2,4,6-Trichlorophenol: Old Chemical, New Questions
Looking Back: A Journey Shaped by Science and Caution
Long before most folks heard of endocrine disruptors or dioxin output, chemists discovered 2,4,6-Trichlorophenol. Work with chlorinated phenols began in the late 19th century, part of a push for stronger disinfectants and wood preservatives. Over the decades, this compound became a staple in protecting timber from rot and mold in construction, railroads, utility poles, and old barns across regions with humid climates. University libraries and environmental archives store case studies spanning generations, showing how demand for materials like this often rose alongside booming infrastructure projects.
Unpacking the Product: How It Looks and What Makes It Tick
This chemical crops up as a white to yellow-orange crystal or powder. Most chem labs catch the sharp, medicinal odor right away. Its structure—three chlorine atoms attached to a phenol ring—gives it strength, yet also plays a part in environmental persistence and toxicity. It dissolves in common organic solvents, but not in water, guiding how people store and use it. It resists heat and resists breaking down, reasons it dominated the wood protection market for decades but also reasons for environmental concern.
Technical Details That Matter on the Ground
2,4,6-Trichlorophenol usually gets shipped in drums or heavy bags, labeled for toxicity and environmental hazard. Tech specs boil down to purity grades, melting point near 69°C, and storage recommendations to keep it from the sun and open flame. Labels need to be clear, not just for regulators but for everyone from storage staff to truck drivers to folks handling treated wood. GHS pictograms and warnings signal risk, shaped by a history of accidents and chronic exposure studies. What gets missed sometimes are the day-to-day details—fine dust settling in storage sheds, vapor exposure during high humidity, and the slow leak into surrounding soil.
Making the Stuff: Preparation with Purpose and Trade-Offs
Most facilities make 2,4,6-Trichlorophenol using chlorination of phenol in acidic water—what chemists call electrophilic aromatic substitution. Producers often use chlorine gas over phenol, sometimes with catalysts to speed up the process and boost yields. This route gives high selectivity for the 2,4,6 positions. Along with the product, these reactions create byproducts, including other chlorophenols and trace pollutants that turn into headaches for plant operators and regulators. Scrubbing and wastewater management, guided by decades of trial and error, work to minimize environmental release, yet the process plants still face close scrutiny.
The Chemistry Does Not Stop: Reactive and Versatile
Chemists appreciate that 2,4,6-Trichlorophenol can serve as a precursor or intermediate. It helps build more complex molecules—herbicides, dyes, and even antiseptics in the past. Reaction with bases forms salts that dissolve better in water, a double-edged sword when considering mobility in soils and water bodies. Under heat and basic conditions, it sometimes transforms into polychlorinated dibenzo-p-dioxins and dibenzofurans—names that ripple across environmental reports for their toxicity and persistence. So each step in a reaction chain invites another layer of responsibility, not just in the lab, but through the supply chain right down to waste disposal.
Names on the Label: Calling It What It Is
Besides “2,4,6-Trichlorophenol,” suppliers and chemists use “TCP,” “Phenol, 2,4,6-trichloro-,” and sometimes old trade names from long-discontinued formulations. Synonyms matter in regulatory filings, shipping manifests, and international trade. People working across countries or in legacy industries sometimes find confusion here, especially when old safety sheets use defunct names or when regulatory agencies switch nomenclature.
Staying Safe: Standards and Daily Realities
Industrial users follow bitter lessons learned over years—proper gloves, goggles, respirators in confined or poorly ventilated spaces. Major regulators flag it as a hazardous substance. Material safety data sheets get updated after nearly every high-profile incident or new toxicity report. Emergency wash stations stand close by in treatment plants or timber yards where 2,4,6-Trichlorophenol sprays or dips get carried out. Still, field reports describe stinging eyes, rashes, headaches—classic exposures even with training and personal protective equipment. Standards promote air monitoring and spill management backed by robust enforcement, not just window-dressing compliance but ongoing, proactive efforts.
Real-World Use: Why Communities Still Hear About It
Timber treated with 2,4,6-Trichlorophenol can last decades outdoors. Paints and protective coatings once leaned on it for durability, especially before alternatives emerged. Pesticide policies shaped how much ended up in farming and garden supply aisles, sparking debates over runoff, residue, and food safety. In my own experience working alongside an environmental consulting team, residues linger decades later near old treatment plants and under derelict barns where treated piles rot. This chemical sparks fear and lawsuits in communities near legacy industrial sites, especially where children’s playgrounds replaced railroad yards. Public concern is not a relic of the 1970s—soil testing and groundwater monitoring still catch traces where cleanup lagged.
Research and What Still Stumps Scientists
Academic papers and government agencies keep expanding the list of possible effects. Researchers measure how 2,4,6-Trichlorophenol breaks down, where it goes in water, soil, and air, and how it affects cellular function. The story does not end with measuring levels—scientists track hormone disruptions, immune response changes, and developmental impacts in animal studies. Debate continues over what “safe” means, especially for low-level, chronic exposure typical in neighborhoods next to old treatment plants. International studies compare practices and cleanup standards, feeding into environmental justice campaigns. People working on remediation sometimes share frustration: progress lags behind scientific certainty, and the funding needed for wide-area soil remediation only trickles in after findings hit the news.
Understanding Toxicity: What the Data Shows
Few substances linger longer in toxicology textbooks. 2,4,6-Trichlorophenol gets absorbed through skin, lungs, and digestion, hitting liver and kidneys first. Animal studies flag cancer risk, immune impacts, and developmental hazards. Byproducts—especially dioxins—raise even more concern, with decades of research showing cancer links at very low concentrations. Workers in sawmills and chemical plants reported high rates of skin, liver, and respiratory issues, and case-control studies link some cancers to direct exposures. Regulators worldwide list exposure limits, but the truth is, even low levels present a risk that deserves respect and vigilance. I have seen how new parents living near old treated rail lines call for more transparency and testing, fed up with slow government action.
A Look Ahead: Change, Substitution, and Unfinished Business
Alternatives to 2,4,6-Trichlorophenol now get the spotlight: borates, copper-based wood treatments, and mechanical preservation sit side by side with calls for reduced reliance on chemicals overall. Bioremediation, using fungi and bacteria to break down what older methods left behind, gather attention at research conferences and field sites. Local governments and environmental groups form partnerships to push for wider cleanup, stricter monitoring, and transparent risk communication. Markets shift to new solutions only when public pressure and clear science align. Moving forward, lasting change takes investment: better training, honest conversations about lingering pollution, and support for communities still carrying the toxic legacy. Experience tells me these chemicals never truly vanish from landscapes or headlines. What changes is the political will to solve the problem—by holding polluters accountable and inviting community leadership in setting new standards.
What is 2,4,6-Trichlorophenol used for?
Real-World Uses of 2,4,6-Trichlorophenol
The name 2,4,6-Trichlorophenol doesn’t exactly roll off the tongue, but it touches far more corners of daily life than most people realize. I remember growing up in a small town, everyone around me used to talk about treating wood fences and decks to keep them from rotting in the wet seasons. Dad always said, “If you skimp on the chemicals, you buy a new fence next year.” Turns out, wood preservatives often contain compounds like 2,4,6-Trichlorophenol, which guard against decay fungi and bacteria. This stuff helps wood products last longer when exposed to moisture and insects, especially across parts of the country where humidity beats down year-round.
It’s not just wood that benefits from this chemical. 2,4,6-Trichlorophenol forms the backbone for other chemicals. Companies use it as a raw material for manufacturing pesticides, disinfectants, and dyes. Walk down any hardware store aisle and barnyards still rely on old-school pesticides to keep grains from being eaten or soiled by pests. 2,4,6-Trichlorophenol's chlorinated structure packs a serious punch against troublesome fungi and bacteria, and that’s why it shows up in so many compounds designed to tackle crop and animal pathogens.
Underlying Risks and Real Conversations
Plenty of folks have never heard of 2,4,6-Trichlorophenol until scientists started raising questions about what happens when these chemicals move beyond fences and farm fields. Researchers found links between this compound and potential health risks—including problems with the immune system and possible cancer connections. As someone who spent summers pulling weeds from treated fields without gloves, it feels uneasy to learn that skin contact or inhaling dust might have consequences decades down the line.
Regulators have taken action in many countries, adding restrictions on how much of these chemicals can be used and handled. Factories face serious scrutiny about what gets dumped into rivers and how much chemical residue sits on lumber or crops. There’s evidence of the chemical lingering in environmental samples, and chlorophenols like 2,4,6-Trichlorophenol break down slowly, which raises questions about safe disposal and long-term soil quality. As communities test their wells and local waterways, more people speak up about wanting transparency from industries and governments alike.
What Responsible Use Looks Like
The key isn’t just banning every tool that poses a risk but taking smart and measured approaches. Manufacturers have shifted toward closed systems that reduce worker exposure, and some companies invest in alternative preservatives that carry fewer baggage. Farmers, builders, and hobbyists look for certified products with clear labels and support initiatives to recycle old treated wood safely instead of tossing it on the nearest bonfire. Local governments fund water testing and make those results public, hoping to invite all parties to the table on decisions involving chemical use and neighborhood health.
There’s no perfect fix. Education plays a big role, from basic safety training on farms to updates at hardware stores and in classrooms. Community engagement keeps pressure on industries to improve practices and support better science. As research uncovers more about these chemicals, the hope is for a future with stronger protections and smarter choices—so that families don’t have to wonder whether fence repairs or a new garden patch will come with hidden costs years down the road.
Is 2,4,6-Trichlorophenol hazardous to health?
Looking at the Risks
Ask anyone who has worked around wood preservatives, pesticides, or certain disinfectants and they'll probably mention 2,4,6-Trichlorophenol. It’s a chemical often lurking in industrial settings, making its way into workplaces and, sometimes, neighborhoods. Inhaling the fumes or getting the powder on your skin isn't rare either, especially for folks handling treated lumber or some outdated agricultural products.
Research shows some real concerns. The U.S. Environmental Protection Agency flags 2,4,6-Trichlorophenol as a probable human carcinogen. This comes from lab tests where animals developed cancers after repeated exposures. It's not just the big cancer headline that matters. The chemical can cause burning eyes, irritation of nose and throat, and there are reports of headaches and dizziness in people who've spent time around it without proper protection. Occasionally workers describe skin rashes or burns—never a good sign.
More Than Just a Workplace Problem
People might think risks only belong in factories, but this chemical drifts into the larger world. It can seep into well water near old disposal sites or break down slowly in contaminated soil. Families in some communities learned this the hard way when chemical leaks traveled long after factories shut down. The bitterness is strongest with parents who look back wondering if their kids' asthma or skin issues tie to an invisible hazard they never knew was there.
Back in my college days near an old lumber plant, neighbors talked nervously about glowing puddles after rain—an unnatural sight tied to years of chemical use. That neighborhood had higher rates of odd illnesses and skin complaints. No one held proof, but worry became part of daily life. Years later, studies showed higher cancer clusters there compared to towns without contaminated water or soil.
Why Protection Still Falls Short
People assume workers wear gloves and respirators, but mistakes slip through. Old processing equipment, rushed shifts, and poor enforcement leave gaps. A single spill kicks up fumes or dust that linger. In places with weak regulation, protective gear collects more dust on shelves than on people’s hands. Regulations in one country barely match another, making global trade of treated products risky for unsuspecting consumers.
It’s not about paranoia—just listening to decades of doctors and industrial hygienists calling for stronger rules and regular monitoring. Those calls often run up against tight budgets or skepticism from decision makers, but ignoring warnings only leads to steeper bills in healthcare and cleanup.
What Works: Smarter Limits and Cleaner Alternatives
The track record changes when companies switch to less hazardous chemicals or redesign products so fewer preservatives are needed. Strong safety training mixed with up-to-date protective equipment cuts down most health incidents. Community right-to-know laws push companies to report spills quickly, so neighbors can act. Cleanup of old dump sites, while expensive, stops chemicals from spreading. Every time a region tightens up regulation and offers safer disposal, rates of related illnesses start dropping.
For everyday folks and workers alike, information stays power. Learning where chemicals land and asking questions about local industrial history makes a difference. Communities that push for periodic water and soil testing catch problems before they grow. This turns a slow environmental problem into something people can tackle before it brings more harm.
What are the safety precautions when handling 2,4,6-Trichlorophenol?
Why 2,4,6-Trichlorophenol Deserves Respect
2,4,6-Trichlorophenol grabs attention because it can hit health hard if handled without proper care. This stuff doesn’t just smell sharp—it can soak through skin, rise up as irritating fumes, and cause both immediate and lasting trouble. The International Agency for Research on Cancer lists it as possibly carcinogenic, and routine exposure links to issues like headaches, dizziness, skin rashes, and irritation of the nose and throat. These risks stack up quickly in labs, warehouses, and industrial plants.
Protective Gear Takes Priority
Nothing replaces solid gloves, goggles, a face shield, and chemical-resistant aprons. Nitrile or neoprene gloves won’t crumble when meeting a strong organic like 2,4,6-Trichlorophenol. Uncovered skin just asks for burns, rashes, or even deeper damage. Eye protection matters, too—splashes or dust can cause injury right away. Splash-proof goggles shield eyes from droplets, while a face shield blocks out sudden sprays.
Some situations need a respirator built for organic vapors, especially if the job stirs up dust or heats the chemical enough to send fumes into the air. Just cracking a window won’t cut it. Good ventilation drops exposure but wearing the right mask seals any gaps in protection. The National Institute for Occupational Safety and Health (NIOSH) recommends cartridge-type respirators for jobs where air levels run high.
Work Practices Mean More Than Rules
Using 2,4,6-Trichlorophenol safely takes more than reading a label or glancing at a messy safety poster. My own time in chemistry labs taught me that shortcuts set up problems down the line. Measure out enough for the task, stash leftovers in containers that really close, and slap on labels that actually tell people what's inside. Nobody wants a mystery spill on the counter or in storage.
Not every workplace treats chemical hygiene like it should, and some employees skip steps thinking nothing bad’s ever happened before. Clear protocols—like having showers and eyewash stations ready, double-checking that you’re not tracking chemical dust elsewhere, and knowing how to clean a spill fast—save pain and cash. A splash on clothing shouldn’t send someone hunting for an emergency manual.
Disposal Respects Environment and Law
Pouring 2,4,6-Trichlorophenol down the sink or tossing it in the trash adds poison to water and soil. That often breaks federal or local laws, and it threatens the community in ways most folks don’t see right away. Hazardous waste rules demand sealed containers and documentation, tracked all the way from use to disposal. Partnering with companies certified for hazardous material handling takes away the guesswork.
Spill kits—stocked with absorbent pads, neutralizing agents, and thick gloves—sit on shelves everywhere work with this compound happens. It's not overkill. The Environmental Protection Agency reminds businesses and researchers every year that quick cleanup keeps both people and wildlife safer. Mistakes become less costly when everybody knows who to call and what steps to take.
Building Safer Workplaces
Experience shows that safety habits stick better when training goes beyond legal checklists. Short workshops, live demos, and letting new hires practice with dummy chemicals helps everyone feel ready instead of anxious. Supervisors setting the tone and modeling safety makes it much harder for shortcuts to take over. Safety turns from a chore into a daily effort that benefits the whole crew.
2,4,6-Trichlorophenol makes big demands because it poses big risks. The right prevention, solid personal habits, and a workplace culture that puts people ahead of shortcuts form the best defense. If health and job security matter, cutting corners just isn’t worth it.
How should 2,4,6-Trichlorophenol be stored?
Why Storage Demands Respect
Anyone working with 2,4,6-Trichlorophenol sees right away this isn’t a substance you toss onto a shelf and forget. Having handled similar chemicals in real-world settings, I know personal and environmental safety can turn on a few simple choices. Mouthful of a name aside, 2,4,6-Trichlorophenol carries risks that deserve serious attention—the health dangers are real, and long-term effects can stack up if storage gets sloppy.
Health and Risk Factors Involved
People who’ve spent years in labs or chemical storage often know: even a small slip-up can mean exposure. This compound puts out a strong, unpleasant odor even at low concentrations. Inhalation or direct skin contact can touch off headaches, skin irritation, or worse. The U.S. Environmental Protection Agency lists it as a possible human carcinogen, and it doesn’t break down quickly in the environment. Manufacturing and research teams can’t afford mistakes around it, both for their health and for the people living nearby.
Keeping It Secure: What Works
Solid chemical safety starts with the right kind of storage container. In my experience and following regulatory guidance, high-density polyethylene drums or similar corrosion-resistant containers are the go-to. Avoiding reactive metals matters; this chemical doesn’t play nice with lightweight metals or aluminum. Containers must stay tightly sealed—not only to trap those sharp vapors but also to stop leaks and spills.
Temperature makes a difference, too. Cool, ventilated, and shaded spaces give the best results. 2,4,6-Trichlorophenol breaks down slowly, but direct sunlight speeds things up and lifts the chances of accidental release. Heat and moisture push up pressure inside the containers and may warp plastics, so good airflow and leak-proof floors help prevent headaches down the road.
Environmental Safeguards
In places I’ve worked, chemical storage always included spill containment—think heavy-duty trays or bunds that catch escapes before they go any further. Teams need to keep this chemical away from drains and groundwater, since contaminated water can travel and stick around. When someone disregards this, local wildlife and water treatment plants get the bill for that mistake.
Labeling and Documentation Count
Properly labeling storage containers turns chaos into order. Big, clear hazard symbols cut through confusion, especially when emergency teams respond. Safety data sheets, kept close by, help anyone nearby understand what to do in case of a spill or accidental contact. Audits by occupational safety groups often flag gaps here, so taking extra care with record-keeping isn’t wasted effort.
Training: The Human Factor
Technology and labels go so far, but people still make the biggest impact. Fire drills and spill response plans rarely seem important until something goes wrong. I’ve seen seasoned workers respond with calm during emergencies, and every company with good training programs churns out fewer horror stories. Regular refreshers, especially for anyone new, close the gaps that develop when routines slip or new chemicals enter the mix.
Looking Forward
New methods for chemical storage—from smart sensors to automated climate control—offer extra layers of protection, especially for large-scale operations. Still, personal responsibility and teamwork guide day-to-day safety. Respect for 2,4,6-Trichlorophenol involves more than a locked door; it’s about planning, constant awareness, and a willingness to speak up if something looks off.
Store smart. Protect people and the environment—it all starts in the storage room.What is the chemical formula of 2,4,6-Trichlorophenol?
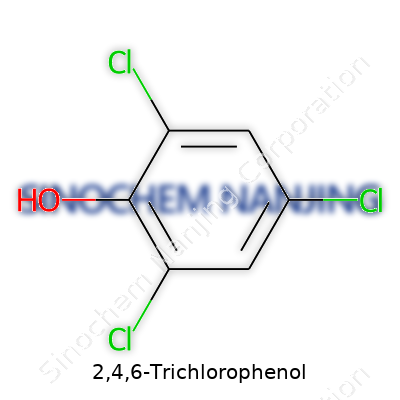
Unpacking a Simple Formula: C6H3Cl3O
2,4,6-Trichlorophenol has a chemical formula of C6H3Cl3O. That formula reflects three chlorine atoms plugging on the second, fourth, and sixth carbons of a phenol ring. Chemistry’s shorthand like this packs a lot of story in a series of letters and numbers. Someone picking up a bottle labeled “2,4,6-Trichlorophenol” should know that what’s inside isn’t some mystery mix. There’s a clear structure, and that matters from health, safety, and environmental standpoints.
Why Names and Formulas Count
Years ago in a college lab, I watched as a student mistook trichlorophenol for plain phenol, only to start coughing from the much stronger fumes. That lesson stuck with me: names aren’t just labels, they’re warnings and promises at once. A single atom swapped into an aromatic ring can change everything from boiling point to how the body reacts to an exposure. C6H3Cl3O is heavier, less volatile, and more toxic than basic phenol. It doesn’t break down easily, and that’s critical in work where safety goggles and gloves are not optional props, but your best chance to walk away healthy.
Health and the Environment
Exposure to trichlorophenol has drawn concern from health agencies worldwide. Studies link this compound to symptoms ranging from skin rashes to more serious organ effects after repeated or high-level contact. The U.S. Environmental Protection Agency groups it among hazardous substances to watch for in groundwater. Wastewater from chemical plants or wood treatment yards often drags trichlorophenol into rivers and streams, putting people and wildlife at risk.
An important fact: this chemical’s formula might look like a small tweak from similar compounds, but the health impacts change a lot. A trichlorinated phenol isn’t the same as pentachlorophenol, and each demands its own handling rules, disposal plans, and cleanup strategies. Even burning waste with this compound can produce dioxins, chemicals that don’t just go away. Cleanup, in my experience, means mobilizing experts and following strict protocols set by environmental law, not just papering over contamination with sand or soil.
Looking for Solutions
Science keeps searching for safer alternatives to chlorinated phenols. In wood preservation, for example, companies test new biocides with fewer environmental side effects. Regulating bodies have tightened storage and disposal requirements, with success stories showing that stricter rules can drop pollution levels in affected communities. Researchers are also working on ways to break down persistent compounds in the environment—using bacteria or advanced oxidation methods to zap those tough chlorine-carbon bonds.
Safer practices don’t just rely on new science, though. Labelling, clear communication, and job training make a world of difference. In workplaces, posting both names and formulas gives people the details they need to handle chemicals responsibly. Schools can teach young scientists to look past assumptions and check the molecular details before acting—because reading a formula right the first time means fewer accidents later on.
The Takeaway from a Simple Formula
Knowing chemical formulas isn’t some gatekeeping detail for elite scientists; it’s everyday protection for workers, the public, and ecosystems. Something as short as C6H3Cl3O reminds us that science, safety, and policy all add up. In a world facing challenges from contamination and industrial waste, getting the details right matters at every step.
| Names | |
| Preferred IUPAC name | 2,4,6-Trichlorophenol |
| Other names |
Dowcide 2 Phenaclor Captol Trichlorophenol T.C.P. Tretoclorol |
| Pronunciation | /ˌtraɪˌklɔːrəˈfiːnɒl/ |
| Identifiers | |
| CAS Number | 88-06-2 |
| Beilstein Reference | 803468 |
| ChEBI | CHEBI:3479 |
| ChEMBL | CHEMBL422 |
| ChemSpider | 7148 |
| DrugBank | DB07930 |
| ECHA InfoCard | 03be9a03-5664-4bdf-b628-6c3a018cb2b3 |
| EC Number | 1.14.13.20 |
| Gmelin Reference | 82238 |
| KEGG | C01343 |
| MeSH | D014258 |
| PubChem CID | 6879 |
| RTECS number | GZ2000000 |
| UNII | U2073INE9E |
| UN number | UN2811 |
| CompTox Dashboard (EPA) | DTXSID5020719 |
| Properties | |
| Chemical formula | C6H3Cl3O |
| Molar mass | 197.45 g/mol |
| Appearance | White to yellowish crystals |
| Odor | Phenolic; medicinal |
| Density | 1.485 g/cm³ |
| Solubility in water | 2.8 g/L (20 °C) |
| log P | 3.69 |
| Vapor pressure | 0.004 mmHg (25°C) |
| Acidity (pKa) | 7.2 |
| Basicity (pKb) | 7.9 |
| Magnetic susceptibility (χ) | -68.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.635 |
| Viscosity | 1.46 mPa·s (25 °C) |
| Dipole moment | 2.68 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 186.2 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -357.1 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1386.8 kJ/mol |
| Pharmacology | |
| ATC code | D08AE06 |
| Hazards | |
| Main hazards | Harmful if swallowed, causes severe skin burns and eye damage, toxic to aquatic life with long lasting effects. |
| GHS labelling | GHS07, GHS05, GHS06 |
| Pictograms | GHS06,GHS05,GHS09 |
| Signal word | Danger |
| Hazard statements | H302, H315, H318, H335 |
| Precautionary statements | P261, P280, P301+P312, P305+P351+P338, P330, P501 |
| NFPA 704 (fire diamond) | 2,4,6-Trichlorophenol NFPA 704: "3-2-0 |
| Flash point | 116°C |
| Autoignition temperature | 230 °C |
| Explosive limits | Explosive limits: 2.2–12% |
| Lethal dose or concentration | LD50 oral rat 580 mg/kg |
| LD50 (median dose) | LD50: 820 mg/kg (oral, rat) |
| NIOSH | DN0875000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of 2,4,6-Trichlorophenol: "5 mg/m3 |
| REL (Recommended) | 2.5 mg/m3 |
| IDLH (Immediate danger) | IDLH: 100 ppm |
| Related compounds | |
| Related compounds |
Phenol 2,4,6-Tribromophenol 2,4,6-Trifluorophenol 2,4-Dichlorophenol 2,6-Dichlorophenol |

