Looking at 2,4,6-Trinitrobenzoic Acid: A Chemical’s Journey from Laboratory Curiosity to Modern Applications
Historical Development
The story of 2,4,6-Trinitrobenzoic acid kicks off somewhere in the landscape of nineteenth-century chemistry. Back in those days, chemists learned to wield powerful nitrating agents, hammering benzene rings into ever more explosive and vibrant forms. Trinitro compounds like picric acid and TNT grabbed headlines for their use in munitions and industry. Tucked among these, 2,4,6-Trinitrobenzoic acid came to light not as a main explosive, but as a sort of chemical cousin. I first learned about it in the cramped stacks of a university library, leafing through dusty journals and patent documents that hinted at its early recipes and mishaps. People looked at it for its reactivity—those three nitro groups attached to an aromatic ring offer a showpiece in functional group manipulation.
Product Overview
Chemists define 2,4,6-Trinitrobenzoic acid by its structure: a benzene ring with three nitro groups and a single carboxylic acid. It’s not one you see every day unless you specialize in niche organic synthesis, but it holds unique interest for those digging into aromatic nitration and the history of energetic materials. Looking past the technical jargon, it’s clear that this molecule owes much of its appeal to its harmonious—but tensely poised—combination of explosive potential and utility as a synthetic building block. Some people lump it together with explosives, but its controlled use extends into analyses and specialty chemical transformations.
Physical & Chemical Properties
This compound shows up as a yellow crystalline solid, its color a classic hallmark of nitrated aromatics. The nitro groups tug on the electron cloud around the ring, making the whole molecule electron-poor and a little edgy with respect to further reactions. As I recall from my own benchwork, you don’t handle this sort of material lightly—sensitivity, particularly to friction or shock, warrants respect. It doesn't dissolve as readily in water as some simpler acids; organic solvents like acetone or alcohols tend to do a better job. Chemists use melting point and infrared spectra to confirm its purity and structure, another reminder that even with advanced science, touchstones of classic organic chemistry remain essential.
Technical Specifications & Labeling
Labeling for 2,4,6-Trinitrobenzoic acid rarely strays from a strict script: hazard warnings, storage advisories, and purity details dominate. Regulatory standards demand clear hazard identification—oxidizing warning symbols, protocols for accidental release, respiratory protections, the whole works. Based on my experience with nitroaromatic compounds, proper labeling isn’t just a matter of bureaucracy; anyone working in a tightly regulated lab breathes easier knowing that every shelf and storage fridge keeps you out of avoidable trouble. Purity grades matter, as does knowledge of possible contaminants that could compromise laboratory results or trigger unintended reactivity.
Preparation Method
Classic syntheses produce 2,4,6-Trinitrobenzoic acid by nitrating a precursor such as benzoic acid. Mixing concentrated nitric and sulfuric acids—a time-tested nitrating duo—nudges the reaction forward. The exothermic nature and potential hazards force even seasoned chemists to plan every step, from chilling reagents to managing the quenching process. In my own work, I remember the tension of dripping viscous acid into another flask, counting on slow addition to keep runaway reactions at bay. Yield and purity always depend on patience, temperature control, and careful post-reaction purification, usually by recrystallization. Environmental concerns over spent acids have spurred research into cleaner processes, aligning chemical practice more closely with today’s demand for sustainability.
Chemical Reactions & Modifications
The trinitro arrangement on the benzene ring pushes the limits of electron withdrawal, so further substitution or reduction reactions become selective and sometimes slow. Reduction can transform it to amino derivatives, while the acid group offers a handle for esterification and salt formation. My time in academic organic labs taught me the importance of subtle manipulation—introducing protecting groups, exploring metal-catalyzed couplings, anything to tweak the molecule for downstream research. Many pathways homed in on attaching more elaborate groups or testing selective reductions, which, despite frustration, often produced remarkable new tools for broader synthetic strategies.
Synonyms & Product Names
2,4,6-Trinitrobenzoic acid sometimes appears in literature and catalogs under alternative monikers: sym-Trinitrobenzoic acid, TNB acid, or just TNB. Such variety springs from historical naming conventions more than marketing. Students and professionals alike sometimes trip over multiple names for the same compound, a frustration anyone who’s chased down research references knows well. One way through the confusion: always double-check the structure, not just the word. Science relies on a shared language, but it doesn’t always read like a single dictionary.
Safety & Operational Standards
Safety starts with recognizing that all nitroaromatics, especially highly nitrated ones, pose significant risks. 2,4,6-Trinitrobenzoic acid doesn't match the volatility of TNT, but nobody sane ignores its sensitivity to heat, spark, or impact. Proper training matters. In my time overseeing undergraduate teaching labs, strict routines covered even the smallest-scale manipulations: fume hoods always on, safety goggles firmly planted, gloves checked for integrity, and a clear understanding of waste disposal. These are not relics of a litigious age; mishaps cost lives. Industry standards and research protocols count on both in-person training and documentation, ensuring that new scientists understand both the power and peril of what they hold.
Application Area
While some see 2,4,6-Trinitrobenzoic acid as a curiosity, targeted applications show up in analytical chemistry and materials research. It surfaces in diagnostic procedures to test for reactive functional groups, sometimes making a guest appearance in explosives research, either as a performance benchmark or breakdown product. Chemistry classrooms have used it to teach about nitration, functional group reactivity, and synthetic design—visual proof that real molecules drive the field, not just textbook schemes. Emerging research continually probes for new tricks, harnessing the nitro groups in developing organic electronic materials or exploring structure-activity relationships in pharmaceuticals, even if commercial applications remain niche.
Research & Development
Research ambitions circle around improved synthesis, greener chemistry, and safer handling. Laboratories, both academic and industrial, keep chasing routes that use milder conditions and minimize hazardous by-products. The regulatory climate pushing green chemistry reminds everyone that yesterday’s strategies don’t always fit today’s world. There’s a quiet race to find catalysts or solvents that break the reliance on strong acids and lessen waste disposal headaches. My colleagues and I swapped stories about failed runs and accidental discoveries, proof that innovation moves step by step, not always in a straight line. Journals today reflect this search for balance—between reactivity and safety, efficiency and environmental stewardship.
Toxicity Research
The toxicity profile of highly nitrated aromatics, including 2,4,6-Trinitrobenzoic acid, makes sober reading. Ingestion or skin contact brings risks of irritation, while chronic exposure may harm vital organs. Toxicological research focuses on metabolic pathways and breakdown products, as some nitro compounds convert into even more dangerous forms in the body. I’ve met researchers deeply wary of nitroaromatic exposure because of accidental spills that left persistent stains and left them feeling ill. Regulations and workplace standards aren’t just paperwork—they exist to keep scientists, technicians, and even students out of the emergency room. Modern labs require good engineering controls, training, and ongoing monitoring; the price of ignoring red flags is far too high.
Future Prospects
Looking ahead, 2,4,6-Trinitrobenzoic acid occupies a crossroads. Environmental pressure pushes against the use of legacy nitration chemistry; at the same time, the demand for specialty aromatic compounds keeps research interest alive. The push for alternative synthetic routes—maybe biocatalysis, maybe electrochemical methods—could cut down on waste and hazards. Academic groups and industrial R&D both see opportunities for innovation in materials science and analytical chemistry, even as regulations tighten. Every advance in safety, sustainability, or performance offers a case study for training the next generation of chemists. These changes don’t happen overnight, and for many in the field, the journey from dangerous curiosity to useful, safer compound feels like a long but necessary road.
What is 2,4,6-Trinitrobenzoic Acid used for?
Beyond the Chemistry Bench
2,4,6-Trinitrobenzoic acid rarely grabs headlines, but the compound leaves its mark in sciences and industry. Built around a benzene ring dotted with three nitro groups and a carboxyl group, this molecule packs a punch both in reactivity and practical uses. My first interaction with this substance happened during an undergraduate lab, where its crystalline orange form challenged my ideas about what chemicals “safe for the bench” really looked like. Proper storage and respect aren’t optional. This isn’t something to take lightly, as the structural cousin picric acid occasionally pops up as a newsworthy safety risk or forgotten bottle in a high school closet. Safety first, always.
Power in Analytical Applications
For analytical chemists, 2,4,6-Trinitrobenzoic acid serves as a tool for detecting and quantifying amino acids and other nitrogen-based compounds. Lab teams exploit its tendency to form colored salts with basic groups, which means spectroscopic determination gets much simpler. Every time a lab uses it, someone’s research pushes ahead—be it medical diagnostics, forensic science, or food safety checks. Spectroscopy depends on reliable reagents; research teams rely on robust, consistent results. Getting things right the first time saves hours in troubleshooting and lets more meaningful work happen. Having precise methods builds trust in science, from a university lab bench to a government compliance office. People want their medicine safe and their food uncontaminated—for these goals, trinitrobenzoic acid lends more than a helping hand.
Link to Explosives Research and Synthesis
A compound featuring three nitro groups doesn’t just walk into the lab for detection tasks. The same nitro-heavy design creates interest in explosives research, particularly for developing calibrants and standards for analytical instruments. Security teams and customs labs need robust, consistent ways to spot explosives; 2,4,6-Trinitrobenzoic acid offers a stable, well-characterized alternative to the more sensitive substances like trinitrotoluene (TNT). Testing equipment with a known, predictable standard improves both safety and reliability. Jobs depend on this accuracy, from transportation safety inspectors to investigative chemists. Mistakes can mean missed threats or false alarms, putting lives and livelihoods at risk.
Educational and Research Training
Chemical education makes good use of compounds that demonstrate real, observable reactions. 2,4,6-Trinitrobenzoic acid shows budding chemists the power of nitro groups, the impact on acidity, and the subtleties of molecular structure—lessons charted in vivid color on the bench. This kind of “in-your-hands” learning churns out researchers who remember safety protocols, understand limits, and respect reactive molecules. Universities use it for advanced organic synthesis and to demonstrate complex analytical techniques. Future scientists sharpen their skills, building habits that later keep everyone safer around even riskier chemicals.
Environmental and Health Considerations
All this usefulness comes with responsibilities. Like many nitro-aromatic compounds, 2,4,6-Trinitrobenzoic acid requires careful disposal and environmental controls. Mistakes spill into water and soil, harming aquatic life and even entering food chains. Labs and factories employ careful waste management to prevent contamination. Talking about chemical safety isn’t just fear-mongering—the stakes aren’t just academic. Families living near industry want assurance that safeguards don’t buckle under cost-cutting or neglect. Regulatory oversight tracks every step, pushing companies and scientists toward greener, safer alternatives where possible.
Pushing for Responsible Science
The story of 2,4,6-Trinitrobenzoic acid—like so many other chemicals—spins out across research, industry, and community. Using it with respect, oversight, and full awareness of the risks builds trust not just in chemistry, but in those who study and commercialize it. As a chemical trusted enough to measure explosives and simple enough to teach basic reactivity, it deserves both our curiosity and our caution.
What are the safety precautions for handling 2,4,6-Trinitrobenzoic Acid?
What Makes 2,4,6-Trinitrobenzoic Acid Demanding?
Anyone who’s handled nitroaromatic compounds knows there’s no room for shortcuts. 2,4,6-Trinitrobenzoic acid stands out for its explosive potential and toxic nature. In the lab, you feel the tension—your muscles tighten as you measure out a yellowish powder, knowing a slip could cause a serious accident. There’s science behind the caution. The compound’s three nitro groups pack a big punch, and even a modest jostle or bit of careless static sparks trouble. My fingers once tingled just reading about an incident in a university lab: improper handling led to a minor explosion, and the lesson stuck.
Personal Protective Gear: Dress for the Job
Standard goggles and gloves don’t cut it here. Splash-proof goggles shield eyes, but I go beyond with a wraparound face shield. There’s a comfort in knowing no splash can sneak past. Chemical-resistant gloves form the next line of defense, but not any old nitrile will do—thicker, certified materials reduce risk. Lab coats mean more than professionalism. I keep mine snapped tight with no exposed skin. Boot covers and flame-resistant aprons round out the look, and it's a small price to pay for peace of mind. It’s not about fear—it’s about respect for a chemical that gives no second chances.
Environmental Controls
Open air labs belong to the past. Even a well-ventilated room can fill with dangerous fumes before you notice. The right containment comes from a fume hood with strong airflow and a sturdy sash. I remember hearing an extractor fan whirr louder than usual during a synthesis run—it was a small reminder of the system’s value. Mechanical ventilation, grounded benches, and anti-static mats cut down static—no snapping wool sweaters here. Flame sources, even a simple heating mantle, stay far away. Everything happens slow, deliberate, methodical. Cleanup materials and spill kits rest only a step away, not hidden on a shelf across the room.
Storage Wisdom
You never stash 2,4,6-trinitrobenzoic acid next to strong acids or bases. Sealed, shatterproof containers get top shelf status, well out of reach of sunlight, heat, or vibration. I double-bag every solid sample, just like handling old picric acid bottles. A temperature log hangs on the refrigerator door, checked daily. Regulatory guidelines set firm limits—not or suggestions, but obligations. Recordkeeping isn’t paperwork—it’s insurance for everyone who works in the building. Shortcuts add up over time, and old samples that linger too long in a drawer become the next headline.
Handling and Disposal
Precision matters more than speed. Small batches only, measured with non-sparking spatulas—plastic or ceramic tools earn their spot in the drawer. No one works alone, not even on overtime. Someone else in the room can call for help, grab a shower station, or dial emergency services. Disposal follows strict hazardous waste protocols—not down the drain, not mixed with general trash. Labeled waste bins, secure transport, and professional disposal partners who specialize in energetic materials save lives. Forgetting a step, cutting a corner—these mistakes haunt.
Building a Safety Culture
Rules in the binder don’t mean much if you don’t teach new people the why. I’ve learned the best-run labs talk about what can happen, not just what should. Training drills, open conversations about close calls, and real-time feedback help everyone spot problems before they start. Team members don’t just rely on supervisors; they check on each other. Regular equipment tests and chemical inventory reviews catch problems early. The instinct to double-check, to ask—those routines matter. Safe handling isn’t a one-person job. It’s a community promise, built on shared habits and lived experience.
What is the chemical structure and molecular formula of 2,4,6-Trinitrobenzoic Acid?
Looking at the Heart of Its Chemistry
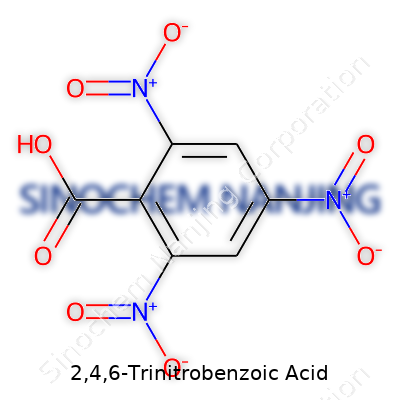
2,4,6-Trinitrobenzoic acid stands out as a strong example of how adding nitro groups changes the world of aromatic compounds. This molecule counts three nitro groups fixed to a benzoic acid backbone, all sitting at the critical positions on the benzene ring. The backbone, benzoic acid, already draws plenty of attention in organic chemistry textbooks due to its carboxylic acid group. Now, by sticking nitro groups at the 2, 4, and 6 spots, you get a substance with new chemical and physical properties.
The Chemical Structure Breaks Down Like This
The chemical formula for 2,4,6-Trinitrobenzoic acid is C7H3N3O8. You see seven carbons, three hydrogens, three nitrogens, and eight oxygens lining up for the job. The benzoic acid portion supplies the carboxyl functional group (COOH), while the three nitro groups (NO2) settle onto the 2nd, 4th, and 6th carbons of that benzene ring.
To picture the structure, start with a classic benzene ring—a hexagon showing alternating double bonds. Place the carboxylic acid group on carbon #1. The nitro groups sit right next to it, skipping every other carbon so they land at position 2 (ortho), 4 (para), and 6 (ortho, opposite side), crowding the ring with electron-withdrawing presence.
What All These Nitros Mean for Chemistry
After spending time tinkering in undergraduate labs, the way nitro groups reshape aromatic compounds always kept things lively. Nitro groups pull electrons away from the ring. This makes the compound more acidic compared to regular benzoic acid, and also gives it a real punch in terms of chemical reactivity. The electron deficiency makes the ring more interested in nucleophiles, and less interested in typical electrophilic substitution you’d see in plain benzene chemistry.
In simple words, these three nitro groups turbo-charge the molecule’s properties. That means 2,4,6-trinitrobenzoic acid winds up with a lower pKa, making it more ready to give up its proton from the carboxylic acid group. This fact holds plenty of importance in reactions involving salts, or in methods looking to fine-tune acidity or reactivity in synthesis.
Why This Structure Matters
I remember separating nitrobenzoic acid isomers during a sophomore organic class. The trinitro version always caught notice because it takes more effort both to synthesize and to handle. Chemicals with this level of nitro substitution often walk the line between academic interest and practical hazards—sometimes drawing the attention of explosives research, industrial dyes, or as neat case studies in designed acidity. Researchers rely on the detailed understanding of atomic placement and the effect of such groups to plan out safer industrial procedures or even find new routes for creating pharmaceutical agents. Knowing the nuances of this molecule helps chemists build safer workplaces and better waste treatment too, since strong acids with nitro groups need special care during disposal.
Looking Ahead to Safer, Smarter Chemistry
Given trinitrobenzoic acid’s reactivity, safety steps cannot get skipped. Chemical knowledge matters most when it drives better safety habits. Simple labels and MSDS sheets warn of risk, but real understanding sparks careful planning and respect for lab safety. Strong acids and energetic nitro compounds require vented storage, attentive handling, and good training. Building this muscle memory helps future chemists avoid common mistakes or exposure incidents. Getting structure right supports these efforts, giving both researchers and industry teams a solid base to make better decisions—from designing reaction routes to managing environmental cleanup after spills.
How should 2,4,6-Trinitrobenzoic Acid be stored?
Keeping Powerful Chemicals in Check
A lot of folks who work in labs, universities, or manufacturing rarely talk about 2,4,6-Trinitrobenzoic Acid outside of technical handbooks, but it deserves a real conversation. The stuff’s not something you want to mishandle. Years back, I watched a simple classroom demo turn into a fire scare because a teacher underestimated nitro compounds. Situations like that stick with you; mishandling can hurt people, ruin work, and trigger bigger emergencies.
Why All the Rules?
This compound comes from the same chemical family as other strong explosives. On paper, 2,4,6-Trinitrobenzoic Acid isn’t as dangerous as TNT, but unchecked heat or friction still turns it risky. Research from the CDC and chemical safety organizations points to the potential for spontaneous ignition, eye and nose irritation, and environmental headaches if this compound leaks. To keep people and property safe, most guidelines stress respect for stability, coolness, and separation from life’s normal clutter.
Solid Storage Starts with Common Sense
Taking care of this acid isn’t about some mystical lab ritual—it’s about straightforward habits. Always keep it in a dark, dry place. Moisture can break down the acid and boost reactivity. Heat, even from direct sunlight, messes with stability and eats away at plastics or old storage containers. I learned to trust thick glass jars with tight-sealing lids, and to stash them in metal cabinets lined with acid-resistant trays. Labels should list full chemical names, hazard symbols, and the date the jar was filled. A lazy label can cost someone their eyesight or worse.
Don’t Store Everything Together
Temptation says one cabinet saves space, but mixing this compound with acetone, strong acids, bases, or flammable solvents multiplies risks. I watched a storage room manager fume about “wasted space” until he saw a near miss—a leaking bottle sitting six inches from ammonia cleaner. Trusted safety sheets and chemical compatibility charts don’t exist just for compliance; they exist because people have gotten badly hurt making simple storage mistakes.
Think About Ventilation and Security
Not every workspace has a state-of-the-art fume hood. Still, some airflow helps prevent vapors from building up after accidental spills. Open windows won’t cut it; hidden acid fumes stick around and corrode metal shelves or eat at labels. I’ve worked in small institutions where a basic exhaust fan and an open flame arrestor meant the difference between peaceful afternoons and hours-long evacuations.
Disposal and Emergency Prep
Running a tight chemical inventory can feel tedious, but it pays off. Expired or unused 2,4,6-Trinitrobenzoic Acid shouldn’t join everyday trash. Partnering with hazardous waste services makes more sense than risking fines or contaminating landfills. Spill kits, absorbent pads, and printed emergency plans belong within arm’s reach. Nobody thinks they’ll ever need them, yet everybody’s grateful once a container cracks or an earthquake rattles loose a shelf.
Responsibility and Culture
Safety grows out of habit and learning together. New lab techs, students, and even seasoned pros do better when trained with real scenarios, not just checklists. Respect for what a chemical can do always trump convenience. Nothing builds credibility in any lab or company faster than a spotless safety record and a visible commitment to proper storage.
Is 2,4,6-Trinitrobenzoic Acid hazardous to health or the environment?
Looking Closer at a Powerful Molecule
I once watched the anxiety spread through a high school chemistry lab after a student realized he’d spilled a little-used reagent on his desk: 2,4,6-Trinitrobenzoic acid. Nobody seemed sure whether to panic or sigh in relief. That memory comes back now whenever the conversation turns to how chemical hazards are handled outside the classroom.
This particular compound often flies under the radar. Its relatives, like picric acid, attract more headlines because they’ve been infamous for explosions and military use. But trinitrobenzoic acid brings its own set of health and environmental headaches. Substances made by stacking nitro groups onto a benzene core are rarely friendly to living things. The more nitro groups, the more toxic, with greater push to both shock sensitivity and long-term persistence in soil and water.
Risks to Health: Skin, Breathing, and the Bloodstream
Direct contact with 2,4,6-Trinitrobenzoic acid isn’t a minor matter. Skin or eye exposure can cause real irritation — anyone who’s misjudged their glove size learns fast. Worse, inhaling dust or fumes from the powder raises the possibility of headaches, nausea, and even cyanosis, which means your blood can’t carry oxygen efficiently. Chronic exposure pounds on the liver and kidneys. Workers who handle this sort of chemical in industrial settings need proper training and personal protection every single day.
What Happens to Nature?
This molecule doesn’t sit and wait to be washed away like some common salts. Its three nitro groups resist breakdown in the environment. Once released, 2,4,6-Trinitrobenzoic acid can linger in soil and leach into groundwater. This means there’s a danger of contamination traveling farther than the original spill. Aquatic life faces real risk if run-off carries traces of this compound to streams—nitroaromatic substances hurt fish and aquatic insects, sometimes shutting down their central nervous systems or causing deformities.
From a personal standpoint, few things frustrate me more than seeing forgotten drums of chemical waste in derelict factories. These sites threaten neighbors and wildlife long after the business closes. That persistent risk isn’t theoretical; a single forgotten barrel can poison wells and kill off entire generations of amphibians. The EPA has flagged various nitro compounds as priority pollutants precisely because of this kind of long-lasting harm.
What Practical Steps Lower the Risk?
Solving problems tied to tough chemicals like this one requires more than just paperwork. Companies using or producing 2,4,6-Trinitrobenzoic acid have to store it carefully, disposing of waste at licensed facilities. Even better, switching to safer alternatives can lower exposure risks across the board. Better labeling and regular audits help flag hidden dangers before someone gets hurt.
For the public, awareness makes a difference. Understanding what hazardous chemicals are stored nearby lets communities ask sharper questions and demand real protections. After a neighbor reported a leaky warehouse in my town, local officials updated emergency plans and increased inspections. That tiny step might have saved a lot of pain down the road.
History shows that careful management and steady attention to old chemical stockpiles prevent problems for people and nature alike. With trinitrobenzoic acid, ‘better safe than sorry’ is more than a motto—it’s the line between safety and tragedy.
| Names | |
| Preferred IUPAC name | 2,4,6-trinitrobenzoic acid |
| Other names |
TNBA Trinitrophenylcarboxylic acid 2,4,6-Trinitrobenzenecarboxylic acid |
| Pronunciation | /ˌtraɪˌnaɪtroʊˌbɛnˈzoʊɪk ˈæsɪd/ |
| Identifiers | |
| CAS Number | 1983-11-9 |
| Beilstein Reference | 1207932 |
| ChEBI | CHEBI:41264 |
| ChEMBL | CHEMBL127038 |
| ChemSpider | 23735969 |
| DrugBank | DB08294 |
| ECHA InfoCard | 100.007.593 |
| EC Number | 209-103-6 |
| Gmelin Reference | 60738 |
| KEGG | C19182 |
| MeSH | D014278 |
| PubChem CID | 66220 |
| RTECS number | TY3150000 |
| UNII | 8K713H3UJP |
| UN number | UN1320 |
| CompTox Dashboard (EPA) | DTXSID4059482 |
| Properties | |
| Chemical formula | C7H3N3O8 |
| Molar mass | 259.12 g/mol |
| Appearance | Yellow crystalline powder |
| Odor | Odorless |
| Density | 1.76 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 1.96 |
| Vapor pressure | 1.9 x 10^-7 mmHg (25°C) |
| Acidity (pKa) | 0.65 |
| Basicity (pKb) | 1.49 |
| Magnetic susceptibility (χ) | -41,000·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.665 |
| Viscosity | 1.47 mPa·s (25 °C) |
| Dipole moment | 2.7 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 175.2 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -41.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1282.0 kJ/mol |
| Hazards | |
| Main hazards | Harmful if swallowed, causes serious eye irritation, may cause respiratory irritation, may cause skin irritation. |
| GHS labelling | GHS02, GHS06, GHS09 |
| Pictograms | GHS07,GHS09,GHS02 |
| Signal word | Danger |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P261, P280, P301+P312, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 2,4,6-Trinitrobenzoic Acid NFPA 704: 2-2-3 |
| Flash point | 190 °C |
| Autoignition temperature | The autoignition temperature of 2,4,6-Trinitrobenzoic Acid is "300 °C". |
| Lethal dose or concentration | LD50 oral rat 660 mg/kg |
| LD50 (median dose) | LD50 (median dose): 240 mg/kg (oral, rat) |
| NIOSH | TR1575000 |
| PEL (Permissible) | PEL: 1.5 mg/m3 |
| REL (Recommended) | 0.1 mg/m³ |
| IDLH (Immediate danger) | IDLH: Not listed |
| Related compounds | |
| Related compounds |
2,4,6-Trinitrotoluene Picric acid Trinitrobenzenesulfonic acid |

