A Deep Dive into 2,4,6-Trinitroaniline: History, Chemistry, and Where We Go from Here
How Science Built Up 2,4,6-Trinitroaniline
Looking at the story of 2,4,6-Trinitroaniline is like leafing through the pages of chemical history, where each step forward mirrored the growing knowledge and urgency around nitroaromatic compounds. Chemists worked to extend what they learned from trinitrotoluene and picric acid, drawn by the promise of groundbreaking energetic materials. Early on, scientists were compelled by the logic that packing nitro groups onto the aniline core could produce something powerful, maybe useful for propellants or explosives, and potentially as a building block for dyes. The development of 2,4,6-Trinitroaniline wasn’t just about curiosity. There was a race to make substances that could tip the balance in times of war or drive innovation in industrial chemistry. Knowledge gained from synthesizing and handling these dangerous, finicky compounds paved the way for advancements in modern synthetic techniques and safety protocols within chemical manufacturing.
Getting to Know 2,4,6-Trinitroaniline
Anyone who has worked in a chemistry lab probably remembers the shock of vivid color when nitrating aromatic rings, and 2,4,6-Trinitroaniline stands out in this memory. Here, chemists join three nitro groups to the aromatic ring of aniline, with each group demanding its own space and influencing how the whole molecule behaves. This compound goes by other names, among them Trinitroaniline and TNA. Its history even gave it a place in the dusty shelves of old explosives research, never quite as famous as TNT but never forgotten by those who study energetics. The product offers more than just energy release — it challenges the understanding of stability, reactivity, and safety in powerful chemical structures.
Physical and Chemical Properties
Many chemists recall how nitroaromatic compounds have a way of filling the room. Trinitroaniline, in its typical form, appears as a crystalline solid with a distinct yellow-orange color, a telltale sign that nitro groups dominate the molecular structure. It doesn’t dissolve easily in water, which protects it a bit from sudden mishaps caused by humidity. Its melting point is high enough to demand care but low compared to some relatives in the energetic materials class. Exposure to heat, friction, or a sharp mechanical shock can trigger a rapid, violent reaction. For those who measure such things, the detonation velocity and power won’t match military explosives like RDX, but that’s not the point—the sensitivity and reactivity define its reputation.
Technical Specifications and Labeling
Regulations push companies and researchers to handle, store, and label this substance with respect and caution. There’s no escaping the need to mark every container with clear hazard symbols and indications of primary risks: explosive potential above all, but also possible acute toxicity. Anyone opening a storage locker or a drum knows instantly that this isn’t just another reagent for a bench-top experiment. TNA demands chemical fume hoods, blast shields, and storage far from sources of heat or open flame. Regulatory frameworks may classify it tightly, treating it almost as a material of warfare, and in many regions, specific permits gatekeep access. This isn’t just bureaucracy—for anyone who has lived through an unexpected chemical incident, the reasons are clear and just.
How Chemists Prepare It
The heart of the synthesis lies in controlled, slow nitration. Chemists usually start with aniline, carefully drip-feeding it into a mixture of nitric acid and sulfuric acid. This process generates a lot of heat, taxing the patience of the operator. An accident here doesn’t just ruin a day—it can be catastrophic. The air fills with sharp acidic fumes as the nitro groups anchor themselves onto the aniline core, often requiring careful cooling and staged additions so that explosive runaways don’t occur. Once the reaction finishes, the yellow crystals precipitate out, and the product stays under acid until it’s safe to handle. The water washing and drying steps test everyone’s nerves, too, since even the damp powder can be dangerous.
Chemical Reactions and Opportunities for Modification
Despite TNA’s fame as a powerful, stubborn molecule, it still offers opportunities for further chemistry. Skilled researchers might reduce nitro groups, swap them for amines, or use the compound as an intermediate to prepare dyes. In experienced hands, it turns from a hazardous end-point to a stepping stone on the road to advances in pigment chemistry and the synthesis of advanced materials. Nitration and substitution reactions can be tuned to selectively produce tri- or dinitroaniline, making the field rich for those who want to tweak physical properties, sensitivity, or color for tailored uses. But this isn’t paint-by-numbers work. Surprises abound, and new researchers learn quickly that bench safety and planning matter more than bravado.
Synonyms and Product Names: Why Names Matter
Chemical identity can be slippery. In the literature, 2,4,6-Trinitroaniline shows up under monikers like TNA, Trinitro-1-aminobenzene, or even the trade term “Picramide,” tying it to the more notorious picric acid family. Researchers turn to ChemID, CAS numbers, and IUPAC for clarity, but the tangle of historic names reminds everyone just how much chemistry has evolved. Mislabeling and confusion aren’t small risks—the wrong bottle in the wrong setting multiplies hazards. Textbooks and advanced guides stress getting the names right, since regulatory authorities use those identifiers to decide who gets access and what restrictions travel with the product.
Safety and Operational Standards — No Room for Complacency
Any conversation about TNA leads quickly to stories of cautious respect in the lab. Researchers handling this compound never work alone, and the room adopts a tense focus that makes even old hands double-check their PPE. Real tragedy has followed when training lagged, or procedures got sloppy. Modern standards apply layers of control, from remote-handling tools to blast-resistant workspaces, and even then, seasoned chemists know that constant vigilance is the greatest protection. Training programs draw lessons from historic accidents, reinforcing the message that this isn’t a compound for shortcuts or improvisation. Safety datasheets lay out strict operational protocols, and regulatory agencies require regular audits and accident reporting to help identify what works and what must change.
Where It Serves: Applications That Matter
While its explosive history gets most of the attention, 2,4,6-Trinitroaniline finds uses in research circles working on advanced energetic materials. Some see potential for TNA-derived compounds in niche detonator applications or as spectral markers for tracing material movement. Dyes based on its structure attract attention in specialized textile and pigment research, although toxicity and stability issues steer most industrial uses toward safer alternatives. In teaching, TNA serves as a sharp example of both molecular ingenuity and what happens when safety isn’t just a footnote. Researchers in fields like forensic science have explored how traces of TNA and analogs can reveal hidden evidence in security investigations.
Current R&D: Pushing Boundaries and Setting Barriers
Active research often circles back to the question: how can we leverage the energy and reactivity of TNA’s structure with less risk? Chemists have worked to modify substituents and optimize crystalline forms to improve shelf life and reduce sensitivity. Some labs focus on using TNA as a model compound for computational studies on energetic materials, since its reactivity can help tune explosive simulations and predictive models. Advances in detection technology, encouraged by growing security threats, use TNA as a test target to fine-tune sensors for field deployment. The compound’s stubbornness and volatility spark both respect and creativity among those who try to harness or neutralize its risks.
Toxicity Research: Lessons Learned and Challenges Ahead
Handling TNA raises a long list of toxicological concerns. Laboratory studies show that its nitro group-packed structure leads to metabolic complications, especially in the liver and kidneys, when significant exposure occurs. The dust and vapors irritate mucous membranes, and in severe cases could lead to systemic poisoning or methemoglobinemia by altering blood chemistry. These risks have kept occupational health experts vigilant, mandating closed systems and regular screenings in facilities where TNA gets regular use. Some animal research points to mutagenic potential, urging chemists to seek alternatives whenever possible. Environmental health studies warn that releases of nitroaromatic compounds like TNA can linger in soil and water, affecting microbial life and working their way up the food chain, so regulations grow stricter every year.
What Matters for the Future
The place for 2,4,6-Trinitroaniline in tomorrow’s science looks different than it once did. Modern research tackles two challenges: finding ways to tame or replace nitroaromatics in critical applications, and turning historic lessons into safer work for the next generation. Advances in green chemistry push hard for drop-in replacements that offer the color, stability, or energetic output needed, without the legacy health and environmental costs. Some hope to build better sensors, develop antidotes, or engineer disposal methods with less risk, all based on deep study into the structure and behavior of TNA. That work pushes the boundaries of what’s possible, and every new paper reminds us that scientific progress comes with both promise and responsibility.
What is 2,4,6-Trinitroaniline used for?
The Core Uses of 2,4,6-Trinitroaniline
People working in chemistry rarely mention 2,4,6-Trinitroaniline to casual friends. Its name doesn’t roll off the tongue, but its punch matters to certain industries. The compound draws most of its attention from its explosive potential. Structurally, it looks like trinitrotoluene (TNT), except it swaps a methyl group for an amino group. This tweak shapes how it’s handled, stored, and put to use.
Some factories rely on 2,4,6-Trinitroaniline as a research tool for explosive science. It offers a chemical that researchers can study to better understand how explosives behave under certain conditions. The research helps armament engineers and safety inspectors plan for better stability and accident response in military or mining environments. My own college days in a chemistry lab showed just how tight the regulations could get—the smallest vial of any nitro compound required not just paperwork, but regular inspection.
Why It Draws Attention
This compound appears as a bright yellow powder, which hints at its potency. It doesn’t show up in mainstream products or household goods. Its main importance comes from serving as a model compound. Scientists analyze, test, and design safer alternatives using chemicals like 2,4,6-Trinitroaniline as benchmarks. The risk of mishandling never sits far from the conversation, since even a small batch can create a serious danger if storage conditions slip.
Chemical suppliers take major steps before handing it over. Most countries restrict buying and selling explosives and near-explosive materials, and 2,4,6-Trinitroaniline sits close to that line. The reasons tie to public safety. Accidents happen, sometimes with tragic results, in illegal or uncontrolled experiments. Safe storage and strict oversight become more than a formality—they protect lives.
Impacts on Science and Security
Detection and identification of explosives depend heavily on reference compounds. This chemical helps train bomb detection dogs, teach machine sensors to respond properly, and calibrate lab instruments. Forensic chemists use it to cross-check samples from crime scenes or accident sites. Criminal misuse stays in the back of everyone’s mind. The dark market for homemade devices has, in more than one documented case, included compounds like this.
On the science side, that same explosive power opens doors for controlled demolition, rescue operations, and even space applications. I remember reading NASA research papers where small, predictable initiators were needed to separate stages on rockets safely. Compounds like 2,4,6-Trinitroaniline helped design those systems.
Paths to Safer Use
Governments and industries work to keep these explosives under control. Rules covering licensing, background checks, secure storage, and regular audits serve a real need. The chemical industry partners with authorities to spot suspicious purchases and track shipments. In the lab, teachers drill students about gloves, goggles, and blast shields. Scientists have developed less sensitive alternatives for some tasks, pushing the field toward safer chemistry.
For the everyday person, 2,4,6-Trinitroaniline plays no direct role. Its legacy sits between science, safety, and security—a reminder that the tools we build for progress can always bring risk and responsibility along for the ride.
Is 2,4,6-Trinitroaniline explosive or hazardous?
The Story Behind the Compound
In chemistry circles, the moment someone says “trinitro-”, ears perk up. Out in the wider world, though, chemicals with complicated names usually just sound alarming. 2,4,6-Trinitroaniline doesn’t feature in front-page headlines, but some folks find themselves wondering if something in the “nitro” family deserves a space in the danger zone alongside TNT.
Diving into 2,4,6-Trinitroaniline, the structure resembles a few familiar names — most people have heard of trinitrotoluene (TNT) and picric acid. Both carry nitro groups, both have explosive records, especially under certain conditions. 2,4,6-Trinitroaniline throws a nitro group onto an aniline backbone, which still matters for its stability. Not every compound with nitro groups blows up when you look at it wrong — real chemistry gets a little more tangled than that.
Stability and Safety Concerns
Many chemists have spent their careers hunting for patterns that explain which molecules explode and which don’t. While 2,4,6-Trinitroaniline counts as energetic, its sensitivity isn’t the same as some notorious explosives. I’ve read studies where researchers used impact tests, and it turns out 2,4,6-Trinitroaniline needs a significant push — not quite as easy to detonate as TNT, but not exactly safe to toss around, either.
Often, the dangers come down to how things get handled and stored. Dust from nitro compounds can be a risk in the air, especially if sparks are flying nearby. The solid itself, under normal conditions, won’t erupt just sitting on a shelf, but you still wouldn’t want to grind or heat it up with no safety steps. Chemical repositories keep it under tight regulation for a reason.
More than just explosions, health hazards come into play. Direct contact with skin, eyes, or inhaling dust isn’t just a minor nuisance. Nitro-aromatic compounds like this have a way of causing methemoglobinemia, a blood condition where oxygen delivery drops. I remember a safety seminar where we looked at cases of chemical burns and poisonings from carelessly handled anilines and nitro materials. Gloves and masks aren’t just for show—they actually make a difference. In labs, training means treating compounds like this with respect. That mindset saves real people from real harm.
Why It Matters
Keeping tabs on substances like 2,4,6-Trinitroaniline protects more than just scientists. Law enforcement and hazmat teams want to know which materials could become threats, accidentally or on purpose. Schools and workplaces run chemical inventories so accidents aren’t waiting to happen. Neighbors near industrial plants have a right to transparency about what goes on around them. These efforts tie straight into public trust. A single mishandled batch could turn a quiet warehouse into next week’s tragedy.
Responsible Chemistry—What Works
Strong safety culture doesn’t happen overnight. Companies invest in proper containers, locked storage, and serious training for all staff. Government regulators keep a running list of high-risk chemicals, pushing for better labeling and tracking. Research teams invent safer alternatives where they can—sometimes swapping out hazardous compounds for less risky ones in explosives, dyes, or manufacturing processes.
No system is perfect, but close calls remind everyone that risk is more than theory. Practicing hazard communication never counts as overkill. Once, my university lab ran a mock “spill response” to keep everyone on their toes. Chemical safety drills might feel boring, but they pay off the moment something real goes wrong.
The Takeaway
2,4,6-Trinitroaniline isn’t nearly as infamous as some other chemicals, but its hazards aren’t just rumors. Anyone who works with or around substances like this needs solid safety habits, because good intentions don't stop an explosion or a trip to the ER. Better information, strict oversight, and respect for chemistry’s quirks go a long way—protecting everyone, from trained professionals to people just living nearby.
What is the molecular formula of 2,4,6-Trinitroaniline?
Chemical Identity
2,4,6-Trinitroaniline holds the molecular formula C6H3N4O6. Sometimes, the details in these formulas might look like a jumble of letters and numbers. But for any lab worker—chemist or student—getting each one right prevents a ton of confusion. This specific arrangement gives clues to both the power and risks that come with 2,4,6-Trinitroaniline.
Structure Tells a Story
The formula isn’t thrown together at random. It comes from a benzene ring, common across many aromatic compounds, but this one arms itself with three nitro groups—each slotted on at the second, fourth, and sixth positions—and a single amino group. Altogether, this structure packs a punch, both literally and in terms of chemistry. If you’ve ever worked with organic synthesis, you know three nitro groups slap some serious instability into the compound. That potential for reactivity drives reasons for strict handling in the lab.
Safety and Practical Lessons
The world doesn’t need more stories about accidental explosions in makeshift laboratories. Adding nitro groups, as you’d find in 2,4,6-Trinitroaniline, means each batch turns more sensitive. One error—a stray spark, wrong chemical combination, or rough handling—can lead to disaster. Back in college, the safety officer described nitroaromatic compounds as “temperamental.” They demand respect, not shortcuts.
Training remains the best solution. Anyone handling substances with the C6H3N4O6 makeup gets a rundown on blast shields, grounding, and personal protection. Even the paperwork matters—a label left half-readable or missing an emergency protocol invites disaster. Companies who treat chemical registries as a formality eventually pay the price. Real-world discipline in safety can and does save lives.
Function and Impact
Some compounds get notoriety for their instability or explosive force. 2,4,6-Trinitroaniline, with its packed nitro groups, fits that category. It’s often studied alongside old-school explosives, not because everyone wants to blow something up, but because researchers still search for new methods to neutralize, recycle, or even reinvent such molecules.
This molecule also pushes conversations about environmental responsibility. Waste from nitroaromatic compounds threatens waterways, soil, and ecosystems. Any time one of these gets made, the by-products demand careful disposal. Strict environmental rules loom for good reason. The molecular formula—so simple on paper—reminds researchers and industries to think twice about what’s flushed or dumped.
Looking Ahead
Most synthetic chemists now lean toward green chemistry principles. If a molecule like 2,4,6-Trinitroaniline enters development, companies ask if the old method truly deserves air time. Can new pathways reduce waste or hazards? More labs adapt protocols to lower their footprint, not just to satisfy government rules—but to avoid ruinous accidents and environmental damage.
Carrying the correct molecular formula isn’t trivia. It means each batch, each process, gets tracked properly. Each person working with sensitive compounds has the details straight. Clarity saves time, resources, and sometimes much more.
How should 2,4,6-Trinitroaniline be stored safely?
A Volatile Compound, Not Just a Label
Many folks see chemical names like 2,4,6-Trinitroaniline and think only labs or defense workers need to worry about them. The truth is, across many industries, people run into substances like this far more often than they might expect. I spent a few years working in a chemistry lab, where a strong respect—bordering on fear at times—surrounded anything with “nitro” in its name. These compounds don't give much warning before they become dangerous.
Real Risks Deserve Real Respect
Start with the basics: 2,4,6-Trinitroaniline contains three nitro groups attached to a single benzene ring. That structure makes it not just energetic, but touchy as a fox on a bad day. People have learned hard lessons about storing energetic compounds. Heat, static electricity, or just a jar falling off a shelf can set off a disaster. One small mistake, and you could be looking at an explosion or a fire that won’t go out easily.
Small Steps Make a Big Difference
Safe storage comes down to more than just tossing a jar on the closest shelf. I remember a colleague who always checked the grounding straps twice before setting anything on a workbench. Keeping 2,4,6-Trinitroaniline in a designated, explosion-proof safety cabinet goes a long way. Flammable safety cabinets, rated for explosive material, should sit in a well-ventilated room, far from sunlight or any source of heat. Avoid metal-on-metal contact or letting jars knock together.
The best practice involves storing it in small amounts only. Splitting larger quantities into several jars reduces risk for everyone. Each container needs a tight seal and a chemical-resistant label. It’s tempting to skip these steps on a busy day, but those five seconds of effort have saved lives more times than most people know. Good signage in the storage area keeps anyone from reaching in blindly.
Know What’s Around You
The company I worked for ran annual drills. People treated them like a chore. The first time an unsafe bottle of energetic chemicals ruptured nearby, those same people became advocates for safety. If a spill or worse happens, emergency response needs to arrive before the situation turns deadly. A working fire suppression system—especially one rated for chemical fires—sits high on the must-have list.
Never store 2,4,6-Trinitroaniline near acids, bases, metals, reducing agents, or strong oxidizers. These almost guarantee an unwanted reaction. A clear list posted outside the storage area, spelling out incompatible materials, makes sure guests and newcomers won’t make mistakes born out of ignorance.
Education and Vigilance
Some of the worst accidents happen not from carelessness, but because someone didn’t know how touchy a compound could be. Training never stops. New employees, seasoned chemists, facility managers—everybody gets briefed, retrained, and kept sharp. Those who handle chemicals like this need more than rules; they need to know the consequences, and understand why each protocol exists.
The goal remains simple: Get the work done, go home safely, and avoid adding another tragic story to the pile. Proper cabinets, small containers, trusted labels, and informed people—these all work together to keep everyone out of harm’s way. Sometimes it just takes someone speaking from experience to remind the team what’s truly at stake.
What are the handling precautions for 2,4,6-Trinitroaniline?
Recognizing the Risks
2,4,6-Trinitroaniline stands out in any lab or industrial setting due to its energetic and toxic nature. It’s a substance that has turned curious afternoons into disasters when people ignored safety protocols. Anyone who remembers high school chemistry knows that compounds with three nitro groups don’t forgive laziness. At its core, trinitroaniline’s structure is similar to other nitroaromatics—prone to explosion if mistreated, always a chemical to handle with real respect, not just gloves and goggles.
The Realities of Handling
Anything containing multiple nitro groups brings a risk of shock, friction, impact, static, or heat-induced detonation. I once watched a senior chemist cringe as someone whisked a beaker too briskly down a bench. Not every lab worker will admit it, but a casual attitude is just asking for trouble with these compounds. Even small amounts demand minimal movement, soft force, and deliberate actions—no exceptions.
Many labs require the use of antistatic mats, properly grounded equipment, and scoops made from non-sparking materials. That’s not overkill—one static spark could set off a tragedy. Chemical training drills it into you early: store trinitroaniline in small samples, always in tightly sealed, appropriate containers, and stash those in secure, cool spaces. I’ve seen old fridges retrofitted as chemical safes, some humming in the corner beside warning signs.
The Importance of Protective Equipment
No one should trust plain gloves and safety glasses for trinitroanilines. Splash-resistant lab coats, face shields, and heavy-duty nitrile or butyl gloves belong on the shopping list. When I worked in a facility that handled explosives precursors, we doubled gloves—just to reduce the odds of skin exposure. It’s not only about preventing burns or poisoning; skin contact or inhalation could lead to allergic reactions, long-term health effects, or acute toxicity.
Trinitro compounds like this one can cause everything from dizziness and headache to more severe symptoms such as methemoglobinemia, affecting oxygen delivery in the body. Inhalation threats bring the risk of lung damage or systemic toxicity. Good ventilation matters—a fume hood is the only place for manipulations with open containers. After every transfer or weighing, hands go straight to decontamination sinks.
Safe Disposal and Emergency Preparedness
Hazardous waste protocols matter just as much as handling. Trinitroaniline doesn’t take well to improvisation. Specialized disposal containers, arranged pick-up by licensed contractors, and absolute records of every gram’s fate—that’s how you avoid regulatory trouble and health risks. I’ve seen too many stories of “forgotten” jars found years later, each one a potential bomb.
Nobody likes to think of accidents, but being prepared beats panicking. Showers and eyewash stations belong within a few steps of any work area. There should always be an up-to-date spill and fire response plan on the wall and staff who know it cold. Some labs even run drills; everyone learns where the blast shields are and how to isolate a spill.
Culture of Responsibility
Across the world, chemical safety depends as much on attitude as on equipment. From big industry to small research labs, taking shortcuts invites disaster with 2,4,6-Trinitroaniline. I’ve heard old timers say, “Treat every day like one careless second could end careers—or lives.” Their caution isn’t paranoia. With this compound, it’s the voice of experience, and the reason most facilities forbid lone working or unsupervised experiments with energetic chemicals.
Instead of fear, respect and habit should drive every action—from opening the storage cabinet to signing out waste. Real safety comes not from rules on paper, but from constant skill and vigilance, supported by solid training and clear-headed leadership. Chemistry can open doors, but safe handling of powerful compounds like trinitroaniline keeps them open for everyone.
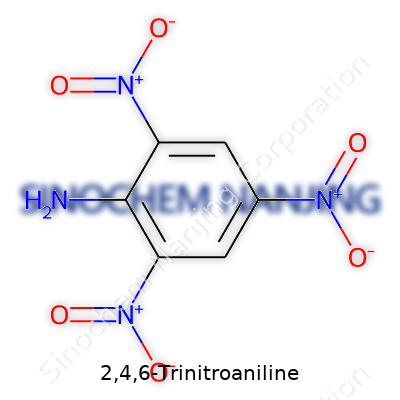
| Names | |
| Preferred IUPAC name | 2,4,6-Trinitrophenylaniline |
| Other names |
2,4,6-TNA 2,4,6-Trinitrophenylamine Picramide 1-Amino-2,4,6-trinitrobenzene |
| Pronunciation | /ˌtraɪˌnaɪtroʊ.əˈnɪlɪn/ |
| Identifiers | |
| CAS Number | 201-20-9 |
| Beilstein Reference | 1526987 |
| ChEBI | CHEBI:53335 |
| ChEMBL | CHEMBL32138 |
| ChemSpider | 15202 |
| DrugBank | DB13520 |
| ECHA InfoCard | 25e80379-084f-48e3-830a-bffdbe04b9a7 |
| EC Number | 209-758-0 |
| Gmelin Reference | 79475 |
| KEGG | C18647 |
| MeSH | D015266 |
| PubChem CID | 66242 |
| RTECS number | BZ9625000 |
| UNII | 00DT43ME8N |
| UN number | UN1321 |
| CompTox Dashboard (EPA) | DTXSID4021129 |
| Properties | |
| Chemical formula | C6H3N4O6 |
| Molar mass | 276.14 g/mol |
| Appearance | yellow crystalline powder |
| Odor | Odorless |
| Density | 1.73 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | 1.51 |
| Vapor pressure | 0.000001 mmHg (25°C) |
| Acidity (pKa) | 7.0 |
| Basicity (pKb) | 12.3 |
| Magnetic susceptibility (χ) | -41.4·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.680 |
| Viscosity | 1.44 cP (25°C) |
| Dipole moment | 3.70 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 282.2 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 111.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1426.8 kJ/mol |
| Hazards | |
| Main hazards | explosive, toxic if swallowed, causes serious eye irritation, may cause respiratory irritation |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS02,GHS03,GHS06 |
| Signal word | Danger |
| Hazard statements | H301 + H311 + H331: Toxic if swallowed, in contact with skin or if inhaled. H319: Causes serious eye irritation. H335: May cause respiratory irritation. H302: Harmful if swallowed. |
| Precautionary statements | Precautionary statements: "P210, P261, P280, P305+P351+P338, P370+P378 |
| NFPA 704 (fire diamond) | 2,4,6-Trinitroaniline: 2-3-2 |
| Flash point | 173 °C |
| Autoignition temperature | 485 °C |
| Lethal dose or concentration | LD50 oral rat 175 mg/kg |
| LD50 (median dose) | 390 mg/kg (rat, oral) |
| NIOSH | TT8750000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | REL: 0.1 mg/m3 |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
2,4,6-Trinitrotoluene 2,4,6-Trinitrophenol 2,4,6-Trinitrobenzoic acid |

