2,4,6-Trinitroresorcinol: A Look at Its History, Chemistry, and Role in Industry
Historical Development
2,4,6-Trinitroresorcinol, often called styphnic acid, tells an intriguing story that reaches back over a century. Chemists discovered its explosive capabilities in the late 1800s, at a time when the world sought new compounds to fuel military advancements and deepen scientific understanding. Through the world wars and into modern laboratories, researchers learned how to produce this compound with greater precision and purity. Knowledge about its hazards grew along the way, shaping tough safety rules and technical expectations. This compound once held key roles beyond weaponry as well, especially in the development of pyrotechnics and specialized chemical reagents.
Product Overview
Styphnic acid draws attention mainly because of its explosive properties and colorful appearance. Its intense yellow crystals pack a punch, not only due to their chemistry but also because of the responsibilities they demand in storage and handling. Scientists and engineers often turn to this compound when looking for a material with reliable detonation energy or when conducting specific chemical syntheses. Often used in initiation devices and as a component in certain primers, it found its way into products only after decades of cautious research and defect prevention.
Physical & Chemical Properties
Styphnic acid presents as bright yellow crystalline solid, notable for its high density and sensitivity to impact and friction. It dissolves a little in water but shows more solubility in organic solvents like acetone or ethanol. This compound contains hydroxyl and nitro groups on a benzene backbone, which together create its reactive profile. Its physical nature means it sometimes clumps or bridges, especially under less-than-ideal moisture control. Those yellow crystals signify danger but also hint at the underlying energy locked within. Chemical stability varies depending on impurities or age, which drives strict quality checks and process monitoring.
Technical Specifications & Labeling
Manufacturers shape their batch processes around achieving high-purity material, typically above 98 percent by weight for research and industrial applications. Strict batch characterization, including melting point determination and moisture analysis, forms the basis of most technical standards. Labeling requires sharp focus, both for communication and regulation—hazard pictograms, detailed handling instructions, and clear signal words always appear on containers. Regulatory agencies enforce specific classifications because even small mistakes may cascade into major hazards. These specifications aren’t just paperwork; they form the cornerstone of safe movement from manufacturing sites to end users.
Preparation Method
Making styphnic acid usually starts with resorcinol and involves nitration with concentrated nitric acid. Controlling temperature and reaction time becomes essential during synthesis to optimize yields and prevent runaway reactions. Seasoned chemists can tell stories of how even subtle process tweaks can make or break a batch; years of experience and careful scale-up work proved the difference between safe production and disaster. The process also generates significant acidic byproducts, demanding robust waste neutralization that adds to operational costs but keeps environmental worries in check.
Chemical Reactions & Modifications
2,4,6-Trinitroresorcinol doesn’t just stop at being an explosive. Researchers use it as a building block for further chemical exploration. Reduction, alkylation, and metal-salt formation are common secondary reactions, enabling the production of compounds with tailored detonative or photochemical properties. These modifications demand precise parameter control and deep understanding of mechanism and reactivity. Laboratories take extra care during these reactions since side products can sometimes eclipse the original compound in terms of hazard or unpredictability. For example, the formation of styphnates—metal salts of this acid—permits fine-tuning of sensitivity and ignition temperature, serving roles in advanced primer formulations.
Synonyms & Product Names
Across trade literature and regulatory documents, 2,4,6-trinitroresorcinol carries various aliases. Styphnic acid stands as the most common label, but you might also see tri-nitroresorcinol, or simply TNR. Some old texts reference its forms as yellow acid or evocative names tied to its historical use in certain detonators or industrial processes. This web of names can confuse newcomers, so standardized reporting and chemical indexing play vital roles in chemical safety and commerce.
Safety & Operational Standards
Handling styphnic acid comes with little room for error. I have seen how operators at chemical plants rely fully on training, discipline, and well-maintained gear to work with this material. Even a lightly jarred flask or uncontrolled static discharge may turn an ordinary workday into a crisis. Robust local ventilation, heavy personal protective equipment, and antistatic precautions turn into best friends for anyone operating near this compound. Regulatory authorities take a no-nonsense approach, supporting frequent inspections and records review. Storage apart from incompatible substances and away from sources of ignition or vibration forms the bedrock of operational safety. Working with compounds like styphnic acid gives one a lasting respect for chemistry’s power and its consequences.
Application Area
The reach of styphnic acid spans much farther than explosives alone. While it helped shape old-school military primers and blasting caps, refinements in its chemistry gave rise to dependable ignition compounds and tailored photochemical reagents. Some laboratories use it as a colorimetric indicator in analytical testing because of its signature color and reactivity toward certain ions. Artisans in pyrotechnics appreciate the unique hues it brings to flame tests and colored smokes. Seeking out new ways to harness its potential draws in researchers from fields as varied as materials science, forensics, and even environmental testing, where detection sensitivity sometimes relies on compounds just like this.
Research & Development
Innovation in trinitroresorcinol chemistry focuses on safer processing and expanded applications. Teams around the globe study ways to cut down on waste in production and look for greener reagents without sacrificing purity or performance. Analytical chemists keep improving ways to test trace impurities and accidental isomers, making detection cheaper and more reliable. Military and industrial customers trigger big investments in formulations that reduce sensitivity or environmentally hazardous fallout. Talent flows into both bench research and practical engineering, showing how an old compound can still prompt a new generation of discoveries, particularly in formulating less sensitive but still reliable explosives and markers.
Toxicity Research
Debates about toxicity continue to surround 2,4,6-trinitroresorcinol. Its harmful nature stretches well beyond the risk of explosive detonation. Animal studies and case reports flag issues such as skin and eye irritation, organ toxicity, and possible carcinogenic or mutagenic effects with prolonged contact. Experts stress careful monitoring in workplaces, paired with medical surveillance and air quality testing, to clamp down on chronic exposure risks. Conventional wisdom tells us to treat any exposure as dangerous, reinforcing the use of tight containment and punctual cleanup. Decades of toxicological research taught industrial users to never take shortcuts on personal protective gear or spill protocols.
Future Prospects
Looking ahead, styphnic acid stands at a crossroads. On one hand, its energy-rich profile gives it persistent value in highly technical applications, both in the lab and in the field. On the other, growth in environmental regulation and the pursuit of less hazardous materials nudges researchers to imagine cleaner and safer alternatives. Advances in computational chemistry and synthetic methodology may reveal new uses for the nitroaromatic backbone that avoid the pitfalls of historical applications. Young chemists entering the workforce today tackle questions their predecessors never imagined, blending tradition with new priorities such as sustainability and green engineering. The journey from battlefield to laboratory bench continues, with 2,4,6-trinitroresorcinol as both lesson and inspiration in the evolving world of chemical science.
What is 2,4,6-Trinitroresorcinol used for?
From Laboratory Curiosity to Key Ingredient
In my days around scientific circles, 2,4,6-Trinitroresorcinol, more frequently called styphnic acid, often cropped up in technical discussions about military and industrial chemistry. This yellow, crystalline compound carries a stigma – folks hear “nitro” and immediately think explosives. In truth, its story stretches beyond Hollywood-style pyrotechnics, digging into real-world problems for both defense researchers and anyone who cares about safety and progress.
Why It Matters in Energetic Materials
Styphnic acid’s biggest claim to fame comes from its role in explosive compounds. Owing to its multiple nitro groups, it carries a punch, but engineers and chemists picked it not just for power, but for reliable ignition properties. It plays a part in the mixture for priming explosives, specifically in primers used in munitions. Police and military organizations have counted on dependable, stable primer mixes for well over a century. Few ordinary folks think about what initiates a cartridge in service pistols or keeps safety fuses predictable — this is exactly where styphnic acid makes a difference.
A key chemistry fact: styphnic acid forms an essential component in lead styphnate. Lead styphnate serves as a go-to primer in cartridge ammunition, replacing older, less stable compounds that often misfired or degraded quickly. While environmental and health concerns about lead remain strong, it still offers unmatched characteristics for many armed forces around the globe.
Beyond Explosives: Laboratory Science and Testing
I’ve come across this compound in labs far removed from weapons research. Analytic chemists regard styphnic acid as a benchmark — its sharp color changes prove handy during spectroscopic analysis. It’s a trusted standard, offering reference points for calibrating sensitive equipment. Still, this remains a niche avenue. Most chemical supply houses stock the stuff for researchers rather than factories.
Safety, Health, and Environmental Costs
Working with styphnic acid never felt casual. The compound stands out for its sensitivity, requiring solid knowledge and precaution. Handling it sloppily spells disaster — even small static charges or friction can trigger accidents. Most chemistry departments contain very strict protocols and training sessions before anyone can even touch a container. Routine use comes with gloves, safety goggles, and robust ventilation.
Its manufacture and disposal bring up big environmental headaches. Lead styphnate in particular leaves toxic residues. Extended exposure can lead to health problems for workers, not to mention the long-term buildup of lead in soil and water when ammunition decomposes. These toxic byproducts sparked serious debates over time. Cleaner alternatives, like lead-free primers, keep popping up as potential replacements, but lead styphnate’s reliability makes it hard to retire overnight.
Looking Ahead: Innovation vs. Tradition
Calls for environmental safety and worker health have pushed the search for greener, less toxic substitutes. Research groups around the world experiment with potassium-based and organic compound primers. They’re aiming for consistency and power without poison. For now, traditions hold strong, especially where reliability trumps environmental risks. Enduring change takes both regulatory pressure and scientific breakthroughs — neither one rushes.
The value of 2,4,6-Trinitroresorcinol lies in its proven dependability. There’s a reason armies and industries stuck with it so long. Anyone championing a safer world faces the tough job of innovating, convincing, and demonstrating. That battle continues both in the lab and out on the firing range.
Is 2,4,6-Trinitroresorcinol explosive or hazardous?
Getting to Know the Chemical
2,4,6-Trinitroresorcinol, also called styphnic acid, isn’t something that pops up on the average shopping list. Chemists recognize it for a couple of reasons: its knack for giving off a dazzling yellow color and its reputation for packing a punch—literally. If you’ve worked in a lab or ever handled chemicals meant for specialized tests, you might remember the peculiar tang in the air and how safety protocol gets strict around any nitroaromatic compounds. Styphnic acid lands right in that category, sitting beside more notorious names like TNT, but with its own traits and dangers.
The Explosive Fact
There’s no getting around the truth: 2,4,6-Trinitroresorcinol is explosive. Anyone calling it otherwise is sidestepping decades of accident reports and safety bulletins. Military engineers and pyrotechnicians learned this the hard way. The compound’s solid, crystalline form packs a wallop. Even a slight mishap—like static from clothing or a jolt from a dropped tool—can trigger a reaction. During the height of the twentieth century, ammunition makers blended it into primer mixtures for cartridges and mines, often as a replacement for mercury-based explosives because of toxicity issues there.
People sometimes downplay lab-scale risks. One leftover spatula, not cleaned well after measuring out a small pile of trinitroresorcinol, can quickly become a lesson in complacency. Chemical catalogues stamp a glaring “explosive” warning on every bottle for good reason. These aren’t empty warnings. In places where safety standards get ignored, injuries follow—maybe from shrapnel, more often from burns and sudden noise trauma. Groups who maintain bomb disposal units train staff to recognize this chemical by sight and handle it with the same respect as dynamite or picric acid.
Health Risks
The explosion risk isn’t where the story ends. Overexposure in a poorly ventilated space brings up headaches, skin irritation, and even symptoms affecting bone marrow after chronic contact. Some studies with workers showed problems with their hemoglobin. The bright yellow stain stuck to their fingers—hard to scrub away—served as a constant reminder that even small errors could stick with you longer than expected.
Smart Solutions
Education goes a long way. Chemistry programs and industrial labs should keep training up-to-date, not just during orientation but as a regular thing. People forget, especially when new students or junior staff try to impress by working fast. Clear rules—like not working alone, avoiding metal tools, and never letting powder accumulate in open containers—stop real accidents from happening. Proper ventilation, emergency eye washes, and explosion-proof storage keep the biggest risks at bay.
On a larger scale, shifting away from compounds like 2,4,6-Trinitroresorcinol makes sense when safer materials pop up on the horizon. Some modern detonators rely on less sensitive substances, while others use engineering controls to stop accidental initiation. Regulatory agencies can speed that trend along by supporting research and helping companies transition.
The bottom line here: you can’t ignore what’s sitting in the bottle. This chemical has history, both as a tool and a hazard. Treating it with the caution it demands keeps people safe, whether you’re troubleshooting ammunition or just teaching in a university lab.
What are the storage requirements for 2,4,6-Trinitroresorcinol?
Handle With Serious Respect
Dealing with energetic chemicals takes more than reading a few lines from a safety data sheet. Anybody who’s spent time around 2,4,6-Trinitroresorcinol, better known as styphnic acid, knows the material can surprise even the careful. This stuff helped propel military shells for a reason — it’s powerful, sensitive, and unforgiving about sloppy practice.
Keep it Cool, Keep it Dry
Heat has a knack for waking up energetic compounds. It doesn’t take a scientist to see the sense in placing styphnic acid away from boilers, steam lines, sunlit windows, or rooms where summer means baking temperatures. Most facilities stick to a climate-controlled storage room under 25 degrees Celsius. Humidity also matters here. Damp conditions can mess with the dry crystals, encouraging slow decomposition or clumping. A dehumidifier and solid ventilation system give peace of mind.
Solid Containment and Clear Segregation
A thick steel cabinet with explosion venting isn’t overkill. I’ve watched containers sweat through their seams after a few months on a wooden shelf — the acid loves to creep and chew on soft packaging. Glass jars with PTFE-lined screw caps, or proper polyethylene bottles, avoid both accidental reactions and slow leaks. Put each container on its own spill tray. Spills, even tiny ones, have ended with entire labs clearing out.
Don’t Mix With Trouble
Experience has a loud voice: keep the oxidizers and fuels apart. Styphnic acid stores well on its own, far from anything like paper, solvents, strong reducing agents, or base chemicals. I’ve seen a black mark on brickwork from a bottle left too close to a cracked container of sodium azide — small disaster, big lesson. If possible, put a chemically resistant barrier or even a separate locked cabinet between acid and any possible reactants.
Access Isn’t a Free-for-All
Any lab with a culture of shared keys or unlocked chemical rooms always seems to pick up bad luck. Training shapes good habits: logging every gram in and out, making users sign off after handling, and running regular checks for leaks or pressure build-ups. Records matter most after someone finds residue on a glove or a shelf. Companies with strong track records always treat styphnic acid as a high-value target, only letting in folks who’ve learned how to respect its risks.
Returning to the Community and the Environment
Professional disposal costs money, but skimping only shifts risk onto someone else’s shoulders. Old, yellowing stockpile? Don’t try flushing it down a drain or burning it outside. Find a certified hazardous waste company and document the handoff. That’s not just about avoiding citations. Emergency crews remember the folks who made their jobs easier — and the ones who left them surprises.
Solid Storage Is Honest Safety
Every step in proper styphnic acid storage builds a buffer between trouble and daily operations. Safe storage means less chance of fire, injury, or unexpected shutdowns. Learning from mishaps — not just your own — shapes habits that shield people, equipment, and reputations. Good practice looks boring, but smart labs see boring as a badge of honor.
What are the physical and chemical properties of 2,4,6-Trinitroresorcinol?
A Hands-On Introduction
Folks in the lab often call 2,4,6-Trinitroresorcinol by another name—Styphnic acid. Once you handle it, you won’t forget. It’s one of those crystalline solids with a yellow cast, plenty dense, but deceivingly simple at a glance. The stuff doesn’t put on a big show until you dig a little deeper. Its real claim to fame comes from both its raw physical makeup and the way its nitro groups pack so tightly onto its structure.
Physical Properties: What You See, What You Get
The powder feels gritty between your fingers—think household salt but denser. Stuff weighs in pretty heavily at about 1.83 grams per cubic centimeter. Pour a little bit out, it doesn’t have much of a smell to speak of, but it will leave a trace on anything white thanks to its deep yellow color.
This compound melts at about 178 degrees Celsius. Go much hotter, and you’ll be dealing with decomposition long before you get a good liquid. That low melting point by energetic material standards sets off alarm bells for anyone thinking of applying heat. It shows just how unstable it can get.
Solubility paints another picture. Drop a scoop into water? You’ll see it dissolve somewhat, but not completely. It does better in hot water. In alcohol or ether, solubility doesn’t stand out, but ammonia soaks it up easily, creating intensely colored solutions.
Chemical Properties: Not a Compound You Ignore
Chemically, 2,4,6-Trinitroresorcinol stands out for its three nitro groups attached to an aromatic ring. That setup puts it squarely in the family of explosives. Those nitro groups don’t just sit quietly, either. They bring instability—eagerly releasing energy when kicked off by friction, heat, or shock.
You don’t want to mix this stuff with metals like lead or potassium, as it can form even more sensitive compounds called styphnates. That comes into play in primer manufacturing—styphnate salts have powered everything from percussion caps to munitions primers. The practical upshot is real: even a small quantity mixed wrong turns into a real hazard in the wrong hands.
Acids and bases treat this compound differently. Strong bases like sodium hydroxide break it down. Strong acids, on the other hand, don’t bother it much—one reason production relies on acidic conditions.
Safety, Environmental Impact, and Solutions
Nearly everyone who’s worked with this compound feels the stress of handling it. Breathing in dust can cause headaches and other issues. Drawing from my own time in research labs and from case studies, I always say gloves, mask, and minimal exposure time are non-negotiable. Improper storage brought trouble in more than one historical incident—the stuff responds poorly to impact and static just as fast as fire.
The hazards reach beyond just the lab. Disposal remains a concern. Leftover trinitroresorcinol contaminates soil and water. Microbial remediation offers a path forward, with some bacteria showing promise in breaking it down safely. Cleaner manufacturing processes, strict containment, and updated waste treatment keep accidents out of the headlines and away from the environment.
Chemists and manufacturers have to respect the powerful nature of this material. Using alternative priming compounds with better safety records and making room for greener chemistry in research and industry can help lower risk and environmental load. Skill, training, and respect for those yellow crystals carry every bit as much weight as any procedure in the manual.
How should 2,4,6-Trinitroresorcinol be handled and disposed of safely?
Understanding the Substance
2,4,6-Trinitroresorcinol carries a reputation for its energetic punch. Explosives experts call it styphnic acid, but most people don’t come across it outside research, military, or specialized industries. Its power isn’t just on paper—styphnic acid is sensitive, reactive, and makes short work of safety shortcuts. My first encounter with energetic materials, years ago, drove home just how much respect these substances demand. You suit up, stay sharp, and remember every safety lesson your mentors drilled into you.
Keeping It Contained
Handling 2,4,6-Trinitroresorcinol shouldn’t ever feel casual. Storage calls for a cool, dry, well-ventilated spot with zero sparks or static. Storing it alongside incompatible chemicals, especially reducing agents and bases, adds unnecessary risk. Back in grad school, a friend learned this the scary way: one misplaced sample led to a small but unforgettable incident. Thankfully, proper protocols stopped that from going further.
Gloves and goggles aren’t negotiable. Dust from trinitroresorcinol burns, irritates, and sneaks into lungs if you’re not careful. Dust control matters—a slip can turn a calm workday into a crisis. Anyone near the compound should understand the properties and potential hazards. The importance of good training came up again and again during site visits; trained hands spot risks and react before a situation escalates.
Safe Use Means Vigilance
Tool choice matters. Forgetting non-sparking tools or letting static sneak up is an invitation for disaster. Years in chemical labs have drilled that in for me: wooden or plastic spatulas, antistatic surfaces, grounding straps for operators, and always an awareness of the charge building up from your own movement. It’s not about paranoia, it’s about keeping people and facilities intact.
Disposal: Responsibility Doesn’t Stop at Use
Some folks think disposal is simpler than handling the stuff. It’s not. Unused trinitroresorcinol can’t just go down a drain or get tossed with the rest. Deactivation stands as the best bet—often by slowly reducing the compound with sodium sulfite or another suitable reducing agent, under professional supervision. Watching an experienced chemist at work, they never rush, always double-check readings, pH, temperature. Any deviation, they stop. No heroics.
Incineration at licensed hazardous waste facilities provides another safe avenue. But not every facility accepts explosive-contaminated waste. Coordination with certified chemical disposal companies is essential. At every step, solid documentation keeps track of quantity, process, and endpoint. I’ve watched newcomers fumble the paperwork and the headaches that follow. Good logs help everyone down the line know what’s coming.
Training and Communication: Lifelines in the Workplace
Regular training and drills drive home both the stakes and the safe paths. Workers who speak up about deteriorating packaging, odd smells, or unexpected residues create a safer workplace. Safety culture grows through voice and shared vigilance, not just written protocol. I’ve seen plants with strong team communication catch near-misses before they become real emergencies.
Addressing Gaps: Where Change Still Matters
Access to strong disposal solutions means everyone involved—from researchers to hazardous waste carriers—needs ongoing technical support and updated training. Safety standards exist, but without enforcement, corners get cut. My time consulting for a midsize manufacturing shop showed me the holes first hand—lack of fresh supplies, expired gloves, training materials collecting dust. Management that invests in fresh gear and regular refreshers reduces accidents, plain and simple.
2,4,6-Trinitroresorcinol reminds us daily: don’t underestimate chemical power. Careful stewardship from purchase to final neutralization protects not just workers, but whole communities. In this field, responsibility never clocks out.
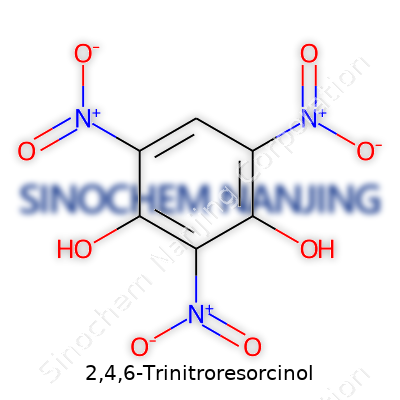
| Names | |
| Preferred IUPAC name | 2,4,6-trinitrobenzene-1,3-diol |
| Other names |
Styphnic acid Trinitropyroresorcinol 2,4,6-Trinitro-1,3-benzenediol |
| Pronunciation | /ˌtraɪˌnaɪtroʊrɪˈzɔːrsɪˌnɒl/ |
| Identifiers | |
| CAS Number | 82-71-3 |
| Beilstein Reference | 1908980 |
| ChEBI | CHEBI:75670 |
| ChEMBL | CHEMBL468326 |
| ChemSpider | 12033 |
| DrugBank | DB14024 |
| ECHA InfoCard | ECHA InfoCard: 100.002.718 |
| EC Number | 208-257-7 |
| Gmelin Reference | 66852 |
| KEGG | C18807 |
| MeSH | D014273 |
| PubChem CID | 6760 |
| RTECS number | UX9625000 |
| UNII | PY24YOD76K |
| UN number | UN0150 |
| CompTox Dashboard (EPA) | DTXSID2021003 |
| Properties | |
| Chemical formula | C6H3N3O8 |
| Molar mass | 272.11 g/mol |
| Appearance | Yellow crystalline powder |
| Odor | Odorless |
| Density | 1.76 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | -0.06 |
| Vapor pressure | 4.47E-7 mmHg at 25°C |
| Acidity (pKa) | 6.6 |
| Basicity (pKb) | 6.79 |
| Magnetic susceptibility (χ) | -51.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.678 |
| Viscosity | 1.14 cP (20°C) |
| Dipole moment | 3.27 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 200.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -181.0 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1398 kJ mol⁻¹ |
| Pharmacology | |
| ATC code | V03AB34 |
| Hazards | |
| Main hazards | Explosive; may cause fire or explosion; strong oxidizer; toxic if swallowed, inhaled, or absorbed through skin; causes irritation to skin, eyes, and respiratory tract. |
| GHS labelling | GHS02, GHS06, GHS03 |
| Pictograms | GHS01,GHS03,GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H201, H301, H311, H331, H373 |
| Precautionary statements | P210, P220, P221, P280, P305+P351+P338, P370+P378 |
| NFPA 704 (fire diamond) | 2,4,6-Trinitroresorcinol: 3-1-1 |
| Flash point | 180 °C |
| Autoignition temperature | > 300 °C |
| Lethal dose or concentration | LD50 oral rat 119 mg/kg |
| LD50 (median dose) | 132 mg/kg (rat, oral) |
| NIOSH | TT2300000 |
| PEL (Permissible) | PEL: Not established |
| REL (Recommended) | 0.1 mg/m³ |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
Trinitrophenol Trinitrobenzene Styphnic acid Resorcinol Nitroresorcinol Picric acid |

