 1.36×10⁻⁴ g/100 mL (at 20 °C) |
| log P | 1.60 |
| Vapor pressure | 1 mmHg (at 80 °C) |
| Acidity (pKa) | pKa = -0.29 |
| Basicity (pKb) | pKb = 13.81 |
| Magnetic susceptibility (χ) | -0.000076 |
| Refractive index (nD) | 1.546 |
| Viscosity | 25.3 mPa·s (80 °C) |
| Dipole moment | 2.98 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 302.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −67 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3406 kJ·mol⁻¹ |
| Hazards | |
| Main hazards | Explosive; fire and blast hazard; toxic by inhalation, ingestion, or skin absorption; may cause skin irritation and allergic reactions. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS01,GHS02,GHS03,GHS06 |
| Signal word | Danger |
| Hazard statements | H201, H278, H302, H332, H351, H373 |
| Precautionary statements | P210, P261, P273, P280, P370+P378, P401, P501 |
| NFPA 704 (fire diamond) | 3-4-2-W |
| Flash point | 82 °C (180 °F; 355 K) |
| Autoignition temperature | 303 °C |
| Explosive limits | Explosive limits: 0.9–1.5% |
| Lethal dose or concentration | Lethal dose or concentration: LD50 oral (rat): 795 mg/kg |
| LD50 (median dose) | 775 mg/kg |
| NIOSH | TT2975000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of 2,4,6-Trinitrotoluene: "1.5 mg/m³ (as TNT) |
| REL (Recommended) | 0.1 mg/m3 |
| IDLH (Immediate danger) | 100 mg/m³ |
| Related compounds | |
| Related compounds |
TNT Dinitrotoluene Nitrotoluene Trinitrobenzene Trinitrophenol (Picric acid) Tetryl Amatol Trinitroanisole |

