Looking Closer at 2,4,6-Trinitrochlorobenzene: A Chemical Story Worth Telling
Historical Development
Many of us don’t give much thought to where certain industrial chemicals come from, but 2,4,6-Trinitrochlorobenzene (TNCB) has a story rooted in early 20th-century chemistry. Long before computers let us simulate molecular behavior, organic chemists relied on steady hands, trial runs, and plenty of patience. TNCB entered the scene as a derivative from the rich tradition of nitroaromatic compounds—chemicals built on the sturdy backbone of benzene with nitro and chlorine groups introduced through controlled reactions. The demand for such molecules ramped up during the age of dye chemistry and as explosives saw military and industrial uses in the mid-20th century. The recipe has barely shifted for decades, a testament to how effective those old methods have remained. When people talk about legacy chemicals, TNCB certainly fits the bill, tracing a path from early synthesis experiments into today’s specialty labs.
Product Overview
As a yellow crystalline powder with a sharp chemical odor, TNCB doesn’t try to hide its industrial pedigree. Its distinctive smell and striking coloration come from the three nitro groups and a single chlorine atom welded onto a benzene ring. Chemists I know recognize it for its punchy presence in the lab. The compound has emerged as a key intermediate for more complex molecules, performing duties that range from polymer stabilization to dye production. It rarely stands alone but usually serves as a building block, handed off to the next stage in a chain of chemical transformations that underpin countless products.
Physical & Chemical Properties
TNCB shows off its stability and reactivity in equal measure. It melts at temperatures that suggest robust bonds, but those nitro groups also flag significant explosiveness. Inexperienced hands often underestimate how suddenly this powder can react, particularly under friction or high heat. Its low solubility in water paired with high solubility in organic solvents might seem routine to an old hand, but this trait steers how chemists use it in syntheses, testing workflows, and engineering new derivatives. The dense nature of its crystals, sharp melting points, and serious eye and skin irritant profile set it apart from blander organic compounds that sit quietly on reagent shelves.
Technical Specifications & Labeling
Chemicals like TNCB force everyone to pay attention to accuracy. Contaminants or off-spec batches risk flaring into hazardous situations. Makers and handlers stress purity levels well over 97 percent for most applications, with moisture content and trace impurities closely monitored. Every lab container sports labels warning of toxic fumes, skin sensitization, and explosion risk, not simply as regulatory formality but as daily reminders to anyone reaching for the jar. Labels must be legible, resistant to chemicals, and list all hazards plainly because shortcuts in labeling can spell disaster—and I've seen more than one chemist burned by “assuming” what's in a bottle.
Preparation Method
The standard route starts with chlorobenzene that gets hit with a nitrating mixture—usually a blend of concentrated nitric and sulfuric acids—under carefully controlled temperatures. This stepwise process brings up memories of long days in lab courses hunched over ice baths, watching for runaway reactions. The three nitro groups don’t jump onto the ring all at once, so reaction conditions take some fine-tuning to avoid overnitration, tarry by-products, or even mini-explosions. Purification can be a slog, full of recrystallization and filtration cycles, with sharp odors hanging in the air long after the work wraps up.
Chemical Reactions & Modifications
What fascinates many researchers about TNCB is its ability to serve as a reactive launching point. Those nitro and chlorine groups practically invite chemical tinkering. Common modifications include reduction to amino groups for use in agrochemicals or further substitution reactions that lay the groundwork for advanced pharmaceutical ingredients. Every step opens a door to derivatives with new properties—think better colorfast dyes or more potent antimicrobial agents. TNCB's role as a reactive intermediate means it's rarely the end goal, but often the push a synthesis needs to cross the finish line. In the right hands, a bottle of TNCB turns into a chemical Swiss army knife.
Synonyms & Product Names
People in the field use different names for TNCB—sometimes it shows up as trichloronitrobenzene, other times as 1-chloro-2,4,6-trinitrobenzene. These names track the same core structure but hint at the culture of chemical shorthand. This variation crops up in academic papers, patents, and shipping manifests, making attention to detail a constant for those moving between regulatory filings and benchwork. Every chemist I know adds synonyms to their mental dictionary, since a missed translation could lead to mixing up materials or even ordering the wrong compound entirely.
Safety & Operational Standards
Handling TNCB means never cutting corners. Standard gloves, goggles, and fume hoods become minimum expectations, not optional upgrades. Regular air monitoring keeps vapor exposure in check, since inhaling nitroaromatics brings risks nobody wants to gamble on. Disposal protocols demand specialized training and hazardous waste channels, with zero tolerance for leaks or sloppy handling. The compound’s legacy as both an irritant and potential explosive etched a culture of vigilance into labs and factories alike. Experience has taught me that even well-drilled safety habits can slip, especially during late-night runs, so double checks and team communication form the backbone of safe operations.
Application Area
TNCB rarely advances to an end product on its own, but its fingerprints show up across several industries. The dye industry leans on TNCB for producing brilliant, colorfast molecules that hold up under light, washing, and time. In the world of polymer chemistry, it introduces new functional groups or serves as a cross-linker, improving thermal and chemical properties of plastics used in electronics and automotive parts. Some agricultural chemical makers employ it as a starting material for fungicides or pesticides. These uses aren’t just historical footnotes—they reflect persistent demand and evolving applications as new technologies call for specialty intermediates.
Research & Development
Academic and corporate labs both push the bounds of what TNCB can do. I’ve seen teams tinker with greener synthesis routes to cut back on waste or find catalysts that shift yields higher with less heat. Recent work explores using TNCB analogues in the hunt for high-energy materials, while others probe its utility in fabricating advanced pigments or electronic materials. Research doesn’t happen in a vacuum, and as regulatory scrutiny and the cost of waste disposal climb, innovation focuses on efficiency, safety, and environmental stewardship. The open questions today—around selective functionalization or novel reactivity—drive new lines of inquiry with every passing year.
Toxicity Research
TNCB isn’t something anyone would call benign. Studies show it triggers strong skin sensitization, serving as a benchmark compound in allergy research. Animal testing has uncovered connections between chronic exposure and health effects, providing fuel for strict occupational guidelines. Environmental toxicologists keep close tabs on its fate in waterways, since nitroaromatics tend to resist simple breakdown and threaten aquatic life. Industry and academia alike continue to probe long-term risks, while regulatory agencies hammer down on acceptable exposure levels, workplace controls, and storage rules. This isn’t abstract concern—real workers in real plants depend on up-to-date science for their well-being.
Future Prospects
Looking ahead, the future of TNCB sits at the intersection of caution and innovation. Green chemistry principles are pushing producers toward milder, less wasteful manufacturing methods. Improved analytics promise faster detection of exposures and contamination, both in labs and out in the world. On the innovation side, new derivatives keep surfacing, feeding the endless pipeline of specialty chemicals for electronics, imaging, and smart materials. The combination of regulatory tightening and technical advancement drives everyone—scientists, safety officers, and manufacturers alike—to rethink old habits and search for smarter pathways. The story of TNCB will keep evolving as long as chemists keep hunting for molecules that do more with less risk and waste.
What is 2,4,6-Trinitrochlorobenzene used for?
Chemical With a Purpose
A lot of people see a daunting chemical name and pull back, but 2,4,6-Trinitrochlorobenzene pulls more weight than most realize. This compound finds its most important use in making explosives. It does not do much by itself, yet serves as a major ingredient to build more complex chemicals for controlled detonations. In many military and mining applications, engineers depend on those chemicals to break hard rock or demolish old buildings safely.
Root Ingredient for Other Products
While chemists in the lab see a powder, manufacturers see something like a bridge. They often use it to make dyes and pigments, letting factories generate colors that last longer in tough conditions. Certain products use it to produce specialty rubbers and plastics, making gear less likely to crack under pressure or extreme cold. That’s not so different from building better tires for rough roads or plastic components that outlast their competition in industrial settings.
Risks That Demand Respect
Anyone who deals with explosives or hazardous chemicals can share stories about close calls. 2,4,6-Trinitrochlorobenzene brings risks you cannot ignore. It can irritate skin, eyes, and lungs. You absolutely do not want to eat, breathe, or spill it. The environmental side matters too—spills and runoff can harm plants, animals, even water supplies. Researchers have seen links between some nitro compounds and health problems, putting extra pressure on companies to handle this stuff the right way.
Why Oversight Means Safety
People sometimes complain about environmental rules, but many forget what happens if those rules disappear. Factories once dumped waste from nitro chemicals straight into the ground, poisoning the land and water. Thanks to stricter laws, today’s operations require sound storage and careful transportation. Trained handlers, tough containers, and regular inspections now stand between communities and disaster. If you’ve ever seen a chemical warning sign on a truck barreling down the highway, you know real steps keep everyone safer.
Working Toward Cleaner Alternatives
No one can erase those dangers with wishful thinking. Some scientists are trying to invent cleaner processes or swap out nitro compounds for greener options. University labs and government agencies are running tests on new materials. They’ve made steady progress on alternatives to diphenylamine or dinitrobenzenes, which sometimes start as 2,4,6-Trinitrochlorobenzene. Those substitutes work for some jobs, but not everything—especially when strength and reliability matter most.
Facing the Future Together
All this boils down to choices. We count on certain chemicals to make the modern world turn, from safety gear to bridges and tunnels carved by explosives. Nearly everyone benefits, but those benefits come with risks that demand constant attention. If society wants to limit harm, nothing beats tough rules and ongoing research. Asking questions and supporting science keeps everyone a little safer, from the lab worker to the family next door. Maybe the bigger lesson—always know what you’re working with, respect it, and refuse to look the other way.
What are the safety precautions when handling 2,4,6-Trinitrochlorobenzene?
Understanding the Risks
Working with 2,4,6-Trinitrochlorobenzene brings a set of real risks. This compound isn’t something you splash around in an open lab, thinking a fume hood will sort it all out. It’s an aromatic compound with three nitro groups and one chlorine atom. Those nitro groups ring alarm bells for chemists because they make the material energetic, meaning it reacts strongly if mishandled. Breathing in its dust or fumes leads to serious breathing problems, and even contact with skin is no joke—blisters, redness, and possible long-term effects if it sits on you too long.
Personal Protective Equipment: Suit Up, Don’t Skip Steps
For anyone handling trinitrochlorobenzene, skipping gloves or safety goggles isn’t just a lazy habit; it invites trouble. Regular latex gloves barely stand a chance, and those thin, blue surgical gloves don’t fare much better. I learned to use thick nitrile or neoprene gloves, which offer stronger protection against the harshness of nitroaromatic chemicals. Safety goggles should give full eye coverage, not just the front. A lab coat comes standard, but go for one with cuffs that close tight—loose fabric soaks up chemicals, spreading danger.
Ventilation: Fume Hoods Don't Mean Free-for-Alls
Some younger chemists figure a fume hood is a green light to handle almost anything. That’s a bad guess, especially here. Any open work or weighing must happen in a real, certified fume hood—one that draws air directly away from the user. Older hoods with poorly sealed windows don’t cut it. Daily airflow checks, using test papers or meters, kept me safer and let me spot a failing system before it failed me. If fumes start escaping anyway, review how you open chemical containers and whether ventilation fans work at full speed.
Storage: Containment Stops Accidents Before They Happen
No matter how careful you act during handling, storage remains a huge factor in safety. 2,4,6-Trinitrochlorobenzene gets stored in clearly labeled glass containers with secure tops. I always kept mine well away from organics or any reducing agents, since those mix-ups start unexpected fires and toxic clouds. Fireproof cabinets, not basic shelves, cut down the risk if something does go wrong. Labels need dates and hazard warnings large enough to read without squinting.
Cleanup: Spills Require Immediate Action
Even the best-prepared person faces the occasional spill. Quick action keeps a bad situation from getting worse. I never swept or wiped with standard paper towels—special absorbent pads suited for hazardous chemicals soak up the mess without sending dust airborne. For any larger spill, get folks out of the area, turn up the vent, and use appropriate personal protective equipment before starting cleanup. All waste goes into dedicated, labeled hazardous waste containers, not regular trash bins.
Training and Vigilance: No Substitute for Experience
Reading a few safety bulletins won’t make anyone ready to handle energetic chemicals safely. In my own training, shadowing someone with years of experience added more value than any textbook. Routinely review safety protocols, refresh your own habits, and question shortcuts—complacency often ends in emergencies. If something feels off, it probably is. Speak up and double-check. Lives and careers have ended from thinking a shortcut would save time.
Solutions: Clear Systems, Reliable Gear, Real Practice
Labs that put real effort into safety culture go beyond checklists. They keep every personal protective item stocked, invest in reliable hoods, organize clear storage, and require hands-on spill response training. Digital reminders about inspections and up-to-date emergency instructions save time and prevent confusion. Safety isn’t just about compliance; it’s about keeping everyone healthy and able to return home each night. Treat 2,4,6-Trinitrochlorobenzene with the respect it demands, and it won’t punish you for a careless mistake.
What is the chemical structure of 2,4,6-Trinitrochlorobenzene?
Unpacking the Structure
People who spend some time in a chemistry lab, or even just poking around a good textbook, have run across compound names that seem deliberately crafted to trip up the tongue. 2,4,6-Trinitrochlorobenzene proves to be one of those mouthfuls, but what it lacks in easy pronunciation it makes up for in a pretty fascinating structure.
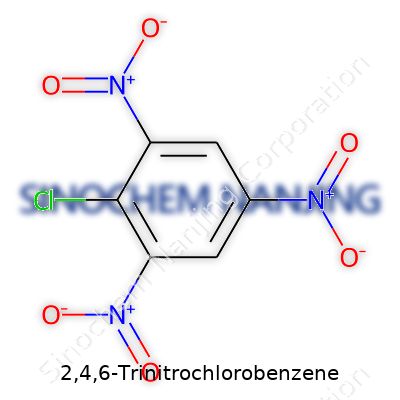
The name spells out what you’re dealing with. This chemical starts with a benzene ring—think six carbon atoms arranged in a flat hexagon, joined by alternating single and double bonds. On this ring, three groups called nitro groups (NO2) sit on the second, fourth, and sixth carbon atoms, while a chlorine atom clings to the first carbon. The basic skeleton follows this formula: C6H2Cl(NO2)3. Each nitro group draws electron density away from the ring, which ends up making the whole thing far less reactive than plain benzene. Place a chlorine on a ring like this and you don’t just have a neat drawing—you’ve got a molecule ready to behave differently from anything you'd find under your kitchen sink.
Real-World Relevance
It’s always important to question why a specific arrangement matters. I remember the moment in an undergraduate lab when a classmate asked, “What’s the big deal about all these tiny tweaks to molecules?” The answer comes fast when you realize how much a single substituent can flip the script on a compound’s reactivity. Toss three nitro groups on a benzene ring and add chlorine, and suddenly you've built something pretty explosive—literally and figuratively.
Researchers use 2,4,6-Trinitrochlorobenzene to study how electron-withdrawing groups change a molecule’s life story. Its explosive potential demands rigorous handling, which stirs memories of lecturers pressing the idea that safety never gets old-fashioned in chemistry. Every tweak to a chemical structure forces scientists to rethink how they use or dispose of materials. This chemical offers a textbook case of how adding or swapping even a single atom can transform a compound’s use, from research material to possible industrial intermediate.
Safety, Environment, and Responsibility
Years of chemical mishaps in schools and factoriesshowed why everyone—from top researchers to lab assistants—should understand the nuts and bolts of what they’re working with. The three nitro groups don’t just look serious on a structural formula; they carve out real consequences for stability and explosive potential. Leaving these dangers unchecked leads straight to headlines about industrial accidents. That reality grounds the need for training, investment in safety equipment, and procedures designed to catch mistakes before anyone pays a heavy price.
Disposal brings another set of questions. Compounds like this don’t quietly degrade into harmless dust. They linger. If labs or factories send trinitro compounds into the environment, nearby communities shoulder the risk. Responsible chemical handling—involving incineration, neutralization, or other proven methods—keeps these substances out of water and soil. These practices take discipline and funding, but they also speak to wider values. Protecting people and the world around us always comes first.
Better Paths Forward
I’ve watched colleagues wrestle with replacing hazardous chemicals using smarter substitutions or synthesizing alternatives with better safety records. For 2,4,6-Trinitrochlorobenzene, researchers continue pushing for new protocols, greener chemistry models, and tougher oversight for the places that use or produce such compounds. The challenge never fully vanishes, but staying honest about the risks keeps people pushing for solutions that safeguard workers, neighbors, and future generations. At its core, deep knowledge about chemicals leads to progress.
How should 2,4,6-Trinitrochlorobenzene be stored?
What’s on the Shelf Really Matters
Lots of folks see a bottle with a label and imagine everything’s under control. In a chemical storeroom, assumptions make accidents. 2,4,6-Trinitrochlorobenzene isn't something you just put next to lab soap or shove under a counter. It's a powerful compound, known to pack some real volatility, and even seasoned people can forget how quickly things go sideways with these chemicals. My own years in labs taught me never to let familiarity blind you to real danger.
Volatility Calls for More Than Locked Doors
2,4,6-Trinitrochlorobenzene, used in dyes and explosives, reacts sharply with heat, impact, and sparks. One summer, the air conditioning broke down in our department; someone shrugged off rising storage room temperatures. Later, a bottle’s seal popped, filling the air with a sickly odor. No explosion that day — but we were lucky. This experience sticks with me as a blunt lesson: even small lapses, like letting temperatures rise above 25°C, invite risk.
No Shortcuts: Proper Containers and Location
Some might reach for any old glass jug, but that’s rolling the dice. Only containers that resist corrosion, with tightly sealed tops and clear hazard labeling, make sense. Once, I found 2,4,6-Trinitrochlorobenzene in an old, cracked jar tucked in a wooden cabinet — a cocktail for disaster if you ask anyone who’s worked with unstable organics. Dry, cool, well-ventilated spots away from light and ignition sources are the only smart option. Even humidity and sunlight push this compound closer to instability.
Don’t Turn Your Back on Segregation
Mixing storage is just asking for trouble. At a university, old bottles used to fill every shelf. Someone placed trinitrochlorobenzene within arm’s reach of oxidizers and acids. A minor slip — reaching for the wrong container — could have sparked a fire, or worse. Chemicals with the nitro group shouldn’t sit anywhere close to acidic or oxidizing substances. Segregating based on hazard class isn’t just textbook advice; it’s common sense learned the hard way.
Monitoring and Training: No Place for Guesswork
Labels fade and knowledge fades just as quickly if teams turn over often. Labs that get it right perform routine checks. Stop and check the logs: Who last opened the bottle? When did someone check the seal or label? Stock control with proper records stops nasty surprises. It only takes one person with half the details to set off a costly cleanup or put someone in the ER. Regular training — not just page-flipping through safety manuals — works far better. In conversations with safety officers, it’s clear they spot more mistakes from old habits than from rookie moves.
Improve, Don’t Gamble
Skip improvisation. Invest in fail-proof ventilation and monitored storage. Businesses and academic labs can work with suppliers who offer safe packaging and up-to-date documentation. Government guidance is everywhere, but personal responsibility matters more than any checklist. From what I’ve seen, safe storage isn’t about fancy warnings — it’s about making each person accountable, from new lab worker to department head.
Bottom Line
Stories about chemical accidents often echo the same failures — shortcuts, old containers, missed checks. Real safety means treating every chemical with the respect it demands, trusting experience, and holding each other to standards that don’t bend when things get busy. If that doesn’t happen, lessons come the hard way.
Is 2,4,6-Trinitrochlorobenzene hazardous to health or the environment?
What’s Hiding in the Name?
People don’t run into 2,4,6-Trinitrochlorobenzene at the grocery store, but this chemical pops up in niche industrial settings, usually among dyes, explosives, or pesticides. The yellowish powder looks harmless, but this isn’t just another unremarkable compound. Research and practical experience show that chemicals in the nitrobenzene family can mess with both human health and nature.
Health Has the Final Say
Any chemical with three nitro groups and a chlorine ring brings certain risks. Breathing in its dust or having bare skin touch it spells out trouble—headaches, dizziness, even fainting for those unprotected workers. Older research into similar substances tells us that nitrobenzene compounds can damage the liver, nervous system, and eyes. If a substance like this lands in water or soil, its toxic bite doesn’t always fade away quickly.
Ask someone who has ever worked in a chemical plant or dealt with old munitions—there’s a clear sense that waste dust and runoff hang around for years, and the headaches linger with them. Even tiny, repeated exposures pile up over time. The tightening in the chest, skin rashes, and unexplained fatigue aren’t always easy to trace back to this one compound, but these symptoms tell a story of long-term impacts.
Piling Up in Soil and Water
After these chemicals spill, get buried, or move through drains, the real challenge emerges. 2,4,6-Trinitrochlorobenzene sticks with clay and silt, refusing to break down easily. Certain bacteria can tackle some pollutants, but this chemical resists. It carries a risk for more than one season—a full-blown problem if it makes contact with crops or gets washed into streams.
Wildlife picks up the slack for humans’ mistakes. Toxic chemicals start in the water, but fish and frogs send those nasties further along the food chain. Birds eating those tainted critters build up even more of this compound in their bodies. Over time, the whole river system runs the risk of thinning numbers, odd birth defects, and fragile eggs in birds.
Solutions Need Sweat and Science
Cleaning up a mess this tough takes a lot more than tossing down sawdust or flushing the waste out to a treatment plant. Worker education sits at the top of the list—clear warnings, tight PPE rules, and tough monitoring by real people, not just paperwork. Companies can’t hide from spill records or medical reports, and new digital tracking tools keep a close eye on waste streams.
Safe disposal matters. Incinerators with the right filters can catch nearly all of the dangerous fumes, but only if run honestly. Storage tanks need regular checks. Outside the fence, watchdog groups test neighborhood soil and waterways, with labs willing to call out the presence of lingering toxics.
Policies from top agencies—real-world rules, not just thick binders on a shelf—help block this chemical from reaching the general public. Enforcing strict controls and regular audits means pushing for greener alternatives, too. Industries must invest in safer replacements to stop dangerous waste from being the norm.
The Everyday Takeaway
Nobody wants more red tape, but in the world of 2,4,6-Trinitrochlorobenzene, a little oversight saves a lot of headaches—literal and legal. For now, knowledge, transparency, and vigilance offer the strongest shields against its hazards.
| Names | |
| Preferred IUPAC name | 1-chloro-2,4,6-trinitrobenzene |
| Other names |
Picryl chloride 1-Chloro-2,4,6-trinitrobenzene Trinitrochlorobenzene |
| Pronunciation | /ˌtraɪˌnaɪtroʊˌklɔːrəˈbɛnziːn/ |
| Identifiers | |
| CAS Number | 88-77-7 |
| Beilstein Reference | 87864 |
| ChEBI | CHEBI:34760 |
| ChEMBL | CHEMBL14036 |
| ChemSpider | 10907 |
| DrugBank | DB14081 |
| ECHA InfoCard | 03a32896-9e2f-444c-8ae1-8e2c2f2c7df6 |
| EC Number | 209-088-3 |
| Gmelin Reference | Gmelin 7435 |
| KEGG | C14305 |
| MeSH | D014272 |
| PubChem CID | 13854 |
| RTECS number | BZ9625000 |
| UNII | 81U132OLJT |
| UN number | UN1321 |
| CompTox Dashboard (EPA) | DTXSID0021804 |
| Properties | |
| Chemical formula | C6H2ClN3O6 |
| Molar mass | 260.53 g/mol |
| Appearance | Yellow crystalline powder |
| Odor | Odorless |
| Density | 1.742 g/cm³ |
| Solubility in water | Insoluble |
| log P | 2.94 |
| Vapor pressure | 0.000016 mmHg (25°C) |
| Acidity (pKa) | -0.38 |
| Basicity (pKb) | 8.65 |
| Magnetic susceptibility (χ) | -60.7·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.639 |
| Dipole moment | 3.97 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 318.1 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -16.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1075.2 kJ·mol⁻¹ |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and eye irritation, may cause respiratory irritation, toxic to aquatic life. |
| GHS labelling | GHS02, GHS06, GHS09 |
| Pictograms | GHS06,GHS09 |
| Signal word | Danger |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P261, P280, P305+P351+P338, P304+P340, P312 |
| NFPA 704 (fire diamond) | 3-3-0 |
| Flash point | 122 °C (closed cup) |
| Autoignition temperature | 190 °C |
| Lethal dose or concentration | LD50 oral rat 308 mg/kg |
| LD50 (median dose) | LD50 (median dose): 308 mg/kg (oral, rat) |
| NIOSH | SN2985000 |
| PEL (Permissible) | PEL: 1.5 mg/m³ |
| REL (Recommended) | 0.1 mg/m3 |
| IDLH (Immediate danger) | IDLH: 5 mg/m3 |
| Related compounds | |
| Related compounds |
2,4,6-Trinitroaniline 2,4,6-Trinitrophenol (Picric acid) 2,4,6-Trinitrotoluene (TNT) 2,4,6-Trinitrobenzoic acid 1,3,5-Trinitrobenzene 2,4-Dinitrochlorobenzene |

