Understanding 2,4,6-Tribromoaniline: Chemistry’s Quiet Powerhouse
The Story Behind 2,4,6-Tribromoaniline
History often tells us more about what we value than we realize. 2,4,6-Tribromoaniline climbed onto the chemical stage during the expansion of organic synthetic chemistry in the late 19th and early 20th centuries. Early chemists, interested in manipulating the aniline scaffold, learned that introducing bromine atoms at specific positions (the 2, 4, and 6 spots on the benzene ring) changed not only the physical appearance but also the reactivity and applications of the molecule. As someone who’s handled aromatic amines in the lab, I always marvel at how a few atoms’ difference can have such outsized impact. Researchers at the time were pursuing dyes, pharmaceutical intermediates, and new agents for research, and 2,4,6-trihalogenated compounds like this one popped up as versatile pieces in their toolkits. Academic curiosity and industrial need often go hand in hand, and so the compound’s use spread across both settings.
A Look at the Molecule Itself
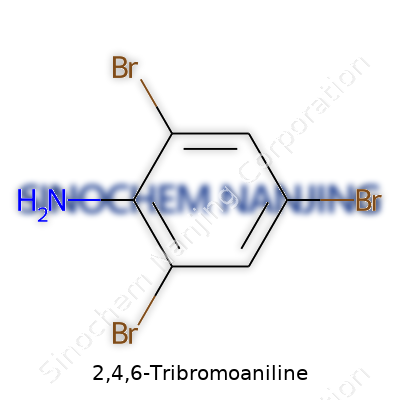
The structure seems simple—a benzene ring with three bromines sitting at positions 2, 4, and 6, along with an amine group. This gives it a C6H2Br3NH2 skeleton. The brick-red to brownish color of its crystals reminds you something interesting is locked away inside. Solid at room temperature, it doesn’t give off much odor. With three heavy bromine atoms and a relatively low melting point (usually in the 150-160°C range), it fits the physical profile you might expect for a polybrominated aromatic. It’s only sparingly soluble in water, reminding chemists to reach for organic solvents when handling it. Reactivity is where things get lively: the electron-withdrawing effect of bromine tames the activity of the amine, a quirk that keeps the molecule stable under most bench-top conditions. Years working with these compounds makes you appreciate how much brute force—heat, catalysts, or strong acids—are sometimes needed to break them down or modify them.
Preparing 2,4,6-Tribromoaniline: Tradition Meets Precision
Old-school chemistry texts describe its preparation in straightforward terms. The typical route begins with aniline (cheap, widely available) reacted with excess bromine in an appropriate solvent; bromine drops into the ortho and para positions thanks to the directing power of the amino group, so you land on 2,4,6-tribromoaniline almost exclusively. Practically, controlling temperature and bromine addition rate makes all the difference—move too fast and you lose selectivity, bring in too much heat and you invite tar or side-products. Filtering out the product after cooling and recrystallizing from ethanol offers a decent yield. Today’s labs might tweak the process for environmental or safety concerns, but the essential chemistry remains almost unchanged over a century. The method’s directness appeals to anyone who likes chemical reactions that just work, with minimal fuss.
Labeling, Names, and Synonyms—Clarity in a Complex World
Chemicals go by plenty of names, and 2,4,6-tribromoaniline’s list features both systematic and informal ones. Synonyms like tribromoaniline, tribromoaminobenzene, or even by their CAS number, all point you to the same brick-red powder. In any lab I’ve worked, we trust both the label and our own instincts—one colleague’s horror story involved grabbing the wrong aniline derivative and scrambling to clean up an unwanted byproduct. Accurate labeling remains fundamental, especially as chemical supply chains globalize and move faster; mislabeling means, at best, wasted experiments and, at worst, risk of harm.
Frontiers in Chemical Reactivity and Modification
2,4,6-Tribromoaniline stands out for its versatility, but that comes from its bromines and amine group playing unique roles. Direct dehalogenation can transform it into less-brominated anilines, or coupling with other starting materials opens paths toward heterocycles and more complex molecules. N-derivatization—adding groups to the nitrogen—shifts the molecule’s identity again, allowing entry into dye chemistry or pharmaceutical precursor work. Most modifications require precise conditions, since brominated aromatics resist change unless given the right push. The molecule delivers for those who stubbornly chase challenging syntheses, but it rewards patience and good technique. Reliable reports detail electrophilic substitutions, nucleophilic displacement on the bromine positions, and cross-coupling reactions as consistent ways to breathe new life into the compound.
Applications: Bridging Lab Bench and Industrial Scale
Applications for 2,4,6-tribromoaniline go far beyond textbook curiosity. In practice, it serves as a solid precursor in the dye and pigment industry, supporting vivid hues that wouldn’t stick around without such robust aromatic scaffolds. The presence of those bromine atoms makes it effective for synthesizing fire retardants—brominated aromatics disrupt the combustion cycle, so you see them used as additives in plastics, textiles, and insulation materials. Academic chemists value the compound for preparing more elaborate materials, including ligands for catalysis or molecular building blocks in targeted pharmaceutical synthesis. For students, running the tribromination reaction offers a vivid demonstration of aromatic substitution, bridging concepts learned in lecture with real-world outcomes seen in the flask.
Thinking About Safety—Lessons from the Lab
Personal experience has taught me the respect these compounds command. 2,4,6-Tribromoaniline falls under the umbrella of "handled with care" aromatic amines. Gloves, goggles, and proper fume hood technique are baseline practice, especially since any aromatic amine can sometimes be absorbed through skin or volatilize in an open lab. Safety data points to hazards with both dust inhalation and skin contact, and disposing of brominated wastes requires diligence because incineration can generate toxic byproducts like dioxins or furans. Years spent teaching new lab users underscores how much safer everyone is when storage containers are clearly labeled, spill protocols rehearsed, and personal protective equipment worn without exception.
Pushing the Frontier: Research, Development, and Future Prospects
Researchers keep finding new niches for compounds like 2,4,6-tribromoaniline. In materials science, there’s growing interest in using highly brominated aromatics as starting points for new polymers or as catalysts. The pharmaceutical sector keeps exploring halogenated scaffolds looking for novel activity. Advances in cross-coupling chemistry let scientists break and remake bonds no one could touch a few decades ago, so the list of possible derivatives expands year by year. On the toxicity front, environmental chemists keep close watch—worries over persistence and bioaccumulation shape regulations and drive search for greener alternatives. I’ve seen new grads gravitate toward research targeting less hazardous synthesis routes and faster, cleaner reactions, which is good for both bench chemists and the planet. Looking ahead, policy pressure, commercial needs, and academic innovation will keep pushing the story of 2,4,6-tribromoaniline forward.
Unraveling Toxicity—What Science Tells Us
Toxicity research into brominated aromatics uncovers a bag of issues. Studies find these molecules generally resist natural degradation and can persist in soil and water. Some can interfere with endocrine function or act as mild irritants, though 2,4,6-tribromoaniline doesn’t top the danger charts like its cousins in the polybrominated diphenyl ether or biphenyl classes. Lab tests for acute exposure focus on liver and kidney stress in rodents, contamination of waterways, and the risk of accidental inhalation or skin loading. Staying on top of the relevant literature demands both scientific curiosity and a sense of responsibility—a single lapse in waste handling or a poorly documented experiment carries risk for both individuals and broader communities. From what I’ve seen, continuous review and serious commitment to green chemistry hold the most promise for lowering long-term risks.
What the Future Holds
The future of 2,4,6-tribromoaniline will ride on our need for robust, customizable molecules in sectors from electronics to medicine. Regulatory shifts and consumer demand for less-toxic components put pressure on both suppliers and end-users to find safer, more sustainable alternatives—even when performance is hard to match. Where past generations valued brute chemical force, today’s priorities turn more toward minimizing environmental footprint, improving worker safety, and fully understanding the fate of every molecule released. Efforts aimed at repurposing industrial byproducts, minimizing or recycling brominated wastes, and developing targeted dehalogenation catalysts may open new paths, blending chemical innovation with stewardship. The molecule’s history and current importance show how much progress depends on honest assessment of risks, transparent reporting, and open-minded pursuit of better ways to meet human needs.
What are the main uses of 2,4,6-Tribromoaniline?
Digging Into Its Roots in Chemical Synthesis
Some chemicals serve as building blocks for countless products around us, and 2,4,6-tribromoaniline is one of those quiet workhorses. Technicians rely on it in organic synthesis, piecing together more complex molecules for research labs and factories. The three bromine atoms attached to the aniline ring give it a special ability to act as a starting point for other halogenated compounds. This chemical opens possibilities for crafting dyes, pharmaceuticals, or specialty polymers where traditional starting materials fall short.
Developing Pharmaceuticals and Fine Chemicals
Most people don’t see where medicines begin, but chemists in pharmaceutical research often lean on 2,4,6-tribromoaniline when they aim to produce brominated intermediates. These intermediates become ingredients for active pharmaceutical compounds. Sometimes, subtle tweaks to a molecule’s structure—like those offered by this brominated aniline—turn an inactive compound into something with real clinical promise. Data from research journals and the chemical supply industry confirm its growing use in the development pipeline for new drug candidates.
Dye and Pigment Production
Color manufacturers need reliable ways to make consistent, vivid hues. Thanks to the bromine atoms on its ring, 2,4,6-tribromoaniline works well as a stepping stone in the production of specialty dyes and pigments. Its aromatic base stands up to demanding processing steps, yielding colorants for textiles, plastics, inks, and coatings. The colorfastness and chemical resistance of these dyes bring value, translating into materials that last longer or withstand harsh conditions. That’s a win for both manufacturers and end users who expect better-looking, longer-lasting goods.
Role in Agrochemical Formulation
Agrochemical chemists have found that halogenated anilines often help them build pesticides and herbicides that remain stable and active under field conditions. 2,4,6-Tribromoaniline steps into these roles as an efficient intermediate, letting formulators tune molecular properties like volatility and water solubility. These tweaks often mean more effective pest control and reduced losses for farmers, translating into safer food supplies and better yields. Industry case studies highlight its use in select insecticides and fungicides, especially newer ones that must pass stricter regulatory checks.
What About Environmental and Safety Considerations?
Any discussion about a chemical like 2,4,6-tribromoaniline brings up important safety questions. Direct handling requires careful control—no room for shortcuts with personal protective gear and ventilation since exposure can cause skin irritation or worse. Regulatory bodies, including the EPA and European Chemicals Agency, keep their eyes on substances in this family because of potential environmental persistence. Companies using or producing this chemical do best when they follow best practices in waste management and invest in greener chemistry where possible. Real progress comes from collaboration between researchers, regulatory agencies, and manufacturers pushing for safer, cleaner processes.
Finding the Balance for the Future
In my years of following developments in chemical manufacturing, responsibility comes up as the backbone of progress. The history of 2,4,6-tribromoaniline shows that with the right balance—innovation paired with care for worker safety and environmental impact—the benefits reach all sectors of society. Science throws interesting challenges our way, but experience suggests the most sustainable advances come from continual learning, transparent reporting, and honest risk assessment. As chemistry moves forward, stories like this remind us why informed choices matter.
What is the chemical formula and molecular weight of 2,4,6-Tribromoaniline?
Chemical Details: Getting the Basics Right
2,4,6-Tribromoaniline stands as a brominated aromatic amine, a quiet performer in complex chemical syntheses and research circles. Its chemical formula is C6H4Br3N and its molecular weight comes out to 345.82 g/mol. These numbers may seem like dry facts, but both play crucial roles in applications and lab safety. Those values help a chemist set up accurate molar calculations, choose proper reagents, and assess hazards. Whether for pharmaceutical research, material synthesis, or educational labs, knowing what you’re handling is essential. Mistaking a formula in a reaction can throw off yields or, worse, produce unintended byproducts. Miscalculating the molecular weight can lead to dosing errors or dangerous outcomes if scale-up or new compounds are the goal.
Why Brominated Aromatic Amines Matter
Compounds like 2,4,6-Tribromoaniline don’t draw big attention, but they sit behind breakthroughs in dye chemistry, pharmaceutical intermediates, and agrochemical development. Bromine atoms on the aromatic ring change both reactivity and toxicity. The amine group, sticking up at the para position, offers a classic entry point for further functionalization. I once worked on an undergraduate organic synthesis where a mislabelled bottle led the team to use an unsubstituted aniline rather than a brominated analogue. The entire reaction route had to be modified mid-lab, and our final product failed the purity test. It drove home the point that overlooking little numbers like atomic weights and formulas means more than just “bad math”—it messes with the chemistry and safety, too.
Hazards Tied to Formula and Structure
Bringing three heavy bromine atoms onto a benzene ring pushes up both the density and potential hazards of 2,4,6-Tribromoaniline. One missed calculation, and exposure limits get crossed in an instant—especially for amateur chemists who mix up molar equivalents when setting up a reaction. The brominated structure means more persistence in the environment and tougher clean-up procedures. Proper identification and use of this compound can help prevent exposures, spills, and tricky waste management headaches. It’s smart to read safety data sheets and lean on trusted resources like PubChem or Sigma-Aldrich for updated safety protocols.
Applying This Knowledge: Safer, Smarter Chemistry
Chemistry classrooms and research labs both benefit from teaching with accurate chemical details. A solid grip on formulas and weights makes for safer experimental setups, fewer stopped reactions, and confidence in scale-up. For teachers, it pays to double-check chemical inventories and lesson materials at the start of each semester. Keeping clear records and referencing established scientific databases can spare a lot of trouble down the road.
Students and researchers alike get a better shot at breakthroughs when the basics are nailed down. The great discoveries rarely start with a headline compound; they begin with careful attention to known values—like the 345.82 g/mol on a modest bottle of 2,4,6-Tribromoaniline. Investing a few minutes in cross-checking those details has saved me a lot of headaches over the years. If you know what you’re working with, you’re already one step closer to insightful, reproducible, and safe research.
How should 2,4,6-Tribromoaniline be stored and handled safely?
Knowing What You’re Dealing With
Anyone who has spent time in a chemistry lab understands the uneasy relationship between curiosity and caution. 2,4,6-Tribromoaniline isn’t a household staple. It’s a specialty compound, showing up mostly in research and industry settings. The triple bromine atoms aren’t just there for decoration; this molecule means business. If you set it on an open bench, you could be risking more than just minor irritation—think along the lines of respiratory and skin hazards. I’ve seen too many people brush off the importance of chemical safety until it’s too late. Once, a misplaced reagent led to an emergency eyewash sprint. It only takes one close call to realize the standards exist for good reason.
Storage — Not Your Average Cupboard Task
A bag in a drawer won’t cut it. This compound requires a cool, dry home away from direct sunlight, sparks, or open flames. Moisture can do a number on its stability and if it degrades, those byproducts might pack their own punch. From my years working with brominated organics, I always trusted glass containers with tight seals more than plastic alternatives. Polyethylene bags may seem convenient, but vapors have a way of slipping out or dissolving right through. Lab flammables cabinets get crowded, but storing reactive solids separately lowers risk. Never let food or drinks get anywhere near it — cross-contamination is more common than people admit.
Keep storage areas well-ventilated. If you keep this material next to volatile acids, things can escalate if there’s a spill or leak. Labels mean nothing if they fade or get covered with fingerprint grime, so a fresh marker and clear hazard signs save headaches later. In shared spaces, listing storage details in an online inventory system helps everyone know what they’re working with.
Each Step Matters During Handling
Lab coats and nitrile gloves form the first barrier, with splash goggles and a face shield for concentrated quantities. Never risk short sleeves or cut-rate gloves—brominated compounds soak through like water through a screen door. I’ve seen colleagues skip goggles “just for a second,” only to regret it. If you feel the urge to work outside a fume hood, resist it. Dust from even a few milligrams can cause throat or skin irritation, and inhaling the fine powder turns an ordinary day upside-down. Keep the container close to the working spot, use a powder funnel, and work slow. The urge to dump and rush gets folks in trouble every time.
Spill kits stocked with absorbent materials and neutralizers sit at an arm’s reach in any lab I’d trust. Double-bag waste and close chemical containers tight after each use. Emergency showers and eyewash stations need a clear line of access — no rolling carts or clutter blocking your route. Accident response drills make a big difference. I’ve practiced them dozens of times, and during a real spill, muscle memory can save years off your stress level.
Training and Ongoing Vigilance
Every person who works with chemicals deserves full training. This goes way beyond reading a label or glancing over a safety data sheet. Staff briefings with hands-on demonstrations, clear written instructions posted right where materials are handled, and a culture where people look out for each other build trust. Safety can’t be a one-off reminder or a checkbox. Audits, refreshers, and a system where anyone can speak up about unsafe practice make the difference between a safe day and a disaster.
Rules around 2,4,6-Tribromoaniline aren’t optional. With proper containers, alert habits, and stubborn attention to every small detail, the risks drop. The science is important, but protecting people always comes first.
What are the possible health hazards and safety precautions for 2,4,6-Tribromoaniline?
What Makes 2,4,6-Tribromoaniline Risky?
Working in a lab, you learn quickly there are some chemicals you never take lightly. 2,4,6-Tribromoaniline isn’t as famous as chlorine or sulfuric acid, but that doesn’t mean it doesn’t come with real hazards. This compound, often used in dye, pharmaceutical, or pesticide manufacturing, has a pretty specific formula that gives it strong chemical properties. Inhaling its dust or vapors can irritate the respiratory tract, and even short-term exposure can make your throat feel raw. Skin exposure? That often leaves a rash or red patches. Getting it in your eyes is worse—burning, watering, sometimes blurred vision for hours.
Over time, working with or around this chemical without taking it seriously adds up. Chronic exposure may lead to persistent dermatitis or breathing issues. Many industrial health records show employees exposed to aromatic amines like this compound sometimes develop allergies or heightened sensitivity to smaller doses. Some aromatic amine relatives have shown carcinogenic effects in rodents, and while specific cancer data on 2,4,6-Tribromoaniline remain limited, it’s never smart to gamble with long-term risks.
Taking Precautions That Actually Work
There’s a big difference between following safety 'by the book' and actually keeping yourself safe. After years working in shared labs, I’ve watched people cut corners just to finish a batch or skip PPE because it feels inconvenient. That almost always ends in regrets when the discomfort hits or, worse, an accident happens. What kept me and my coworkers healthy?
Start with personal protective equipment—enclosed goggles, thick nitrile gloves, and proper lab coats aren’t there for show. 2,4,6-Tribromoaniline tends to cling to clothing and sticks around on unwashed hands. For this reason, simple habits like not eating or drinking while working and washing up after each job make more of a difference than you’d think.
Ventilation matters a lot. In my own experience, local exhaust fans and fume hoods keep air clear. If a space feels stuffy, it’s a warning sign. I’ve seen cases where one open beaker starts to spread enough chemical smell people notice headaches in less than an hour.
Emergency Response and Long-Term Safety
Spills happen to even the most careful. If someone knocks over a flask or drops a bottle, fast containment limits exposure. Lab protocols say to use absorbent pads, avoid direct contact, and get rid of waste in sealed chemical disposal bins. Never sweep up dry powder without a mask, because that raises dust. Safety showers and eyewash stations make a big difference if things go wrong.
Companies that use 2,4,6-Tribromoaniline should run honest risk assessments. Training new hires and holding regular drills gets everyone ready for the real thing. Up-to-date material safety data sheets posted in work areas encourage informed decisions. Tracking symptoms through occupational health clinics helps catch problems early.
Nobody sets out to get hurt at work. Tight standards, careful handling, and respect for chemical risks keep health problems from becoming long-term regrets. Knowing the risks and acting on them—every day—goes a lot further than a shelf full of manuals gathering dust.
Is 2,4,6-Tribromoaniline available in different purity grades or quantities?
Purity: Not Always One-Size-Fits-All
Anyone who’s bought chemicals for a lab knows the story—purity counts. For 2,4,6-Tribromoaniline, a brominated aromatic amine often used in synthesis or research, the label can read anything from technical grade to high-purity, sometimes 97% or even over 99%. Purity defines how much actual 2,4,6-Tribromoaniline ends up in your beaker versus the rest—unreacted materials, byproducts, dust from the process, maybe even moisture if the packaging let some in.
Go for the low-purity stuff and you risk throwing your experiment off. That tiny bit of unknown that sneaks in can send a reaction sideways—sometimes subtly, sometimes in full smoke-alarm territory. People handling trace analysis or pharma-focused research often pay up for purer compounds, because a quirky blip on the gas chromatograph is the last thing anyone wants to chase for an afternoon. Big companies producing large batches for polymers or flame retardants may aim for technical grade to save on costs, betting that extra impurities won’t derail the batch.
Quantities: From Grams to Drums
2,4,6-Tribromoaniline is not a household staple, but you can pick it up in a range of sizes if you know where to look. Research suppliers list quantities as small as a gram. Synthesis teams don’t want to fork out for a bucket when they need a pinch. You might see offers stretching up to kilograms or, for larger industry, full drums. Cost per gram drops fast as quantity climbs—a fact any grad student stretching a grant budget knows all too well.
Handling a delicate material in kilo quantities brings its own bag of problems. The chemical’s not especially toxic according to the data, but decent personal protective equipment stays a must. Packaging sometimes has to shield from light or moisture, and buyers working outside city centers deal with extra shipping hurdles. A tiny lab in a remote area pays more or waits longer. Sometimes the cost isn’t just what’s on the supplier’s invoice, but the days spent chasing a tracked package across customs.
Regulations and Sourcing: More Than Just Shopping Around
Any tale of chemical procurement wouldn’t be complete without plenty of red tape. For some locations, getting chemicals like 2,4,6-Tribromoaniline past regulations calls for detailed paperwork. Customs agents and safety inspectors both care what gets imported, how it gets stored, and where it will eventually find use. Companies need documentation on purity, batch numbers, and sometimes even the synthetic route taken before the chemical reaches the lab.
Major suppliers spell out purity levels with certificates of analysis. Anyone serious about reproducibility or regulatory compliance collects and keeps these certificates in case inspectors come knocking. Labs tied to government contracts know this drill. Enthusiasm fades fast if a supplier fudges the specs or skips testing before shipping. Inconsistent material puts projects at risk and eats into research budgets.
Real-World Solutions and Best Practice
Based on experience, establishing supplier relationships pays off. Trusted vendors answer questions about purity quickly and provide documentation upfront. Working with reputable companies often means fewer surprises down the line. Buying in excess only makes sense if storage conditions are up to scratch—temperature, darkness, moisture, and safety all become factors.
Researchers sometimes check the purity themselves using techniques like melting points or NMR, especially if early results seem off. Investing time in this sort of due diligence may seem like overkill, but it saves a whole lot of frustration. And always, clear labeling and careful logging of lot numbers keep future headaches away. In research and production, keeping an honest eye on purity, quantity, and documentation turns chemical procurement from a gamble into a routine part of the job.
| Names | |
| Preferred IUPAC name | 2,4,6-tribromoaniline |
| Other names |
2,4,6-Tribromobenzenamine 2,4,6-Tribromo-1-aminobenzene |
| Pronunciation | /ˌtraɪˌbroʊmoʊˈænɪliːn/ |
| Identifiers | |
| CAS Number | 583-56-8 |
| Beilstein Reference | 1209085 |
| ChEBI | CHEBI:87224 |
| ChEMBL | CHEMBL137873 |
| ChemSpider | 117176 |
| DrugBank | DB03261 |
| ECHA InfoCard | 100.018.851 |
| EC Number | 221-967-2 |
| Gmelin Reference | 80389 |
| KEGG | C14322 |
| MeSH | D017089 |
| PubChem CID | 12041 |
| RTECS number | BW6650000 |
| UNII | 6D82G6074W |
| UN number | UN2811 |
| CompTox Dashboard (EPA) | DTXSID4042211 |
| Properties | |
| Chemical formula | C6H4Br3N |
| Molar mass | 330.89 g/mol |
| Appearance | White to pale cream crystalline powder |
| Odor | Odorless |
| Density | 2.44 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 2.89 |
| Vapor pressure | 1.89E-4 mmHg at 25°C |
| Acidity (pKa) | 2.09 |
| Basicity (pKb) | pKb = 8.1 |
| Magnetic susceptibility (χ) | -80.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.756 |
| Viscosity | 400 cP (20°C) |
| Dipole moment | 3.34 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 158.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −46.4 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2200.6 kJ/mol |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and eye irritation, may cause respiratory irritation |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P261, P264, P271, P272, P280, P302+P352, P321, P333+P313, P362+P364, P501 |
| NFPA 704 (fire diamond) | 2,4,6-Tribromoaniline: NFPA 704 = 2-1-0 |
| Flash point | > 315°C |
| Lethal dose or concentration | LD50 (oral, rat): >2000 mg/kg |
| LD50 (median dose) | LD50 (median dose): >5,000 mg/kg (oral, rat) |
| NIOSH | JN8225000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.1 mg/m3 |
| Related compounds | |
| Related compounds |
Aniline 2-Bromoaniline 4-Bromoaniline 2,4-Dibromoaniline 2,6-Dibromoaniline 2,4,6-Trichloroaniline 2,4,6-Trimethylaniline |

