2,4,6-Trichloroaniline: A Close-Up Look at Its Story and Importance
Historical Development
Chemistry often carries the names and signatures of decades past. 2,4,6-Trichloroaniline surfaced from a period when the chemical industry pressed for new molecules with fresh potential, especially during the mid-20th century. Companies searched for ways to improve dyes, pesticides, and pharmaceuticals, leading them through aromatic chlorination routes. Early papers and industry notes show heavy interest in anilines, since their straightforward benzene backbone gave chemists a blank canvas for modification. The presence of three chlorines wasn’t an accident — consistent chlorination improved both chemical stability and biological activity, two traits deeply valued in agricultural and pharmaceutical settings back then.
Product Overview
2,4,6-Trichloroaniline isn’t the flashiest compound on the periodic table, but it pulls more weight than it gets credit for. The molecule sits as a fine crystalline solid, pale yellow or off-white, a far cry from the brightly colored dyes or reactive poisons that might result from its use. It’s made its mark as an intermediate, a necessary link in complicated syntheses for agrochemicals and specialty products. Some folks working in applied chemistry view this molecule sort of like flour in a bakery — hardly the finished good, but without it there'd be a lot less hitting the shelves.
Physical & Chemical Properties
Chemists start with what they can see, touch, or smell. 2,4,6-Trichloroaniline carries a distinct, sometimes lingering odor and doesn’t mind staking a claim on your skin with a yellow stain. Its melting point, above room temperature, keeps it from being a volatile risk in average settings. Looking at solubility, you find it's stubborn in water but dissolves more willingly in organic solvents. Its dense atomic structure means it holds up in storage, and high chlorination blocks degradation, so samples hang onto their original form over time. From firsthand lab work, this stubbornness sometimes means solvents and glassware spend a lot of time in the wash.
Technical Specifications & Labeling
Some will spend hours poring over specification sheets and purity records. Most production batches land above 98% purity, whether for research or final manufacturing. Labels flag its notable hazards: environmental persistence, toxicity to aquatic life, warnings about skin and eye contact. Import and transit rules pop up, given the molecule’s reputation and potential for misuse. Labels tie back to international standards — those GHS diamond labels and UN numbers don’t arise for no reason, and anyone working hands-on with 2,4,6-Trichloroaniline soon learns to respect them to avoid trouble, not just with regulators but also with their own health.
Preparation Method
The lab process for 2,4,6-Trichloroaniline draws on classic aromatic chemistry. Most syntheses start with aniline, a molecule both famous and infamous for its place in chemical history. Chemists introduce chlorines through controlled halogenation — too little and yields drop, too much and byproducts run rampant. Reaction temperatures, solvent choices, and chlorine sources turn into a balancing act. Years ago, many labs relied on direct chlorination, but concerns about byproducts like polychlorinated dibenzo-p-dioxins forced new, cleaner methods. That struggle — cleaner reactions, safer workspaces, and better control — tells a lot about how the industry edges toward green chemistry, step by painstaking step.
Chemical Reactions & Modifications
Once on hand, 2,4,6-Trichloroaniline doesn’t just sit on a shelf. Its three chlorines encourage a variety of further transformations. Nucleophilic substitutions, reduction reactions, and coupling processes transform it into more complex molecules. Chemists have harnessed those reactivity patterns for decades, especially in pesticide development or the chase for new pharmaceutical scaffolds. The benzene ring’s tendency to host substitutions means careful planning during reaction setup. Real-world work often mirrors this — I’ve watched teams tweak solvents and catalysts, searching for selectivity while minimizing nasty byproducts, and success brings real-world improvements rather than just academic footnotes.
Synonyms & Product Names
Those who read technical papers quickly spot a flock of aliases. You might encounter “1-amino-2,4,6-trichlorobenzene” or the slightly less revealing “TCA.” The product sometimes carries shorthand labels in inventory lists, reflecting habits and standards from different labs or countries. While this creates a headache during cross-border shipments or sharing research, it also reflects chemistry’s patchwork history of naming and renaming. It’s a reminder to double-check when ordering, since a mix-up in synonyms brings anything from minor delays to major safety protocol headaches.
Safety & Operational Standards
Few seasoned chemists underestimate the dangers lurking behind names like 2,4,6-Trichloroaniline. Strong gloves and eye protection aren’t for show, considering the compound’s track record for skin irritation and toxic effects. Good ventilation, fume hoods running at full strength — these become part of everyday workspaces. Regulatory authorities set clear exposure limits, referencing peer-reviewed studies on acute and chronic effects. Some universities and companies go further, holding regular safety drills and maintaining up-to-date spill kits. From personal experience, folks who take shortcuts often discover why those standards exist, sometimes the hard way.
Application Area
The reach of 2,4,6-Trichloroaniline runs across several big-ticket sectors. Crop protection stays high on the list, with the molecule feeding into the synthesis of specialized herbicides and fungicides. Its tough, stable frame makes it valuable in polymer and dye manufacturing. Environmental chemists study it as both a product and a byproduct, tracking its movement through soil and water systems, aware that persistence isn’t always a good thing. Industrial research tends to explore new uses or modifications, searching for ways to enhance activity or reduce harm in both agricultural fields and municipal systems.
Research & Development
Modern R&D work focused on 2,4,6-Trichloroaniline looks beyond just synthesizing it efficiently. Teams explore how structural adjustments influence properties, aiming for compounds with higher selectivity and lower toxicity. Green chemistry principles guide newer projects, pushing for reduced waste, safer reagents, and milder reaction conditions. The scientific journals fill with report after report analyzing impurities, yield optimization, or structure-activity relationships. For academic research, the molecule serves as a model for studying electron distribution in aromatic systems, contributing to our understanding of reactivity and environmental behavior. Companies in research alliances often share their protocols or findings, hoping to push the field forward without putting sensitive IP at risk.
Toxicity Research
Toxicologists treat 2,4,6-Trichloroaniline with genuine caution. Lab and field studies point to risks — acute effects kick in at low exposures, causing everything from skin rashes to more serious organ impacts. Long-term studies raise flags about bioaccumulation and the molecule traveling up food chains. Governments have responded with stricter discharge limits and environmental monitoring requirements. It’s not just concern for humans, either; aquatic ecosystems show the greatest vulnerability. Some of the best advice comes from environmental chemists who urge regular monitoring of effluent streams and push for better cleanup tech. Real-life cases of mishandling led to stricter protocols, with persistent lobbying by watchdog groups keeping the issue alive in both the regulatory and public spheres.
Future Prospects
The future for 2,4,6-Trichloroaniline remains tied to balancing usefulness and risk. Demand from agricultural and specialty chemical markets isn’t vanishing, so safe production and responsible application carry more weight than ever. Researchers aim to develop analogues with less environmental impact, and regulatory bodies highlight the need for new tech in waste treatment. Promising work involves bioremediation — bacteria and plants offering hope for cleaning up sites affected by persistent chlorinated compounds. Emerging catalysis and greener synthetic methods give some optimism, striving for a world where needed chemicals exist without the heavy clean-up bill. The story of 2,4,6-Trichloroaniline isn’t just about chemistry, it reflects broader conversations on responsibility, risk, and the legacies left behind by everyday industrial choices.
What are the main uses of 2,4,6-Trichloroaniline?
Real-World Applications Rooted in Chemistry
Anyone who has spent time in a chemistry lab or in industrial production learns to respect the power of specialty chemicals and intermediates. 2,4,6-Trichloroaniline, a compound with three chlorine atoms attached to an aniline ring, fits into this category. Its chemical structure opens the door to several practical uses, especially in fields that affect daily life more than most people realize.
Turning Building Blocks Into Beneficial Products
Chemists often chase after reliable intermediates to streamline the manufacture of complex goods. 2,4,6-Trichloroaniline stands out as a trusted intermediate in the synthesis of dyes, agricultural chemicals, and pharmaceuticals. In one of my first industry jobs, everyone in the plant talked about the importance of this compound in producing advanced colorants. Dyes incorporating this molecule end up in textiles, plastics, and inks. Textile factories, for instance, rely on consistent color-fastness and vibrancy. Compounds like this help deliver those qualities.
Pesticides and Herbicides: A Double-Edged Sword
Farmers look for solutions to control weeds and pests. Chemical companies use 2,4,6-Trichloroaniline to manufacture several herbicides and fungicides. Many wheat and corn fields across the world stay productive because these chemicals help protect crops from blight and disease. I’ve met agricultural extension workers who juggle food supply challenges with concerns about runoff and accumulation of residues in soil and water. These real consequences remind users and regulators to keep a watchful eye on environmental and health safety profiles.
Pharmaceutical Synthesis: Feeding Innovation
Some important medicines begin as fairly simple organic molecules. 2,4,6-Trichloroaniline helps scientists build complex examples, especially in the field of antimicrobial agents. Years ago, I worked alongside chemists synthesizing new molecules for research stages. Many trial drugs and research targets grew from the “skeletons” built using this compound. Research journals describe it as a springboard for molecules that disrupt bacterial functions in clinical trials.
Environmental and Health Considerations: A Balancing Act
Every tool in industrial chemistry brings trade-offs. Research at major universities and government agencies keeps uncovering information about the toxicity, persistence, and breakdown of 2,4,6-Trichloroaniline. Chronic exposure in manufacturing workers calls for careful monitoring, with guidelines from OSHA and similar organizations worldwide. Wastewater treatment plants now run pilot projects to try and filter or break down residual organochlorine compounds before water returns to rivers. Groups focused on public health and safety keep pushing for green chemistry options that use safer alternatives or closed-loop recycling.
Ways Forward: Smarter Chemistry and Safer Practices
Improving reactor efficiency and containment methods can limit exposure and waste in industrial facilities. Research teams are searching for biodegradable alternatives that break down without harming ecosystems. Plugging in automation and better filtration can ease some risks. Building trust with communities, keeping up with the latest regulatory changes, and investing in cleaner protocols reflects not just technical wisdom but a sense of responsibility. Learning from these efforts can help other chemicals follow safer paths from lab benches to the market.
What are the safety precautions for handling 2,4,6-Trichloroaniline?
Why 2,4,6-Trichloroaniline Demands Respect
People who spend time working near chemicals quickly learn not to trust the seemingly simple ones. 2,4,6-Trichloroaniline gets used a fair bit in research, synthesis, and specialty chemical manufacturing. Its strong smell and crystalline texture might not give away much, but there’s risk tucked beneath the surface—breathing the dust, touching it, or letting it mix with other materials can bring real harm to your health.
Everyday Hazards: Skin, Lungs, and Eyes
Contact with 2,4,6-Trichloroaniline often means skin irritation, especially if you’ve got sensitive patches or open cuts. It absorbs faster through broken skin, so gloves that fit and don’t tear matter more than people think. Splashes in the eyes sting and can lead to lasting problems. Inhaling sneaky, powdery particles may irritate your lungs, causing wheezing or coughing, especially among folks with asthma or allergies.
Smart Precautions Worth Practicing
Putting simple barriers between yourself and chemical contact takes stress off everyone in the lab or workshop. Nitrile gloves, safety goggles with side shields, and a lab coat with long sleeves form a reliable base layer. Fix any cracks in goggles and toss gloves right after handling. I’ve seen coworkers wipe sweat away with gloves that carried residue—after that, nobody forgot to swap them out.
Work inside a chemical fume hood, never on an open bench. Airflow inside the hood keeps invisible particles from drifting your way. Even in short sessions, indoor air quickly fills with more than you expect. Clean up right after, sealing waste in a labeled, leakproof bag for disposal. Never leave open containers overnight—vapors and dust build up fast.
Some days, supplies run short. If the only gloves left feel thin or the fume hood sputters, don’t push your luck. Postponing the work until things are fixed makes a difference. People rarely regret waiting but often regret rushing.
Staying Informed: Know Your Risks
New hires often breeze past safety sheets. It’s tempting to skim, but I’ve learned firsthand that knowing exactly what reactions to expect—skin, steam, or catastrophic mixing with another chemical—brings real confidence. Safety Data Sheets for 2,4,6-Trichloroaniline spell out reactions to acids, bases, or heat sources. Read them closely, keep them nearby, and ask questions if something feels unclear.
Good training shows how to rinse spills with water and baking soda, bag up wipes and gloves, and make the right emergency call. Know where the eyewash station and shower stand. Walking through the steps before an accident means fewer panicked decisions when one happens.
Building a Culture That Values Carefulness
In busy labs or factories, speed often gets celebrated more than safety. Taking a moment to point out broken goggles or cleaning up after a spill pays off. Pressure to meet deadlines fades, but the memory of a bad chemical injury sticks with you. People trust coworkers who practice safety and share tips. Taking extra steps might slow things down slightly. At the end of the day, everyone heads home healthy, and that’s the standard worth upholding every time.
What is the chemical formula and molecular weight of 2,4,6-Trichloroaniline?
Chemical Formula Basics
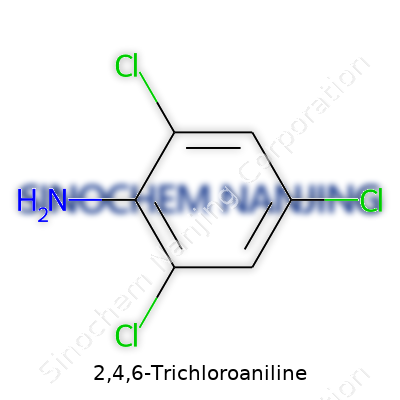
2,4,6-Trichloroaniline means what it says: an aniline ring with three chlorine atoms hanging off at specific spots on the benzene ring. Chemists make sense of this by using its chemical formula, C6H4Cl3NH2.
You get six carbons from the benzene base, four hydrogens because three have been switched out with chlorines, three chlorines, and the NH2 group—the signature of an aniline. Each part tells a story about how reactive and distinctive this compound can get.
Molecular Weight
People often think molecular weight is just classroom trivia, but it's not. Laboratory folks, pharmacists, and chemical engineers rely on this number every single day. For 2,4,6-Trichloroaniline, the calculation adds up like this:
- Carbon (C): 12.01 × 6 = 72.06
- Hydrogen (H): 1.01 × 4 = 4.04
- Chlorine (Cl): 35.45 × 3 = 106.35
- Nitrogen (N): 14.01 × 1 = 14.01
- Hydrogen (from NH2): 1.01 × 2 = 2.02
Add those together and you get a molecular weight of about 197.55 g/mol. That number matters more than folks realize. Anyone measuring doses, calculating yields, or doing environmental checks depends on an accurate molecular weight.
What 2,4,6-Trichloroaniline Means in Real Life
It only takes a quick look at regulatory watchlists to see why this compound gets attention. As someone who's worked in labs where synthetic dyes and herbicide ingredients make their rounds, I've seen firsthand the attention paid to substances like this one. You’re looking at an intermediate that’s not only crucial for making specialty chemicals, but also flagged for potential health impacts.
Many pollution headlines trace back to aromatic amines and chlorinated derivatives. These chemicals don’t just vanish—they stick around in soil and water, and research has shown persistence can sometimes mean trouble for wildlife and humans. The chlorines amplify toxicity, and watching how even a simple change—like three chlorines instead of none—transforms a compound offers a quick chemistry lesson in why molecular details matter.
Why Care About Details Like These?
Working in chemical safety, you end up valuing precision—someone’s mislabelled bottle or a misread label can derail a whole project or worse, create a genuine hazard. A clear formula and the correct molecular weight help avoid accidents and mistakes. For a supplier, these details help set up safe handling procedures. For wastewater engineers, these clues hint at how aggressive the cleanup process might need to be. Researchers stuck on stubborn chromatograms need sharp chemical info to pin down their unknowns.
Companies and agencies talk a lot about "responsible stewardship." In reality, this means triple-checking substances like 2,4,6-Trichloroaniline. Making sure labs label everything correctly and that downstream users know what they're dealing with helps prevent bigger headaches later. One overlooked digit in molecular weight, or a skipped safety sheet, and suddenly it's not just chemistry anymore—it's a policy and public health issue.
Looking for Solutions
Safer chemistry doesn't come from crossing your fingers; it comes from checking your facts. Substituting hazardous intermediates, investing in greener manufacturing routes, and enforcing clearer labeling can all lower the risks associated with compounds in this family. My experience says nothing beats good record-keeping, open training, and steady updates to guidelines to keep up with what we keep learning about substances like 2,4,6-Trichloroaniline.
How should 2,4,6-Trichloroaniline be stored?
Understanding the Real Risks
People often downplay the hazards that certain chemicals bring into labs and storage rooms. 2,4,6-Trichloroaniline falls in that camp. Its name sounds like a line from a chemistry exam, but the risks aren’t so academic. This compound can irritate skin and eyes, cause respiratory trouble, and spill risks stick around longer than most folks think. So the way a facility stores this substance shapes both workplace safety and long-term health for workers.
Security Over Convenience
Leaving 2,4,6-Trichloroaniline containers half-open or stacked carelessly can spell trouble. Vapors, even in small amounts, pose danger over time. Store it in tightly sealed containers that don’t react with the compound—glass or certain high-quality plastics. The storage area needs to breathe easily, meaning a well-designed fume hood or ventilated space keeps building up vapor at bay. In older storage rooms that rely on old-school vent fans, air may recirculate instead of moving outside, trapping risk inside four walls.
Don’t Mix With the Wrong Crowd
Putting chemicals on any shelf that’s empty tempts fate. 2,4,6-Trichloroaniline reacts badly with oxidizers, acids, and strong bases. Mixing it up on the same rack with bleach or ammonia-based products leads to dangerous reactions if a spill happens. Use dedicated shelving marked for chlorinated compounds or organic solids. Label shelves clearly so new staff and old pros both know exactly what’s nearby. Mistakes almost always happen at busy times or during clean-ups, not when everyone is on alert.
Temperature and Moisture: Silent Saboteurs
Heat and humidity ruin any plan for safe storage. 2,4,6-Trichloroaniline likes cool and dry spaces, away from direct sunlight or locker lights that run hot all day. Humid conditions let small amounts break down, leading to slow leaks and risks that don’t always show up right away. Folks in tropical climates or older buildings need to check for leaks, damaged seals, or unexpected chemical changes more often. Risk isn’t some theoretical problem; you can smell it before you see it.
Staying Ready For Trouble
Accidents don’t send warning signs. Every space that stores 2,4,6-Trichloroaniline should have eye wash stations and safety showers close by, plus gloves and goggles built for chemical work—not simple dust masks from a hardware store. Spill kits sit useless if hidden behind boxes or locked cabinets. Staff must walk through spill response regularly, not just once during orientation. If local fire departments don’t know what’s on site, nobody gets proper help in an emergency.
Documentation and Real Accountability
Paperwork often gets ignored until trouble shows up. Every time someone moves, opens, or disposes of 2,4,6-Trichloroaniline, logs make the difference between a quick fix and a mystery. Chemical inventory systems, digital or on paper, help keep tabs on quantities and storage locations. Surprise visits by inspectors go smoother with detailed records. Simple honesty—if a compound seems off, report it right away—keeps everyone safer. Safety isn’t about following rules; it’s about sending people home just as healthy as they arrived.
Looking Ahead
Lab culture shapes how storage gets handled. Facilities that prioritize safety train new staff thoroughly and hold each other accountable. Installing environmental monitors keeps track of any vapor leaks early. Workers walk in every day knowing simple steps help prevent problems down the line. Experience teaches everyone the same lesson: shortcuts make today’s job easier, but bring tomorrow’s regret.
What are the potential health hazards of 2,4,6-Trichloroaniline exposure?
Everyday Risks Hiding in the Lab and Workplace
Anyone who’s worked around chemical plants, research labs, or certain manufacturing lines has probably brushed up against some intimidating ingredients. Chemicals like 2,4,6-Trichloroaniline fly under the radar—unless you know a bit of organic chemistry, you’re not going to spot its dangers just from the name. But this compound deserves some attention, because it’s a regular player in producing dyes, pesticides, and pharmaceuticals. People can run into it without even knowing, simply by being near the places that handle it.
How 2,4,6-Trichloroaniline Can Hurt
I’ve seen the way seasoned lab workers reach for gloves and fume hoods without a second thought. They know many of these aromatic amines can hit hard, and 2,4,6-Trichloroaniline is no exception. Even skin contact can cause irritation or allergic reactions—a rash or burning that keeps spreading if someone doesn’t wash up fast. Breathing dust or vapors leads to coughing, sore throat, and sometimes headaches. Anyone who’s handled strong, chlorine-laden chemicals recognizes that smell. Once those tiny particles get in the airways, it’s hard to ignore them.
The real concerns start showing up over repeated or high-level exposure. Some studies link similar chlorinated anilines to anemia, particularly types where red blood cells break down faster than the body can replace them. Signs include fatigue, dizziness, and skin that looks just a bit too pale. Long-term, experiments on animals point toward changes in liver and kidney function, hinting at stresses that don’t always come to the surface until it’s late in the game. The International Agency for Research on Cancer hasn’t placed 2,4,6-Trichloroaniline in its most dangerous categories, but that should never suggest it’s harmless—cautious handling makes sense.
Looking Out for Yourself and Your Crew
Safety lines sometimes get crossed because people forget how unpredictable some chemical reactions can be, or they trust air scrubbers to do the whole job. In real workplaces, spills happen, and tiny leaks sneak past detection. Getting into a rhythm—always wearing protection, always working in ventilated spaces—makes those small accidents just a blip instead of a crisis. I remember long days in a university lab, where the most experienced staff checked hoods and gloves without lecturing anyone about it; they set the tone just by doing it themselves.
Tracking air and surface contamination with regular tests acts as an early warning. Simple things like well-marked storage and updated safety sheets crowd out confusion when emergencies pop up. Sometimes people cut corners on training, thinking it slows work down, but time spent showing someone how to handle reactive chemicals pays back a hundredfold. Personal experience tells me there’s a world of difference between “We’ve always done it that way” and “We double-check because we know what can go wrong.”
Moving Toward Stronger Protection
Tighter occupational rules reduce the odds of health trouble popping up years later. Federal agencies recommend keeping exposure as low as technology allows, not just ticking boxes on forms. Substituting safer compounds whenever possible provides another buffer, but industry change rarely comes overnight. Until that shift happens, having a culture that values open communication about near-misses and exposures helps everyone spot patterns early. Sharing close calls—without punishment—means near misses turn into lessons instead of headlines.
People trust each other with safety every day in labs and factories. Even if 2,4,6-Trichloroaniline rarely makes news, its health hazards deserve respect and steady attention.
| Names | |
| Preferred IUPAC name | 2,4,6-Trichlorobenzenamine |
| Other names |
2,4,6-Trichlorobenzenamine 2,4,6-Trichlorophenylamine |
| Pronunciation | /ˌtraɪˌklɔːroʊˈænɪliːn/ |
| Identifiers | |
| CAS Number | 634-93-5 |
| 3D model (JSmol) | `3D Model (JSmol) string for 2,4,6-Trichloroaniline:` ``` NC1=CC(Cl)=CC(Cl)=C1Cl ``` |
| Beilstein Reference | 77199 |
| ChEBI | CHEBI:38762 |
| ChEMBL | CHEMBL16604 |
| ChemSpider | 12051 |
| DrugBank | DB13167 |
| ECHA InfoCard | 19b9976590-2ace-4c32-bde1-d00b418f477d |
| EC Number | 202-450-1 |
| Gmelin Reference | 787 |
| KEGG | C06581 |
| MeSH | D014277 |
| PubChem CID | 6925 |
| RTECS number | CAS7177 |
| UNII | K0R02IUG3J |
| UN number | 2811 |
| CompTox Dashboard (EPA) | DTXSID2038636 |
| Properties | |
| Chemical formula | C6H4Cl3N |
| Molar mass | 197.46 g/mol |
| Appearance | White to pale cream solid |
| Odor | Odorless |
| Density | 1.56 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | 2.9 |
| Vapor pressure | 0.00013 mmHg (25°C) |
| Acidity (pKa) | 3.68 |
| Basicity (pKb) | 7.45 |
| Magnetic susceptibility (χ) | -62.2·10^-6 cm³/mol |
| Refractive index (nD) | 1.661 |
| Viscosity | 1.48 cP (25°C) |
| Dipole moment | 2.87 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 276.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -68.5 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -1968.1 kJ·mol⁻¹ |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and serious eye irritation, may cause an allergic skin reaction, toxic to aquatic life with long lasting effects |
| GHS labelling | GHS05, GHS07, GHS09 |
| Pictograms | GHS06,GHS09 |
| Signal word | Warning |
| Hazard statements | H301, H315, H319, H332, H373 |
| Precautionary statements | P261, P264, P270, P271, P301+P312, P330, P405, P501 |
| NFPA 704 (fire diamond) | 2,4,6-Trichloroaniline: "2-2-0 |
| Flash point | 99°C |
| Autoignition temperature | 240 °C |
| Lethal dose or concentration | LD50 oral rat 850 mg/kg |
| LD50 (median dose) | LD50 (median dose): 1300 mg/kg (oral, rat) |
| NIOSH | SN3675000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.5 mg/m3 |
| IDLH (Immediate danger) | 50 mg/m3 |
| Related compounds | |
| Related compounds |
Aniline 2-Chloroaniline 4-Chloroaniline 2,4-Dichloroaniline 2,6-Dichloroaniline 2,4,6-Tribromoaniline |

